Abstract
Over the past decade, the idea of targeting the endocannabinoid system to treat anxiety disorders has received increasing attention. Previous studies focused more on developing cannabinoid receptor agonists or supplementing exogenous cannabinoids, which are prone to various adverse effects due to their strong pharmacological activity and poor receptor selectivity, limiting their application in clinical research. Endocannabinoid hydrolase inhibitors are considered to be the most promising development strategies for the treatment of anxiety disorders. More recent efforts have emphasized that inhibition of two major endogenous cannabinoid hydrolases, monoacylglycerol lipase (MAGL) and fatty acid amide hydrolase (FAAH), indirectly activates cannabinoid receptors by increasing endogenous cannabinoid levels in the synaptic gap, circumventing receptor desensitization resulting from direct enhancement of endogenous cannabinoid signaling. In this review, we comprehensively summarize the anxiolytic effects of MAGL and FAAH inhibitors and their potential pharmacological mechanisms, highlight reported novel inhibitors or natural products, and provide an outlook on future directions in this field.
Introduction
Anxiety disorders represent prevalent mental health conditions characterized by persistent psychogenic anxiety, somatic anxiety, and sleep disorders. According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), the classification of anxiety disorders includes Generalized Anxiety Disorder, Phobias (including specific phobias and social anxiety disorder), Panic Disorder, as well as other anxiety-related conditions such as Separation Anxiety Disorder and Selective Mutism.Citation1 The global prevalence of anxiety disorders ranges from 7.3% to 28.0%, making them a significant concern worldwide.Citation2 Specifically, in China, anxiety disorders stand out as the most prevalent mental illness, with a lifetime incidence of 7.6%.Citation3
The World Health Organization underscores the significance of anxiety disorders by ranking them as the sixth leading contributor to global disability, establishing it as a pressing health and wellness concern that demands attention.Citation4 Regrettably, the existing repertoire of anxiolytic medications faces challenges in adequately addressing the needs of the extensive population affected by anxiety disorders.Citation5 Benzodiazepines and selective serotonin reuptake inhibitors (SSRIs) antidepressants are the primary drugs employed clinically for the treatment of anxiety disorders.Citation6,Citation7 However, their anxiolytic effects are secondary indications rather than primary, and the drugs, especially benzodiazepines, are subject to stringent controls and possess potential addictive properties.Citation8 Consequently, the development of novel drugs targeting anxiety disorders remains a focal point in the ongoing research and development of psychotropic medications.
Traditionally, anxiolytic drug development focused on neurotransmitter-based hypotheses, yielding medications like eszopiclone and buspirone.Citation9,Citation10 Recent attention has turned to novel approaches, particularly targeting the endocannabinoid system (ECS). The ECS, integral to mood regulation, has been implicated in the pathogenesis of both anxiety and depression, underscoring its potential as a novel therapeutic frontier.Citation11 Novel drug designs that target the ECS are considered to be the most promising candidates for the treatment of anxiety disorders.Citation12
Studies explore ECS modulation for anxiolysis, including increasing endogenous cannabinoids (eCBs) secretion and inhibiting their hydrolase enzymes. However, challenges arise due to the unique characteristics of endocannabinoids. Efforts to develop cannabinoid receptor agonists face obstacles like pharmacological intensity and adverse effects. A more promising strategy involves inhibiting eCB hydrolases, notably monoacylglycerol lipase (MAGL) and fatty acid amide hydrolase (FAAH), indirectly activating cannabinoid receptors. Clinical trials with FAAH inhibitors, such as JNJ-42165279, have shown positive anxiolytic potential.Citation13 This paper comprehensively summarizes the anxiolytic effects of eCB hydrolase inhibitors, focusing on underlying mechanisms and highlighting novel inhibitors or natural products. The insights from existing studies aim to provide new perspectives for the development of innovative anxiolytic drugs.
Endocannabinoid System
The ECS is one of the crucial regulatory systems in the central nervous system, and consists of cannabinoid receptor 1 (CB1R), cannabinoid receptor 2 (CB2R), eCBs, and their corresponding synthesizing and degrading enzymes. eCBs are classified into three categories according to chemical structure: 1) esters: such as 2-arachidonoylglycerol (2-AG), anandamide (AEA); 2) amides: such as palmitoylethanolamide (PEA), oleoylethanolamide (OEA); 3) ethers: 2-arachidonyl glyceryl ether (noladin ether or 2-AGE).Citation14,Citation15 Among them, AEA and 2-AG are more abundant and widely distributed in the human body, while specific eCBs such as OEA and 2-AGE can only be detected in particular regions of brain tissue.Citation16 Notably, 2-AG is agonistic for both CB1R and CB2R, whereas AEA selectively has a high affinity for CB1R but almost no activity on CB2R.Citation17
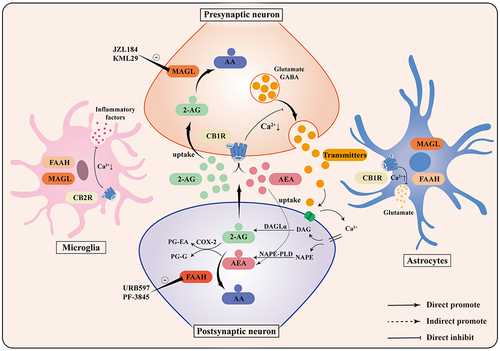
The ways of synthesis, transport and inactivation of 2-AG and AEA in their respective target tissues are also different. 2-AG is mainly produced by 1-oleoyl-2-arachidonoyl-sn-glycerol (OAG) and 1-stearoyl-2-arachidonoylglycerol (SAG) via diacylglycerol lipase α (DAGLα), whereas AEA produced by N-acylphosphatidylethanolamine (NAPE) catalysed by N-acylphosphatidylethanolamine-specific phospholipase D (NAPE-PLD).Citation11 Degradation and inactivation of eCBs mainly involve two pathways: hydrolysis and oxidation. MAGL, a specific hydrolase of 2-AG, is mainly distributed on the axons of presynaptic neurons and is able to terminate retrograde eCB signaling generated by postsynaptic neuronal activity. Approximately 85% of 2-AG in the brain is hydrolyzed and metabolized to arachidonic acid and glycerol by MAGL.Citation18 AEA was mainly degraded by FAAH into free arachidonic acid and ethanolamine. FAAH, a serine hydrolase mainly present in the endoplasmic membrane, is widely distributed in the central nervous system and hydrolyses various fatty acid amides, including AEA. In another oxidation pathway, the critical enzyme involved is cyclooxygenase-2 (COX-2), catalyzing the synthesis of prostaglandin ethanolamide (PG-EA) and prostaglandin glycerol (PG-G) from 2-AG and AEA.Citation11 briefly illustrates the composition of the endocannabinoid system, and the synthesis and degradation pathways of endocannabinoids.
Figure 1 Composition of the endocannabinoid system. The endocannabinoid system consists of CB1R, CB2R, endocannabinoids, and their corresponding synthesizing and degrading enzymes. 2-AG and AEA, the primary endocannabinoids, are produced on demand and are synthesized from the postsynaptic terminals by DAGLα and NAPE-PLD, respectively, to activate presynaptic cannabinoid receptors. CB1R activation inhibits presynaptic neurotransmitter release and promotes astrocytic glutamate release. CB2R activation reduces microglial inflammatory factor production. 2-AG and AEA are enzymatically degraded to AA by MAGL and FAAH hydrolases, and can also be oxidatively degraded to PG-EA and PG-G by COX-2.
Numerous studies have demonstrated that the anxiolytic effects could be exerted by increasing eCB secretion, applying exogenous cannabinoids, agonizing cannabinoid receptors, and decreasing eCB hydrolase activity.Citation19–21 However, unlike classical neurotransmitters and neuropeptides, eCBs are not stored within synaptic vesicles, but are produced on demand and immediately released from neurons. At the same time, the eCBs synthesis pathways are numerous and complex, and the rate-limiting enzymes are plentiful and poorly defined, leading to insignificant gains through the strategy of increasing the activity of eCB synthetases. In addition, drug development targeting cannabinoid receptor agonists or adding exogenous cannabinoids is susceptible to exclusion from clinical trials or even post-market withdrawal due to stronger pharmacological activity, irreversible binding, poor receptor selectivity, and increased risk of adverse effects.Citation22,Citation23 Therefore, the most prominent strategies may be to search for inhibitors of eCB hydrolases MAGL and FAAH, especially reversible inhibitors that indirectly activate cannabinoid receptors by increasing synaptic gap levels of 2-AG and AEA, respectively.Citation24
2-AG Hydrolase MAGL Inhibitors in Treatment for Anxiety
2-AG, an abundant brain eCB, surpasses AEA levels by 200 times.Citation25 Clinical studies revealed that serum 2-AG levels are significantly reduced in patients with post-traumatic stress disorder (PTSD), which was previously often categorized as an anxiety disorder.Citation26 Additionally, it has been found that increasing 2-AG levels through exercise can alleviate anxiety symptoms.Citation27 This findings are consistent with rodent studies,Citation28,Citation29 where higher MAGL levels have been strongly linked to the production of anxious behaviors.Citation30 Inhibition of MAGL activity enhances central synaptic 2-AG-mediated phasic and tonic signaling, which has positive implications in anxiety relief, reduction of stress-induced anxiety susceptibility, and fear extinction.Citation31
Anxiolytic Effects of MAGL Inhibitors
Due to MAGL’s pivotal role in regulating 2-AG levels and synaptic transmission, efforts to enhance 2-AG signaling for therapeutic purposes have centered on inhibiting MAGL enzyme activity. Numerous pharmacological studies highlight that systemic or local administration of MAGL inhibitors can effectively reduce anxiety-like behaviors induced by acute or chronic stress (). For example, aberrant excitation of glutamatergic neurons in basolateral amygdala (BLA)-prelimbic prefrontal cortex (plPFC) neural circuit in mice subjected to chronic stress, accompanied by abnormal 2-AG-CB1R signal, and administration of a MAGL inhibitor reversed anxiety-like behavior.Citation32 Overexpression of MAGL in hippocampal glutamatergic neurons also increases anxiety-like behavior in animals.Citation33 In addition, anxiety-like behaviors induced by traumatic brain injury and alcohol withdrawal improved after administration with MAGL inhibitor.Citation34
Table 1 Summary of the Anxiolytic Effects of MAGL Inhibitors in Preclinical Studies
However, it is important to note that 2-AG levels do not consistently correlate negatively with anxiety disorders. Acute stress given to healthy people increased circulating concentrations of AEA in vivo but had no significant effect on 2-AG.Citation45 In chronic restraint stress mice, 2-AG levels in the cingulate cortex (ACC), caudate putamen (CP), nucleus accumbens (NAc), and piriform cortex (PIR) all increased.Citation46 Similarly, 2-AG content increased in the amygdala of chronic stress-induced anxiety mice.Citation41,Citation47 The researchers explained that the above phenomenon was due to CB1R desensitization, as it did not affect the anxiolytic effect of applying JZL184. Bedse G’s research also supports the idea that increased 2-AG is a compensatory response to counteract anxiety-like behaviors induced by stress, and that 2-AG pharmacological enhancers can augment this response to more effectively counteract the adverse effects of stress.Citation38 Therefore, the benefit of elevating 2-AG pharmacologically through MAGL inhibitors in the treatment of anxiety disorders is definitive.
Interestingly, instead of demonstrating anxiolytic effects, full knockout MAGL significantly reduced the duration in the light box in mice.Citation48 The study found that the CB1R receptor was significantly downregulated in MAGL knockout mice, possibly due to enhanced levels of innate endogenous cannabinoids leading to CB1R desensitization and the emergence of anxiety-like behavior. Consistent with these findings, CB1R density and functional responses were reduced in chronic MAGL inactivation and MAGL KO mice.Citation49 In conclusion, congenital and chronic MAGL inactivation may lead to CB1R desensitization and feedback down-regulation, which may inhibit the downstream anti-anxiety effect of CB1R, but more in-depth studies are needed.
Anxiolytic Mechanisms of MAGL Inhibitors
Although preclinical studies support the anxiolytic effects of MAGL inhibitors, their specific downstream molecular mechanisms remain poorly understood. As shown in , we summarized the potential mechanisms of MAGL from the following three aspects based on the above pharmacological studies.
Figure 2 Schematic representation of the mechanism of anxiolytic action mediated by MAGL inhibitors. Briefly, the anxiolytic effects of MAGL are related to its maintenance of Glutamate/GABA balance, inhibition of neuroinflammation, and regulation of corticosterone levels.
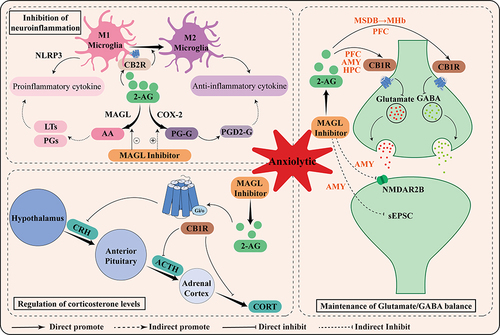
Maintenance of Glutamate/GABA Balance
Glutamate and GABA are representative excitatory and inhibitory transmitters in the central nervous system, which cause neurons to generate corresponding excitatory and inhibitory currents through inter-synaptic transmitter transmission. Typically, neuronal excitation/inhibition (E/I) is in a dynamic balance. Once this balance is disturbed, especially when it tends to be excitatory, it can lead to the development of anxiety disorders. The E/I balance in the PFC, a brain region responsible for executive function, stress and emotion regulation, seems to play an important role in the anxiolytic effects of MAGL inhibitors.Citation50 PFC projection neurons are involved in the development of anxiety by modulating neural signaling in downstream brain regions during exposure to stress.Citation51 Systemic administration of the MAGL inhibitor, KML29, increased vmPFC excitability but could be blocked by CB1R or GABA receptor antagonist, which supports the conclusion that activation of the CB1R leads to transient inhibition of GABA release and long-term inhibition of inhibitory transmission.Citation43 Consistent with this finding, the CB1R agonist 5F-AMB attenuates glutamatergic and GABAergic synaptic transmission in mPFC L5 pyramidal neurons, leading to an E/I imbalance.Citation52
Meanwhile, the amygdala, an essential part of the limbic system that controls emotion, plays a crucial role in the regulation of anxiety, and maintaining its E/I balance is of great significance.Citation53 MAGL inhibitors inhibit Glutaminergic neurotransmission in the amygdala region. It was found that increased expression of multiple glutamate receptors in the amygdala, including mGluR1, mGluR5, and NMDAR1, was accompanied by elevated expression of MAGL in an alcohol exposure-induced anxiety model,Citation30 and that JZL184 could exert anxiolytic effects by indirectly activating the CB1R to reduce NMDAR2B expression.Citation54 Bedse et al found that acute stress increased the frequency of spontaneous excitatory postsynaptic currents (sEPSC) in basolateral amygdala neurons and was positively correlated with anxiety-like behaviors, with the above reversed after administration with JZL184.Citation38
Notably, 2-AG is recognized as a potent agonist of CB1R. CB1R is found in both glutamatergic and GABAergic neurons, but the relative abundance of glutamatergic and GABAergic neurons in various brain regions varies, potentially causing functional alterations in distinct brain regions. Lysine-specific demethylase 1 (LSD1) suppressed hippocampal MAGL transcript levels and activated CB1R to inhibit glutamate release in response to anxiety.Citation55 JZL184 enhanced the inhibitory effect of 2-AG on the release of GABA from the medial septum and nucleus of the diagonal band (MSDB) axons to the medial habenula (MHb) and produced anxiolytic effects.Citation56
Inhibition of Neuroinflammation
Neuroinflammation is considered to be a trigger for behavioral changes and cognitive deficits in several psychiatric disorders, including anxiety disorders. Prolonged exposure to stress imbalances the central immune system and affects the secretion and function of immune cells and cytokines.Citation57 Currently, MAGL inhibitors play an important role in the treatment of central and peripheral inflammation.Citation58,Citation59 The study showed that JZL184 reduced LPS-induced expression of IL-1β, IL-6, TNF-α, and IL-10 in the prefrontal cortex and spleen.Citation60
Dual metabolism of 2-AG is closely relevant to the anti-inflammatory effects exerted by MAGL inhibitors. Arachidonic acid (AA), the main metabolite of 2-AG hydrolysis by MAGL, is a crucial precursor of proinflammatory prostaglandins and leukotrienes involved in inflammatory responses and immune initiation. The latest single-cell sequencing found that MAGL KO mice had significantly upregulated genes related to immunity and inflammation in microglia and astrocytes, which enable glial cells to react rapidly to insults.Citation61 Specifically knocking out MAGL in astrocytes reverses LPS-induced inflammatory activation and is not blocked by the CB1R agonist SR141716.Citation62 Similarly, CB1R/CB2R antagonists did not block JZL184 from inhibiting LPS-induced neuroinflammation.Citation63 This suggests that the anti-inflammatory effect produced by MAGL inhibition is a direct result of reduced prostaglandin, rather than a profile result of enhanced endocannabinoid signaling. In addition, enhanced inhibition of 2-AG hydrolysis can promote the oxidation pathway mediated by COX-2 to produce prostaglandin glycerides (PG-Gs), with PGD2-G exhibiting anti-inflammatory activation.Citation64
CB2R, which can be fully activated by 2-AG, is mainly expressed in brain microglia and associated with neuroinflammation.Citation65 Many studies have confirmed the anxiolytic effect of CB2R agonists, and found that the use of CB2R agonist AM1241 inhibited the over-activation of PFC microglia by inhibiting the NLRP3 pathway, thereby improving anxiety-like behavior.Citation66 In addition, CB2R activation also promoted the transformation of microglia to M2 anti-inflammatory phenotype, creating positive feedback by releasing more 2-AG and AEA.Citation67,Citation68 Thus, the effects of MAGL inhibitors in improving anxiety may be related to increasing the response of glial cells to external stimuli and promoting the polarization transformation of microglia.
Regulation of Corticosterone Levels
The dysfunction of the hypothalamic-pituitary-adrenal (HPA) axis, the main regulator of stress response, is one of the biological mechanisms of anxiety. Increased corticosterone is an important marker of HPA axis activation. Studies have shown that ECS bi-directionally regulates the function of the HPA axis. MAGL inhibitors reduced high corticosterone levels in mice 30 min after chronic restraint stress but restored higher corticosterone levels after 120 min, whereas these changes were not observed in CB1R knockout mice.Citation69 The reason for these phenomena was the predominant inhibition of HPA axis activation during the early phase of JZL184 injection, whereas the sustained activation of CB1R elevated circulating corticosterone levels with increasing 2-AG concentrations. Aliczki et al found that metiramone, a cortisol synthesis blocker, reversed the increased open-arm time in the EPM test in mice with JZL184, suggesting that the anxiolytic effect of JZL184 is accompanied by activation of the HPA axis.Citation70 Glucocorticoids released after exposure to stress activate CB1R signaling in the mPFC, inhibit GABA release, and act on the HPA axis negative feedback process to appropriately reduce corticosterone secretion.Citation71 Therefore, MAGL inhibitors may exert anxiolytic effects through CB1R-dependent glucocorticoid increases.
Novel MAGL Inhibitors and Natural Product Development
Given the significant pharmacological effects and higher safety of MAGL inhibitors, there is growing attention towards the development of novel inhibitors, with substantial efforts underway to identify safer, more effective, and reversible MAGL inhibitors. Research has primarily focused on developing novel inhibitors through structural modification of lead compounds or the use of fluorescent probes. Finding MAGL inhibitors from natural products has also been a strategic approach. Encouragingly, the majority of natural products reported to have MAGL inhibitory activity are reversible, enhancing drug safety with their gentle effects.
Reversible MAGL Inhibitors
A total of 96 patents related to MAGL inhibitors were searched (https://patentscope.wipo.int/). Initially developed MAGL inhibitors were eliminated due to poor inhibition and lack of selectivity for MAGL. Later, Cravatt et al discovered JZL184, which was irreversible, inhibitory, and selective, was widely used in experimental studies. JZL184 is a potent tool compound, but it is difficult to use clinically due to the fact that it exhibits some cross-reactivity of FAAH at high doses (40 mg/kg) and some species differences in MAGL inhibitory potency.Citation72 Subsequently, KML29 was developed with better selectivity and good neuroprotection in a stroke model and good analgesic activity in a migraine model.Citation73,Citation74
However, JZL184, KML29, NAM, SAR629 and CK37 are all irreversible inhibitors of MAGL, which are prone to physical dependence, endocannabinoid dependent synaptic plasticity impairment and cross-tolerance to exogenous CB1 agonists.Citation75 JJKK048 is the first reported reversible, highly selective MAGL inhibitor with powerful analgesic effects and does not cause side effects similar to cannabis.Citation76 (R)-3t, a synthetic selective and reversible MAGL inhibitor, reduced arachidonic acid levels and increased 2-AG levels in the brain after gavage in mice.Citation77 Recently discovered M-18c, with an IC50 value of 662.6 nM for MAGL, attenuates LPS-induced acute kidney injury by inhibiting NLRP3-associated inflammation.Citation78 Compared with irreversible inhibitors, reversible inhibitors have better pharmacokinetic advantages and offer substantial advantages in drug safety, tolerability and efficacy, with better development prospects and clinical applications.
Structural Modifications Based on Lead Compounds
Virtual screening based on molecular docking was applied to discover novel reversible MAGL inhibitors. For example, Afzal O performed virtual docking of the ZINC database with 21 million compounds and screened seven potential activities, of which ZINC24092691 showed significant inhibitory activity.Citation79 DC630-8Citation80 and CL6aCitation81 also showed good inhibitory activity based on virtual docking. However, the screened compounds have poor drug-forming properties due to low screening efficacy. Therefore, many studies of structural modification based on lead compounds and functional group replacement have been added to the search for reversible inhibitors. Arylformylpiperidine derivatives developed by Zhi et al exhibit good reversible inhibitory properties and significantly ameliorate rifampicin-induced depressive-like behavior, providing support for MAGL as a potential therapeutic target for depression.Citation82 In addition, classical MAGL inhibitors such as CAY10499, JZL184, and ABX-1431 (also known as Lu AG06466) are classified as carbamate derivatives. Tiziano Tuccinardi’s team discovered a series of new compounds with MAGL-inhibitory properties based on benzylpiperidine derivatives, such as compound 13 with antipancreatic cancer effects,Citation83 compounds 28 and 29 with low in vivo toxicity and high selectivity,Citation84 diphenylsulfide-benzoylpiperidine derivatives with anticancer activity.Citation85
Looking for potential MAGL inhibitory activity in existing drugs is also a strategy. Disulfiram, an aldehyde dehydrogenase inhibitor used primarily in the treatment of chronic alcoholism, was shown to be a MAGL inhibitor that irreversibly inhibits MAGL through carbamoylation of Cys208 and Cys242 located near the MAGL active site.Citation86 However, due to the presence of some FAAH inhibitory activity, Omran synthesized compounds targeting MAGL but lacking anti-FAAH activity by replacing the two ethyl groups in the disulfide.Citation87 Recently, cetirizine and levetiracetam have also been found to have potential MAGL inhibitory activity, with IC50 values of 9.3931 µM and 3.0095 µM, respectively, and demonstrated some analgesic and anti-inflammatory activity.Citation88
Consideration of pharmacokinetic distribution is crucial in developing MAGL inhibitors. LEI-515, a recently discovered peripherally restricted reversible MAGL inhibitor, interestingly increased 2-AG levels only in peripheral organs but not in the mouse brain, hinting at potential applications in peripheral diseases.Citation89 Additionally, the compound properties can be improved by structural modification or application of nanocarriers if the pharmacokinetics are not ideal. Muhammad Adeel’s team developed the first nano-formulation of a MAGL inhibitor, MAGL23, which showed promising anti-tumour activity by using albumin-complexed nanocrystals that increased its solubility in water from less than 0.01 mg/mL to 0.82 mg/mL.Citation90
Probe Development
Developing MAGL inhibitors based on active molecular probes is also a new strategy. Activity-based protein profiling (ABPP) technology, which uses active site-directed covalent probe molecules to detect the functional state of enzyme activities in complex proteomes, has been applied to various enzyme classes.Citation91 Cisar et al identified and optimised a highly effective, selective and centrally permeable oral MAGL inhibitor, ABX-1431 (Lu AG06466), from a carbamate library by ABPP technology, which has entered Phase II clinical trials.Citation92 In addition, some newly screened compounds such as quinoid diterpene and β-carbolines,Citation93 as well as newly synthesised structural modifiers based on benzylpiperidine and benzylpiperazine,Citation84 were confirmed for their potential MAGL inhibitory activity by ABPP. The ABPP technique was also applied in discovering and mapping the distribution of eCB hydrolase activity, and MAGL enzyme activity was found to be strongest in the PFC region.Citation94 In conclusion, the ABPP has made it possible to visualize the spatio-temporal release of eCB hydrolases with high spatial resolution.
In recent years, many target-labeled radioactive probes have been developed based on MAGL inhibitors, some of which have even entered clinical trials. This technology can be used to image the in vivo distribution of MAGL and provide a method for subsequent disease diagnosis and treatment. PET imaging using 18F-T-401 was the first to image and quantify the distribution of MAGL in the human brain and found that MAGL was highest in the cerebral cortex, intermediate in the thalamus and nucleus accumbens, and lowest in the white matter and brainstem.Citation95 He et al developed a modified compound 7 based on morpholin-3-one derivatives, which may be a potential MAGL PET tracer, and successfully mapped the MAGL distribution pattern on rodent brain in vitro radioautography using the fixation method of direct 11CO2 synthesis.Citation96 Based on a unique 4-piperidinylazetidine diamide scaffold, Cheng et al developed a reversible and peripherally specific radiolucinated MAGL PET ligand, [18F]FEPAD, which has excellent specificity and selectivity for MAGL in brown adipose tissue, a tissue known to be metabolically active.Citation97
Natural Product Development
Chemically synthesized MAGL inhibitors face challenges in clinical use due to their potent pharmacological effects and safety concerns. The current focus is shifting towards natural products and botanicals to discover milder, safer, and more effective reversible MAGL inhibitors. Pritimerin and euphol were the first identified natural products with reversible MAGL inhibitory activity.Citation98 Four triterpenoid constituents, including pritimerin and euphol, have been reported to significantly inhibit human recombinant MAGL activity, of which pritimerin ameliorated mechanical pain in mice with a concentration-dependent manner.Citation99 Protium copal, commonly used as incense by the Maya, displayed significant MAGL inhibitory activity, alleviating anxiety-like behaviors in rats, and this effect was blocked by a CB2R blocker.Citation100 The compound 8-prenylnaringenin in Humulus lupulus L. reversibly inhibited MAGL and reduces neuroinflammation, promising for Alzheimer’s disease.Citation101 Extracts from Myristica fragrans exhibited anxiolytic and antidepressant effects, with significant MAGL inhibitory activity.Citation102
Taking a computer-aided drug design (CADD) approach, combined with molecular docking, accelerates the discovery of MAGL inhibitors from natural products. Through the establishment of the pharmacophore model Phar-MAGL, combined with molecular docking and Ligplot analysis, NP-2/8-PN (IC50 = 9.5 ± 1.2 μM), NP-5 (IC50 = 14.5 ± 1.3 μM), and NP-3 (IC50 = 15.2 ± 1.4 μM) were successfully screened for their promising in vitro inhibitory activities of MAGL.Citation101 Interestingly, 8-PN also had a positive metamorphic modulatory effect on GABAA that was not mediated through a high-affinity benzodiazepine binding site, and its potential anxiolytic effect could be further investigated.Citation103 Phenylethanoid glycosides from C. phelypeaeCitation104 and Jewenol A from S. pseudorosmarinusCitation105 also demonstrated good MAGL inhibitory activity through enzymatic assays and molecular docking. Screening based on existing efficacy can enhance hit rates, as demonstrated in the study by Mei et al, who screened the MAGL inhibitory activity of 12 Chinese herbal medicines commonly used for analgesia, identifying Corydalis yanhusuo as the most effective.Citation106 Forsythiaside, a phenolic acid glycoside in Forsythia suspensa, inhibits COX-2 and MAGL, demonstrating neuroprotective effects in Alzheimer’s disease by increasing hippocampal 2-AG content.Citation107 summarizes some reported natural products with MAGL inhibiting activity.
Table 2 Summary of MAGL Inhibitors Derived from Natural Products
AEA Hydrolase FAAH Inhibitors in Treatment for Anxiety
Human and rodent studies consistently show a negative correlation between anxiety levels and AEA concentrations.Citation45,Citation110 Reduced AEA levels are strongly associated with PTSD severity, and moderate aerobic exercise has been found to increase AEA levels.Citation111 Stress-induced anxiety-like behaviors in rodents coincide with significant reductions in brain AEA levels, while microinjection of methanandamide (an AEA analogue) into the rat prefrontal cortex produces anxiolytic effects.Citation112 Alternatively, inhibition of AEA synthase promotes anxiety production. In NAPE-PLD-deficient mice, dorsal hippocampal AEA is significantly reduced and induces anxiety-like behavior.Citation113 FAAH is widely distributed in major neurons, including pyramidal cells in the BLA and hippocampus. Increased FAAH activity and decreased AEA levels have been found in mice subject to chronic restraint stressCitation114 and in Marchigian Sardinian alcohol-preferring rats with innate anxiety.Citation115 Therefore, enhancing AEA signaling by inhibiting FAAH activity is a potential strategy for the treatment of anxiety disorders.
Anxiolytic Effects of FAAH Inhibitors
Several new compounds developed for FAAH inhibitors have entered clinical trials with promising prospects (https://classic.clinicaltrials.gov/ct2/home). JNJ-42165279 produces central and peripheral FAAH inhibition, significantly increases AEA levels in cerebrospinal fluid and plasma, and has not been found to have any safety concerns.Citation116 In the latest clinical pilot study, attenuation of amygdala, bilateral anterior cingulate gyrus, and bilateral insula activation during an emotional face processing task was found after 4 days of administration of JNJ-42165279 (100 mg) to 43 subjects, which is in line with the effects previously observed with anxiolytics.Citation117 Meanwhile, in another clinical study, JNJ-42165279 (25 mg/d) was effective in improving anxiety-like symptoms after 12 weeks of administration to patients with social anxiety disorder.Citation13 For PF-04457845, it was well tolerated,Citation118 attenuated anxiety effects in healthy subjects facing stress,Citation119 and weakened cannabis withdrawal symptoms.Citation120
Several preclinical studies have shown that pharmacological inhibition of FAAH activity exhibited remarkable anxiolytic effects in different animal models of anxiety (). Meanwhile, genetic evidence also supports the above results. It was found that FAAH whole genome knockout C57BL/6J mice are not induced with anxiety-like behavior by chronic restraint stress.Citation114 In contrast, increased FAAH expression leads to the development of anxious behaviors in animals. Specific overexpression of FAAH in hippocampal glutamatergic neurons using AAV vectors significantly reduced AEA and PEA levels and increased anxiety-like behaviors, which may be related to enhanced LTP in glutamatergic neurons, leading to increased glutamate release.Citation121 Similarly, specific overexpression of FAAH in PFC significantly reduced AEA levels and had anxiogenic effects.Citation112
Table 3 Summary of the Anxiolytic Effects of FAAH Inhibitors in Preclinical Studies
Single nucleotide polymorphism (SNP) in the FAAH gene, particularly the C385A (rs324420) allele, is noteworthy due to its significant association with FAAH expression. The polymorphism consists of the replacement of cytosine (C) by adenine (A) at nucleotide position 385, which translates into an amino acid exchange in which proline (Pro) replaces threonine (Thr) in codon 129. Mutations in the A allele result in decreased FAAH activity and increased AEA levels.Citation131 Healthy male adults carrying AC heterozygotes have higher levels of AEA and lower anxiety scores than CC homozygotes,Citation132 and carriers of the A allele have been found to have more robust neural network connectivity and lower anxiety levels in adolescents.Citation133 Similarly, knock-in of the FAAH C385A gene in mice significantly ameliorated anxiety-like behavior, which was associated with reduced FAAH expression, elevated AEA levels, and enhanced eCB signaling.Citation134 However, additional studies have shown that the FAAH C385A genetic variant in children increased the risk of anxiety.Citation135 Therefore, when FAAH inhibitor therapy is taken in the future, it may be necessary to consider the genotype of the patient’s C385A to adopt a safer and more effective therapeutic strategy.
Anxiolytic Mechanisms of FAAH Inhibitors
As shown in , FAAH inhibitors have partly the same anxiolytic mechanism as MAGL inhibitors due to similar functions. Except for maintaining Glutamate/GABA balance, suppressing neuroinflammation, and regulating the HPA axis, the promotion of neurogenesis and plasticity has also been suggested to be related to the anxiolytic effects of FAAH inhibitors.
Figure 3 Schematic representation of the mechanism of anxiolytic action mediated by FAAH inhibitors. Briefly, the anxiolytic effects of FAAH are related to its maintenance of Glutamate/GABA balance, suppression of neuroinflammation, regulation of HPA axis, and promotion of neurogenesis and plasticity.
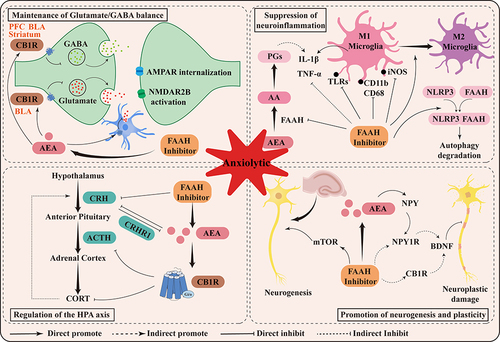
Maintenance of Glutamate/GABA Balance
Similar to MAGL inhibitors, FAAH inhibitors also exert anxiolytic effects by regulating the E/I balance in various brain regions. As we mentioned, AEA selectively exhibits a high affinity for CB1R. CB1R exists in high density in the axonal terminals of Glutaminergic neurons and GABAergic neurons. When activated by AEA signals, G protein-mediated signal cascades are activated, thereby inhibiting the opening of voltage-gated calcium channels and membrane hyperpolarization caused by increased potassium channel opening.Citation136 Thus, FAAH inhibition restores the E/I balance and exerts anxiolytic effects primarily through CB1R alteration of brain Glutamatergic and GABAergic signaling.
Within the amygdala, constitutive signaling of AEA is present at the glutamatergic terminal CB1R, which limits excitatory transmission in the BLA and central amygdala (CeA).Citation137 Expression of FAAH in the postsynaptic terminal is higher in the BLA compared to the CeA.Citation138 The loss of AEA signaling in the amygdala leads to increased glutamate release, which increases the activity of postsynaptic output neurons, triggering anxious behaviors and stress responses.Citation139 Inhibition of FAAH prevented the rapid loss of AEA signalling caused by stress, which countered the effects of different types of stress, including elevated anxiety.Citation124 PF-04457845 similarly reversed the dysregulation of amygdala E/I balance in rats, reducing anxiety-like behaviors and increasing social behaviors.Citation140,Citation141 Interestingly, in contrast to activating neuronal CB1R to inhibit glutamate release, AEA could activate astrocytic CB1R to promote glutamate release, leading to NMDAR2B activation and AMPAR internalization, which induced long-term depression (LTD) and inhibited BLA pyramidal neurons to produce anxiolytic effects.Citation142 Therefore, the excitatory balance of BLA may depend on the level of glutamate release in neurons and astrocytes.
The striatum is also rich in CB1R and is involved in the regulation of anxiety. FAAH inhibition of accumulated AEA modulates excitatory and inhibitory neurotransmission in the striatum.Citation143 Social defeat stress induction decreased the sensitivity of striatal GABAergic synapses, but not glutamatergic, to CB1R activation and was restored by UBR597.Citation144 URB597 also prevented quinolinic acid-induced neuroexcitotoxic damage and preserved striatal structural integrity.Citation145
Suppression of Neuroinflammation
FAAH inhibitors have also been reported to have robust anti-inflammatory effects. For example, URB937 reversed increased plasma inflammatory factor levels in social failure stress rats.Citation128 URB597 also reversed the expression of peripheral and cerebral proinflammatory cytokines under stress induced by LPS.Citation146 Studies have additionally shown that increased FAAH activity in LPS induced social behavioral deficits in adolescent rats and that imposition of PF04457845 reversed social behavioral changes.Citation147
The central anti-inflammatory effects of FAAH inhibitors have been linked to the modulation of microglia polarisation phenotype.Citation148 AEA is an important signalling molecule in regulating microglia function, promoting their anti-inflammatory gene expression and inhibiting pro-inflammatory cytokine release.Citation67,Citation149 The FAAH inhibitors PF3845 and URB597 both inhibited the production of prostaglandin E2 and pro-inflammatory gene expression in the BV2 microglial cell line, and the inhibitory effect of PF3845 was more pronounced.Citation150 URB597 improved the morphological characteristics of rat hippocampal microglia and promoted the transformation of microglia into an anti-inflammatory phenotype.Citation151 In addition, PF3845 also inhibits the expression of inducible nitric oxide synthase and COX-2 and promotes the shift of M1 pro-inflammatory phenotype to M2 anti-inflammatory phenotype.Citation152
Modulation of Toll-like receptors (TLRs) may be another pathway. TLRs are intimately involved in the nervous system’s immune response and are thought to underlie and exacerbate neurological disorders. Systemic administration of URB597 or PF3845 both inhibited TLR3/4-induced associated neuroinflammation in the PFC and hippocampus.Citation153 URB597 also reduced the TLR3-mediated increase in the expression of microglia/macrophage activation markers CD11b/CD68 and significantly alleviated anxiety-like behavior in rats.Citation125 Furthermore, recent studies revealed that FAAH has membrane anchoring and stabilising effects on NLRP3, and that URB597 and PF-04457845 inhibit NLRP3-FAAH interactions and induce autophagic NLRP3 degradation, suppressing the inflammatory phenotype.Citation154
Regulation of the HPA Axis
Patients with anxiety disorders were accompanied by high levels of adrenocorticotropic hormone (ACTH), glucocorticoids in the blood, and overall hyperactivity of the HPA axis.Citation155 The level of AEA is negatively correlated with the activation of the HPA axis. It has been found that reduced homeostasis of AEA after repeated stressor exposure leads to basal overproduction of corticosterone.Citation156 The use of the hydrolase inhibitor URB937 reversed high plasma corticosterone levels 24 hours after social failure.Citation128
The underlying mechanisms of FAAH inhibitors suppress HPA axis activation may be mediated by AEA/CB1R. In stressed rats, AEA levels in the amygdala were negatively correlated with serum corticosterone concentrations, and repeated corticosterone injections also resulted in a stress response. Meanwhile, injection of UBR597 into the BLA reduced stress-induced corticosterone secretion, and this effect was blocked by the CB1R antagonist AM251.Citation157 The administration of AM251 to rats similarly increased their plasma concentrations of ACTH and CORT.Citation158
Alternatively, corticotropin-releasing hormone (CRH) may be a candidate for linking the AEA to the HPA axis. The FAAH inhibitor URB597 reduced HPA axis hyperactivation and anxiety-like responses to stressCitation159 and dose-dependently down-regulated stress-induced CRH mRNA expression in the paraventricular nucleus of the hypothalamus.Citation160 CRH administration rapidly reduced AEA levels in the amygdala but not 2-AG, and induced anxiety-like behavior and HPA activation, both of which were reversed by URB597.Citation161 Interestingly, the increase in FAAH activity induced by the administration of CRH was not accompanied by an increase in FAAH protein levels or mRNA levels, which may be related to dynamic changes in enzyme activity or consistent with the specific coupling of the CRHR1 and FAAH regions. Up-regulation of CRH was associated with CORT, with CRH mRNA significantly elevated in the rat PFC after CORT administration and dependent on CRHR1 signaling to regulate amygdala AEA content but not 2-AG.Citation162 Therefore, the relationship between CRH, AEA and HPA axis is not completely linear, indicating a potential crosstalk that requires further exploration.
Promotion of Neurogenesis and Plasticity
Impaired neurogenesis as well as altered neuroplasticity are commonly seen in anxiety disorders caused by chronic stress. Studies have shown that neural progenitor cell division is affected by eCB signalingCitation163 and enhanced neurogenesis is always associated with elevated eCBs.Citation164 Repeated injections of cannabidiol, an exogenous cannabinoid, attenuated anxiety-like behavior by inhibiting FAAH activity and also promoted hippocampal neurogenesis and dendritic remodeling.Citation165 Moreover, HIV-1 Gp120-mediated impaired neurogenesis was rescued by FAAH gene deletion.Citation166 URB597 increased neuroplasticity by modulating long-term potentiation in the hippocampal CA1 region and the amygdala, attenuating fear memory.Citation167 JNJ5003 significantly reversed chronic restraint stress-induced dendritic expansion and increased spine density in BLA, promoting synaptic remodeling and reducing anxiety-like behavior.Citation114,Citation168 URB532 and URB597 similarly prevented the reduction in AEA, the dendritic hypertrophy of the BLA, and the increase in anxiety-like behavior induced by stress.Citation168 The above studies illustrate that the use of FAAH inhibitors enhances brain neurogenesis as well as interneuronal transmission.
mTOR and neuropeptide Y (NPY) signaling may be associated with enhanced neurogenesis and neuroplasticity by FAAH inhibitors. mTOR signaling is essential for maintaining hippocampal neurogenesis and protecting against stress-induced impairment of neuroplasticity, and inhibition of this signaling increased anxiety-like behaviors in mice.Citation169 Recent studies have found that URB597 has an inverted U-shaped anxiolytic quantity-effect relationship in mice exposed to social defeat stress, and that its anxiolytic effect can be blocked by rapamycin (an mTOR antagonist), while its anxiogenic dose reduces the number of newborn neurons.Citation122 NPY is widely distributed in the nervous system and is particularly highly expressed in NAc and BLA.Citation170 Numerous studies have demonstrated that NPY levels are negatively correlated with anxiety,Citation171 which may be related to neuroprotective effects by promoting neurogenesis and neuroplasticity,Citation172 and decreasing amygdala excitability.Citation173 NPY has been reported to be involved in the neuroplastic protective effects of AEA, and inhibition of its expression antagonizes the effects of URB597 in suppressing PTSD behavior.Citation174
Novel FAAH Inhibitors and Natural Product Development
Unlike the urgent need to find reversible inhibitors of MAGL, irreversible FAAH inhibitors, such as URB597, PF3845, and PF04457845, do not exhibit significant toxic side effects and are well tolerated clinically. Therefore, the structural modification and optimization of FAAH inhibitors are more directed towards improving the drug-forming properties like solubility and central permeability for better bioavailability. ARN14633 and ARN14280 are novel analogues of URB597 with improved solubility and bioavailability effectively alleviating anxiety-like behavior in rats exposed to predator-evoked fear models.Citation175,Citation176 Structural modifications based on lead compounds, as well as extractions from natural products, are equally pivotal in the development of novel FAAH inhibitors.
Structural Modifications Based on Lead Compounds
In recent years, the development of novel FAAH inhibitors is mainly based on the skeletons of piperazine, isatin, oxazole and carbamate for structural modification and performance optimisation. Among them, piperazine-based FAAH inhibitors account for a relatively large proportion. Compound 4i (IC50= 0.12 μM) developed on the basis of indole-2-carbonyl piperazinourea derivatives possessed desirable antidepressant, analgesic and anti-inflammatory effects.Citation177 Among the piperazinourea derivatives with thiadiazole portion, the compounds with 4-chlorobenzyl (19) and 4-fluorobenzyl (20) tails on the piperazine side were found to be the most effective in inhibiting FAAH, with IC50 of 0.13 and 0.22 µM, respectively.Citation178 Heteroaryl ureas with a thickened bicyclic diamine core exhibited better FAAH inhibitory activity compared to compounds constructed with a piperazine core.Citation179
Jaiswal et al designed a series of isatin derivatives using the contemporary scaffold hopping approach in which compound 8c possessed antidepressant and anxiolytic effects without any neurotoxicityCitation180 and used the Dihydroindole-2,3-dione derivatives as lead compounds to further search for FAAH inhibitors with good pharmacological properties.Citation181 JZP327A (IC50 = 11 nM), synthesized using 1,3,4-oxadiazol-2-ones as a scaffold, is a highly selective, slowly reversible FAAH inhibitorCitation182 with good analgesic effects.Citation183 FAAH inhibitors developed on the basis of carbamates also exhibit good selectivity, reversibility, water solubility and some neuroprotective effects.Citation184
Three-dimensional quantitative structure-activity relationship (3D-QSAR) model, which can better represent the structure-activity relationship of ligand-enzyme interaction and help to develop more effective compounds, has been widely used in FAAH inhibitor development. Zięba et al constructed two 3D-QSAR models based on 31 FAAH inhibitors containing the 1,3,4-oxadiazol-2-one structure, which contribute to the design of novel, more potent, and more indicative FAAH inhibitors.Citation185 Lorca et al also constructed a similar model based on piperazine-carboxamide scaffold and designed 10 new compounds with highly predicted FAAH inhibitory activity.Citation186 Application of QSAR model and molecular docking technology to screen potential FAAH inhibitors in current clinical drugs also deserves attention. Montelukast, Repaglinide, Refenacin, Raloxifene and Buclizine are considered to have potential FAAH inhibitory activity, but further in vivo and in vitro validation is required.Citation187
Dual-Target FAAH Inhibitors
Dual-target FAAH inhibition increases disease specificity. For example, Ibu-AM68Citation188 and Flu-AM4Citation189 both have dual FAAH and COX-2 inhibition, which circumvents the gastrointestinal response to NSAIDs, and have anti-inflammatory or analgesic effects. Development of dual FAAH/ChE inhibitors targeting the neuroprotective effects of FAAH inhibitors as promising candidates for the treatment of Alzheimer’s disease.Citation190,Citation191 Dual FAAH/sEH inhibitors with the piperidinylsulfonamide portion as the pharmacophore have been shown to have good inhibitory effects on neuropathic pain and inflammation.Citation192 In addition to dual inhibition, inhibition/excitation can also exist simultaneously. UCM1341 inhibits FAAH while activating melatonin receptors, producing anti-inflammatory effects to provide neuroprotection.Citation193 Compounds designed to target FAAH inhibition/activation of CB2R also have potent neuroinflammatory inhibitory effects.Citation194
Furthermore, FAAH inhibitors also have central and peripheral inhibition selectivity. For example, ASP3652, which entered clinical trials, is a well-tolerated peripheral reversible FAAH inhibitor that reduces lower urinary tract symptoms but no efficacy in the improvement of patients’ pain symptoms, possibly related to its lack of central inhibition.Citation195 Surprisingly, URB937, a peripherally restricted FAAH inhibitor, was unable to cross the blood-brain barrier but also possessed central activities such as anxiolysisCitation128 and analgesia.Citation196 This result suggests the presence of peripheral and central crosstalk, with anxiolysis possibly related to sympathetic efferents and analgesia possibly related to reduced afferents for injurious pain, requiring further experimental verification.
Off-Target Effects of FAAH Inhibitors
However, although FAAH inhibitors show relatively excellent pharmacological activity, their off-target effects should not be overlooked. FAAH is a serine hydrolase that uses highly conserved serine residues in its active site as nucleophilic reagents to catalyse the hydrolysis of its substrate. Most FAAH inhibitors exert their inhibitory effects by binding and modifying catalytic serine residues, which can also inhibit other serine hydrolases.Citation197 For example, URB597, BMS-1, OL-135 and LY2077855 all have low selectivity and show a variety of off-target effects, the main off-target being carboxylesterases.Citation197 Unfortunately, the off-target effects of FAAH inhibitors have been disregarded and even carried over into clinical trials, resulting in a tragic lesson. BIA 10–2474, an irreversible FAAH inhibitor entered Phase I clinical trials in 2016, was urgently called off due to resulting in the death of one volunteer and mild to severe neurological symptoms in four volunteers.Citation198 Subsequent studies showed that the cause of the clinical incident of BIA 10–2474 may be that its off-target protein PNPLA6 is strongly associated with organophosphorus neurotoxicity.Citation199 Another irreversible FAAH inhibitor, PF04457845, has entered a Phase 2 trial with no serious adverse events.Citation120 Therefore, the off-target effects must be evaluated to ensure clinical safety when developing novel FAAH inhibitors.
As mentioned above, ABPP technology can also be used to screen FAAH enzyme inhibitors. Otrubova et al assessed the FAAH inhibition performance of a series of N-acyl pyrazole derivatives by ABPP, minimizing off-target activity.Citation200 Lamani et al demonstrated that FAAH inhibitors 9 and 31 were highly selective for brain FAAH and protective against kainic acid-induced excitotoxicity by ABPP method.Citation201 JZP-327A, a slowly reversible FAAH inhibitor with over 900-fold selectivity for MAGL and COX isozymes, was shown by ABPP to have higher FAAH selectivity compared to other serine hydrolases.Citation182 Generally, evaluating the potential pharmacological activity of novel inhibitors by ABPP technology enables timely circumvention of off-target effects and minimization of compound side effects.
Natural Product Development
Various active ingredients with FAAH inhibitory activity were also found in natural products (). Among them, flavonoids inhibited the activity of FAAH significantly. Daidzein, silybin and chickpea bractein A showed high FAAH inhibitory activity both in vivo and in vitro.Citation202 Kaempferol inhibited FAAH activity in a concentration-dependent manner in vitro, while in vivo experiments further confirmed that acting on FAAH reduced anxiety-like behavior.Citation203 In addition, isoflavonoids are thought to target the ECS by modulating eCB metabolism. Three isoflavonoids, 7-hydroxyflavone, biochanin-A, and genistein, all bind to the FAAH active site and dose-dependently inhibit FAAH activity, increasing AEA levels in the PFC and decreasing blood corticosterone concentrations.Citation204
Table 4 Summary of FAAH Inhibitors Derived from Natural Products
Terpenoids and phenolic compounds also showed positive FAAH inhibitory activity. All 17 triterpenoids isolated from Ganoderma lucidum exhibited some inhibitory activity against FAAH without cytotoxicity, and FAAH may be a potential target for anti-neuroinflammation.Citation209,Citation220 β-stigmasterol and eugenol extracted from Harpagophytum procumbens, a sesquiterpene and monopostane constituent, respectively, exerted an anti-arthritic effects by inhibiting FAAH expression,Citation214 with β-stigmasterol also being a selective CB2R agonist.Citation221,Citation222 Some phenolic compounds, such as cannabidiol extracted from Cannabis sativa L. and 5′-methoxylicarin A extracted from Myristica fragrans Houtt., have also been reported to inhibit FAAH activity and have anxiolytic efficacy.Citation216
Some other types of natural products also have FAAH inhibition effects. Macamides, a unique series of non-polar long-chain fatty acids N-benzamide isolated from Maca, are mainly alkaloids with neuroprotective properties,Citation223 of which N-Benzyloctadeca-9Z,12Z-dienamide exhibits the best FAAH inhibitory activity and attenuates ischaemic stroke injury,Citation224 but this inhibition is an irreversible inhibitor that exhibits time-dependent inhibition.Citation205 Lavender essential oil, an over-The-counter herbal medicine approved by the European Medicines Agency for the relief of anxiety, displayed FAAH inhibitory activity in vitro and produced effects comparable to diazepam in mice.Citation217 In addition, Xiaoyao Pills, a proprietary Chinese medicine widely used in China for the treatment of depression, was shown to alleviate depression-like behavior in rats by inhibiting FAAH levels in the brain.Citation225
Techniques such as fluorescent probes and molecular docking have been used in recent years to screen potential FAAH inhibitors from natural products. Ginkgolide, the main active ingredient in Ginkgo biloba, was confirmed to bind well to FAAH by molecular docking.Citation226 The aqueous extracts and fatty oils of Platycladi Semen are also predicted to exert anxiolytic effects through FAAH.Citation227 The FAAH-activated fluorescent probe named THPO developed by Tian et al identified a natural inhibitor, neobavaisoflavone, from 68 traditional herbs.Citation228 Similarly, DAND, a FAAH-activated near-infrared fluorescent probe, screened piperine as a novel inhibitor of FAAH and presented excellent anti-inflammatory activity in the vitro experiments.Citation229
Summary and Prospect
The high prevalence of anxiety disorders and socio-economic burden highlights the pressing need for effective treatments, as current options remain limited. Although named with a plant possessing psychoactive properties, ECS is actually one of the crucial regulatory systems of the organism. In recent years, the idea of targeting the ECS for the treatment of anxiety disorders has received increasing attention. Inhibition of hydrolase activity, particularly targeting the dominant enzymes MAGL and FAAH, offers a promising avenue by indirectly increasing eCB levels, thereby avoiding the addictive properties and adverse effects linked to direct exogenous cannabinoid supplementation.
The anxiolytic efficacy of FAAH and MAGL inhibitors has been supported by both clinical and preclinical studies, indicating their potential as promising treatments for anxiety disorders. The pharmacological mechanisms of these inhibitors are primarily associated with maintaining Glutamate/GABA balance, suppressing neuroinflammation, modulating the HPA axis, and promoting neurogenesis. Given the crucial role of neuronal excitatory/inhibitory (E/I) imbalance in the pathology of anxiety disorders, the maintenance of Glutamate/GABA homeostasis is particularly vital. CB1R, the primary mediator of the biological effects of 2-AG and AEA, is extensively expressed in glutamatergic and GABAergic terminals, offering the potential to reverse this imbalance. The clarification of the above mechanisms is not only crucial for drug development and clinical application of endocannabinoid hydrolase inhibitors, but also aids in achieving precision treatment for anxiety disorders. Similarly, more research is necessary to support or further clarify the precise mechanisms of FAAH and MAGL inhibitors in treating anxiety disorders.
Numerous biochemical techniques, including ABPP, fluorescence probes, molecular docking, and 3D-QSAR, have been used to generate novel MAGL and FAAH inhibitors. These techniques have greatly improved the screening efficiency and facilitated the discovery of new compounds. Notably, most of these new compounds are mainly developed for anticancer, analgesic, and anti-inflammatory purposes, and their potential anxiolytic activity remains to be further evaluated. Meanwhile, while significantly reducing the adverse effects associated with direct supplementation of cannabinoids or activation of cannabinoid receptors, the potential cardiotoxicity of some MAGL inhibitors and off-target effects of FAAH inhibitors should not be overlooked. In addition, given the polymorphism in the FAAH C385A gene, the genotype of patients must be thoroughly considered when applying FAAH inhibitors for treatment to achieve precision medicine. In contrast, some natural products isolated from plants have been shown to exert anxiolytic activity by inhibiting FAAH or MAGL, mostly reversibly, which greatly improves the tolerability and safety of the drugs, and thus active compounds derived from natural products would be a good source of novel anxiolytic drugs.
The development of dual MAGL/FAAH inhibitors has also come into the limelight. The newly developed AKU-005 significantly enhanced 2-AG and AEA levels and inhibited neural excitability in rat and human meninges, which is expected to be a new treatment for migraine.Citation230 JZL195, a classical dual MAGL/FAAH inhibitor, embodies an antidepressant activity,Citation231 potent neuroleptic activity,Citation232 weak antihypertensive effectCitation233 and anti-inflammatory activity.Citation234 Regrettably, the anxiolytic effects of JZL195 remain a subject of controversy. Some studies suggested an improvement in anxiety-like behavior in EPM test in mice.Citation35 On the contrary, other research have shown that it fails to reverse restraint stress-induced anxiety-like behaviors or even promotes anxiety-like behaviors.Citation37,Citation235 These discrepancies may stem from variations in experimental conditions and animal models. However, it’s crucial to acknowledge that the potential anxiolytic effects of MAGL/FAAH dual inhibition cannot be conclusively denied.
Furthermore, consistent with the inhibition of hydrolase activity, the levels of synaptic interstitial 2-AG and AEA can also be increased by inhibiting the activity of eCB transporter proteins, and intracellular transport is necessary for eCB hydrolysis. Current studies focus on AEA transporter proteins, which mainly include FABP5, HSP70, and FLAT1.Citation236,Citation237 Recent studies have found that the use of a FABP5 inhibitor, SBFI-103, in either the amygdala or the PFC produces significant anxiolytic effects.Citation238 WOBE437, a natural product-derived inhibitor of AEA reuptake, irreversibly blocks 2-AG, AEA membrane transport, producing anxiolytic effects.Citation239 Similarly, AM404, also an AEA reuptake inhibitor, has been reported to exhibit reliable anxiolytic activity.Citation240,Citation241 Although the above studies demonstrated the anxiolytic promise of targeted transporter proteins, it should not be neglected that they are still in their infancy, and need to be confirmed by further experimental studies.
In summary, there exists great potential to develop a clinically effective, safe, and well-tolerated novel anxiolytic drug from MAGL and FAAH inhibitors compared to direct cannabinoid receptor agonists. This can be achieved by screening and synthesizing new hydrolase inhibitors using advanced technologies, or by exploring active ingredients with robust inhibitory activity from natural products. Structural optimization and modification may further enhance the selectivity and biological activity of these potential drugs.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no conflicts of interest in this work.
Additional information
Funding
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®). American Psychiatric Publisher; 2013.
- Szuhany KL, Simon NM. Anxiety disorders: a review. JAMA. 2022;328(24):2431–2445. doi:10.1001/jama.2022.22744
- Huang Y, Wang Y, Wang H, et al. Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2019;6(3):211–224. doi:10.1016/S2215-0366(18)30511-X
- Inserra A, Piot A, De Gregorio D, Gobbi G. Lysergic Acid Diethylamide (LSD) for the treatment of anxiety disorders: preclinical and clinical evidence. CNS Drugs. 2023;37(9):733–754. doi:10.1007/s40263-023-01008-5
- Skolnick P. Anxioselective anxiolytics: on a quest for the Holy Grail. Trends Pharmacol Sci. 2012;33(11):611–620. doi:10.1016/j.tips.2012.08.003
- Gosmann NP, de Abreu Costa M, de Barros Jaeger M, et al. Selective serotonin reuptake inhibitors, and serotonin and norepinephrine reuptake inhibitors for anxiety, obsessive-compulsive, and stress disorders: a 3-level network meta-analysis. PLoS Med. 2021;18(6):e1003664. doi:10.1371/journal.pmed.1003664
- Murphy SE, Capitão LP, Giles SLC, Cowen PJ, Stringaris A, Harmer CJ. The knowns and unknowns of SSRI treatment in young people with depression and anxiety: efficacy, predictors, and mechanisms of action. Lancet Psychiatry. 2021;8(9):824–835. doi:10.1016/S2215-0366(21)00154-1
- Gosmann NP, de Abreu Costa M, de Barros Jaeger M, et al. Incidence of adverse events and comparative tolerability of selective serotonin reuptake inhibitors, and serotonin and norepinephrine reuptake inhibitors for the treatment of anxiety, obsessive-compulsive, and stress disorders: a systematic review and network meta-analysis. Psychol Med. 2023;53(9):3783–3792. doi:10.1017/S0033291723001630
- Joffe H, Petrillo L, Viguera A, et al. Eszopiclone improves insomnia and depressive and anxious symptoms in perimenopausal and postmenopausal women with hot flashes: a randomized, double-blinded, placebo-controlled crossover trial. Am J Obstet Gynecol. 2010;202(2):171.e1–171.e11. doi:10.1016/j.ajog.2009.10.868
- Mokhber N, Azarpazhooh MR, Khajehdaluee M, Velayati A, Hopwood M. Randomized, single-blind, trial of sertraline and buspirone for treatment of elderly patients with generalized anxiety disorder. Psychiatry Clin Neurosci. 2010;64(2):128–133. doi:10.1111/j.1440-1819.2009.02055.x
- Lowe H, Toyang N, Steele B, Bryant J, Ngwa W. The endocannabinoid system: a potential target for the treatment of various diseases. Int J Mol Sci. 2021;22(17):9472. doi:10.3390/ijms22179472
- Alcaraz-Silva J, Feingold D, Viana-Torre G, et al. The endocannabinoid system as a biomarker for diagnostic and therapeutic applications in depression and anxiety. CNS Neurol Disord Drug Targets. 2023;22(3):417–430. doi:10.2174/1871527321666220405114402
- Schmidt ME, Liebowitz MR, Stein MB, et al. The effects of inhibition of fatty acid amide hydrolase (FAAH) by JNJ-42165279 in social anxiety disorder: a double-blind, randomized, placebo-controlled proof-of-concept study. Neuropsychopharmacology. 2021;46(5):1004–1010. doi:10.1038/s41386-020-00888-1
- Cristino L, Bisogno T, Di Marzo V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat Rev Neurol. 2020;16(1):9–29. doi:10.1038/s41582-019-0284-z
- Murkar A, De Koninck J, Merali Z. Cannabinoids: revealing their complexity and role in central networks of fear and anxiety. Neurosci Biobehav Rev. 2021;131:30–46. doi:10.1016/j.neubiorev.2021.09.002
- Kaczocha M, Haj-Dahmane S. Mechanisms of endocannabinoid transport in the brain. Br J Pharmacol. 2022;179(17):4300–4310. doi:10.1111/bph.15469
- Petrie GN, Nastase AS, Aukema RJ, Hill MN. Endocannabinoids, cannabinoids and the regulation of anxiety. Neuropharmacology. 2021;195:108626. doi:10.1016/j.neuropharm.2021.108626
- Morgan AJ, Kingsley PJ, Mitchener MM, et al. Detection of cyclooxygenase-2-derived oxygenation products of the endogenous cannabinoid 2-arachidonoylglycerol in mouse brain. ACS Chem Neurosci. 2018;9(7):1552–1559. doi:10.1021/acschemneuro.7b00499
- Patel S, Hill MN, Cheer JF, Wotjak CT, Holmes A. The endocannabinoid system as a target for novel anxiolytic drugs. Neurosci Biobehav Rev. 2017;76(Pt A):56–66. doi:10.1016/j.neubiorev.2016.12.033
- Maldonado R, Cabañero D, Martín-García E. The endocannabinoid system in modulating fear, anxiety, and stress. Dialogues Clin Neurosci. 2020;22(3):229–239. doi:10.31887/DCNS.2020.22.3/rmaldonado
- Lama A, Pirozzi C, Severi I, et al. Palmitoylethanolamide dampens neuroinflammation and anxiety-like behavior in obese mice. Brain Behav Immun. 2022;102:110–123. doi:10.1016/j.bbi.2022.02.008
- Fraguas-Sánchez AI, Torres-Suárez AI. Medical Use of Cannabinoids. Drugs. 2018;78(16):1665–1703. doi:10.1007/s40265-018-0996-1
- Cohen K, Weizman A, Weinstein A. Positive and negative effects of cannabis and cannabinoids on health. Clin Pharmacol Ther. 2019;105(5):1139–1147. doi:10.1002/cpt.1381
- Stasiulewicz A, Znajdek K, Grudzień M, Pawiński T, Sulkowska AJI. A Guide to Targeting the Endocannabinoid System in Drug Design. Int J Mol Sci. 2020;21(8):2778. doi:10.3390/ijms21082778
- Gipson CD, Kupchik YM, Kalivas PW. Rapid, transient synaptic plasticity in addiction. Neuropharmacology. 2014;76(Pt B):276–286. doi:10.1016/j.neuropharm.2013.04.032
- Hill MN, Bierer LM, Makotkine I, et al. Reductions in circulating endocannabinoid levels in individuals with post-traumatic stress disorder following exposure to the World Trade Center attacks. Psychoneuroendocrinology. 2013;38(12):2952–2961. doi:10.1016/j.psyneuen.2013.08.004
- Siebers M, Biedermann SV, Bindila L, Lutz B, Fuss J. Exercise-induced euphoria and anxiolysis do not depend on endogenous opioids in humans. Psychoneuroendocrinology. 2021;126:105173. doi:10.1016/j.psyneuen.2021.105173
- Shonesy BC, Bluett RJ, Ramikie TS, et al. Genetic disruption of 2-arachidonoylglycerol synthesis reveals a key role for endocannabinoid signaling in anxiety modulation. Cell Rep. 2014;9(5):1644–1653. doi:10.1016/j.celrep.2014.11.001
- Jenniches I, Ternes S, Albayram O, et al. Anxiety, stress, and fear response in mice with reduced endocannabinoid levels. Biol Psychiatry. 2016;79(10):858–868. doi:10.1016/j.biopsych.2015.03.033
- Sánchez-Marín L, Flores-López M, Pastor A, et al. Acute stress and alcohol exposure during adolescence result in an anxious phenotype in adulthood: role of altered glutamate/endocannabinoid transmission mechanisms. Prog Neuropsychopharmacol Biol Psychiatry. 2022;113:110460. doi:10.1016/j.pnpbp.2021.110460
- Hill MN, Haney M, Hillard CJ, Karhson DS, Vecchiarelli HA. The endocannabinoid system as a putative target for the development of novel drugs for the treatment of psychiatric illnesses. Psychol Med. 2023;53(15):7006–7024. doi:10.1017/S0033291723002465
- Marcus DJ, Bedse G, Gaulden AD, et al. Endocannabinoid signaling collapse mediates stress-induced amygdalo-cortical strengthening. Neuron. 2020;105(6):1062–1076.e6. doi:10.1016/j.neuron.2019.12.024
- Guggenhuber S, Romo-Parra H, Bindila L, et al. Impaired 2-AG signaling in hippocampal glutamatergic neurons: aggravation of anxiety-like behavior and unaltered seizure susceptibility. Int J Neuropsychopharmacol. 2015;19(2):pyv091. doi:10.1093/ijnp/pyv091
- Serrano A, Pavon FJ, Buczynski MW, et al. Deficient endocannabinoid signaling in the central amygdala contributes to alcohol dependence-related anxiety-like behavior and excessive alcohol intake. Neuropsychopharmacology. 2018;43(9):1840–1850. doi:10.1038/s41386-018-0055-3
- Pavón FJ, Polis IY, Stouffer DG, et al. Selective inhibition of monoacylglycerol lipase is associated with passive coping behavior and attenuation of stress-induced dopamine release in the medial prefrontal cortex. Neurobiol Stress. 2021;14:100293. doi:10.1016/j.ynstr.2021.100293
- Alteba S, Mizrachi Zer-Aviv T, Tenenhaus A, et al. Antidepressant-like effects of URB597 and JZL184 in male and female rats exposed to early life stress. Eur Neuropsychopharmacol. 2020;39:70–86. doi:10.1016/j.euroneuro.2020.08.005
- Bedse G, Bluett RJ, Patrick TA, et al. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: comparative profiling of FAAH, MAGL and dual inhibitors. Transl Psychiatry. 2018;8(1):92. doi:10.1038/s41398-018-0141-7
- Bedse G, Hartley ND, Neale E, et al. Functional redundancy between canonical endocannabinoid signaling systems in the modulation of anxiety. Biol Psychiatry. 2017;82(7):488–499. doi:10.1016/j.biopsych.2017.03.002
- Bosch-Bouju C, Larrieu T, Linders L, Manzoni OJ, Layé S. Endocannabinoid-mediated plasticity in nucleus accumbens controls vulnerability to anxiety after social defeat stress. Cell Rep. 2016;16(5):1237–1242. doi:10.1016/j.celrep.2016.06.082
- Lomazzo E, Bindila L, Remmers F, et al. Therapeutic potential of inhibitors of endocannabinoid degradation for the treatment of stress-related hyperalgesia in an animal model of chronic pain. Neuropsychopharmacology. 2015;40(2):488–501. doi:10.1038/npp.2014.198
- Sumislawski JJ, Ramikie TS, Patel S. Reversible gating of endocannabinoid plasticity in the amygdala by chronic stress: a potential role for monoacylglycerol lipase inhibition in the prevention of stress-induced behavioral adaptation. Neuropsychopharmacology. 2011;36(13):2750–2761. doi:10.1038/npp.2011.166
- Kinsey SG, O’Neal ST, Long JZ, Cravatt BF, Lichtman AH. Inhibition of endocannabinoid catabolic enzymes elicits anxiolytic-like effects in the marble burying assay. Pharmacol Biochem Behav. 2011;98(1):21–27. doi:10.1016/j.pbb.2010.12.002
- Worley NB, Varela JA, Gaillardetz GP, Hill MN, Christianson JP. Monoacylglycerol lipase alpha inhibition alters prefrontal cortex excitability and blunts the consequences of traumatic stress in rat. Neuropharmacology. 2020;166:107964. doi:10.1016/j.neuropharm.2020.107964
- Vozella V, Cruz B, Feldman HC, et al. Sexually dimorphic effects of monoacylglycerol lipase inhibitor MJN110 on stress-related behavior and drinking in Marchigian Sardinian alcohol-preferring rats. Br J Pharmacol. 2023;180(24):3130–3145. doi:10.1111/bph.16197
- Dlugos A, Childs E, Stuhr KL, Hillard CJ, de Wit H. Acute stress increases circulating anandamide and other N-acylethanolamines in healthy humans. Neuropsychopharmacology. 2012;37(11):2416–2427. doi:10.1038/npp.2012.100
- Zhai Q, Islam A, Chen B, et al. Endocannabinoid 2-arachidonoylglycerol levels in the anterior cingulate cortex, caudate putamen, nucleus accumbens, and piriform cortex were upregulated by chronic restraint stress. Cells. 2023;12(3):393. doi:10.3390/cells12030393
- Di S, Itoga CA, Fisher MO, et al. Acute stress suppresses synaptic inhibition and increases anxiety via endocannabinoid release in the basolateral amygdala. J Neurosci. 2016;36(32):8461–8470. doi:10.1523/JNEUROSCI.2279-15.2016
- Imperatore R, Morello G, Luongo L, et al. Genetic deletion of monoacylglycerol lipase leads to impaired cannabinoid receptor CB₁R signaling and anxiety-like behavior. J Neurochem. 2015;135(4):799–813. doi:10.1111/jnc.13267
- Schlosburg JE, Blankman JL, Long JZ, et al. Chronic monoacylglycerol lipase blockade causes functional antagonism of the endocannabinoid system. Nat Neurosci. 2010;13(9):1113–1119. doi:10.1038/nn.2616
- Kenwood MM, Kalin NH, Barbas H. The prefrontal cortex, pathological anxiety, and anxiety disorders. Neuropsychopharmacology. 2022;47(1):260–275. doi:10.1038/s41386-021-01109-z
- Liu WZ, Zhang WH, Zheng ZH, et al. Identification of a prefrontal cortex-to-amygdala pathway for chronic stress-induced anxiety. Nat Commun. 2020;11(1):2221. doi:10.1038/s41467-020-15920-7
- Domoto M, Sasase H, Wada S, et al. The synthetic cannabinoid 5F-AMB changes the balance between excitation and inhibition of layer V pyramidal neurons in the mouse medial prefrontal cortex. Psychopharmacology. 2018;235(8):2367–2376. doi:10.1007/s00213-018-4933-5
- Zhang WH, Zhang JY, Holmes A, Pan BX. Amygdala Circuit Substrates for Stress Adaptation and Adversity. Biol Psychiatry. 2021;89(9):847–856. doi:10.1016/j.biopsych.2020.12.026
- Jiang S, Zheng C, Wen G, Bu B, Zhao S, Xu X. Down-regulation of NR2B receptors contributes to the analgesic and antianxiety effects of enriched environment mediated by endocannabinoid system in the inflammatory pain mice. Behav Brain Res. 2022;435:114062. doi:10.1016/j.bbr.2022.114062
- Longaretti A, Forastieri C, Gabaglio M, Rubino T, Battaglioli E, Rusconi F. Termination of acute stress response by the endocannabinoid system is regulated through lysine-specific demethylase 1-mediated transcriptional repression of 2-AG hydrolases ABHD6 and MAGL. J Neurochem. 2020;155(1):98–110. doi:10.1111/jnc.15000
- Vickstrom CR, Liu X, Liu S, et al. Role of endocannabinoid signaling in a septohabenular pathway in the regulation of anxiety- and depressive-like behavior. Mol Psychiatry. 2021;26(7):3178–3191. doi:10.1038/s41380-020-00905-1
- Biltz RG, Sawicki CM, Sheridan JF, Godbout JP. The neuroimmunology of social-stress-induced sensitization. Nat Immunol. 2022;23(11):1527–1535. doi:10.1038/s41590-022-01321-z
- Pasquarelli N, Porazik C, Hanselmann J, Weydt P, Ferger B, Witting A. Comparative biochemical characterization of the monoacylglycerol lipase inhibitor KML29 in brain, spinal cord, liver, spleen, fat and muscle tissue. Neuropharmacology. 2015;91:148–156. doi:10.1016/j.neuropharm.2014.12.001
- Wenzel TJ, Klegeris A. Novel multi-target directed ligand-based strategies for reducing neuroinflammation in Alzheimer’s disease. Life Sci. 2018;207:314–322. doi:10.1016/j.lfs.2018.06.025
- Kerr DM, Harhen B, Okine BN, Egan LJ, Finn DP, Roche M. The monoacylglycerol lipase inhibitor JZL184 attenuates LPS-induced increases in cytokine expression in the rat frontal cortex and plasma: differential mechanisms of action. Br J Pharmacol. 2013;169(4):808–819. doi:10.1111/j.1476-5381.2012.02237.x
- Zhu D, Zhang J, Hashem J, Gao F, Chen C. Inhibition of 2-arachidonoylglycerol degradation enhances glial immunity by single-cell transcriptomic analysis. J Neuroinflammation. 2023;20(1):17. doi:10.1186/s12974-023-02701-4
- Grabner GF, Eichmann TO, Wagner B, et al. Deletion of monoglyceride lipase in astrocytes attenuates lipopolysaccharide-induced neuroinflammation. J Biol Chem. 2016;291(2):913–923. doi:10.1074/jbc.M115.683615
- Nomura DK, Morrison BE, Blankman JL, et al. Endocannabinoid hydrolysis generates brain prostaglandins that promote neuroinflammation. Science. 2011;334(6057):809–813. doi:10.1126/science.1209200
- Alhouayek M, Masquelier J, Cani PD, Lambert DM, Muccioli GG. Implication of the anti-inflammatory bioactive lipid prostaglandin D2-glycerol ester in the control of macrophage activation and inflammation by ABHD6. Proc Natl Acad Sci U S A. 2013;110(43):17558–17563. doi:10.1073/pnas.1314017110
- Kibret BG, Ishiguro H, Horiuchi Y, Onaivi ES. New insights and potential therapeutic targeting of CB2 cannabinoid receptors in CNS disorders. Int J Mol Sci. 2022;23(2):975. doi:10.3390/ijms23020975
- Li J, Wang H, Liu D, et al. CB2R activation ameliorates late adolescent chronic alcohol exposure-induced anxiety-like behaviors during withdrawal by preventing morphological changes and suppressing NLRP3 inflammasome activation in prefrontal cortex microglia in mice. Brain Behav Immun. 2023;110:60–79. doi:10.1016/j.bbi.2023.02.001
- Mecha M, Feliú A, Carrillo-Salinas FJ, et al. Endocannabinoids drive the acquisition of an alternative phenotype in microglia. Brain Behav Immun. 2015;49:233–245. doi:10.1016/j.bbi.2015.06.002
- Young AP, Denovan-Wright EM. The dynamic role of microglia and the endocannabinoid system in neuroinflammation. Front Pharmacol. 2021;12:806417. doi:10.3389/fphar.2021.806417
- Roberts CJ, Stuhr KL, Hutz MJ, Raff H, Hillard CJ. Endocannabinoid signaling in hypothalamic-pituitary-adrenocortical axis recovery following stress: effects of indirect agonists and comparison of male and female mice. Pharmacol Biochem Behav. 2014;117:17–24. doi:10.1016/j.pbb.2013.11.026
- Aliczki M, Zelena D, Mikics E, et al. Monoacylglycerol lipase inhibition-induced changes in plasma corticosterone levels, anxiety and locomotor activity in male CD1 mice. Horm Behav. 2013;63(5):752–758. doi:10.1016/j.yhbeh.2013.03.017
- Hill MN, McLaughlin RJ, Pan B, et al. Recruitment of prefrontal cortical endocannabinoid signaling by glucocorticoids contributes to termination of the stress response. J Neurosci. 2011;31(29):10506–10515. doi:10.1523/JNEUROSCI.0496-11.2011
- Long JZ, Li W, Booker L, et al. Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat Chem Biol. 2009;5(1):37–44. doi:10.1038/nchembio.129
- Yamasaki T, Hatori A, Zhang Y, et al. Neuroprotective effects of minocycline and KML29, a potent inhibitor of monoacylglycerol lipase, in an experimental stroke model: a small-animal positron emission tomography study. Theranostics. 2021;11(19):9492–9502. doi:10.7150/thno.64320
- Della Pietra A, Giniatullin R, Savinainen JR. Distinct activity of endocannabinoid-hydrolyzing enzymes MAGL and FAAH in key regions of peripheral and central nervous system implicated in migraine. Int J Mol Sci. 2021;22(3):1204. doi:10.3390/ijms22031204
- Chanda PK, Gao Y, Mark L, et al. Monoacylglycerol lipase activity is a critical modulator of the tone and integrity of the endocannabinoid system. Mol Pharmacol. 2010;78(6):996–1003. doi:10.1124/mol.110.068304
- Aaltonen N, Kedzierska E, Orzelska-Górka J, et al. In Vivo Characterization of the Ultrapotent Monoacylglycerol Lipase Inhibitor {4-[bis-(benzo[d][1,3]dioxol-5-yl)methyl]-piperidin-1-yl}(1H-1,2,4-triazol-1-yl)methanone (JJKK-048). J Pharmacol Exp Ther. 2016;359(1):62–72. doi:10.1124/jpet.116.233114
- Aida J, Fushimi M, Kusumoto T, et al. Design, synthesis, and evaluation of piperazinyl pyrrolidin-2-ones as a novel series of reversible monoacylglycerol lipase inhibitors. J Med Chem. 2018;61(20):9205–9217. doi:10.1021/acs.jmedchem.8b00824
- Xiang H, Wang Y, Yang L, et al. Novel MAGL inhibitors alleviate LPS-induced acute kidney injury by inhibiting NLRP3 inflammatory vesicles, modulating intestinal flora, repairing the intestinal barrier, and interfering with serum metabolism. Molecules. 2023;28(21):7245. doi:10.3390/molecules28217245
- Afzal O, Kumar S, Kumar R, Firoz A, Jaggi M, Bawa S. Docking based virtual screening and molecular dynamics study to identify potential monoacylglycerol lipase inhibitors. Bioorg Med Chem Lett. 2014;24(16):3986–3996. doi:10.1016/j.bmcl.2014.06.029
- Xiong F, Ding X, Zhang H, et al. Discovery of novel reversible monoacylglycerol lipase inhibitors via docking-based virtual screening. Bioorg Med Chem Lett. 2021;41:127986. doi:10.1016/j.bmcl.2021.127986
- Tuccinardi T, Granchi C, Rizzolio F, et al. Identification and characterization of a new reversible MAGL inhibitor. Bioorg Med Chem. 2014;22(13):3285–3291. doi:10.1016/j.bmc.2014.04.057
- Zhi Z, Zhang W, Yao J, et al. Discovery of Aryl formyl piperidine derivatives as potent, reversible, and selective monoacylglycerol lipase inhibitors. J Med Chem. 2020;63(11):5783–5796. doi:10.1021/acs.jmedchem.9b02137
- Bononi G, Di Stefano M, Poli G, et al. Reversible monoacylglycerol lipase inhibitors: discovery of a new class of benzylpiperidine derivatives. J Med Chem. 2022;65(10):7118–7140. doi:10.1021/acs.jmedchem.1c01806
- Di Stefano M, Masoni S, Bononi G, et al. Design, synthesis, ADME and biological evaluation of benzylpiperidine and benzylpiperazine derivatives as novel reversible monoacylglycerol lipase (MAGL) inhibitors. Eur J Med Chem. 2024;263:115916. doi:10.1016/j.ejmech.2023.115916
- Bononi G, Tonarini G, Poli G, et al. Monoacylglycerol lipase (MAGL) inhibitors based on a diphenylsulfide-benzoylpiperidine scaffold. Eur J Med Chem. 2021;223:113679. doi:10.1016/j.ejmech.2021.113679
- Kapanda CN, Muccioli GG, Labar G, Poupaert JH, Lambert DM. Bis(dialkylaminethiocarbonyl)disulfides as potent and selective monoglyceride lipase inhibitors. J Med Chem. 2009;52(22):7310–7314. doi:10.1021/jm901323s
- Omran Z. New Disulfiram Derivatives as MAGL-Selective Inhibitors. Molecules. 2021;26(11):3296. doi:10.3390/molecules26113296
- Andrei C, Mihai DP, Nitulescu G, et al. Cetirizine and levetiracetam as inhibitors of monoacylglycerol lipase: investigating their repurposing potential as novel osteoarthritic pain therapies. Pharmaceuticals. 2023;16(11):1563. doi:10.3390/ph16111563
- Jiang M, Huizenga MCW, Wirt JL, et al. A monoacylglycerol lipase inhibitor showing therapeutic efficacy in mice without central side effects or dependence. Nat Commun. 2023;14(1):8039. doi:10.1038/s41467-023-43606-3
- Adeel M, Saorin G, Boccalon G, et al. A carrier free delivery system of a monoacylglycerol lipase hydrophobic inhibitor. Int J Pharm. 2022;613:121374. doi:10.1016/j.ijpharm.2021.121374
- Deng H, Li W. Monoacylglycerol lipase inhibitors: modulators for lipid metabolism in cancer malignancy, neurological and metabolic disorders. Acta Pharm Sin B. 2020;10(4):582–602. doi:10.1016/j.apsb.2019.10.006
- Cisar JS, Weber OD, Clapper JR, et al. Identification of ABX-1431, a selective inhibitor of monoacylglycerol lipase and clinical candidate for treatment of neurological disorders. J Med Chem. 2018;61(20):9062–9084. doi:10.1021/acs.jmedchem.8b00951
- Deng H, Zhang Q, Lei Q, et al. Discovering monoacylglycerol lipase inhibitors by a combination of fluorogenic substrate assay and activity-based protein profiling. Front Pharmacol. 2022;13:941522. doi:10.3389/fphar.2022.941522
- Punt JM, van der Vliet D, van der Stelt M. Chemical probes to control and visualize lipid metabolism in the brain. Acc Chem Res. 2022;55(22):3205–3217. doi:10.1021/acs.accounts.2c00521
- Takahata K, Seki C, Kimura Y, et al. First-in-human in vivo imaging and quantification of monoacylglycerol lipase in the brain: a PET study with 18F-T-401. Eur J Nucl Med Mol Imaging. 2022;49(9):3150–3161. doi:10.1007/s00259-021-05671-y
- He Y, Grether U, Taddio MF, et al. Multi-parameter optimization: development of a morpholin-3-one derivative with an improved kinetic profile for imaging monoacylglycerol lipase in the brain. Eur J Med Chem. 2022;243:114750. doi:10.1016/j.ejmech.2022.114750
- Cheng R, Fujinaga M, Yang J, et al. A novel monoacylglycerol lipase-targeted 18F-labeled probe for positron emission tomography imaging of brown adipose tissue in the energy network. Acta Pharmacol Sin. 2022;43(11):3002–3010. doi:10.1038/s41401-022-00912-8
- King AR, Dotsey EY, Lodola A, et al. Discovery of potent and reversible monoacylglycerol lipase inhibitors. Chem Biol. 2009;16(10):1045–1052. doi:10.1016/j.chembiol.2009.09.012
- Al-Romaiyan A, Masocha W. Pristimerin, a triterpene that inhibits monoacylglycerol lipase activity, prevents the development of paclitaxel-induced allodynia in mice. Front Pharmacol. 2022;13:944502. doi:10.3389/fphar.2022.944502
- Merali Z, Cayer C, Kent P, et al. Sacred Maya incense, copal (Protium copal - Burseraceae), has antianxiety effects in animal models. J Ethnopharmacol. 2018;216:63–70. doi:10.1016/j.jep.2018.01.027
- Tung M-C, Fung K-M, Hsu H-M, Tseng T-S. Discovery of 8-prenylnaringenin from hop (Humulus lupulus L.) as a potent monoacylglycerol lipase inhibitor for treatments of neuroinflammation and Alzheimer’s disease. RSC Adv. 2021;11(49):31062–31072. doi:10.1039/d1ra05311f
- El-Alfy AT, Joseph S, Brahmbhatt A, Akati S, Abourashed EA. Indirect modulation of the endocannabinoid system by specific fractions of nutmeg total extract. Pharm Biol. 2016;54(12):2933–2938. doi:10.1080/13880209.2016.1194864
- Benkherouf AY, Soini SL, Stompor M, Uusi-Oukari M. Positive allosteric modulation of native and recombinant GABAA receptors by hops prenylflavonoids. Eur J Pharmacol. 2019;852:34–41. doi:10.1016/j.ejphar.2019.02.034
- Beladjila KA, Berrehal D, De Tommasi N, et al. New phenylethanoid glycosides from cistanche phelypaea and their activity as inhibitors of Monoacylglycerol Lipase (MAGL). Planta Med. 2018;84(9–10):710–715. doi:10.1055/s-0044-100187
- De Leo M, Huallpa CG, Alvarado B, et al. New diterpenes from Salvia pseudorosmarinus and their activity as inhibitors of monoacylglycerol lipase (MAGL). Fitoterapia. 2018;130:251–258. doi:10.1016/j.fitote.2018.09.010
- Mei J, Guo R, Zhang F, et al. Identification of bioactive natural products using yeast: application to monoacylglycerol lipase inhibitor extraction from Corydalis Rhizoma. Biomed Pharmacother. 2022;149:112798. doi:10.1016/j.biopha.2022.112798
- Chen L, Yan Y, Chen T, et al. Forsythiaside prevents β-amyloid-induced hippocampal slice injury by upregulating 2-arachidonoylglycerol via cannabinoid receptor 1-dependent NF-κB pathway. Neurochem Int. 2019;125:57–66. doi:10.1016/j.neuint.2019.02.008
- Talia S, Benarous K, Lamrani M, Yousfi M. Lepidine B from Lepidium sativum seeds as multi-functional anti- alzheimer’s disease agent: in vitro and in silico studies. Curr Comput Aided Drug Des. 2021;17(3):360–377. doi:10.2174/1573409916666200302120305
- Chicca A, Marazzi J, Gertsch J. The antinociceptive triterpene β-amyrin inhibits 2-arachidonoylglycerol (2-AG) hydrolysis without directly targeting cannabinoid receptors. Br J Pharmacol. 2012;167(8):1596–1608. doi:10.1111/j.1476-5381.2012.02059.x
- Vecchiarelli HA, Morena M, Lee TTY, et al. Sex and stressor modality influence acute stress-induced dynamic changes in corticolimbic endocannabinoid levels in adult Sprague Dawley rats. Neurobiol Stress. 2022;20:100470. doi:10.1016/j.ynstr.2022.100470
- Botsford C, Brellenthin AG, Cisler JM, Hillard CJ, Koltyn KF, Crombie KM. Circulating endocannabinoids and psychological outcomes in women with PTSD. J Anxiety Disord. 2023;93:102656. doi:10.1016/j.janxdis.2022.102656
- Rubino T, Realini N, Castiglioni C, et al. Role in anxiety behavior of the endocannabinoid system in the prefrontal cortex. Cereb Cortex. 2008;18(6):1292–1301. doi:10.1093/cercor/bhm161
- Tevosian M, Todorov H, Lomazzo E, et al. NAPE-PLD deletion in stress-TRAPed neurons results in an anxiogenic phenotype. Transl Psychiatry. 2023;13(1):152. doi:10.1038/s41398-023-02448-9
- Hill MN, Kumar SA, Filipski SB, et al. Disruption of fatty acid amide hydrolase activity prevents the effects of chronic stress on anxiety and amygdalar microstructure. Mol Psychiatry. 2013;18(10):1125–1135. doi:10.1038/mp.2012.90
- Stopponi S, Fotio Y, Domi A, et al. Inhibition of fatty acid amide hydrolase in the central amygdala alleviates co-morbid expression of innate anxiety and excessive alcohol intake. Addict Biol. 2018;23(6):1223–1232. doi:10.1111/adb.12573
- Postnov A, Schmidt ME, Pemberton DJ, et al. Fatty acid amide hydrolase inhibition by JNJ-42165279: a multiple-ascending dose and a positron emission tomography study in healthy volunteers. Clin Transl Sci. 2018;11(4):397–404. doi:10.1111/cts.12548
- Paulus MP, Stein MB, Simmons AN, Risbrough VB, Halter R, Chaplan SR. The effects of FAAH inhibition on the neural basis of anxiety-related processing in healthy male subjects: a randomized clinical trial. Neuropsychopharmacol. 2021;46(5):1011–1019. doi:10.1038/s41386-020-00936-w
- Li GL, Winter H, Arends R, et al. Assessment of the pharmacology and tolerability of PF-04457845, an irreversible inhibitor of fatty acid amide hydrolase-1, in healthy subjects. Br J Clin Pharmacol. 2012;73(5):706–716. doi:10.1111/j.1365-2125.2011.04137.x
- Mayo LM, Asratian A, Lindé J, et al. Elevated anandamide, enhanced recall of fear extinction, and attenuated stress responses following inhibition of fatty acid amide hydrolase: a randomized, controlled experimental medicine trial. Biol Psychiatry. 2020;87(6):538–547. doi:10.1016/j.biopsych.2019.07.034
- D’Souza DC, Cortes-Briones J, Creatura G, et al. Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF-04457845) in the treatment of cannabis withdrawal and dependence in men: a double-blind, placebo-controlled, parallel group, phase 2a single-site randomised controlled trial. Lancet Psychiatry. 2019;6(1):35–45. doi:10.1016/S2215-0366(18)30427-9
- Zimmermann T, Bartsch JC, Beer A, et al. Impaired anandamide/palmitoylethanolamide signaling in hippocampal glutamatergic neurons alters synaptic plasticity, learning, and emotional responses. Neuropsychopharmacology. 2019;44(8):1377–1388. doi:10.1038/s41386-018-0274-7
- Fusse EJ, Scarante FF, Vicente MA, et al. Anxiogenic doses of rapamycin prevent URB597-induced anti-stress effects in socially defeated mice. Neurosci Lett. 2024;818:137519. doi:10.1016/j.neulet.2023.137519
- Borges-Assis AB, Uliana DL, Hott SC, Guimarães FS, Lisboa SF, Resstel LBM. Bed nucleus of the stria terminalis CB1 receptors and the FAAH enzyme modulate anxiety behavior depending on previous stress exposure. Prog Neuropsychopharmacol Biol Psychiatry. 2023;125:110739. doi:10.1016/j.pnpbp.2023.110739
- Danandeh A, Vozella V, Lim J, et al. Effects of fatty acid amide hydrolase inhibitor URB597 in a rat model of trauma-induced long-term anxiety. Psychopharmacology. 2018;235(11):3211–3221. doi:10.1007/s00213-018-5020-7
- Flannery LE, Kerr DM, Finn DP, Roche M. FAAH inhibition attenuates TLR3-mediated hyperthermia, nociceptive- and anxiety-like behavior in female rats. Behav Brain Res. 2018;353:11–20. doi:10.1016/j.bbr.2018.06.030
- de Ceglia M, Micioni Di Bonaventura MV, Romano A, et al. Anxiety associated with palatable food withdrawal is reversed by the selective FAAH inhibitor PF-3845: a regional analysis of the contribution of endocannabinoid signaling machinery. Int J Eat Disord. 2023;56(6):1098–1113. doi:10.1002/eat.23917
- Bluett RJ, Gamble-George JC, Hermanson DJ, Hartley ND, Marnett LJ, Patel S. Central anandamide deficiency predicts stress-induced anxiety: behavioral reversal through endocannabinoid augmentation. Transl Psychiatry. 2014;4(7):e408. doi:10.1038/tp.2014.53
- Carnevali L, Barbetti M, Fotio Y, et al. Enhancement of peripheral fatty acyl ethanolamide signaling prevents stress-induced social avoidance and anxiety-like behaviors in male rats. Psychopharmacology. 2023. doi:10.1007/s00213-023-06473-w
- Vecchiarelli HA, Morena M, Keenan CM, et al. Comorbid anxiety-like behavior in a rat model of colitis is mediated by an upregulation of corticolimbic fatty acid amide hydrolase. Neuropsychopharmacology. 2021;46(5):992–1003. doi:10.1038/s41386-020-00939-7
- Marco EM, Rapino C, Caprioli A, Borsini F, Laviola G, Maccarrone M. Potential therapeutic value of a novel FAAH inhibitor for the treatment of anxiety. PLoS One. 2015;10(9):e0137034. doi:10.1371/journal.pone.0137034
- Silva HH, Tavares V, Neto BV, Cerqueira F, Medeiros R, Silva MRG. FAAH rs324420 polymorphism: biological pathways, impact on elite athletic performance and insights for sport medicine. Genes. 2023;14(10):1946. doi:10.3390/genes14101946
- Spohrs J, Ulrich M, Grön G, Plener PL, Abler B. FAAH polymorphism (rs324420) modulates extinction recall in healthy humans: an fMRI study. Eur Arch Psychiatry Clin Neurosci. 2022;272(8):1495–1504. doi:10.1007/s00406-021-01367-4
- Sisk LM, Rapuano KM, Conley MI, et al. Genetic variation in endocannabinoid signaling is associated with differential network-level functional connectivity in youth. J Neurosci Res. 2022;100(3):731–743. doi:10.1002/jnr.24946
- Dincheva I, Drysdale AT, Hartley CA, et al. FAAH genetic variation enhances fronto-amygdala function in mouse and human. Nat Commun. 2015;6:6395. doi:10.1038/ncomms7395
- Marusak HA, Evanski J, Desai S, Rabinak CA. Impact of childhood trauma exposure, genetic variation in endocannabinoid signaling, and anxiety on frontolimbic pathways in children. Cannabis Cannabinoid Res. 2023;8(6):1079–1089. doi:10.1089/can.2022.0144
- Hillard CJ. Chapter one - the endocannabinoid signaling system in the CNS: a primer. In: Parsons L, Hill M editors. International Review of Neurobiology. Vol 125. Endocannabinoids. Academic Press; 2015:1–47. doi:10.1016/bs.irn.2015.10.001
- Yasmin F, Colangeli R, Morena M, et al. Stress-induced modulation of endocannabinoid signaling leads to delayed strengthening of synaptic connectivity in the amygdala. Proc Natl Acad Sci. 2020;117(1):650–655. doi:10.1073/pnas.1910322116
- Gulyas AI, Cravatt BF, Bracey MH, et al. Segregation of two endocannabinoid-hydrolyzing enzymes into pre- and postsynaptic compartments in the rat hippocampus, cerebellum and amygdala. Eur J Neurosci. 2004;20(2):441–458. doi:10.1111/j.1460-9568.2004.03428.x
- Morena M, Patel S, Bains JS, Hill MN. Neurobiological Interactions Between Stress and the Endocannabinoid System. Neuropsychopharmacol. 2016;41(1):80–102. doi:10.1038/npp.2015.166
- Alugubelly N, Mohammed AN, Carr RL. Persistent proteomic changes in glutamatergic and GABAergic signaling in the amygdala of adolescent rats exposed to chlorpyrifos as juveniles. Neurotoxicology. 2021;85:234–244. doi:10.1016/j.neuro.2021.05.012
- Rl C, A N, de L K, et al.. Inhibition of fatty acid amide hydrolase by chlorpyrifos in juvenile rats results in altered exploratory and social behavior as adolescents. Neurotoxicology. 2020:77. doi:10.1016/j.neuro.2020.01.002
- Duan T, Gu N, Wang Y, et al. Fatty acid amide hydrolase inhibitors produce rapid anti-anxiety responses through amygdala long-term depression in male rodents. J Psychiatry Neurosci. 2017;42(4):230–241. doi:10.1503/jpn.160116
- Pavón FJ, Serrano A, Stouffer DG, et al. Ethanol-induced alterations in endocannabinoids and relevant neurotransmitters in the nucleus accumbens of fatty acid amide hydrolase knockout mice. Addict Biol. 2019;24(6):1204–1215. doi:10.1111/adb.12695
- Rossi S, De Chiara V, Musella A, et al. Preservation of striatal cannabinoid CB1 receptor function correlates with the antianxiety effects of fatty acid amide hydrolase inhibition. Mol Pharmacol. 2010;78(2):260–268. doi:10.1124/mol.110.064196
- Aguilera-Portillo G, Rangel-López E, Villeda-Hernández J, et al. The pharmacological inhibition of fatty acid amide hydrolase prevents excitotoxic damage in the rat striatum: possible involvement of CB1 receptors regulation. Mol Neurobiol. 2019;56(2):844–856. doi:10.1007/s12035-018-1129-2
- Kerr DM, Burke NN, Ford GK, et al. Pharmacological inhibition of endocannabinoid degradation modulates the expression of inflammatory mediators in the hypothalamus following an immunological stressor. Neuroscience. 2012;204:53–63. doi:10.1016/j.neuroscience.2011.09.032
- Doenni VM, Gray JM, Song CM, Patel S, Hill MN, Pittman QJ. Deficient adolescent social behavior following early-life inflammation is ameliorated by augmentation of anandamide signaling. Brain Behav Immun. 2016;58:237–247. doi:10.1016/j.bbi.2016.07.152
- Grieco M, De Caris MG, Maggi E, et al. Fatty Acid Amide Hydrolase (FAAH) inhibition modulates amyloid-beta-induced microglia polarization. Int J Mol Sci. 2021;22(14):7711. doi:10.3390/ijms22147711
- Malek N, Popiolek-Barczyk K, Mika J, Przewlocka B, Starowicz K. Anandamide, acting via CB2 receptors, alleviates LPS-induced neuroinflammation in rat primary microglial cultures. Neural Plast. 2015;2015:130639. doi:10.1155/2015/130639
- Tanaka M, Yagyu K, Sackett S, Zhang Y. Anti-inflammatory effects by pharmacological inhibition or knockdown of fatty acid amide hydrolase in BV2 microglial cells. Cells. 2019;8(5):491. doi:10.3390/cells8050491
- Rivera P, Del Mar Fernández-Arjona M, Silva-Peña D, et al. Pharmacological blockade of fatty acid amide hydrolase (FAAH) by URB597 improves memory and changes the phenotype of hippocampal microglia despite ethanol exposure. Biochem. Pharmacol. 2018;157:244–257. doi:10.1016/j.bcp.2018.08.005
- Tchantchou F, Tucker LB, Fu AH, et al. The fatty acid amide hydrolase inhibitor PF-3845 promotes neuronal survival, attenuates inflammation and improves functional recovery in mice with traumatic brain injury. Neuropharmacology. 2014;85:427–439. doi:10.1016/j.neuropharm.2014.06.006
- Henry RJ, Kerr DM, Finn DP, Roche M. FAAH-mediated modulation of TLR3-induced neuroinflammation in the rat hippocampus. J Neuroimmunol. 2014;276(1–2):126–134. doi:10.1016/j.jneuroim.2014.09.002
- Zhu Y, Zhang H, Mao H, et al. FAAH served a key membrane-anchoring and stabilizing role for NLRP3 protein independently of the endocannabinoid system. Cell Death Differ. 2023;30(1):168–183. doi:10.1038/s41418-022-01054-4
- Abelson JL, Liberzon I, Young EA, Khan S. Cognitive modulation of the endocrine stress response to a pharmacological challenge in normal and panic disorder subjects. Arch Gen Psychiatry. 2005;62(6):668–675. doi:10.1001/archpsyc.62.6.668
- Hill MN, McLaughlin RJ, Bingham B, et al. Endogenous cannabinoid signaling is essential for stress adaptation. Proc Natl Acad Sci U S A. 2010;107(20):9406–9411. doi:10.1073/pnas.0914661107
- Hill MN, McLaughlin RJ, Morrish AC, et al. Suppression of amygdalar endocannabinoid signaling by stress contributes to activation of the hypothalamic-pituitary-adrenal axis. Neuropsychopharmacology. 2009;34(13):2733–2745. doi:10.1038/npp.2009.114
- Newsom RJ, Osterlund C, Masini CV, Day HE, Spencer RL, Campeau S. Cannabinoid receptor type 1 antagonism significantly modulates basal and loud noise induced neural and hypothalamic-pituitary-adrenal axis responses in male Sprague-Dawley rats. Neuroscience. 2012;204:64–73. doi:10.1016/j.neuroscience.2011.11.043
- Haller J, Barna I, Barsvari B, et al. Interactions between environmental aversiveness and the anxiolytic effects of enhanced cannabinoid signaling by FAAH inhibition in rats. Psychopharmacology. 2009;204(4):607–616. doi:10.1007/s00213-009-1494-7
- Bedse G, Colangeli R, Lavecchia AM, et al. Role of the basolateral amygdala in mediating the effects of the fatty acid amide hydrolase inhibitor URB597 on HPA axis response to stress. Eur Neuropsychopharmacol. 2014;24(9):1511–1523. doi:10.1016/j.euroneuro.2014.07.005
- Gray JM, Vecchiarelli HA, Morena M, et al. Corticotropin-releasing hormone drives anandamide hydrolysis in the amygdala to promote anxiety. J Neurosci. 2015;35(9):3879–3892. doi:10.1523/JNEUROSCI.2737-14.2015
- Gray JM, Wilson CD, Lee TTY, et al. Sustained glucocorticoid exposure recruits cortico-limbic CRH signaling to modulate endocannabinoid function. Psychoneuroendocrinology. 2016;66:151–158. doi:10.1016/j.psyneuen.2016.01.004
- Duranti A, Beldarrain G, Álvarez A, et al. The endocannabinoid system as a target for neuroprotection/neuroregeneration in perinatal hypoxic–ischemic brain injury. Biomedicines. 2022;11(1):28. doi:10.3390/biomedicines11010028
- Oddi S, Fiorenza MT, Maccarrone M. Endocannabinoid signaling in adult hippocampal neurogenesis: a mechanistic and integrated perspective. Prog Lipid Res. 2023;91:101239. doi:10.1016/j.plipres.2023.101239
- Fogaça MV, Campos AC, Coelho LD, Duman RS, Guimarães FS. The anxiolytic effects of cannabidiol in chronically stressed mice are mediated by the endocannabinoid system: role of neurogenesis and dendritic remodeling. Neuropharmacology. 2018;135:22–33. doi:10.1016/j.neuropharm.2018.03.001
- Avraham HK, Jiang S, Fu Y, et al. Impaired neurogenesis by HIV-1-Gp120 is rescued by genetic deletion of fatty acid amide hydrolase enzyme. Br J Pharmacol. 2015;172(19):4603–4614. doi:10.1111/bph.12657
- Segev A, Korem N, Mizrachi Zer-Aviv T, et al. Role of endocannabinoids in the hippocampus and amygdala in emotional memory and plasticity. Neuropsychopharmacology. 2018;43(10):2017–2027. doi:10.1038/s41386-018-0135-4
- Kathuria S, Gaetani S, Fegley D, et al. Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med. 2003;9(1):76–81. doi:10.1038/nm803
- Koehl M, Ladevèze E, Catania C, Cota D, Abrous DN. Inhibition of mTOR signaling by genetic removal of p70 S6 kinase 1 increases anxiety-like behavior in mice. Transl Psychiatry. 2021;11(1):165. doi:10.1038/s41398-020-01187-5
- Tanaka M, Yamada S, Watanabe Y. The Role of Neuropeptide Y in the Nucleus Accumbens. Int J Mol Sci. 2021;22(14):7287. doi:10.3390/ijms22147287
- Amstadter AB, Koenen KC, Ruggiero KJ, et al. NPY moderates the relation between hurricane exposure and generalized anxiety disorder in an epidemiologic sample of hurricane-exposed adults. Depress Anxiety. 2010;27(3):270–275. doi:10.1002/da.20648
- Decressac M, Barker RA. Neuropeptide Y and its role in CNS disease and repair. Exp Neurol. 2012;238(2):265–272. doi:10.1016/j.expneurol.2012.09.004
- Molosh AI, Sajdyk TJ, Truitt WA, Zhu W, Oxford GS, Shekhar A. NPY Y1 receptors differentially modulate GABAA and NMDA receptors via divergent signal-transduction pathways to reduce excitability of amygdala neurons. Neuropsychopharmacology. 2013;38(7):1352–1364. doi:10.1038/npp.2013.33
- Maymon N, Mizrachi Zer-Aviv T, Sabban EL, Akirav I. Neuropeptide Y and cannabinoids interaction in the amygdala after exposure to shock and reminders model of PTSD. Neuropharmacology. 2020;162:107804. doi:10.1016/j.neuropharm.2019.107804
- Greco R, Demartini C, Zanaboni AM, et al. Potentiation of endocannabinoids and other lipid amides prevents hyperalgesia and inflammation in a pre-clinical model of migraine. J Headache Pain. 2022;23(1):79. doi:10.1186/s10194-022-01449-1
- Fotio Y, Mabou Tagne A, Jung KM, Piomelli D. Fatty acid amide hydrolase inhibition alleviates anxiety-like symptoms in a rat model used to study post-traumatic stress disorder. Psychopharmacology. 2023. doi:10.1007/s00213-023-06358-y
- Shang Y, Wang M, Hao Q, et al. Development of indole-2-carbonyl piperazine urea derivatives as selective FAAH inhibitors for efficient treatment of depression and pain. Bioorg Chem. 2022;128:106031. doi:10.1016/j.bioorg.2022.106031
- Gur Maz T, Turanlı S, Caliskan HB, Çalışkan B, Banoglu E. Development and molecular modeling studies of new thiadiazole piperazine urea derivatives as potential fatty acid amide hydrolase inhibitors. Arch Pharm. 2022;355(8):e2200082. doi:10.1002/ardp.202200082
- Keith JM, Jones W, Pierce JM, et al. Heteroarylureas with fused bicyclic diamine cores as inhibitors of fatty acid amide hydrolase. Bioorg Med Chem Lett. 2020;30(20):127463. doi:10.1016/j.bmcl.2020.127463
- Jaiswal S, Tripathi RKP, Ayyannan SR. Scaffold hopping-guided design of some isatin based rigid analogs as fatty acid amide hydrolase inhibitors: synthesis and evaluation. Biomed Pharmacother. 2018;107:1611–1623. doi:10.1016/j.biopha.2018.08.125
- Jaiswal S, Ayyannan SR. Lead optimization study on indoline-2,3-dione derivatives as potential fatty acid amide hydrolase inhibitors. J Biomol Struct Dyn. 2023;41(19):9632–9650. doi:10.1080/07391102.2022.2145372
- Patel JZ, Parkkari T, Laitinen T, et al. Chiral 1,3,4-oxadiazol-2-ones as highly selective FAAH inhibitors. J Med Chem. 2013;56(21):8484–8496. doi:10.1021/jm400923s
- Greco R, Francavilla M, Demartini C, et al. Activity of FAAH-Inhibitor JZP327A in an Experimental Rat Model of Migraine. Int J Mol Sci. 2023;24(12):10102. doi:10.3390/ijms241210102
- Papa A, Pasquini S, Galvani F, et al. Development of potent and selective FAAH inhibitors with improved drug-like properties as potential tools to treat neuroinflammatory conditions. Eur J Med Chem. 2023;246:114952. doi:10.1016/j.ejmech.2022.114952
- Zięba A, Laitinen T, Patel JZ, Poso A, Kaczor AA. Docking-based 3D-QSAR studies for 1,3,4-oxadiazol-2-one derivatives as FAAH inhibitors. Int J Mol Sci. 2021;22(11):6108. doi:10.3390/ijms22116108
- Lorca M, Valdes Y, Chung H, Romero-Parra J, Pessoa-Mahana CD, Mella J. Three-Dimensional Quantitative Structure-Activity Relationships (3D-QSAR) on a Series of Piperazine-Carboxamides Fatty Acid Amide Hydrolase (FAAH) inhibitors as a useful tool for the design of new cannabinoid ligands. Int J Mol Sci. 2019;20(10):2510. doi:10.3390/ijms20102510
- Zanfirescu A, Nitulescu G, Mihai DP, Nitulescu GM. Identifying FAAH inhibitors as new therapeutic options for the treatment of chronic pain through drug repurposing. Pharmaceuticals. 2021;15(1):38. doi:10.3390/ph15010038
- Deplano A, Karlsson J, Svensson M, et al. Exploring the fatty acid amide hydrolase and cyclooxygenase inhibitory properties of novel amide derivatives of ibuprofen. J Enzyme Inhib Med Chem. 2020;35(1):815–823. doi:10.1080/14756366.2020.1743283
- Deplano A, Karlsson J, Moraca F, et al. Design, synthesis and in vitro and in vivo biological evaluation of flurbiprofen amides as new fatty acid amide hydrolase/cyclooxygenase-2 dual inhibitory potential analgesic agents. J Enzyme Inhib Med Chem. 2021;36(1):940–953. doi:10.1080/14756366.2021.1875459
- Montanari S, Scalvini L, Bartolini M, et al. Fatty Acid Amide Hydrolase (FAAH), Acetylcholinesterase (AChE), and Butyrylcholinesterase (BuChE): networked targets for the development of carbamates as potential anti-Alzheimer’s Disease agents. J Med Chem. 2016;59(13):6387–6406. doi:10.1021/acs.jmedchem.6b00609
- Maleki MF, Nadri H, Kianfar M, et al. Design and synthesis of new carbamates as inhibitors for fatty acid amide hydrolase and cholinesterases: molecular dynamic, in vitro and in vivo studies. Bioorg Chem. 2021;109:104684. doi:10.1016/j.bioorg.2021.104684
- Wilt S, Kodani S, Le TNH, et al. Development of multitarget inhibitors for the treatment of pain: design, synthesis, biological evaluation and molecular modeling studies. Bioorg Chem. 2020;103:104165. doi:10.1016/j.bioorg.2020.104165
- Cammarota M, Ferlenghi F, Vacondio F, et al. Combined targeting of fatty acid amide hydrolase and melatonin receptors promotes neuroprotection and stimulates inflammation resolution in rats. Br J Pharmacol. 2023;180(10):1316–1338. doi:10.1111/bph.16014
- Intranuovo F, Brunetti L, DelRe P, et al. Development of N-(1-Adamantyl)benzamides as novel anti-inflammatory multitarget agents acting as dual modulators of the cannabinoid CB2 receptor and fatty acid amide hydrolase. J Med Chem. 2023;66(1):235–250. doi:10.1021/acs.jmedchem.2c01084
- Wagenlehner FME, van Till JWO, Houbiers JGA, et al. Fatty acid amide hydrolase inhibitor treatment in men with chronic prostatitis/chronic pelvic pain syndrome: an adaptive double-blind, randomized controlled trial. Urology. 2017;103:191–197. doi:10.1016/j.urology.2017.02.029
- Demartini C, Greco R, Zanaboni AM, Francavilla M, Facchetti S, Tassorelli C. URB937 prevents the development of mechanical allodynia in male rats with trigeminal neuralgia. Pharmaceuticals. 2023;16(11):1626. doi:10.3390/ph16111626
- Zhang D, Saraf A, Kolasa T, et al. Fatty acid amide hydrolase inhibitors display broad selectivity and inhibit multiple carboxylesterases as off-targets. Neuropharmacology. 2007;52(4):1095–1105. doi:10.1016/j.neuropharm.2006.11.009
- Kerbrat A, Ferré JC, Fillatre P, et al. Acute neurologic disorder from an inhibitor of fatty acid amide hydrolase. N Engl J Med. 2016;375(18):1717–1725. doi:10.1056/NEJMoa1604221
- van Esbroeck ACM, Janssen APA, Cognetta AB, et al. Activity-based protein profiling reveals off-target proteins of the FAAH inhibitor BIA 10-2474. Science. 2017;356(6342):1084–1087. doi:10.1126/science.aaf7497
- Otrubova K, Chatterjee S, Ghimire S, Cravatt BF, Boger DL. N-Acyl pyrazoles: effective and tunable inhibitors of serine hydrolases. Bioorg Med Chem. 2019;27(8):1693–1703. doi:10.1016/j.bmc.2019.03.020
- Lamani M, Malamas MS, Farah SI, et al. Piperidine and piperazine inhibitors of fatty acid amide hydrolase targeting excitotoxic pathology. Bioorg Med Chem. 2019;27(23):115096. doi:10.1016/j.bmc.2019.115096
- Thors L, Eriksson J, Fowler CJ. Inhibition of the cellular uptake of anandamide by genistein and its analogue daidzein in cells with different levels of fatty acid amide hydrolase-driven uptake. Br J Pharmacol. 2007;152(5):744–750. doi:10.1038/sj.bjp.0707401
- Ahmad H, Rauf K, Zada W, et al. Kaempferol facilitated extinction learning in contextual fear conditioned rats via inhibition of fatty-acid amide hydrolase. Molecules. 2020;25(20):4683. doi:10.3390/molecules25204683
- Zada W, VanRyzin JW, Perez‐Pouchoulen M, et al. Fatty acid amide hydrolase inhibition and N‐arachidonoylethanolamine modulation by isoflavonoids: a novel target for upcoming antidepressants. Pharmacol Res Perspect. 2022;10(5):e00999. doi:10.1002/prp2.999
- Alasmari M, Bӧhlke M, Kelley C, Maher T, Pino-Figueroa A. Inhibition of Fatty Acid Amide Hydrolase (FAAH) by macamides. Mol Neurobiol. 2019;56(3):1770–1781. doi:10.1007/s12035-018-1115-8
- Almukadi H, Wu H, Böhlke M, Kelley CJ, Maher TJ, Pino-Figueroa A. The macamide N-3-methoxybenzyl-linoleamide is a time-dependent fatty acid amide hydrolase (FAAH) inhibitor. Mol Neurobiol. 2013;48(2):333–339. doi:10.1007/s12035-013-8499-2
- Police A, Shankar VK, Pandey P, Rangappa S, Doerksen RJ, Narasimha Murthy S. Novel topical anandamide formulation for alleviating peripheral neuropathic pain. Int J Pharm. 2023;641:123085. doi:10.1016/j.ijpharm.2023.123085
- Shanmugasundaram D, Roza JM. Effect of broad-spectrum hemp extract on neurobehavioral activity on the immobilization stress-induced model in Sprague Dawley rats. ScientificWorldJournal. 2023;2023:3425576. doi:10.1155/2023/3425576
- Lin YX, Sun JT, Liao ZZ, et al. Triterpenoids from the fruiting bodies of Ganoderma lucidum and their inhibitory activity against FAAH. Fitoterapia. 2022;158:105161. doi:10.1016/j.fitote.2022.105161
- Li DW, Liu M, Leng YQ, et al. Lanostane triterpenoids from Ganoderma lucidum and their inhibitory effects against FAAH. Phytochemistry. 2022;203:113339. doi:10.1016/j.phytochem.2022.113339
- Thors L, Alajakku K, Fowler CJ. The “specific” tyrosine kinase inhibitor genistein inhibits the enzymic hydrolysis of anandamide: implications for anandamide uptake. Br J Pharmacol. 2007;150(7):951–960. doi:10.1038/sj.bjp.0707172
- El-Mekkawy S, Shahat AA, Alqahtani AS, et al. A polyphenols-rich extract from Moricandia sinaica Boiss. Exhibits analgesic, anti-inflammatory and antipyretic activities in vivo. Molecules. 2020;25(21):5049. doi:10.3390/molecules25215049
- Thors L, Belghiti M, Fowler CJ. Inhibition of fatty acid amide hydrolase by kaempferol and related naturally occurring flavonoids. Br J Pharmacol. 2008;155(2):244–252. doi:10.1038/bjp.2008.237
- Mariano A, Di Sotto A, Leopizzi M, et al. Antiarthritic effects of a root extract from Harpagophytum procumbens DC: novel insights into the molecular mechanisms and possible bioactive phytochemicals. Nutrients. 2020;12(9):2545. doi:10.3390/nu12092545
- Gonçalves ECD, Assis PM, Junqueira LA, et al. Citral inhibits the inflammatory response and hyperalgesia in mice: the role of TLR4, TLR2/Dectin-1, and CB2 cannabinoid receptor/ATP-sensitive K+ channel pathways. J Nat Prod. 2020;83(4):1190–1200. doi:10.1021/acs.jnatprod.9b01134
- El-Alfy AT, Abourashed EA, Patel C, Mazhari N, An H, Jeon A. Phenolic compounds from nutmeg (Myristica fragrans Houtt.) inhibit the endocannabinoid-modulating enzyme fatty acid amide hydrolase. J Pharm Pharmacol. 2019;71(12):1879–1889. doi:10.1111/jphp.13174
- Sanna MD, Les F, Lopez V, Galeotti N. Lavender (Lavandula angustifolia Mill.) essential oil alleviates neuropathic pain in mice with spared nerve injury. Front Pharmacol. 2019;10:472. doi:10.3389/fphar.2019.00472
- De Petrocellis L, Ligresti A, Moriello AS, et al. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br J Pharmacol. 2011;163(7):1479–1494. doi:10.1111/j.1476-5381.2010.01166.x
- Capasso R, Borrelli F, Aviello G, et al. Cannabidiol, extracted from Cannabis sativa, selectively inhibits inflammatory hypermotility in mice. Br J Pharmacol. 2008;154(5):1001–1008. doi:10.1038/bjp.2008.177
- Li D, Leng Y, Liao Z, et al. Nor-triterpenoids from the fruiting bodies of Ganoderma lucidum and their inhibitory activity against FAAH. Nat Prod Res. 2023;37(4):579–585. doi:10.1080/14786419.2022.2078817
- Chicca A, Caprioglio D, Minassi A, et al. Functionalization of β-caryophyllene generates novel polypharmacology in the endocannabinoid system. ACS Chem Biol. 2014;9(7):1499–1507. doi:10.1021/cb500177c
- Irrera N, D’Ascola A, Pallio G, et al. β-Caryophyllene Mitigates Collagen Antibody Induced Arthritis (CAIA) in mice through a cross-talk between CB2 and PPAR-γ receptors. Biomolecules. 2019;9(8):326. doi:10.3390/biom9080326
- Wu H, Kelley CJ, Pino-Figueroa A, Vu HD, Maher TJ. Macamides and their synthetic analogs: evaluation of in vitro FAAH inhibition. Bioorg Med Chem. 2013;21(17):5188–5197. doi:10.1016/j.bmc.2013.06.034
- Taboada-Rosell K, Castro-García FA, Medina-Saldivar C, Cruz-Visalaya SR, Pacheco-Otalora LF. The novel FAAH inhibitor, MCH1, reduces the infarction area in the motor cortex-related region but does not affect the sensorimotor function or memory and spatial learning in rats exposed to transient middle cerebral artery occlusion. Brain Res. 2024;1822:148636. doi:10.1016/j.brainres.2023.148636
- Zhang ZW, Han P, Fu J, et al. Gut microbiota-based metabolites of Xiaoyao Pills (a typical Traditional Chinese medicine) ameliorate depression by inhibiting fatty acid amide hydrolase levels in brain. J Ethnopharmacol. 2023;313:116555. doi:10.1016/j.jep.2023.116555
- Samandar F, Tehranizadeh ZA, Saberi MR, Chamani J. CB1 as a novel target for Ginkgo biloba’s terpene trilactone for controlling chemotherapy-induced peripheral neuropathy (CIPN). J Mol Model. 2022;28(9):283. doi:10.1007/s00894-022-05284-8
- Xie J, Li Y, Liang Y, Kui H, Wang C, Huang J. Integration of non-targeted metabolomics with network pharmacology deciphers the anxiolytic mechanisms of Platycladi Semen extracts in CUMS mice. J Ethnopharmacol. 2023;315:116571. doi:10.1016/j.jep.2023.116571
- Tian X, Liu T, Li L, et al. Visual high-throughput screening for developing a fatty acid amide hydrolase natural inhibitor based on an enzyme-activated fluorescent probe. Anal Chem. 2020;92(14):9493–9500. doi:10.1021/acs.analchem.9b05826
- Tian M, Tian Z, Yao D, et al. A NIR fluorescent probe for fatty acid amide hydrolase bioimaging and its application in development of inhibitors. J Mater Chem B. 2021;9(32):6460–6465. doi:10.1039/d1tb01054a
- Della Pietra A, Krivoshein G, Ivanov K, et al. Potent dual MAGL/FAAH inhibitor AKU-005 engages endocannabinoids to diminish meningeal nociception implicated in migraine pain. J Headache Pain. 2023;24(1):38. doi:10.1186/s10194-023-01568-3
- Dong B, Shilpa BM, Shah R, et al. Dual pharmacological inhibitor of endocannabinoid degrading enzymes reduces depressive-like behavior in female rats. J Psychiatr Res. 2020;120:103–112. doi:10.1016/j.jpsychires.2019.10.010
- Adamson Barnes NS, Mitchell VA, Kazantzis NP, Vaughan CW. Actions of the dual FAAH/MAGL inhibitor JZL195 in a murine neuropathic pain model. Br J Pharmacol. 2016;173(1):77–87. doi:10.1111/bph.13337
- Toczek M, Ryszkiewicz P, Remiszewski P, et al. Weak hypotensive effect of chronic administration of the dual FAAH/MAGL inhibitor JZL195 in spontaneously hypertensive rats as revealed by area under the curve analysis. Int J Mol Sci. 2023;24(13):10942. doi:10.3390/ijms241310942
- Abohalaka R, Karaman Y, Recber T, Onder SC, Nemutlu E, Bozkurt TE. Endocannabinoid metabolism inhibition ameliorates ovalbumin-induced allergic airway inflammation and hyperreactivity in Guinea pigs. Life Sci. 2022;306:120808. doi:10.1016/j.lfs.2022.120808
- Manduca A, Morena M, Campolongo P, et al. Distinct roles of the endocannabinoids anandamide and 2-arachidonoylglycerol in social behavior and emotionality at different developmental ages in rats. Eur Neuropsychopharmacol. 2015;25(8):1362–1374. doi:10.1016/j.euroneuro.2015.04.005
- Fowler CJ. Transport of endocannabinoids across the plasma membrane and within the cell. FEBS J. 2013;280(9):1895–1904. doi:10.1111/febs.12212
- Nicolussi S, Gertsch J. Endocannabinoid transport revisited. Vitam Horm. 2015;98:441–485. doi:10.1016/bs.vh.2014.12.011
- Jones MJ, Uzuneser TC, Clement T, et al. Inhibition of fatty acid binding protein-5 in the basolateral amygdala induces anxiolytic effects and accelerates fear memory extinction. Psychopharmacology. 2023. doi:10.1007/s00213-023-06468-7
- Chicca A, Nicolussi S, Bartholomäus R, et al. Chemical probes to potently and selectively inhibit endocannabinoid cellular reuptake. Proc Natl Acad Sci U S A. 2017;114(25):E5006–E5015. doi:10.1073/pnas.1704065114
- Bortolato M, Campolongo P, Mangieri RA, et al. Anxiolytic-like properties of the anandamide transport inhibitor AM404. Neuropsychopharmacology. 2006;31(12):2652–2659. doi:10.1038/sj.npp.1301061
- Kwee CMB, Leen NA, Van der Kamp RC, et al. Anxiolytic effects of endocannabinoid enhancing compounds: a systematic review and meta-analysis. Eur Neuropsychopharmacol. 2023;72:79–94. doi:10.1016/j.euroneuro.2023.04.001
