Abstract
The purpose of this study was to compare the relative efficacy and safety of different antibiotic drugs and recommend superior regimens in the treatment of bronchitis. With respect to the antibiotic comparisons against quinolones in terms of intention-to-treat patients, we concluded that quinolones had advantages over placebo, β-lactams, sulfonamides, and double β-lactams. Concerning treatment methods for clinically evaluable patients, quinolones demonstrated better performance than β-lactams and sulfonamides. The secondary effects of macrolides, quinolones, and double β-lactams were significantly more adverse than β-lactams with odds ratios (ORs) of 1.5 (95% credible interval [CrI] =1.1–2.0), 1.7 (95% CrI =1.2–2.3), and 2.7 (95% CrI =1.8–4.1), respectively. Significant differences in the prevalence of diarrhea as a secondary effect were only identified among the comparisons of double β-lactams against β-lactams and macrolides (OR =5.0, 95% CrI =2.1–12.0; OR =3.0, 95% CrI =1.7–5.4, respectively). Quinolones can be recommended as the superior treatment for bronchitis, in accordance with our cluster analysis with surface under the cumulative ranking curve. The primary outcomes of network meta-analysis indicated that quinolones showed the best performance among the 8 treatments studied, although β-lactams showed the lowest risk of adverse side effects. Quinolones are recommended as the primary treatment option for bronchitis patients, having taking into account the success rates and safety profiles of the eight drugs studied here.
Introduction
Bronchitis is a disease commonly caused by bacteriaCitation1 like Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalisCitation2 as well as by viruses.Citation3 Acute bronchitis (AB), chronic bronchitis (CB), and acute exacerbations of chronic bronchitis (AECB) are the three main types of bronchitis. Patients usually experience symptoms such as coughing, wheezing dyspnea, greater sputum volume, rales, and rhonchi.Citation4 The illness is a major public health concern, due to its remarkably high morbidity rate. Also of concern, the disease can induce long-term effects such as a decline in lung function and a higher risk of cardiovascular disease.Citation5–Citation10 Thus, the morbidity rate and severe health conditions caused by bronchitis constitute a serious burden for the modern health care system.
This disease has been studied for a long time, and various types of drugs have been developed or identified for the treatment of bronchitis including mucolytics and antibiotics; however, antibiotics have been used more commonly than mucolytics in both clinical trials and treatment of bacterial bronchitis. It has been noted that the administration of antibiotics is currently regarded as a considerable step in the treatment of AECB.Citation11 In addition, antibiotics have shown superior efficacy in their ability to reduce cardinal symptoms compared to other type of intervention.Citation12 For instance, AECB patients are commonly treated with many oral agents that actively act as antibacterials against pathogens, including fluoroquinolone, β-lactam, tetracycline, macrolide, or trimethoprim-sulfamethoxazole.Citation2 To the best of our knowledge, there are seven major groups of antibiotic treatments available in the treatment of bronchitis: β-lactams, macrolides, tetracyclines, quinolones, sulfonamides, double β-lactams, and double sulfonamides. The safety and tolerability profiles of tetracyclines,Citation13 moxifloxacin,Citation4,Citation14–Citation16 and clarithromycinCitation17 strongly suggest that these are superior compared to other drugs when administered in the treatment of bronchitis-related symptoms. However, clinical trials have shown that the commonly used β-lactams drug, phenethicillin, has demonstrated little efficacy compared to placebo.Citation18 Gemifloxacin, a type of quinolone with a powerful capacity in eliminating respiratory tract pathogens, enabled patients with AECB to remain recurrence-free after 26 weeks.Citation19 Post-therapy cure rates among AECB patients treated with telithromycin and axetil were 86.4% and 83.1%, respectively.Citation2 These results suggest a strong efficacy with respect to the treatment of AECB. Roxithromycin and doxycycline also displayed satisfactory outcomes, with clinical success rates of 81% and 80% and an adverse effect incidence rate of 12.2% and 33%, respectively. It has been recommended that roxithromycin should be considered the preferred antibiotic treatment for bronchitis due to its efficacy and safety characteristics.Citation20 Other related studiesCitation17,Citation21 have attempted to combine two or three different regimens, such as sulfadiazine and trimethoprim, to obtain a higher success rate. However, the overuse of various antibiotics in treating bacterial diseases including bronchitis has already led to a rise in antibiotic resistance worldwide.Citation2 This indicates the need for a wiser regulation and selection process for the use of antibiotics in the treatment of bacterial diseases. More well-focused and safer regimens for bacterial infections are needed, as this will reduce the potential risk of causing multiple drug resistance strains.
As such, researchers have been attempting to identify the most suitable antibiotic drug for the treatment of bronchitis with the highest efficacy and fewest adverse effects. Several meta-analyses (MAs) have already provided us with clear insight into the comparative efficacy and safety of different antibiotics, such as amoxicillin,Citation22 erythromycin,Citation23 macrolides,Citation24 quinolones,Citation24 moxifloxacin,Citation25,Citation26 penicillins,Citation27 and gemifloxacinCitation28 through head-to-head comparison methods. Nevertheless, we still know little about the relative efficacy of other medical treatments used to treat bronchitis. Thus, the antibiotic family of treatment options is still not comprehensively understood, as it has not been compared to other families of treatment options. Aside from this, assessment results have even demonstrated significant heterogeneity among the same antibiotic drugs, which further complicates the drug selection process.
However, the shortcomings extant in current conventional MAs and individual studies may be made up for by conducting a network meta-analysis (NMA) which offers indirect evidences between different regimens based on direct evidence provided by clinical trials. This new statistical method is now applied frequently in the field of pharmaceutical screening and evaluationCitation29–Citation33 and has proven to be an effective way to recognize the overall superiority and inferiority of different interventions.
The objective of this work is to compare the relative efficacy and safety of different antibiotic drugs and recommend superior regimens in the treatment of bronchitis, especially bacteria-induced bronchitis. It is hoped that the results of this NMA will provide insight into efficient treatment options as well as help to avoid the misuse of less effective and more harmful antibiotics. As far as we know, this is also the first NMA conducted on bronchitis-targeted antibiotic interventions.
Methods
Database and search strategies
We searched China National Knowledge Internet, PubMed, and Embase for relevant articles up to April 10, 2016, without restrictions on language. Keywords such as “bronchitis,” “anti-bacterial agents,” “quinolones,” “beta-lactams,” “macrolides,” “mucolytics,” “expectorants,” and “trimethoprim” as well as their correlated expressions were included in our search strategy. The reference lists of all retrieved articles were also reviewed manually to identify any relevant studies that may have been overlooked. Parallel literature screenings were carried out by two reviewers independently.
Inclusion criteria
All included articles met the following criteria: 1) randomized controlled trials (RCTs); 2) sufficient relevant data existed concerning the outcomes (the intention-to-treat [ITT] patients’ treatment success rate, clinically evaluable (CE) patients’ treatment success rate, adverse effects, and diarrhea); and 3) patients were diagnosed with bronchitis for at least three consecutive months or had a history of bronchitis.
Data extraction
Two investigators independently extracted relevant data from the included articles, including the name of the experiment conditions of sample subjects, dose and duration of treatment, and the drugs used in the treatment. Any disagreements between the two investigators were subject to a third party discussion until a consensus was established.
Statistical analysis
This NMA was carried out with a random-effects model of Bayesian framework analysis using WinBUGS (V1.4.3, University of Cambridge) and R software (V3.3.1). ORs and their 95% credible intervals (95% CrIs) were applied for comparisons between two target therapies on each outcome. Node splitting plots were constructed to show the consistency and inconsistency between direct and indirect evidence. A P-value of <0.05 indicated a significant difference. Heat plots were also graphed to better demonstrate the heterogeneity level. A warmer color indicated stronger heterogeneity concerning the relevant comparison. In addition, the surface under the cumulative ranking curve (SUCRA) was calculated to rank each therapy on 4 outcomes, including ITT patient treatment success rate, CE patient treatment success rate, adverse effects, and diarrhea. A higher SUCRA value corresponds to a higher level of efficiency and safety. Finally, cluster plots were drawn to make the process of choosing the most appropriate therapy more efficient.
Results
Basic characteristics of eligible studies
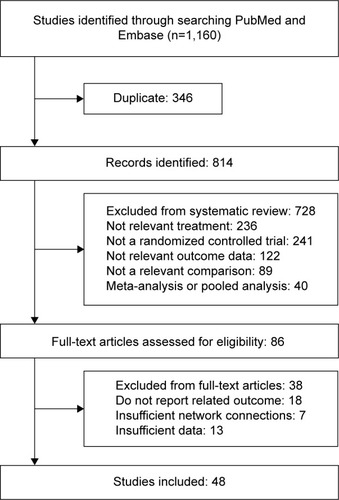
A total of 1,160 studies were identified by using the search strategies detailed above. Finally, 48 eligible studies were selected for data extraction. The literature screening process is shown in . Initially, 346 studies were categorized as ineligible due to duplication. Out of 814 full-text articles, we identified 86 and rejected the others due to irrelevant treatment, outcome data, comparison, or not being RCT studies. Finally, only 48 of the 86 articles, with a total of 14,448 participants were retrieved.Citation2,Citation4,Citation12–Citation21,Citation34–Citation69 Among the 48 studies, 29 of the randomized trials were double blind, 5 were single blind, and the others were open-label or unspecified. The most popular treatments assessed were quinolones and macrolides. Other relevant characteristics of these studies are shown in .
Table 1 Study and patient characteristics
NMA results of ITT patient treatment success and CE patient treatment success rates
shows the network comparisons between placebo and antibiotics on ITT patient treatment success rates and CE patient treatment success rates. Adverse effects including diarrhea are shown in .
Figure 2 Network meta-analysis results for the endpoints of ITT patient treatment success and CE patient treatment success rates. The network plots show direct comparison of different therapies, with node size corresponding to sample size. The number of included studies for specific direct comparison governs the thickness of solid lines. ORs with 95% CrIs are applied to evaluate the efficacy outcomes. Note that in the upper half of the table, column treatments are compared against row treatments, whereas in the lower half of the table, row treatments are compared against column treatments. Bold data represents significant results.
Abbreviations: ITT, intention-to-treat; CE, clinically evaluable; OR, odds ratio; CrI, credible interval.
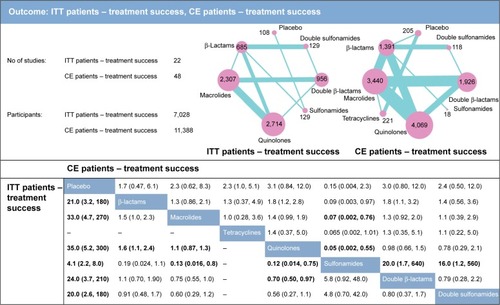
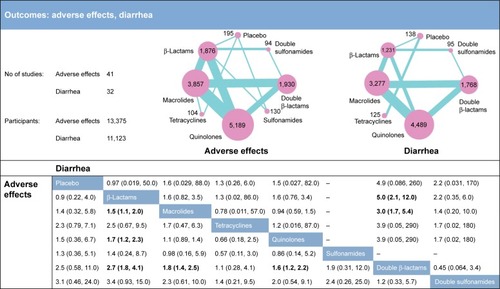
Figure 3 Network meta-analysis results for the endpoints of adverse effects and diarrhea. The network plots show direct comparison of different therapies, with node size corresponding to sample size. The number of included studies for specific direct comparison governs the thickness of solid lines. ORs with 95% CrIs are applied to evaluate the efficacy outcomes. Note that in the upper half of the table, column treatments are compared against row treatments, whereas in the lower half of the table, row treatments are compared against column treatments. Bold data represents significant results.
As illustrated in , β-lactams, macrolides, quinolones, sulfonamides, double β-lactams, and double sulfonamides were all more successful than placebo in treating ITT patients. From comparisons between quinolones and other treatments, we could conclude that quinolones had an advantage over placebo (OR =35.0, 95% CrI =5.2–300.0), β-lactams (OR =1.6, 95% CrI =1.1–2.4), sulfonamides (OR =8.3, 95% CrI =1.3–71.4), and double β-lactams (OR =1.4, 95% CrI =1.0–2.0). Aside from this, sulfonamides seemed to be significantly less efficient than macrolides (OR =0.13, 95% CrI =0.016–0.8).
We detected no significant difference between the effectiveness of placebo and antibiotics on CE patients. However, β-lactams demonstrated statistically lower efficacy when compared with quinolones and double β-lactams (OR =1.8, 95% CrI =1.2–2.8; OR =1.8, 95% CrI =1.1–3.2), while sulfonamides showed less overall benefits than β-lactams (OR =0.09, 95% CrI =0.003–0.97). Sulfonamides were also inferior to macrolides and quinolones with ORs of 0.07 (95% CrI =0.002–0.76) and 0.05 (95% CrI =0.002–0.55), respectively. In addition, both double β-lactams and double sulfonamides showed higher efficacy compared to sulfonamides (OR =20.0, 95% CrI =1.7–640; OR =16.0, 95% CrI =1.2–560, respectively). This suggested to us that quinolones and macrolides were a suitable treatment option for ITT bronchitis patients, while quinolones and double β-lactams proved superior to other antibiotics as a treatment option in CE bronchitis patients.
NMA results of all adverse effects and diarrhea
As to the secondary outcomes of all adverse effects shown in , macrolides, quinolones, and double β-lactams were three drugs which showed significantly higher risk of inducing adverse effects compared to β-lactams (OR =1.5, 95% CrI =1.1–2.0; OR =1.7, 95% CrI =1.2–2.3; OR =2.7, 95% CrI =1.8–4.1). In addition, double β-lactams were more harmful than macrolides and quinolones with ORs of 1.8 (95% CrI =1.4–2.5) and 1.6 (95% CrI =1.2–2.2), respectively.
also offers evidence concerning diarrhea as an adverse effect. We did not conduct a study on diarrhea as a side effect of sulfonamides due to a lack of data. The only significant differences were identified in the comparisons of double β-lactams against β-lactams and macrolides (OR =5.0, 95% CrI =2.1–12.0; OR =3.0, 95% CrI =1.7–5.4, respectively). Based on the above safety findings, β-lactams seemed to be the most tolerable therapy as it was the least likely to induce adverse effects including diarrhea. When adverse effects were taken into consideration, double β-lactams seems to be the therapy with the worst adverse effects in bronchitis.
Consistency assessments
In this study, node splitting plots () and net heat plots () were used to assess the consistency level between direct and indirect evidence. No significant discrepancy was found between direct and indirect evidence on ITT patient treatment success rates and CE patient treatment success rates. However, there was inconsistency in the comparison between direct and indirect evidence from quinolones versus β-lactams, quinolones versus macrolides, and macrolides versus β-lactams. Their associated P-values of adverse effects and diarrhea were <0.05, as demonstrated in . The red blocks shown in also indicate that a strong inconsistency was detected in the above noted comparisons.
Figure 4 Node-splitting results for ITT patient treatment success, CE patient treatment success, adverse effects, and diarrhea.
Abbreviations: ITT, intention-to-treat; CE, clinically evaluable; OR, odds ratio; CrI, credible interval.
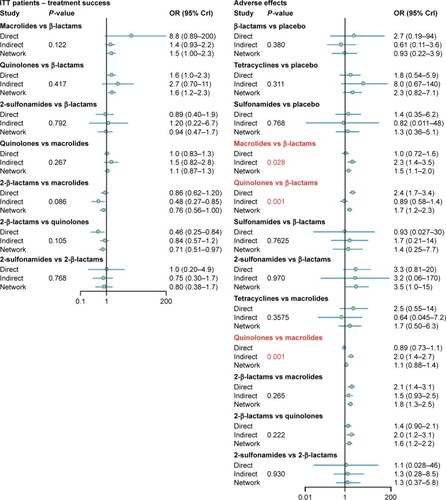
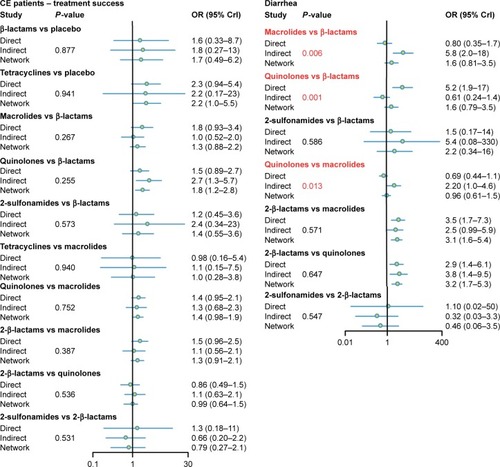
Figure 5 Net heat plot. The size of the gray squares indicates the contribution of direct evidence (shown in the column) to the network evidence (shown in the row). The colors are associated with the change in inconsistency between direct and indirect evidence (shown in the row). Blue colors indicate an increase of inconsistency and warm colors indicate a decrease.
Treatment rankings and cluster analysis with SUCRA
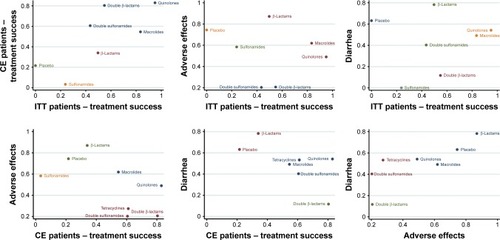
SUCRA rankings of each therapy on 4 indexes are demonstrated in and . First, quinolones and macrolides proved to be the top two most successful antibiotic treatments for ITT bronchitis patients. They were both associated with a medium risk of adverse effects including diarrhea, as indicated in . Moreover, quinolones and double β-lactams were effective in treating CE bronchitis patients but double β-lactams seemed to pose the highest overall risk of inducing adverse effects including diarrhea, as illustrated in the cluster plots in . β-Lactams ranked first in reducing both overall adverse effects and diarrhea. All in all, the ranking and cluster analyses suggest quinolones as the superior treatment for bronchitis among antibiotics, considering that it produced an even balance between efficacy and safety performance.
Table 2 Surface under the cumulative ranking curve (SUCRA) results
Figure 6 SUCRA results.
Abbreviations: ITT, intention-to-treat; CE, clinically evaluable; SUCRA, surface under the cumulative ranking curve.
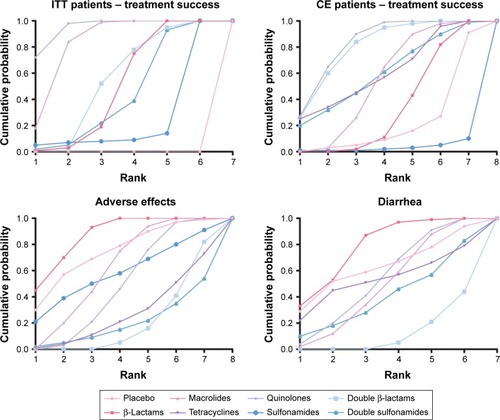
Figure 7 Clustered ranking plot of the network. The plot is based on cluster analysis of SUCRA values. Each plot shows SUCRA values for two outcomes: ITT patient treatment success, CE patient treatment success, adverse effects, and diarhoea. Each color represents a group of treatments which belongs to the same cluster. Treatments lying in the upper right corner are more effective and safer than the other treatments.
Discussion
Primary outcomes
Based on our findings, quinolones and macrolides had the highest patient success rates, thus are recommended as the two most efficient drugs in treating ITT patients. In the meantime, quinolones and double β-lactams showed significant advantages over other types of antibiotics in terms of CE patient success rate. It should be noted that quinolones are a family of synthetic broad-spectrum antibiotic drugs extracted from natural sources which function as antimicrobials.Citation70,Citation71 The core mechanism of their antibiotic ability is in preventing bacterial DNA from unwinding and duplicating, and thus, they have drastic effects on the reproduction cycles of bacteria cells.Citation72
Macrolides have also been suggested as an effective treatment option for ITT bronchitis patients. This family of antibiotics also originates from natural products and can reduce the activity of bacteria by inhibiting the protein formation process of the bacteria.Citation73,Citation74 Double β-lactams are a combination of β-lactams, and moxicillin/clavulanate is the most commonly used combination. It was reported that combining two different β-lactams could produce a higher spectrum of action and efficacy compared to what is achievable with a single β-lactam treatment.Citation22 This is also the reason for the inefficiency of some β-lactams in the treatment of bacteria-induced bronchitis.
The strong curative capacity of quinolones is displayed in various extant studies, showing that moxifloxacin, a quinolone, was superior to β-lactamsCitation14 and macrolides such as clarithromycin.Citation16 An MA study targeting the treatment of oral gemifloxacin at 320 mg daily for AECB patients also confirmed the superiority of quinolones compared to the other approved antibiotics tested.Citation28 In another MA study by Siempos et al,Citation24 quinolones was again proved to be more effective in removing bacteria than macrolides, which was consistent with our findings.
A similar level of efficacy was observed between macrolides and tetracyclinesCitation20 as well as between macrolides and double β-lactamsCitation17,Citation24,Citation42,Citation44,Citation52,Citation61 on the basis of both clinical and MA studies. However, our study did show that macrolides demonstrated slightly higher efficacy all other treatments apart from quinolones in terms of ITT patient success rate. Double β-lactams was the second most effective regimen with respect to CE patient success rate, and there was also evidence suggesting a similar efficacy rate of this antibiotic to double sulfonamides like co-trimoxazole,Citation65 which might be due to the small sample size of only 49 as well as the particularly older age of patients (average 62.6 years) involved in the included double sulfonamide research. In addition, the low efficacy of β-lactams indicated in our study corresponded with other findings,Citation18,Citation25,Citation26 and no superiority concerning the efficacy of this treatment was demonstrated.
More broadly, the MA study carried out by Dimopoulos et alCitation75 on various types of bronchitis-related antibiotics compared the relative efficacy and tolerability of first-line (ie, amoxicillin, trimethoprim/sulfamethoxazole, and doxycycline) and second-line antibiotic regimens (ie, amoxicillin/clavulanic, macrolides, cephalosporins, and quinolones), concluding that the second-line agents were more potent than the first-line agents, which strongly supported our conclusions.
Secondary outcomes
Safety or tolerability characteristics were also critical in the process of drug selection analysis. In this NMA, we analyzed all the adverse effects as an aggregate as well as assessed diarrhea as an adverse effect individually. The results indicated that patients receiving double β-lactams had the highest possibility of experiencing adverse effects including diarrhea compared to all other treatments included. Conversely, the most widely used β-lactams proved to have the lowest risk of causing side effects.
Discrepancy between direct and indirect evidence was detected in the pairings of quinolones versus β-lactams, quinolones versus macrolides, and macrolides versus β-lactams, which might be attributed to the fact that the RCTs included in our study not only used various drugs from the same antibiotic group but also different dosages of the same treatment. Although elements of this issue existed elsewhere, this kind of heterogeneity was more commonly detected in the three groups noted above.
After a careful examination of the results and conclusions of the related studies, we found that the findings from other studies were similar to what we identified concerning secondary outcomes of antibiotics commonly used in the treatment of bronchitis. With respect to the comparison of macrolides versus β-lactams, one study showed the response rate of common adverse events was 12.8% versus 11.8% for diarrhea and 8.9% versus 3.2% for nausea in telithromycin and cefuroxime axetil patients, respectively,Citation2 showing that macrolides were slightly less tolerable than β-lactams, which was similar to what this NMA illustrated. Many studies also showed corresponding evidence to our finding that double β-lactams were associated with a higher risk of inducing drug-related adverse effects on subjects when macrolides were compared to a combination of β-lactams (such as double β-lactams).Citation17,Citation42,Citation52,Citation61 For example, one study demonstrated that the frequency of adverse effects regarding telithromycin (a type of macrolide) was only half that of amoxicillin/clavulanate.Citation42 However, double sulfonamides, such as co-trimazine, were proven to be well tolerated when treating AECB patients.Citation21 This was contradictory to our findings, where this group ranked the second lowest in terms of adverse effects. This inconsistency might be due to the fact that the mentioned study was published in 1984, and the associated treating methods, bacterial susceptibility, and patient backgrounds may be quite different from current studies.
Although quinolones and macrolides were recommended as optimal treatments for bronchitis on the basis of the primary outcomes of our NMA, their medium adverse effects including diarrhea lowered their application value. US Centers for Disease Control and Prevention found that patients treated with fluoroquinolones, a type of quinolone, experienced adverse events more often than with macrolides.Citation76 With respect to macrolides, their safety was also questioned by one study, which demonstrated that they could lead to debilitating myopathy.Citation77 One possible explanation of this could be that some macrolides are potent inhibitors of the cytochrome P450 system.Citation73 The most harmful antibiotic regimen was double β-lactams, such as amoxicillin/clavulanate and pivampicillin/pivmecillinam. In our study, they were shown to increase the risk of yeast infection and diarrhea and are recommended to be used only within 14 days by the UK Committee on Safety of Medicines.Citation22 Our findings with respect to the adverse effects of various antibiotics offered us a systematic review of the relative safety of these treatments for bronchitis, which to some degree offsets the lack of associated wide-range safety comparisons in the current literature.
Strengths and limitations
Through this research, we obtained an overall understanding of the efficacy as well as safety outcomes from a total of eight groups of interventions including placebo. Unlike the previous individual studies and MAs, this study managed to screen out the favorable treatments for bronchitis pertaining to both CE and ITT success rates and also suggested the drugs with the highest and lowest adverse effects for each. A relatively large sample size and comprehensive antibiotic screening basis (a total of 36 antibiotics belonging to seven groups) added to the reliability and the comprehensiveness of our findings. It should be noted that all of the included studies were RCTs, the outcomes of which are generally expected to be of higher reliability.
Nevertheless, the results of this NMA should be interpreted with the consideration of several limitations. First, we included all three types of bronchitis (AB, CB, and AECB) in our study along with subjects of various ages, drugs of different dosages, and durations. All these variables can influence the applicability and feasibility of the ideal intervention. To be more specific, as it has been stated in the introduction that virus is also the cause of bronchitis. Indeed, AB is primarily caused by virusesCitation78 and therefore does not respond to antibiotics while AECB is associated with both bacteria and viruses.Citation79 This might lead to efficacy variation in the antibiotics among different subgroups of patients. Thus, a future subgroup NMA might be needed to supplement these results. Another drawback of this study is that bacterial susceptibility has not been taken into account, which also partly influences the efficacy evaluation of antibiotics. Many of the studies are also fairly old (9 before 1990, most of the others between 1990 and 2010, and only one after 2010). As we know, the resistance of bacteria to current antibiotics is highly likely to have changed over these periods,Citation80 further affecting the efficacy of antibiotic treatments. As such, a lack of recent RCTs could lower the reliability of the NMA results. In addition, regional variation in resistance might also exist,Citation81 which would mean that treatment suggestions may vary among different countries and regions. Stratification by regions is also thus needed in subsequent research.
Prospect
In this NMA, we included two primary outcomes: ITT patient success rate and CE patient success rate. As was indicated in our findings, the rankings of antibiotics based on these two types of patients were quite different on the basis of ITT and CE patients’ success rate. This inconsistency largely depended on the type of patients involved. In the future, a weighted composition might be applied to combine ITT and CE patient profiles together to form a more inclusive antibiotic recommendation in the treatment of bronchitis.
Studies concerning the antibiotic treatments for bronchitis have often failed to report enough of the adverse effects which appeared during the study. This is a deficiency when considered in contrast to an abundance of efficacy profilesCitation82 and could affect the process of screening out an efficacious and safe treatment option. The related discrepancy observed in our NMA also calls for more large-sample RCTs of antibiotic treatments, especially sulfonamides, double sulfonamides, and tetracyclines. Future studies should address these potential problems.
In conclusion, our NMA identified quinolones and macrolides as strong choices in the treatment of ITT bronchitis while quinolones and double β-lactams performed best in the treatment of CE bronchitis patients. Double β-lactams proved to have the highest risk of adverse events while β-lactams were the least likely to cause adverse effects when prescribed to bronchitis patients. In addition, quinolones and macrolides both showed median adverse effects across all the included antibiotics. Thus, considering both treatment success rates and safety profiles, quinolones were recommended as the most preferable treatment option.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Acknowledgments
We would like to acknowledge the reviewers for their helpful comments on this paper. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure
The authors report no conflicts of interest in this work.
References
- PapiABellettatoCMBraccioniFInfections and airway inflammation in chronic obstructive pulmonary disease severe exacerbationsAm J Respir Crit Care Med2006173101114112116484677
- ZervosMJHeyderAMLeroyBOral telithromycin 800 mg once daily for 5 days versus cefuroxime axetil 500 mg twice daily for 10 days in adults with acute exacerbations of chronic bronchitisJ Int Med Res200331315716912870368
- MandellGLBennettJEDolinRBook Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. Volumes 1 and 24th edChurchill Livingstone1995
- SchabergTBallinIHuchonGBassarisHHampelBReimnitzPA multinational, multicentre, non-blinded, randomized study of moxifloxacin oral tablets compared with co-amoxiclav oral tablets in the treatment of acute exacerbation of chronic bronchitisJ Int Med Res200129431432811675905
- DonaldsonGCHurstJRSmithCJHubbardRBWedzichaJAIncreased risk of myocardial infarction and stroke following exacerbation of COPDChest201013751091109720022970
- Soler-CatalunaJJMartinez-GarciaMARoman SanchezPSalcedoENavarroMOchandoRSevere acute exacerbations and mortality in patients with chronic obstructive pulmonary diseaseThorax2005601192593116055622
- DonaldsonGCSeemungalTABhowmikAWedzichaJARelationship between exacerbation frequency and lung function decline in chronic obstructive pulmonary diseaseThorax2002571084785212324669
- KannerREAnthonisenNRConnettJELower respiratory illnesses promote FEV(1) decline in current smokers but not ex-smokers with mild chronic obstructive pulmonary disease: results from the lung health studyAm J Respir Crit Care Med2001164335836411500333
- SeemungalTADonaldsonGCPaulEABestallJCJeffriesDJWedzichaJAEffect of exacerbation on quality of life in patients with chronic obstructive pulmonary diseaseAm J Respir Crit Care Med19981575 Pt 1141814229603117
- AnthonisenNRManfredaJWarrenCPHershfieldESHardingGKNelsonNAAntibiotic therapy in exacerbations of chronic obstructive pulmonary diseaseAnn Intern Med198710621962043492164
- FalagasMEAvgeriSGMatthaiouDKDimopoulosGSiemposIIShort- versus long-duration antimicrobial treatment for exacerbations of chronic bronchitis: a meta-analysisJ Antimicrob Chemother200862344245018467303
- ZervosMMartinezFJAmsdenGWRothermelCDTreadwayGEfficacy and safety of 3-day azithromycin versus 5-day moxifloxacin for the treatment of acute bacterial exacerbations of chronic bronchitisInt J Antimicrob Agents2007291566117189096
- BuchananJBuchananWWMelroseAGMcGJPriceAULong-term prophylactic administration of tetracycline to chronic bronchiticsLancet19582704971972313588985
- WilsonRAllegraLHuchonGShort-term and long-term outcomes of moxifloxacin compared to standard antibiotic treatment in acute exacerbations of chronic bronchitisChest2004125395396415006954
- DeAbateCAMathewCPWarnerJHHeydAChurchDThe safety and efficacy of short course (5-day) moxifloxacin vs. azithromycin in the treatment of patients with acute exacerbation of chronic bronchitisRespir Med200094111029103711127487
- ChodoshSDeAbateCAHaverstockDAneiroLChurchDShort-course moxifloxacin therapy for treatment of acute bacterial exacerbations of chronic bronchitis. The Bronchitis Study GroupRespir Med2000941182710714475
- MartinotJBCarrWDCullenSA comparative study of clarithromycin modified release and amoxicillin/clavulanic acid in the treatment of acute exacerbation of chronic bronchitisAdv Ther200118111111512528
- JohnstonRNLockhartWSmithDHCadmanNKA trial of phenethicillin in chronic bronchitisBr Med J19612525898598614452159
- WilsonRSchentagJJBallPMandellLA comparison of gemifloxacin and clarithromycin in acute exacerbations of chronic bronchitis and long-term clinical outcomesClin Ther200224463965212017408
- De VliegerADruartMPuttemansMRoxithromycin versus doxycycline in the treatment of acute exacerbations of chronic bronchitisDiagn Microbiol Infect Dis1992154 Suppl123S127S1617923
- LeoneGLanzaRPaolettiVParlapianoCVincentelliGMA clinical trial of co-trimazine (sulfadiazine + trimethoprim) in flare-ups of chronic bronchitisJ Int Med Res1984121196363154
- GilliesMRanakusumaAHoffmannTCommon harms from amoxicillin: a systematic review and meta-analysis of randomized placebo-controlled trials for any indicationCMAJ20151871E21E3125404399
- BentSSaintSVittinghoffEGradyDAntibiotics in acute bronchitis: a meta-analysisAm J Med19991071626710403354
- SiemposIIDimopoulosGKorbilaIPMantaKFalagasMEMacrolides, quinolones and amoxicillin/clavulanate for chronic bronchitis: a meta-analysisEur Respir J20072961127113717301097
- LiuKXXuBWangJEfficacy and safety of moxifloxacin in acute exacerbations of chronic bronchitis and COPD: a systematic review and meta-analysisJ Thorac Dis20146322122924624286
- MiravitllesMMolinaJBrosaMClinical efficacy of moxifloxacin in the treatment of exacerbations of chronic bronchitis: a systematic review and meta-analysisArch Bronconeumol2007431222817257560
- KorbilaIPMantaKGSiemposIIDimopoulosGFalagasMEPenicillins vs trimethoprim-based regimens for acute bacterial exacerbations of chronic bronchitis: meta-analysis of randomized controlled trialsCan Fam Physician2009551606719155372
- ZhangLWangRFalagasMEChenLALiuYNGemifloxacin for the treatment of community-acquired pneumonia and acute exacerbation of chronic bronchitis: a meta-analysis of randomized controlled trialsChin Med J (Engl)2012125468769522490497
- ZhuGQZouZLZhengJNSystematic review and network meta-analysis of randomized controlled trials: comparative effectiveness and safety of direct-acting antiviral agents for treatment-naive hepatitis C genotype 1Medicine (Baltimore)2016959e300426945424
- ZhaoBCJiangHYMaWYAlbendazole and corticosteroids for the treatment of solitary cysticercus granuloma: a network meta-analysisPLoS Negl Trop Dis2016102e000441826849048
- ZhangMYZhuGQShiKQSystematic review with network meta-analysis: comparative efficacy of oral nucleos(t)ide analogues for the prevention of chemotherapy-induced hepatitis B virus reactivationOncotarget2016721306423065827121321
- VeronikiAAStrausSEAshoorHMComparative safety and effectiveness of cognitive enhancers for Alzheimer’s dementia: protocol for a systematic review and individual patient data network meta-analysisBMJ Open201661e010251
- TveteIFNatvigBGasemyrJMelandNRoineMKlempMCorrection: comparing effects of biologic agents in treating patients with rheumatoid arthritis: a multiple treatment comparison regression analysisPLoS One2016111e014663326812687
- BlasiFTarsiaPManteroMMorlacchiLCPifferFCefditoren versus levofloxacin in patients with exacerbations of chronic bronchitis: serum inflammatory biomarkers, clinical efficacy, and microbiological eradicationTher Clin Risk Manag20139556423430960
- MartinezFJGrossmanRFZadeikisNPatient stratification in the management of acute bacterial exacerbation of chronic bronchitis: the role of levofloxacin 750 mgEur Respir J20052561001101015929954
- StarakisIGogosCABassarisHFive-day moxifloxacin therapy compared with 7-day co-amoxiclav therapy for the treatment of acute exacerbation of chronic bronchitisInt J Antimicrob Agents200423212913715013037
- WilsonRLanganCBallPOral gemifloxacin once daily for 5 days compared with sequential therapy with i.v. ceftriaxone/oral cefuroxime (maximum of 10 days) in the treatment of hospitalized patients with acute exacerbations of chronic bronchitisRespiratory Medicine200397324224912645831
- SolerMLodeHBaldwinRRandomised double-blind comparison of oral gatifloxacin and co-amoxiclav for acute exacerbation of chronic bronchitisEur J Clin Microbiol Infect Dis200322314415012649711
- AmsdenGWBairdIMSimonSTreadwayGEfficacy and safety of azithromycin vs levofloxacin in the outpatient treatment of acute bacterial exacerbations of chronic bronchitisChest2003123377277712628877
- WeissLROpen-label, randomized comparison of the efficacy and tolerability of clarithromycin, levofloxacin, and cefuroxime axetil in the treatment of adults with acute bacterial exacerbations of chronic bronchitisClin Ther20022491414142512380633
- GrassiCCasaliLCurtiETellariniMLazzaroCSchitoGEfficacy and safety of short course (5-day) moxifloxacin vs 7-day ceftriaxone in the treatment of acute exacerbations of chronic bronchitis (AECB)J Chemother200214659760812583552
- AubierMAldonsPMLeakATelithromycin is as effective as amoxicillin/clavulanate in acute exacerbations of chronic bronchitisRespir Med2002961186287112418583
- GotfriedMHDeAbateCAFogartyCMathewCPSokolWNComparison of 5-day, short-course gatifloxacin therapy with 7-day gatifloxacin therapy and 10-day clarithromycin therapy for acute exacerbation of chronic bronchitisClin Ther20012319710711219483
- AnzuetoAFisherCLJrBusmanTOlsonCAComparison of the efficacy of extended-release clarithromycin tablets and amoxicillin/clavulanate tablets in the treatment of acute exacerbation of chronic bronchitisClin Ther2001231728611219481
- SchouenborgPGerdesNRasmussenHWickers-NielsenNMathiassenEAzithromycin versus pivampicillin in the treatment of acute exacerbations of chronic bronchitis: a single-blind, double-dummy, multicentre studyJ Int Med Res200028310111010983860
- KreisA comparison of moxifloxacin and azithromycin in the treatment of acute exacerbations of chronic bronchitisJ Clin Outcomes Manag20007123337
- FileTSchlemmerBGarauJLodeHLynchSYoungCGemifloxacin versus amoxicillin/clavulanate in the treatment of acute exacerbations of chronic bronchitis. The 070 Clinical Study groupJ Chemother200012431432510949981
- WilsonRKubinRBallinIFive day moxifloxacin therapy compared with 7 day clarithromycin therapy for the treatment of acute exacerbations of chronic bronchitisJ Antimicrob Chemother199944450151310588312
- LanganCEZuckPVogelFRandomized, double-blind study of short-course (5 day) grepafloxacin versus 10 day clarithromycin in patients with acute bacterial exacerbations of chronic bronchitisJ Antimicrob Chemother199944451552310588313
- Van RoyenPBetzWHeyrmanJTaziauxPVan den HauteMPoelmanMDirithromycin versus amoxiclav in the treatment of acute exacerbations of chronic bronchitisJ Int Med Res199725133409027671
- HoepelmanIMMollersMJvan SchieMHA short (3-day) course of azithromycin tablets versus a 10-day course of amoxycillin-clavulanic acid (co-amoxiclav) in the treatment of adults with lower respiratory tract infections and effects on long-term outcomeInt J Antimicrob Agents1997931411469552709
- ZachariahJA randomized, comparative study to evaluate the efficacy and tolerability of a 3-day course of azithromycin versus a 10-day course of co-amoxiclav as treatment of adult patients with lower respiratory tract infectionsJ Antimicrob Chemother199637Suppl C1031138818851
- GrisPOnce-daily, 3-day azithromycin versus a three-times-daily, 10-day course of co-amoxiclav in the treatment of adults with lower respiratory tract infections: results of a randomized, double-blind comparative studyJ Antimicrob Chemother199637Suppl C93101
- SachsAPKoeterGHGroenierKHvan der WaaijDSchiphuisJMeyboom-de JongBChanges in symptoms, peak expiratory flow, and sputum flora during treatment with antibiotics of exacerbations in patients with chronic obstructive pulmonary disease in general practiceThorax19955077587637570411
- BeghiGBerniFCarratuLEfficacy and tolerability of azithromycin versus amoxicillin/clavulanic acid in acute purulent exacerbation of chronic bronchitisJ Chemother1995721461527666122
- VerheijTJHermansJMulderJDEffects of doxycycline in patients with acute cough and purulent sputum: a double blind placebo controlled trialBr J Gen Pract1994443864004048790652
- UlmerWFleroxacin versus amoxicillin in the treatment of acute exacerbation of chronic bronchitisAm J Med1993943A136S141S8452170
- ChodoshSEfficacy of fleroxacin versus amoxicillin in acute exacerbations of chronic bronchitisAm J Med1993943A131S135S8452169
- MertensJCvan BarneveldPWAsinHRDouble-blind randomized study comparing the efficacies and safeties of a short (3-day) course of azithromycin and a 5-day course of amoxicillin in patients with acute exacerbations of chronic bronchitisAntimicrob Agents Chemother1992367145614591324645
- GuayDRCraftJCComparative safety and efficacy of clarithromycin and ampicillin in the treatment of out-patients with acute bacterial exacerbation of chronic bronchitisJ Intern Med199223132953011532615
- DautzenbergBScheimbergABrambillaCComparison of two oral antibiotics, roxithromycin and amoxicillin plus clavulanic acid, in lower respiratory tract infectionsDiagn Microbiol Infect Dis1992154 Suppl85S89S1617930
- BachandRTJrComparative study of clarithromycin and ampicillin in the treatment of patients with acute bacterial exacerbations of chronic bronchitisJ Antimicrob Chemother199127Suppl A91100
- AldonsPMA comparison of clarithromycin with ampicillin in the treatment of outpatients with acute bacterial exacerbation of chronic bronchitisJ Antimicrob Chemother199127Suppl A1011081827095
- GoveRICaytonRMA double-blind comparison of amoxycillin with trimethoprim in acute exacerbations of chronic bronchitisJ Antimicrob Chemother19851544954993891710
- LalSMcGhieDKerfootPA comparison of pivmecillinam/pivampicillin and co-trimoxazole in hospitalized patients with acute exacerbations of chronic bronchitisJ Antimicrob Chemother19841421791846334071
- AndersonGWilliamsLPardoeTPeelETCo-trimoxazole versus cefaclor in acute on chronic bronchitisJ Antimicrob Chemother1981864874896977523
- DaviesDDarkeCSSulfametopyrazine prophylaxis in chronic bronchitisBr J Dis Chest1978723231234359024
- PinesANandiARRaafatHRahmanMAmoxycillin and co-trimoxazole in acute purulent exacerbations of chronic bronchitisChemotherapy19772315864
- StottNCWestRRRandomised controlled trial of antibiotics in patients with cough and purulent sputumBr Med J197626035556559786428
- HeebSFletcherMPChhabraSRDiggleSPWilliamsPCamaraMQuinolones: from antibiotics to autoinducersFEMS Microbiol Rev201135224727420738404
- IvanovDVBudanovSVCiprofloxacin and antibacterial therapy of respiratory tract infectionsAntibiot Khimioter20065152937
- HooperDCEmerging mechanisms of fluoroquinolone resistanceEmerg Infect Dis20017233734111294736
- TensonTLovmarMEhrenbergMThe mechanism of action of macrolides, lincosamides and streptogramin B reveals the nascent peptide exit path in the ribosomeJ Mol Biol200333051005101412860123
- DrainasDKalpaxisDLCoutsogeorgopoulosCInhibition of ribosomal peptidyltransferase by chloramphenicol. Kinetic studiesEur J Biochem1987164153583549307
- DimopoulosGSiemposIIKorbilaIPMantaKGFalagasMEComparison of first-line with second-line antibiotics for acute exacerbations of chronic bronchitis: a metaanalysis of randomized controlled trialsChest2007132244745517573508
- ShehabNPatelPRSrinivasanABudnitzDSEmergency department visits for antibiotic-associated adverse eventsClin Infect Dis200847673574318694344
- SathasivamSLeckyBStatin induced myopathyBMJ2008337a228618988647
- AlbertRHDiagnosis and treatment of acute bronchitisAm Fam Physician201082111345135021121518
- BruntonSCarmichaelBPColganRAcute exacerbation of chronic bronchitis: a primary care consensus guidelineAm J Manag Care2004101068915521160
- LivermoreDMFourteen years in resistanceInt J Antimicrob Agents201239428329422386741
- AndradeSSDGalesACSaderHSAntimicrobial resistance in gram-negative bacteria from developing countriesSosaA de JByarugabaDKAmabileCHsuehPRKariukiSOkekeINAntimicrobial Resistance in Developing CountriesNew YorkSpringer-Verlag2010249266
- LokeYKPriceDHerxheimerASystematic reviews of adverse effects: framework for a structured approachBMC Med Res Methodol200773217615054