Abstract
Background
Estimation suggests that at least 4 million people die, annually, as a result of chronic respiratory disease (CRD). The Global Alliance against Chronic Respiratory Diseases (GARD) was formed following a mandate from the World Health Assembly to address this serious and growing health problem.
Objectives
To investigate the prevalence of CRD in Russian symptomatic patients and to evaluate the frequency of major risk factors for CRD in Russia.
Methods
A cross-sectional, population-based epidemiological study using the GARD questionnaire on adults from 12 regions of the Russian Federation. Common respiratory symptoms and risk factors were recorded. Spirometry was performed in respondents with suspected CRD. Allergic rhinitis (AR) and chronic bronchitis (CB) were defined by the presence of related symptoms according to the Allergic Rhinitis and its Impact on Asthma and the Global Initiative for Obstructive Lung Disease guidelines; asthma was defined based on disease symptoms; chronic obstructive pulmonary disease (COPD) was defined as a post-bronchodilator forced expiratory volume per 1 second/forced vital capacity ratio <0.7 in symptomatic patients, following the Global Initiative for Obstructive Lung Disease guidelines.
Results
The number of questionnaires completed was 7,164 (mean age 43.4 years; 57.2% female). The prevalence of asthma symptoms was 25.7%, AR 18.2%, and CB 8.6%. Based on patient self-reported diagnosis, 6.9% had asthma, 6.5% AR, and 22.2% CB. The prevalence of COPD based on spirometry in patients with respiratory symptoms was estimated as 21.8%.
Conclusion
The prevalence of respiratory diseases and risk factors was high in Russia when compared to available data. For bronchial asthma and AR, the prevalence for related symptoms was higher than self-reported previous diagnosis.
Introduction
Chronic respiratory diseases (CRDs) are recognized as being the major cause for premature death in adult populations worldwide. Preventable and treatable CRDs include chronic obstructive pulmonary disease (COPD), asthma, and respiratory allergies.Citation1
In general, the prevalence of CRD is increasing everywhere and in particular amongst children and the elderly.Citation1 The burden of CRD has major adverse effects on the quality of life and disability of affected individuals. It has been predicted that the global burden of CRD will increase considerably in the future, even though many preventable CRDs can be controlled with adequate management in both developedCitation2 and developing countries,Citation3,Citation4 as well as among deprived populations.Citation5,Citation6 However, CRDs remain under-diagnosed and under-treated.Citation7
To address this global health problem, the Global Alliance against Chronic Respiratory Diseases (GARD) was formed following a mandate from the World Health Assembly.Citation8–Citation12 GARD is a voluntary alliance of organizations, institutions, and agencies working towards a common vision to improve global lung health according to local needs. GARD aims to develop a standard way of obtaining relevant data on CRD and risk factors, encourage countries to implement CRD prevention policies and to make recommendations of simple and affordable strategies for CRD management.Citation7
The rationale of this study was to investigate the prevalence of COPD in patients with respiratory symptoms, as well as the prevalence of bronchial asthma (BA), allergic rhinitis (AR), and chronic bronchitis (CB) in the overall Russian population. The frequency of major risk factors for CRD was also evaluated in the same study population.
Methods
This was a cross-sectional population-based epidemiological study conducted in 2010–2011 across 12 regions () of the Russian Federation.
The aim of the study was to recruit 250 adult (≥18 years) respondents in each major Russian city. As a general procedure, the administrative districts of each region participating in the study were selected based on a stratified random cluster sampling procedure. This stratification ensured appropriate weighted representation of each district’s target population in the study sample. The most current census data from the entire region and from each district were collected from official sources in order to proportionally stipulate the number of participants in each district. In a second stage, streets from each previous selected district were also selected by applying a two-step stratified random cluster sampling procedure with a standard random number generator (Microsoft Excel 2010; Microsoft Corporation, Redmond, WA, USA).Citation13 In the last stage, each selected street had also been randomly assigned specific households that would be approached to take part in the study. When blocks of apartments were selected, only several apartments were chosen from the block, and then the interviewers’ team moved to the next block randomly. To ensure inclusion of respondents who could not be available, rounds were conducted during non-working time.
GARD study received favorable opinion from the National Ethics Committee, Russian Ministry of Health. Prior to initiating a face-to-face interview by a team that consisted of either a nurse or a physician, subjects gave written consent for the use of the anonymized data reported in the questionnaire () and for pulmonary function testing – applicable to respondents with suspected CRD based on self-reported symptoms. Upon availability of hospital records, patient-reported diagnoses were checked.
The presence of BA symptoms was considered if patients experienced an attack of wheezing, or wheezing/whistling that resulted in breathlessness. AR symptoms were adapted from the Allergic Rhinitis and its Impact on Asthma criteria, according to which the presence of running nose with sneezing or nasal obstruction indicates rhinitis and the presence of running nose alone might also indicate rhinitis.Citation14 CB was defined as the presence of cough and sputum production for at least 3 months in 2 years.Citation15
COPD was defined following the Global Initiative for Obstructive Lung Disease definition of post-bronchodilator FEV1/FVC <0.7. The identification of symptomatic patients included clinical diagnosis of dyspnea, chronic cough or sputum production,Citation15 as well as those who were active smokers for more than 1 year, or those exposed to biomass or occupational hazards.
These selection criteria were checked by a doctor/pulmonologist using the GARD questionnaire which was developed in the respondent’s native language. As the questionnaire was self-completed by respondents and did not cover all of the information necessary to check the above criteria, the doctor/pulmonologist briefly interviewed the respondents regarding their medical history to establish the pulmonary origin of dyspnea and collect details on allergies and current health condition. In addition, all subjects who had an acute respiratory viral infection at the time of the interview were excluded to ensure reliable pulmonary function tests. Spirometry was performed at the investigational center and in accordance with international standards, including bronchodilator challenge.Citation16 For post-bronchodilator measurements, investigators were recommended to perform spirometry 15 minutes after two–four puffs of salbutamol (200–400 μg) via metered-dose inhaler with spacer.
Statistical analysis
The primary study endpoint was to establish prevalence of COPD, BA, and AR in accordance with the current diagnostics standards in the representative population. Using a two-tailed binomial test with a significance level of 5%, the study was designed to have 80% power to establish prevalence for each disease under study with the significance level not less than 1% on each side. The calculation of the sample size was made using Stata 12 package (sampsi) (StataCorp LP, College Station, TX, USA). The target sample size of 7,164 respondents was determined using the following assumptions:
Binomial distribution of the prevalence
The maximum prevalence for each study indication according to literature was 20%
Maximum acceptable one-sided error for prevalence determination was 5%, which was a 1% one-sided error for a maximum prevalence of 20%
Two-sided type 1 error of 5%, which was the risk to incorrectly accept the false null hypothesis of non-equivalence of sample prevalence and estimated population prevalence
An 80% probability to detect non-equivalence of the sample prevalence and the estimated population prevalence in case of true non-equivalence
A 10% probability that patients did not show up for the functional testing at the investigational site after the completion of the questionnaire.
The comparison of the qualitative parameters in different groups (by age, sex, etc) was carried out using chi-square test, in the case of two groups, where possible, Fisher’s exact test was used. Taking into consideration the cross-sectional nature of the study, odds ratio was calculated and 95% confidence intervals (CI) for the odds ratio to estimate the statistical significance of associations between risk factors and diseases. As spirometry was performed only in a sub-set population with symptoms and/or risk factors, this association was not calculated. All results were considered statistically significant at the level of P<0.05.
Results
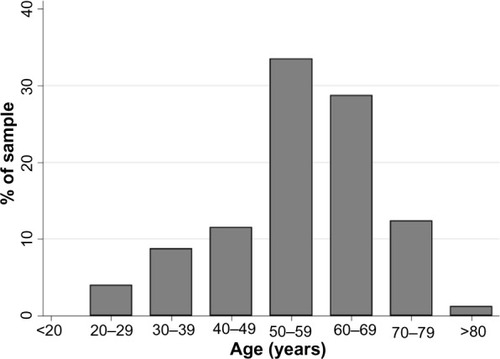
A total of 7,164 questionnaires were completed. The mean age of respondents was 43.4 years, ranging from 18 to 88 years, and 57.2% were female. Of the respondents, 64.2% were employed at the time of the survey, and 3.4% were migrants from other countries.
Respondents’ characteristics are shown in .
Table 1 Demographic characteristics of sample respondents
Prevalence of BA, AR, and CB related symptoms
Prevalence of disease related symptoms is shown in . When respondents were asked if they had ever experienced attacks of wheezing or whistling accompanied with the feeling of breathlessness, 25.7% (95% CI: 24.7–26.7) responded affirmatively. Out of those, 78.7% confirmed they had two or more attacks.
Table 2 Prevalence of respiratory symptoms in the total sample
The presence of a running nose along with the presence of at least one of the symptoms of sneezing or nasal congestion was reported among 18.2% (95% CI: 17.3–19.2) of the study population. Of these, 52.9% also had ocular symptoms. The distribution of respondents with respiratory symptoms, by age group, is shown in .
Figure 1 The distribution of respondents with respiratory symptoms for bronchial asthma, allergic rhinitis, and chronic bronchitis.
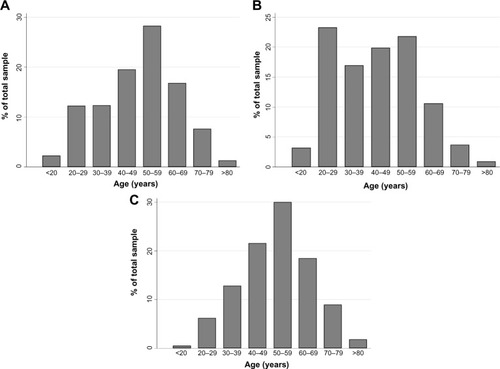
Cough and expectoration, occurring in the majority of the week for more than three consecutive months in a year, lasting more than 2 years, compatible with CB, was experienced by 8.6% of all respondents (95% CI: 7.9–9.3). Frequencies of respiratory symptoms are shown in .
Prevalence of previous diagnosis
Out of all respondents participating in the survey, 6.9% (95% CI: 6.3–7.5) reported a previous diagnosis of BA at some point in their life. Previous AR diagnosis was also reported by 6.5% of respondents (95% CI: 5.9–7.1). The highest percentage was found among those respondents who had a previous diagnosis of CB (22.2%; 95% CI: 21.2%–23.2%).
Of patients with proven COPD based on spirometry results, 51.4% (95% CI: 45.0–57.7) and 6.8% (95% CI: 4.0–10.6) had also self-reported previous CB and emphysema diagnosis, respectively. When analyzing the pool of respondents with symptoms compatible with CB, only 23.5% (95% CI: 21.5–25.7) reported to have a previous diagnosis. Out of those, 25.9% (95% CI: 20.6–31.8) had positive COPD diagnosis after spirometry. Distribution of respondents with COPD within spirometry population, by age group, is shown in .
Prevalence of COPD based on spirometry
Spirometry data was recorded in 16% (251) of the total study sample in patients with suspected CRD. This information was based on both historical and newly performed spirometry data collected as part of the study.
A post-bronchodilator test was performed in 94.4% of these subjects. Subjects with a post-bronchodilator test had a significantly reduced vital capacity and FEV1 when compared to the total population who had spirometry, and also had a significantly higher age (P<0.001).
The prevalence of COPD in patients with respiratory symptoms, or risk factors, was 21.8% (95% CI: 19.5%–24.5%). By extrapolation, the prevalence of symptomatic COPD in the total study population was 15.3%.
Risk factors
The prevalence of smoking was quite high, with 45.7% of the total population responding that they had a smoking history, of which 73.1% were current smokers. Smoking history was measured if patients have consumed at least 200 packs in their life-time. Regarding workplace dust, 22.2% of the total population responded that they had been exposed to workplace dust for more than a year. For indoor use of an open fire for heating or cooking, 34.0% of the total population responded that they used one. The association between these selected risk factors and respiratory symptoms is shown in .
Table 3 Association between risk factors and chronic respiratory diseases
Discussion
The GARD study was the first cross-sectional population-based epidemiological study among a representative sample, using a standardized methodology and validated questionnaire, to evaluate the prevalence of respiratory diseases in several regions of the Russian Federation.
Partial use of data collected from previously completed GARD questionnaires from 2009–2010 was approved by the study steering committee. The GARD questionnaire has been used in several studies and has been shown to be an accurate and reliable diagnostic tool.Citation17,Citation18
The prevalence of respiratory symptoms in the population sampled was found to be high. The percentage of patients with asthma related symptoms was 25.7%, AR 18.2% and CB 8.6%.
Based on spirometry-confirmed diagnosis, 21.8% of respondents with respiratory symptoms had COPD, and, by extrapolations, 15.3% of the overall population suffered from the disease.
CRDs are recognized as a major public health problem with an increasing morbidity and mortality. With such a high burden on the health care system, emphasis on better diagnosis and management of these diseases must be achieved, and reliable epidemiological data on the prevalence and severity of diseases, such as COPD and its exacerbations, are crucial to guide health care policy.Citation19
In the Russian Federation, it has been estimated from earlier epidemiological studies that the prevalence of CRD ranges from 17% to 21%. This includes the prevalence of asthma which ranges from 6%–8% for adults and up to 12% for children, and for COPD between 6%–7%, and other miscellaneous disease of 2%.Citation20
COPD is the fourth cause of death worldwide.Citation21 An estimation from the World Health Organization suggests that COPD will be the third cause of death by 2030.Citation22 The association between COPD and CB may lead to a more severe COPD prognostic, which encompasses a poorer lung function, exacerbation, a worse quality of life and, consequently, a higher economic burden.Citation23 We used the Global Initiative for Obstructive Lung Disease strategy definition of COPD in symptomatic subjects, which represents a simplified case definition for epidemiological purposes, rather than a definitive clinical diagnosis; this may have resulted in patients with COPD not being diagnosed. The limitation of our study is that a large proportion of patients with COPD are asymptomatic; the study may have underestimated the prevalence of COPD as the spirometry was conducted only in symptomatic patients.Citation24
The problem of COPD under-diagnosis is well known. Only about one-third of all cases with COPD are recognized by the health care professional,Citation25–Citation27 and the proportion of undiagnosed cases decreases with increasing disease severity.Citation28 The prevalence of COPD has often been reported in the range of 6%–10% of the total adult population.Citation29 However, for the PLATINO study,Citation18 the crude prevalence of COPD was estimated to be up to 19.7% in population ≥40 years in Montevideo, especially in elderly men. Other studies have also reported prevalence of up to 20%, dependent on the definition used.Citation24,Citation30,Citation31 In the BOLD (Burden of Obstructive Lung Disease) Study, prevalence of non-flow obstruction was observed in up to 80% with variation of COPD prevalence from 0.9% to 15.5% among cities, depending on the disease stage.Citation32
As the main objective of the GARD study was to assess COPD prevalence in symptomatic patients, the estimation of crude prevalence of 15.3% should be analyzed carefully, even though our results are compatible with what has been seen in different populations.
The assumption of this prevalence was based on the fact that symptomatic patients who have not undergone spirometry (due to the exclusion criteria being matched or refusals/further contact failure after the first assessment by questionnaire) would also have the same frequency of cases observed in those patients with similar clinical characteristics who underwent spirometry. For the remaining asymptomatic population who did not meet clinical diagnosis criteria, our assumption was that the result of spirometry would be ≥0.7.
COPD itself is a predictor of mortality as it has been shown that this is significantly higher amongst subjects with COPD compared to subjects without COPD (P<0.001).Citation33 This study reinforces the need for the provision of adequate standard care management in order to improve the quality of life of patients and to decrease exacerbations and hospitalizations.Citation34–Citation36 Our results suggest that prevalence of COPD in Russia is higher than previously suggested, which also results in a greater health issue.
The prevalence of CB in COPD patients varies substantially.Citation23,Citation37 In the GARD study, more than half of the patients with COPD also had CB. In a recent European study,Citation38 CB prevalence varies from 0.7% to 9.7%, going up to 20.1%–56.9% among current smoker respondents, as smoking is one of the major risk factors for developing CB, which can also be seem from our study results. Our findings suggested that prevalence based on CB symptoms is 8.6%, which is consistent with the findings in European populations.
The prevalence of BA may vary considerably in different countries, from 4% to 18% of populations.Citation39 Even though BA is much more present in developed countries, its prevalence in less industrialized regions is growing.Citation40 Our findings suggest that in Russia, the prevalence of BA related-symptoms is higher than originally estimated.Citation20 Asthma is one of the most common chronic diseases in the world and it is estimated to be accountable for about one in every 250 deaths worldwide.Citation41 A study performed by Brogger et al suggested a 3-fold increase in the prevalence of self-reported diagnosis in 26 years, which could be due to a better standard of care and an easier access to physicians.Citation42
As for other CRDs, AR is also experiencing an increase of epidemic proportion,Citation43 which leads to an intensification in the socioeconomic burden of the disease across the world. Prevalence of AR has been reported as high as 21% in Europe.Citation44 Asthma and rhinitis have been reported to have similarities related to their epidemiological and patho-physiological background.Citation45 If untreated, rhinitis may have considerable economic and quality of life implications.Citation46,Citation47
A high proportion of the Russian population is exposed to risk factors which could drive specific public health initiatives. In this study, the odds ratios between BA, CB, and AR and occupational hazard, smoking, and biomass exposure were estimated as an attempt to collect information on the major risk factors for those CRDs. The present study did not intend to assess risk factor related to COPD, nor the difference pattern between genders and cities. The prevalence of major risk factors including smoking, occupational hazards, and biomass exposure was high.
The major effects of smoking on CRDs have been extensively recorded over more than 40 years.Citation48 The associations found in this study were generally consistent with findings from other epidemiological studies where the GARD questionnaire has been used.Citation17,Citation18 It should be noted, however, that this study design (cross-sectional) was not performed to evaluate causality between risk factors and respiratory disease already established in other studies. Among all three risk factors, the occurrence of an occupational hazard demonstrated a statistically significant association with all respiratory symptoms (P<0.0001). Biomass exposure occurrence had a positive association with CB and BA, which was also statistically significant (P<0.0001). The negative association between smoking and AR should be analyzed carefully. Some risk factors may be obscured due to changes in smoke habits due to the occurrence of respiratory symptoms. Also, for those respondents with a history of respiratory symptoms, there is a possibility of reluctance to start smoking. All of these factors may create a bias in the risk factors association analysis due to the cross-sectional design study type.
This study has substantial strengths. To the best of our knowledge, it is the first comprehensive prevalence study conducted in Russia for CRD. Using probabilistic sampling strategy, the study was able to capture the prevalence of symptomatic CRD and the major risk factors in the Russian Federation. There are also potential limitations of the study, which included only symptomatic patients and did not represent the total number of COPD patients. Moreover not all self-reported symptomatic patients underwent spirometry due to the reasons previously explained.
Furthermore, as for any questionnaire based study, the study outcomes are based on the willingness of the respondents to report their diseases. Furthermore, by also having the prevalence of self-reported symptoms we are minimizing the subjectivity of self-report diagnosis due to the different diagnosis criteria that may be used by physicians. There is also a potential bias of non/incomplete-responders, as we did not adjust for subjects who did not complete all of the questions during the visit. In addition, no adjustments for age or sex have been made. Our sample includes 57% females which could have an impact on prevalence as there may be important sex differences on the perception of dyspnea, health status, and physical activity limitation.Citation49
Conclusion
The prevalence of respiratory symptoms in the Russian Federation was found to be high. For asthma, the overall asthmatic symptoms were present in 25.7% of respondents. AR symptoms were presented in 18.2% and CB in 8.6%.
The estimated prevalence of 21.8% for COPD in symptomatic patients and 15.3% in the overall population may still be an underestimate as this was only estimated by spirometry from symptomatic patients. COPD can be present in asymptomatic patients which also may reflect a misrepresentation of prevalence present in this study. There was a considerable discrepancy between self-reported diagnosis and disease based-symptoms for AR and BA, which may suggest that respondents were under-diagnosed. The discrepancy between self-reported symptoms and previous diagnosis highlights the fact that CDR may not be recognized, or may have a late diagnosis which might lead to economic and health impacts. This data will be used to monitor the course and health care utilization of these diseases and to evaluate the impact of future educational programs on assessing and treating patients with CRDs.
Acknowledgments
The authors wish to acknowledge Diana Jones of Cambrian Clinical Associates Ltd for the development of the first draft of the manuscript, editorial suggestions to draft versions of this paper, assembling tables and figures, collating author comments, and referencing. This assistance was funded by GlaxoSmithKline.
The study was supported from the grant provided by GlaxoSmithKline Russia to the contract-research organization “Worldwide Clinical Trials”. Worldwide Clinical Trials has conducted the study and assisted in developing the study protocol to investigators. This study was supported by ZAO GlaxoSmithKline Trading Ltd.
Supplementary material
Disclosure
The authors report no conflicts of interest in this work.
References
- World Health OrganizationGlobal Surveillance, Prevention and Control of Chronic Respiratory Diseases. A Comprehensive ApproachGenevaWorld Health Organization2007 Available from: http://www.who.int/gard/publications/GARD%20Book%202007.pdfAccessed July 17, 2014
- HaahtelaTTuomistoLEPietinalhoAA 10 year asthma programme in Finland: major change for the betterThorax200661866367016877690
- FairallLRZwarensteinMBatemanEDEffect of educational outreach to nurses on tuberculosis case detection and primary care of respiratory illness: pragmatic cluster randomised controlled trialBMJ2005331751975075416195293
- FischerGBCamargosPAMocelinHTThe burden of asthma in children: a Latin American perspectivePaediatr Respir Rev20056181315698808
- EvansRIIIGergenPJMitchellHA randomized clinical trial to reduce asthma morbidity among inner-city children: results of the National Cooperative Inner-City Asthma StudyJ Pediatr1999135333233810484799
- CloutierMMHallCBWakefieldDBBailitHUse of asthma guidelines by primary care providers to reduce hospitalizations and emergency department visits in poor, minority, urban childrenJ Pediatr2005146559159715870660
- BousquetJDahlRKhaltaevNGlobal alliance against chronic respiratory diseasesAllergy200762321622317298337
- KhaltaevNWHO strategy for prevention and control of chronic obstructive pulmonary diseaseExp Lung Res200531Suppl 1555616395859
- World Health OrganizationWHO Strategy for Prevention and Control of Chronic Respiratory DiseasesGenevaWorld Health Organization2001 Available from: http://www.who.int/respiratory/publications/WHO_MNC_CRA_02.1.pdfAccessed July 17, 2014
- World Health OrganizationImplementation of the WHO Strategy for Prevention and Control of Chronic Respiratory DiseasesGenevaWorld Health Organization2002 Available from: http://www.who.int/respiratory/publications/WHO_MNC_CRA_02.2.pdfAccessed July 17, 2014
- World Health OrganizationPrevention and Control of Chronic Respiratory Diseases in Low and Middle-Income African Countries: A Preliminary ReportGenevaWorld Health Organization2004 Available from: http://whqlibdoc.who.int/hq/2003/WHO_NMH_CRA_04.1.pdfAccessed July 17, 2014
- World Health OrganizationPrevention and Control of Chronic Respiratory Diseases at Country Level: Towards a Global Alliance against Chronic Respiratory DiseasesGenevaWorld Health Organization2005 Available from: http://www.who.int/respiratory/publications/WHO_NMH_CHP_CPM_-CRA_05.1.pdfAccessed July 17, 2014
- SilmanAJMacfarlaneGJEpidemiological Studies: A Practical Guide2nd edCambridgeCambridge University Press2002
- Allergic Rhinitis and its Impact on Asthma (ARIA)Management of Allergic Rhinitis and its Impact on Asthma. Pocket GuideARIA2007 Available from: http://www.whiar.org/docs/ARIA_PG_08_View_WM.pdfAccessed July 17, 2014
- Global Initiative for Chronic Obstructive Lung DiseaseGlobal Strategy for the Diagnosis, Management and Prevention of Chronic Obstructive Pulmonary DiseaseGlobal Initiative for Chronic Obstructive Lung Disease2014 Available from: http://www.goldcopd.org/uploads/users/files/GOLD_Report2014_Feb07.pdfAccessed July 17, 2014
- MillerMRHankinsonJBrusascoVStandardisation of spirometryEur Respir J200526231933816055882
- MohammadYShaabanRYassineFExecutive summary of the multicenter survey on the prevalence and risk factors of chronic respiratory diseases in patients presenting to primary care centers and emergency rooms in SyriaJ Thorac Dis20124220320522833827
- MenezesAMPerez-PadillaRJardimJRPLATINO TeamChronic obstructive pulmonary disease in five Latin American cities (the PLATINO study): a prevalence studyLancet20056695001875188116310554
- MartinsPRosado-PintoJdo Céu TeixeiraMUnder-report and underdiagnosis of chronic respiratory diseases in an African countryAllergy20096471061106719210360
- BellevskiyAGARD in RussiaGenevaWorld Health Organization Available from: http://www.who.int/gard/news_events/GARD%20in%20Russia.pdfAccessed July 17, 2014
- PauwelsRARabeKFBurden and clinical features of chronic obstructive pulmonary disease (COPD)Lancet2004364943461362015313363
- http://www.who.int [homepage on the Internet]. World Health OrganizationCOPD predicted to be third leading cause of death in 2030 Available from: http://www.who.int/respiratory/copd/World_Health_Statistics_2008/en/Accessed July 17, 2014
- de OcaMMHalbertRJLopezMVThe chronic bronchitis phenotype in subjects with and without COPD: the PLATINO studyEur Respir J2012401283622282547
- HalbertRJIsonakaSGeorgeDIqbalAInterpreting COPD prevalence estimates: what is the true burden of disease?Chest200312351684169212740290
- LundbäckBLindbergALindströmMNot 15 but 50% of smokers develop COPD? – Report from the Obstructive Lung Disease in Northern Sweden StudiesRespir Med200397211512212587960
- SiafakasNMVermeirePPrideNBOptimal assessment and management of chronic obstructive pulmonary disease (COPD). The European Respiratory Society Task ForceEur Respir J199588139814207489808
- LindbergABjergARönmarkELarssonLGLundbäckBPrevalence and underdiagnosis of COPD by disease severity and the attributable fraction of smoking. Report from the Obstructive Lung Disease in Northern Sweden StudiesRespir Med2006100226427215975774
- LindbergALarssonLGMuellerovaHRönmarkELundbäckBUp-to-date on mortality in COPD – report from the OLIN COPD studyBMC Pulm Med201212122230685
- ViegiGPedreschiMPistelliFPrevalence of airways obstruction in a general population: European Respiratory Society vs American Thoracic Society definitionChest20001175 Suppl 2339S345S10843974
- CelliBRHalbertRJIsonakaSSchauBPopulation impact of different definitions of airway obstructionEur Respir J200322226827312952259
- BatemanEDBousquetJBusseWWStability of asthma control with regular treatment: an analysis of the Gaining Optimal Asthma controL (GOAL) studyAllergy200863793293818588561
- BuistAS1McBurnieMAVollmerWMInternational variation in the prevalence of COPD (the BOLD Study): a population-based prevalence studyLancet2007370958974175017765523
- PaggiaroPLDahleRBakranIFrithLHollingworthKEfthimiouJMulticentre randomised placebo-controlled trial of inhaled fluticasone propionate in patients with chronic obstructive pulmonary disease. International COPD Study GroupLancet199835191057737809519948
- FriedmanMSerbyCWMenjogeSSWilsonJDHillemanDEWitekTJJrPharmacoeconomic evaluation of a combination of ipratropium plus albuterol compared with ipratropium alone and albuterol alone in COPDChest1999115363564110084468
- CalverleyPPauwelsRVestboJCombined salmeterol and fluticasone in the treatment of chronic obstructive pulmonary disease: a randomised controlled trialLancet2003361935644945612583942
- MasoliMFabianDHoltSBeasleyRGlobal Initiative for Asthma (GINA) ProgramThe global burden of asthma: executive summary of the GINA Dissemination Committee ReportAllergy200459546947815080825
- CorhayJLVinckenWSchlesserMBossuytPImschootJChronic bronchitis in COPD patients is associated with increased risk of exacerbations: a cross-sectional multicentre studyInt J Clin Pract201367121294130124246208
- CerveriIAccordiniSVerlatoGVariations in the prevalence across countries of chronic bronchitis and smoking habits in young adultsEur Respir J2001181859211510810
- Global Initiative for AsthmaGlobal Strategy for Asthma Management and PreventionGlobal Initiative for Asthma2014 Available from: http://www.ginasthma.org/local/uploads/files/GINA_Report_March13.pdfAccessed July 17, 2014
- BramanSSThe global burden of asthmaChest2006130Suppl 14S12S16840363
- BatemanEDBousheyHABousquetJCan guideline-defined asthma control be achieved? The Gaining Optimal Asthma Control StudyAm J Respir Crit Care Med2004170883684415256389
- BroggerJBakkePEideGEJohansenBAndersenAGulsvikALong-term changes in adult asthma prevalenceEur Respir J200321346847212662003
- PawankarRBunnagCKhaltaevNBousquetJAllergic rhinitis and its impact on asthma in Asia Pacific and the ARIA Update 2008World Allergy Organ J20125Suppl 3S212S21723268481
- JansonCAntoJBurneyPThe European Community Respiratory Health Survey: what are the main results so far? European Community Respiratory Health Survey IIEur Respir J200118359861111589359
- GaugrisSSazonov-KocevarVThomasMBurden of concomitant allergic rhinitis in adults with asthmaJ Asthma20064311716448958
- FinemanSMThe burden of allergic rhinitis: beyond dollars and centsAnn Allergy Asthma Immunol2002884 Suppl 12711991546
- SchoenwetterWFDupclayLJrAppajosyulaSBottemanMFPashosCLEconomic impact and quality-of life burden of allergic rhinitisCurr Med Res Opin200420330531715025839
- SpencerSJonesPWGLOBE Study GroupTime course of recovery of health status following an infective exacerbation of chronic bronchitisThorax200358758959312832673
- Lopez VarelaMVMontes de OcaMHalbertRJSex-related differences in COPD in five Latin American cities: the PLATINO studyEur Respir J20103651034104120378599