Abstract
Purpose
Dexibuprofen is an enantiomer of ibuprofen with low bioavailability which results from its hydrophobic nature. Nanosuspensions have developed a podium to solve the in vitro dissolution problem that frequently occurs in current research.
Materials and methods
The drug and polymer solutions were mixed in a microchannel fluid reactor and the successive embryonic nanosuspension was decanted into a vial having the polymer solution. The impact of different process and formulation parameters including inlet angle, antisolvent and solvent flow rate(s), mixing time, drug concentration, polymer type and concentration was evaluated.
Results and discussion
Stable dexibuprofen nanocrystals with a particle size of 45±3.0 nm and polydispersity index of 0.19±0.06 were obtained. Differential scanning calorimetry and powder X-ray diffraction confirmed the crystallinity. The key parameters observed were inlet angle 10°, antisolvent to solvent volume of 2.0/0.5 mL/min, 60 minutes mixing with 5 minutes sonication, Poloxamer-407 with a concentration of 0.5% w/v and drug concentration (5 mg/mm). The 60-day stability studies revealed that the nanocrystals were stable at 4°C and 25°C. The scanning electron microscopy and transmission electron microscopy images showed crystalline morphology with a homogeneous distribution.
Conclusion
Stable dexibuprofen nanocrystals with retentive distinctive characteristics and having marked dissolution rate compared to raw and marketed formulations were efficiently fabricated. In future perspectives, these nanocrystals could be converted to solid dosage form and the process can be industrialized by chemical engineering approach.
Introduction
The issue of poor water solubility for a list of drug candidates has become a hard challenge for the drug delivery scientists both from academia and research and development sectors.Citation1 As per the scientist reports, nearly 70% of the applicant drugs in development phase and 40% of marketed APIs available as oral dosage forms are practically insoluble in water.Citation2 The issue of poor water solubility would lead to erratic bioavailability and, consequently, unwanted toxic effects.Citation3 Drug delivery scientists, by their continuous struggle, have developed and designed a range of strategies to boost the solubility and dissolution of medicines in aqueous media, including solid dispersions, solubilization, emulsions, micronization and so on.Citation4 However, these approaches have limited applications because of the production of ionizable groups, stability issues and limited improvement in solubility.Citation5 In addition the added solvents residues, enhances the solubility that can lead to potential toxicity and other unwanted side effects. It is, therefore, the utmost need to develop alternative approaches to enhance the solubility of plethora of drug compounds.Citation6
It has become evident from the present literature that nanosuspensions have developed a podium to solve the in vitro dissolution problem that frequently occurs in current research.Citation7 Nanocrystal can be created by bottom-up and top-down methods.Citation8 In top-down methods, the size reduction occurs by mechanical forces and attrition forces. However, some of the issues associated with top-down methods include long processing time, broad particle size distribution and contamination.Citation9 Bottom-up method involves antisolvent precipitation in which the significant footstep is nucleation.Citation10 The issues associated with the bottom-up methods are uncontrolled particle growth through Ostwald ripening and changes in particle attributes with partial or complete conversion to amorphous state, which can potentially cause phase transition and stability issues.Citation9,Citation11 It is of paramount importance for crystalline nanoparticles to be fabricated because it causes long-term stability.Citation7
Dexibuprofen is the active form of ibuprofen in vitro and in vivo.Citation12 It is rapid acting and used in a decreased quantity as compared to its racemic mixture.Citation13 It is indicated for dental pain, dysmenorrhea, postoperative pain and headache.Citation14 It is a Biopharmaceutics Classification System class II drug. Its aqueous solubility is 51 μg/mL as compared to ibuprofen (21 μg/mL), and its bioavailability is low in the gastric environment, which can be enhanced by increasing its aqueous solubility.Citation15
The basic principle of antisolvent precipitation with syringe pump (APSP) is the addition of drug molecules from organic phase through a syringe pump into aqueous polymer phase and, consequently, nucleation and particle formation occur while the microchannel fluidic reactor (MCFR) has a small channel in which the liquid behavior is in contrast to the conventional flow theory, that is, the liquids follow laminar flow pattern without turbulence and mixing of liquid occurs due to diffusion of molecules across the liquid interface.Citation16 MCFR is a novel advanced low-energy bottom-up approach, which is different from APSP. Basically, in these approaches, the important step is micromixing of antisolvent aqueous polymer solution and organic solvent drug solution, which controls the nucleation, crystal size and attributes of the nanocrystal. In the APSP method, only the solvent drug solution is controlled, while in MCFR, both the solutions (antisolvent and solvent solutions) are controlled, leading to more control on nucleation, particle size, size distribution, stability and characteristics of the nanocrystals.
This study aimed to evaluate the modified MCFR process and produce nanocrystals of dexibuprofen through the modified MCFR method. The hydrocortisone nanoparticles reported by Ali et alCitation17 were produced by MCFR with a particle size above 200 nm. In that method, the crystallized nanoparticles were mixed with the buffer solution composed of polymers using stirring and sonication. However, in this new method, we used a polymeric solution during mixing of the drug solution in MCFR as well as after processing the nanosuspension. Crystallization of the drug in plain water and in polymeric medium are substantially different from each other. In polymeric medium, the nucleation and particle size are controlled by the polymer molecules, which results in low particle size and homogenous size distribution.Citation18 In the current project, the special characteristics of nanocrystals were investigated by executing different assessments which included the particle size, zeta potential, powder X-ray diffraction (PXRD), differential scanning calorimetry (DSC), scanning electron microscopy (SEM), transmission electron microscopy (TEM), stability studies and in vitro dissolution.
Materials and methods
The following were the chemicals and the manufacturers from where they were obtained: dexibuprofen (batch no C102-150100M; Hubei Biocause Helen Pharmaceutical, Hubei, China), Poloxamer-407 (batch no BCBK50056; Sigma-Aldrich, Dorset, UK), polyvinylpyrrolidon-K30 (batch no 08297052 G0; BASF, Ludwigshafen, Germany), hydroxypropyl methyl cellulose (HPMC) viscosity 15cps (batch no 8028213; Shinetsu, Tokyo, Japan). Laboratory distilled water was obtained from the research laboratory of Bradford University.
Preparation of dexibuprofen nanosuspension
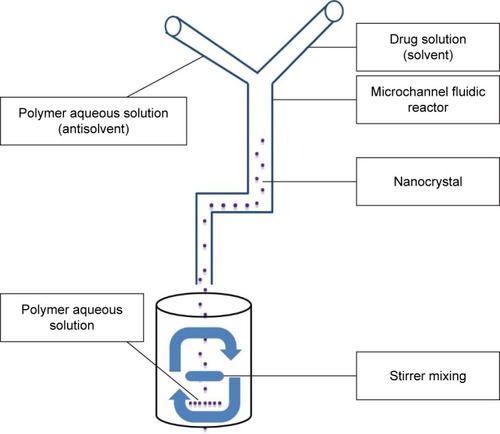
The current method is a modification of the previously published method of Ali et al.Citation17 Dexibuprofen dissolved in ethanol (5 mg/mL) and polymers dissolved in aqueous phase were introduced into MCFR (inlet angle 10° with internal diameter of 0.5 mm) at a flow rate 0.5:2.0 mL/min, respectively. Then, the emergent nanosuspension was poured into a vial having aqueous polymer solution and stirred for 60 minutes (). At the end, the nanosuspension was sonicated for 5 minutes with 100 W ultrasonication (P1200; Crest Ultrasonic, Ewing Township, NJ, USA).Citation19 The impact of five important process parameters including 1) polymer type and its concentration, 2) inlet angle, 3) solvent and antisolvent flow rates, 4) drug concentration and 5) stirring time with and without ultrasonication was assessed at three levels.
Particle size and zeta potential measurements
Particle size measurements of the produced nanosuspension were determined through photon correlation spectroscopy (Zetasizer® NanoS; Malvern Instruments, Malvern, UK). All the samples were subjected to particle size analysis without further dilution. The average particle size diameter (Z.Ave) and the polydispersity index (PDI) were determined from three measurements. Additionally, the zeta potential calculations of the produced nanoparticles were carried out using the Malvern Zetasizer. The clear disposable zeta cells were used for zeta potential measurements. The samples were analyzed in triplicate.
SEM
The morphology of unprocessed dexibuprofen was evaluated using SEM (Quanta 400; FEI, Cambridge, UK). The images were taken at different magnifications levels. Gold coating of the dexibuprofen particles using sputter coater was carried out to obtain clear images.
TEM
The external morphology of the produced nanosuspension was characterized by TEM (JEM-1200EX; JEOL, Tokyo, Japan) performed at 100 kV. Nanosuspension drop was poured on copper grid surface and dried at ambient temperature; then, magnesium uranyl acetate solution was applied as a 2% aqueous solution for negative staining.
DSC analysis
Thermal characteristics of the processed and unprocessed dexibuprofen were assessed using DSC (Discovery; TA Instrument, New Castle, DE, USA). The DSC instrument was calibrated with indium 99% and zinc having melting points 156.6°C and 419.5°C, respectively. Under nitrogen atmosphere, the scanning rate of 10°C/min was used for both raw and nanocrystals. The scan was performed at temperature ranging from 0°C to 100°C. All the samples were analyzed in triplicate.
X-ray powder diffraction analysis
The X-ray powder diffraction of raw and produced nanocrystals was carried out with D5000 diffractometer (Siemens, Munich, Germany) consuming Cu Ka radiation (X=1.5418 A). The samples were measured with the angle range of 5°–50°, step size of 0.05°, count time of 3 seconds per step, rotation during analysis 30 rpm and the generator fixed at 40 kV and 30 mA.
Stability studies
Dexibuprofen nanosuspensions were assessed for their chemical stability at regular time intervals in 1 week (day 0, day 1, day 3, day 4, day 5, day 6, day 7) and the % of drug content was evaluated with UV spectrophotometer (V-630; JASCO) at 222 nm as per the method reported previously.Citation20 Moreover, physical stability study of the produced dexibuprofen nanosuspension was carried out at 25°C and 4°C for 60 days. The growth of the particles was monitored by measuring the particle size and PDI values at regular time intervals.
In vitro dissolution
The dissolution rate of the produced dexibuprofen nanocrystals was compared with those of raw and marketed formulations. The dissolution study was carried out using USP XXIV rotating paddle method. The sample was placed in dissolution vessels containing 900 mL phosphate buffer, pH 7.2, maintained at 37°C±0.5°C with a stirring speed of 100 rpm, which has been reported for nanosuspension.Citation20,Citation21 The samples were collected after 5, 10, 15, 20, 25, 30, 40, 50, and 60 minutes through a filter (Millex®GP; IRL) with a pore size of 0.22 μm and were substituted with equal amount of fresh phosphate buffer solution. The samples were assessed on UV spectrophotometer (V-630; JASCO) at 222 nm.
Statistical analysis
Statistical analysis of the experimentally collected data was done by SPSS 18 (SPSS Inc., Chicago, IL, USA). The data for all drugs were collected in triplicate. The data presented here are as mean ± SD. One-way analysis of variance test (P<0.05) and least significant difference test were used.
Results and discussion
Impact of process conditions on the particle size of dexibuprofen nanoparticles
This is a low-energy method which can produce stable drug nanocrystals. However, process optimization is important to be taken under consideration. Interestingly, a variation in particle size of the dexibuprofen was observed while changing the parameters. The impact of three levels of these parameters on the particle size is described below.
Evaluation of polymers
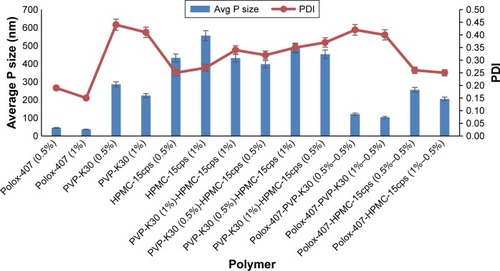
The effect of polymer type and concentration on the particle size of dexibuprofen nanocrystals was assessed (). Poloxamer-407, PVP-K30 and HPMC-15cps were tested in single and combination forms at various concentrations. Poloxamer-407 (0.5% w/v) was found to be the most suitable polymer, and the nanocrystal attained a size of 45±3.0 nm with PDI of 0.19±0.06. Particle size distribution is presented in .
Effect of flow rate of antisolvent and solvent
Antisolvent volume variable and solvent volume constant
Antisolvent and solvent flow rate was assessed and it was observed that it had a dominant effect on nanocrystal size. The nanocrystal of smaller sizes was obtained while changing the antisolvent volume and keeping the drug solution constant. The nanoparticle size achieved at 2.0/0.5 mL/min was 45±3.0 nm with a PDI of 0.19±0.06 (). The relatively smaller particle sizes obtained are attributed to two reasons. The first one is the perfect mixing of the drug solution with the anti-solvent solution because the antisolvent solution volume is high compared to the drug solution.Citation22 The second one is the higher supersaturation level achieved in beginning due to less drug solution and antisolvent concentration, which decreases the solute concentration on the nuclei surfaces.Citation23
Table 1 Impact of process conditions on dexibuprofen nanoparticle size (n=3)
Antisolvent volume constant and solvent volume variable
It was observed that keeping the antisolvent solution constant and increasing the volume of drug solution result in larger particles. The maximum size obtained at 2.0/2.0 mL/min was 803±3.0 nm with a PDI of 0.57±0.19 (). This effect is attributed to increase in drug concentration which inflates the solute fixation on the surface of framed nuclei and, thus, encourages molecular growth and produces bigger crystals.Citation23
Equal volume of antisolvent and solvent
At equal ratios of both solutions, it was observed that higher stream rate (2.0/2.0 mL/min) resulted in larger particle sizes (803±3.0 nm with a PDI of 0.57±0.19; ). The higher particle sizes obtained at higher stream rate of both solutions are due to inadequate blending of the drug solution with the antisolvent solution as the dwelling time in MCFR is very short; so, inadequate diffusion takes placeCitation24 and it may produce a non-uniform region supersaturation, subsequently producing bigger particles with more extensive size dispersion.Citation17
Drug concentration effect
The drug concentration effect was evaluated and it was observed that larger particles sizes were obtained at higher concentrations (). Principally, higher concentration crops the growth of crystal through condensation and coagulation phenomenon. Moreover, at higher concentrations, the viscosity of the drug solution is high, which decreases mutual diffusion of antisolvent and solvent solutions with consequent creation of non-uniform supersaturation that ultimately produces larger particles through agglomeration or crystal growth.Citation25 Furthermore, BaSO4 and prednisolone nanoparticles in MCFR have been produced with the same effect.Citation17,Citation22
Inlet angle of MCFR
Three different inlet angles were investigated for their effect on particle size of the dexibuprofen nanoparticles. The nanoparticles produced by angle 50° were little a bit larger with slightly broader distribution as compared to those at angles 10° and 25° (). It is thought that Keener delta edges (10° and 25°) have sharper channel edges which permit the two liquid streams to meet and then stream down the outlet without creating any interruption to either liquid stream,Citation26 while at an angle of 50°, the liquid streams encounter a more even level creating further disturbance of the meeting liquid streams while forwarding to the vent.
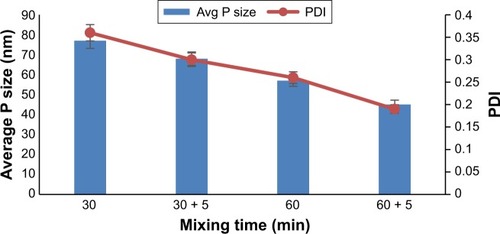
Effect of mixing time
The effect of mixing time on nanosuspension was evaluated and it was observed that 60 minutes mixing produced nanoparticles with smaller sizes as compared to 30 minutes mixing (). Following mixing, the produced nanosuspensions were sonicated with 100 W ultrasonication (P1200) for 5 minutes, which further reduced the size and PDI. Principally, high stirring rate and efficient mixing can potentially produce high level of micromixing, supersaturation and rapid nucleation, and consequently, nanocrystals with relative smaller particle size are produced.Citation27
Stability studies
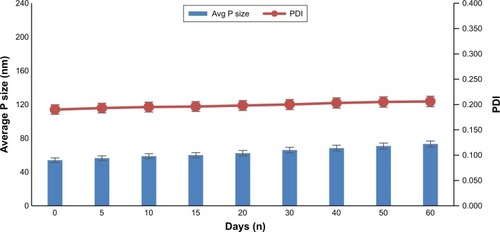
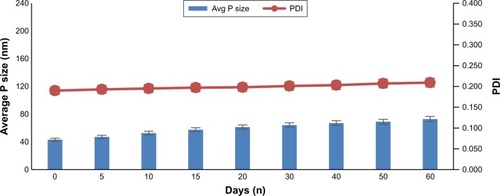
Stability of the nanocrystal is very significant because nanonization enhances surface area with increase in surface free energy, and consequently, stability of nanoparticle is greatly affected. Moreover, little bit of high solubility of the drugs at higher temperature leads to lower supersaturation level with subsequent growth of nanoparticles through Ostwald ripening.Citation28 The van der Waals forces among the nanocrystals lead to agglomeration and, consequentially, weaken the nanosuspensions. The zeta potential value obtained was −22.1±2.2 mV, and the literature reported value for nanosuspensions is ±20 mV.Citation29,Citation30 The chemical stability of the produced nanosuspensions was evaluated for 1 week as per the method reported previously.Citation20 The % of the drug content of dexibuprofen nanosuspension stored for 7 days was 96.9%±2.6%. No sign of degradation was observed. Moreover, the produced nanosuspensions were assessed for their physical stability at 4°C and 25°C for a period of 60 days. It was observed that the nanosuspensions stored at both temperatures were stable and no significant growth occurred ( and ). The 2-month stability data indicate homogenous distribution of nanoparticles and as per Deng et al,Citation34 homogenous distribution prevents Ostwald ripening. Moreover, regarding nanosuspension stability, Freitas and Müller recommended that it should be kept in fridge (2°C–8°C) to retain its particle size.Citation31
Thermal and X-ray analysis
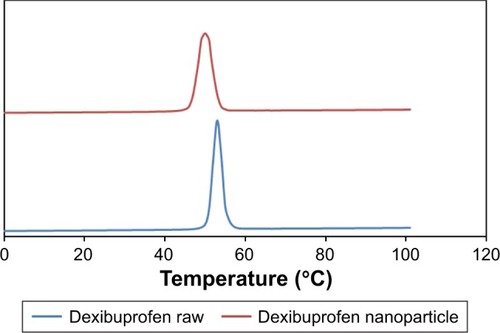
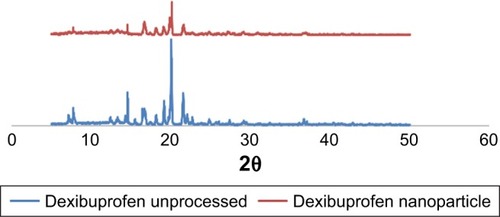
The crystallinity of the nanocrystals was assessed and compared with that of the raw counterparts using DSC and PXRD. These studies confirmed that the nanoparticles were crystalline in nature. The raw drug and the produced nanoparticles had sharp melting endotherm. A reduction in melting point of nanocrystals was observed related to the raw drugs. The melting point of raw dexibuprofen was 53.0°C, while that of dexibuprofen nanocrystal was 50.0°C (). This is because of the small particle size and lower packing density of the nanocrystals lattice compared to those of raw particles.Citation32 Basically, peak intensity shifted to lower level because of minor angle reflection by the nanocrystals. Furthermore, due to sizing effects, the peak for the nanocrystals was also found to be little broadened compared to the raw drug. The PXRD study also confirmed the crystalline characteristics of the nanoparticles. The PXRD diffractograms of the raw dexibuprofen was found to have sharp and high-intensity peaks (). On the other hand, disappearance of some peaks and decrease in the intensity of the peaks as well were observed for nanocrystals. Owing to small particle size effect, the diffraction patterns are different compared to those of raw drug, as the peak intensity has been shifted to a lower level due to minor angle reflection by the nanocrystal.Citation33
Morphological studies
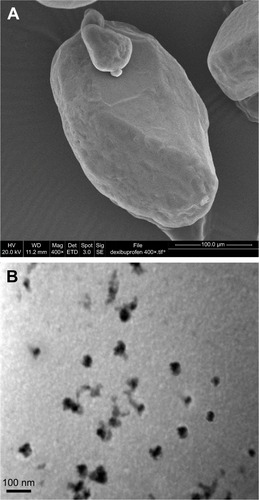
SEM and TEM were used to study the morphology of raw and processed dexibuprofen. The SEM images () showed that the raw drug was crystalline and triangular and irregular in shape. In addition, the images of nanocrystals obtained by TEM () clearly showed that the nanoparticles had a homogenous size distribution with size 50 nm and also confirmed the crystalline nature.
In vitro dissolution
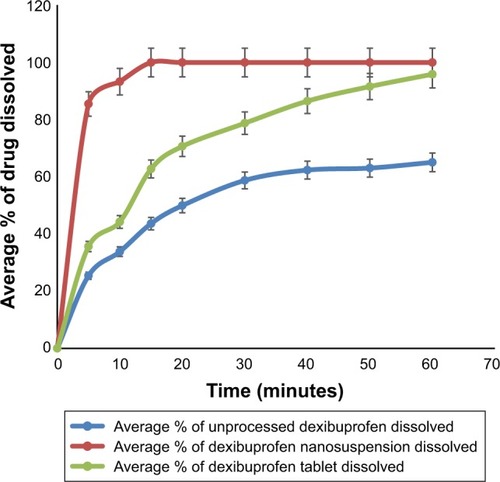
Investigation on the in vitro dissolution of the nanocrystals confirmed enhanced dissolution rate correlated to the raw and marketed tablets of dexibuprofen, that is, 200 mg. shows that ~85.4% of nanocrystals were dissolved in the first 5 minutes. On the other hand, raw dexibuprofen showed a dissolution rate of around 25.4% in the first 5 minutes, while marketed tablets showed around 35.6%. This significant increase in dissolution rate of dexibuprofen nanocrystals indicated that controlled nucleation produced nanocrystals with increased surface area and, consequently, enhanced the dissolution rate.Citation18 Moreover, it is already proved that as per the Ostwald–Freundlich equation, nanoparticles with a size below 100 nm have quicker dissolution rate.Citation28
Conclusion
This study demonstrated that MCFR is a low-energy novel bottom-up technology which can potentially produce stable nanocrystals. However, the experimental and process conditions need to be properly controlled. The optimized process conditions including solvent, antisolvent flow rates, inlet angles, stirring rate and mixing time are the key factors that alter the particle size of the nanoparticles. High antisolvent flow rate, small inlet angle, high mixing time and stirring rate are the favorable factors to produce nanocrystals with a small particle size. Additionally, the appropriate selection of the polymers/surfactant and the low concentration of the drug are the key factors to control the particle size during nucleation. MCFR is a technology which produces the nanocrystals without changes in particle attributes, especially crystallinity of the particles. In future perspectives, this technology can potentially be scaled up. Also, the produced nanocrystals could be converted to the solid dosage form and the process industrialized at a large scale by using chemical engineering approach.
Author contributions
We declare that this work was done by the authors named in this article and all liabilities pertaining to claims relating to the content of this article will be borne by the authors. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Acknowledgments
Jahangir Khan appreciatively acknowledges the support of University of Malakand, HEC Pakistan for its support under the International Research support initiative program and the School of Pharmacy Institute of Life Sciences Research, University of Bradford, UK, for providing resources to accomplish research project.
Disclosure
The authors report no conflicts of interest in this work.
References
- de WaardHHinrichsWLFrijlinkHWA novel bottom-up process to produce drug nanocrystals: controlled crystallization during freeze-dryingJ Control Release2008128217918318423767
- KawabataYWadaKNakataniMYamadaSOnoueSFormulation design for poorly water-soluble drugs based on biopharmaceutics classification system: basic approaches and practical applicationsInt J Pharm2011420111021884771
- PlakkotSde MatasMYorkPSaundersMSulaimanBComminution of ibuprofen to produce nanoparticles for rapid dissolutionInt J Pharm20114151–230731421683776
- ChenHKhemtongCYangXChangXGaoJNanonization strategies for poorly water-soluble drugsDrug Discov Today2011167–835436020206289
- SerajuddinATSolid dispersion of poorly water-soluble drugs: early promises, subsequent problems, and recent breakthroughsJ Pharm Sci199988101058106610514356
- SchwarzCMehnertWLucksJMüllerRSolid lipid nanoparticles (SLN) for controlled drug delivery. I. Production, characterization and sterilizationJournal of Controlled Release19943018396
- GhoshIBoseSVippaguntaRHarmonFNanosuspension for improving the bioavailability of a poorly soluble drug and screening of stabilizing agents to inhibit crystal growthInt J Pharm20114091–226026821371540
- RabinowBENanosuspensions in drug deliveryNat Rev Drug Discov20043978579615340388
- KakranMSahooNGLiLFabrication of drug nanoparticles by evaporative precipitation of nanosuspensionInt J Pharm20103831–228529219781606
- de WaardHFrijlinkHWHinrichsWLBottom-up preparation techniques for nanocrystals of lipophilic drugsPharm Res20112851220122321086152
- PatravaleVBDateAAKulkarniRMNanosuspensions: a promising drug delivery strategyJ Pharm Pharmacol200456782784015233860
- HaoHWangGSunJEnantioselective pharmacokinetics of ibuprofen and involved mechanismsDrug Metab Rev200537121523415747501
- IbuprofenKCDexibuprofen: the superior non-steroidal anti-inflammatory agents for development of pharmaceuticalsInt J Curr Pharm Res2010213
- KaehlerSTPhlepsWHesseEDexibuprofen: pharmacology, therapeutic uses and safetyInflammopharmacology200311437138315035791
- AbdelbaryGMakhloufAAdoption of polymeric micelles to enhance the oral bioavailability of dexibuprofen: formulation, in-vitro evaluation and in-vivo pharmacokinetic study in healthy human volunteersPharm Dev Technol201419671772723937590
- van der WoerdMFerreeDPuseyMThe promise of macromolecular crystallization in microfluidic chipsJ Struct Biol2003142118018712718930
- AliHSYorkPBlagdenNPreparation of hydrocortisone nanosuspension through a bottom-up nanoprecipitation technique using microfluidic reactorsInt J Pharm20093751–210711319481696
- KhanSMatasMDEZhangJAnwarJNanocrystal preparation: low-energy precipitation method revisitedCryst Growth Des201313727662777
- RaoYKumarMApteSFormulation of nanosuspensions of albendazole for oral administrationCurr Nanosci2008415358
- ShahSMUllahFKhanSSmart nanocrystals of artemether: fabrication, characterization, and comparative in vitro and in vivo antimalarial evaluationDrug Des Devel Ther20161038373850
- Jahangir KhanSBShahzebKAyeshaIFabrication and characterization of dexibuprofen nanocrystals using DENA® media millingLat Am J Pharm2018375947952
- Y-FSKimHKovenkliogluSLeeWContinuous nanoparticle production by microfluidic-based emulsion, mixing and crystallizationJ Solid State Chem2007180926252629
- ZhaoHWangJ-XWangQ-AChenJ-FYunJControlled liquid anti-solvent precipitation of hydrophobic pharmaceutical nanoparticles in a microchannel reactorInd Eng Chem Res2007462482298235
- ZhangSYunJShenSFormation of solid lipid nanoparticles in a microchannel system with a cross-shaped junctionChem Eng Sci2008632356005605
- ZhangH-XWangJ-XShaoLChenJ-FMicrofluidic fabrication of monodispersed pharmaceutical colloidal spheres of atorvastatin calcium with tunable sizesInd Eng Chem Res201049941564161
- BrookTLDesign and Fabrication of a Novel Microfluidic Crystalliser for Organic Nanoparticle Production: Investigation of Process Parameters on the Production of Salicylic Acid Nanoparticles for Optimised Drug DeliveryUniversity of Bradford2006
- MatteucciMEHotzeMAJohnstonKPWilliamsRODrug nanoparticles by antisolvent precipitation: mixing energy versus surfactant stabilizationLangmuir200622218951895917014140
- MüllerRHPetersKNanosuspensions for the formulation of poorly soluble drugs: I. Preparation by a size-reduction techniqueInt J Pharm19981602229237
- O’MahonyMLeungAKFergusonSTroutBLMyersonASA process for the formation of nanocrystals of active pharmaceutical ingredients with poor aqueous solubility in a nanoporous substrateOrg Process Res Dev201519911091118
- AliHSYorkPAliAMBlagdenNHydrocortisone nanosuspensions for ophthalmic delivery: a comparative study between microfluidic nanoprecipitation and wet millingJ Control Release2011149217518120946923
- FreitasCMüllerRHEffect of light and temperature on zeta potential and physical stability in solid lipid nanoparticle (SLN™) dispersionsInt J Pharm19981682221229
- PatravaleVBDateAAKulkarniRMNanosuspensions: a promising drug delivery strategyJ Pharm Pharmacol200456782784015233860
- BunjesHKochMHJWestesenKEffect of particle size on colloidal solid triglyceridesLangmuir2000161252345241
- DengJHuangLLiuFUnderstanding the structure and stability of paclitaxel nanocrystalsInt J Pharm201039024224920167270

![Figure 3 Particle size distribution of dexibuprofen nanoparticle (Poloxamer-407 [0.5%]).](/cms/asset/a7ffa564-6da7-475a-8226-32375bd3ad74/dddt_a_12181785_f0003_c.jpg)