Abstract
The incidence and prevalence of metabolic and musculoskeletal diseases are increasing. Type 2 diabetes mellitus (T2DM) is characterized by insulin resistance, inflammation, advanced glycation end-product accumulation and increased oxidative stress. These characteristics can negatively affect various aspects of muscle health, including muscle mass, strength, quality and function through impairments in protein metabolism, vascular and mitochondrial dysfunction, and cell death. Sarcopenia is a term used to describe the age-related loss in skeletal muscle mass and function and has been implicated as both a cause and consequence of T2DM. Sarcopenia may contribute to the development and progression of T2DM through altered glucose disposal due to low muscle mass, and also increased localized inflammation, which can arise through inter- and intramuscular adipose tissue accumulation. Lifestyle modifications are important for improving and maintaining mobility and metabolic health in individuals with T2DM and sarcopenia. However, evidence for the most effective and feasible exercise and dietary interventions in this population is lacking. In this review, we discuss the current literature highlighting the bidirectional relationship between T2DM and sarcopenia, highlight current research gaps and treatments, and provide recommendations for future research.
Introduction
In 2017, it was estimated that 451 million adults globally had diabetes.Citation1 Approximately 90% of these individuals have type 2 diabetes mellitus (T2DM),Citation2 with the highest prevalence observed in older adults.Citation3 As populations age, incidence and prevalence of metabolic and musculoskeletal diseases increase.Citation1,Citation4 Body composition changes during aging, resulting in significant losses of skeletal muscle mass and increased body fat percentage.Citation5 Collectively and independently, low muscle mass and adiposity are associated with higher incidence of metabolic disorders, including T2DM.Citation6,Citation7 “Sarcopenia” describes age-related declines in muscle mass and functionCitation8–Citation12 and has been implicated as both a cause and consequence of T2DM.Citation13 T2DM is characterized by insulin resistance, increased advanced glycation end-products (AGEs), a proinflammatory phenotype and oxidative stress, which can lead to micro- and macro-vascular complications. These characteristics can interfere with normal cellular functioning and cause cell deathCitation14, which may lead to losses in skeletal muscle mass, strength, and function, potentially leading to the development of sarcopenia. Conversely, low muscle mass and function in sarcopenia can lead to poorer glucose disposal and decreased metabolic rate and physical activity, all of which may place older adults with sarcopenia at increased risk for developing T2DM.
Type 2 diabetes mellitus and sarcopenia
Currently, there is no consensus definition of sarcopenia; however, several definitions and criteria for diagnosis have been proposed, some of which are presented in . The most commonly used multi-component operational definition since 2010 is the European Working Group on Sarcopenia in Older People (EWGSOP) definition,Citation8 which has recently been revised.Citation9 The Asian Working Group for Sarcopenia (AWGS) definitionCitation12 was derived from the EWGSOP definition but provides appropriate criteria for Asian populations. The International Working Group on Sarcopenia (IWGS)Citation11 definition is similar to the EWGSOP definition, but includes only low muscle mass and physical performance, not low strength. The only group to use statistical procedures to identify criteria for sarcopenia (low lean mass and handgrip strength), rather than expert consensus, is the Foundation for the National Institutes of Health Biomarkers Consortium Sarcopenia Project (FNIH).Citation10 Unsurprisingly, the different approaches taken to develop sarcopenia definitions results in poor agreement between them.
Table 1 Comparison of current definitions of sarcopenia
Recent meta-analyses indicate sarcopenia prevalence ranges from 10% to 40% in healthy men and women aged ≥60, and this variability is influenced by the operational definition applied and population assessed.Citation15,Citation16 Our understanding of associations between sarcopenia and T2DM is similarly limited by lack of consistency in definitions applied. However, older adults with T2DM do appear to have an increased risk for sarcopenia. The prevalence of sarcopenia in Japanese and Chinese adults (aged ≥65 and >60 years, respectively) with T2DM is 15% using the AWGS definition.Citation17,Citation18 In the Korean Sarcopenic Obesity Study, individuals with T2DM had threefold higher odds of having low skeletal muscle mass relative to weight compared with non-diabetic controls after adjusting for multiple confounders.Citation19 In the Health ABC Study (n=2,675), older women with undiagnosed and confirmed T2DM demonstrated greater losses in thigh muscle cross-sectional area over five years compared with non-diabetic controls (−10.6±1.5 and −9.3±1.2 versus −5.2±0.4 cm2, respectively).Citation20 Interestingly, the English Longitudinal Study of Ageing including 3,404 older adults recently demonstrated that diabetes predicted low handgrip strength eight years later in men (OR: 2.43 [95%CI: 1.5–3.95]), but not women (OR: 1.49 [95%CI: 0.83–2.68]), based on FNIH cut-points.Citation21
In addition to the risk of sarcopenia being increased in T2DM patients, older adults with sarcopenia may have increased risk of developing T2DM. In 113,913 Korean men and 89,854 women (mean age of 40 years), individuals in the lowest skeletal muscle mass relative to body weight quartiles had a twofold greater risk of developing T2DM compared with individuals in the highest quartile.Citation6 Additionally, the Korean Genome Epidemiology Study in 6,895 adults (mean age of 52 years) showed that individuals in the lowest muscle mass index tertile (defined by weight-adjusted appendicular lean mass [ALM]) had twofold higher odds of incident T2DM after adjusting for confounders.Citation22 Furthermore, comorbid presence of obesity and low muscle mass conferred even greater risk of incident T2DM in this population.Citation22 Lower hand grip strength normalized to body weight was also predictive of elevated fasting glucose (>5.56 mmol/L or treatment with glucose-lowering drugs) in 17,703 Chinese men and women aged over 40 years.Citation23
Muscle deficits may affect metabolic health through altered glucose disposal, as skeletal muscle accounts for ~80% of glucose clearance during euglycemic and hyperinsulinemic conditions.Citation24 Therefore, greater quantities of muscle increase glucose disposal and potentially delay the onset of T2DM. Once established, however, T2DM leads to various pathophysiological manifestations that have bidirectional relationships with components of sarcopenia.
Insulin resistance
Insulin resistance is a hallmark of T2DM that results in poor glycemic control, which potentially affects components of sarcopenia. In the MrOS study, men aged ≥65 years without T2DM but in the highest quartile for insulin resistance had two-fold higher odds of losing ≥5% total lean mass over approximately five years.Citation25 In the NHANES study, gait speed decreased across increasing quartiles of insulin resistance in non-diabetic older men, but not women.Citation26 Conversely, in 968 Italian men and women with a mean age of 67 and 68 years, respectively, insulin resistance was negatively associated with handgrip strength normalized to BMI in women, but not men.Citation27 Thus, insulin resistance may be a predictor of poor muscle health, irrespective of diabetes status, although further research is required to clarify sex-specific associations of insulin resistance and components of sarcopenia.
Glycated hemoglobin (HbA1c) is a measure of long-term glycemic control.Citation28 In older Korean men aged ≥65 years with T2DM, poor glycemic control (HbA1c ≥8.5%) was associated with poor lower-limb muscle quality and physical performance.Citation29 Similar associations have been reported for Caucasian and Black individuals; in the Baltimore Longitudinal Study of Ageing, knee extensor strength was lower across increasing quartiles of HbA1c.Citation30 Interestingly, total body and leg lean mass remained the same across all quartiles of HbA1c in adjusted models that included body weight as a covariate. However, this is a potential over-adjustment and could explain the lack of association between HbA1c and lean mass, despite the relationship with muscle strength.Citation30 Loss of muscle strength and mass in individuals with poor glycemic control likely occurs due to increased protein degradationCitation31 and decreased protein synthesisCitation32 Impaired protein metabolism is therefore exacerbated in individuals with T2DM, and particularly in individuals with undiagnosed T2DM, who typically have greater glucose variability. The negative effects of poor glycemic control on muscle health are compounded as it often occurs alongside other adverse characteristics of T2DM, including inflammation.
Inflammation
T2DM is associated with chronic, low-grade systemic inflammation which may negatively affect glucose and muscle homeostasis.Citation33–Citation35 Inflammatory markers including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α) and C-reactive protein (CRP) are often elevated in individuals with T2DM.Citation36,Citation37 These inflammatory markers are associated with adiposity and insulin resistanceCitation38 and are elevated in obese and non-obese individuals with T2DMCitation36 Visceral adipose tissue (VAT) secretes IL-6 and TNF-α,Citation39 and it is therefore likely to be a major contributor to inflammation in T2DM. We recently demonstrated that waist circumference (an indirect measure of VAT) was negatively associated with muscle strength, quality, and performance in 84 overweight and obese older adults.Citation40 However, it is unclear whether these negative associations were driven in part by increased inflammation.
Interestingly, IL-6 is also produced by myocytes during exercise, and transient increases in IL-6 assist in nutrient mobilization and muscle hypertrophy.Citation41–Citation43 Contrary to this, IL-6 has been reported to cause muscle atrophy when localized to the tibialis anterior of Sprague-Dawley rats, independent of systemic inflammation.Citation35 In the Framingham Heart Study consisting of 558 older adults aged 72–92 years, IL-6 predicted two-year lean mass losses in women, but not men.Citation44 In 115 older adults aged >60 years, ALM loss over five years was fivefold and fourfold greater in individuals with IL-6 and CRP concentrations in the top tertile of the cohort, respectively.Citation45 Park et al demonstrated that older adults with T2DM had greater losses in leg muscle mass and strength over three years compared with non-diabetic controls, and these associations were partially attenuated after adjusting for cytokines including IL-6 and TNF-α.Citation46
In addition to being negatively associated with muscle mass, inflammatory markers are also negatively associated with muscle strength, which is now considered the principal component of sarcopenia.Citation9 Visser et al reported that in 2,746 generally well-functioning Black and Caucasian men and women from the USA aged 70–79 years, handgrip strength was between 1.1 and 2.4 kg lower per standard deviation increase in IL-6 concentration.Citation47 TNF-α was negatively associated with grip strength in Black and Caucasian women, but not men.Citation47 In the English Longitudinal Study of Ageing, CRP was negatively associated with handgrip strength in women, and chair stand time in both sexes.Citation48 Taken together, inflammation associated with T2DM may independently affect muscle mass and strength.
Oxidative stress
Aging is associated with reduced antioxidant capacity, which has been implicated in the pathogenesis of sarcopenia.Citation49 T2DM is also associated with increased oxidative stress,Citation50 which can lead to myopathy. Oxidative stress in T2DM can occur through dyslipidemia and altered lipid metabolism, insulin resistance, increased AGEs and mitochondrial dysfunction.Citation50 In diabetic rats, increased oxidative stress impairs muscle repair.Citation51 In 107 pre- and post-menopausal women increased oxidative stress(measured by lipid peroxide concentrations) was negatively associated with skeletal muscle mass adjusted for height (SMI).Citation52 Given that oxidative stress also has the capacity to impair satellite cell differentiation and damage DNA,Citation53 it is evident that myopathy in T2DM is caused by the disruption of numerous cellular pathways, and further research is required to clarify how they work, and determine whether they are perhaps suitable targets for interventions.
Another age- and T2DM-related factor implicated in the progression of metabolic and muscle health deterioration through oxidative stress is mitochondrial dysfunction.Citation54,Citation55 Older individuals have up to 50% lower oxidative capacity per unit of muscleCitation56 and increased mitochondrial DNA mutationsCitation57 compared with younger adults. Interestingly, when compared with BMI-matched controls, individuals with T2DM had a 45% lower phosphocreatine recovery half-time (a measure of mitochondrial function) immediately after exercise.Citation58 Furthermore, this study also reported that fasting plasma glucose and HbA1c were positively associated with phosphocreatine recovery half-time (r=0.42 and r=0.48, respectively).Citation58 Impairments in oxidative capacity lead to decreased metabolism of macronutrients, and, more importantly, poorer physical performance. Interestingly, T2DM has intergenerational effects on mitochondria; young, lean offspring of individuals with T2DM had 38% lower skeletal muscle mitochondrial density compared with insulin-sensitive offspring from non-diabetic parents.Citation59 Additional studies investigating links between T2DM, sarcopenia and mitochondrial dysfunction in humans are required given the important role of mitochondria in muscle function and metabolism.
Advanced glycation end-products
Non-enzymatic reactions between glucose and amino groups in proteins, lipids, and nucleic acids can lead to the formation of AGEs.Citation60 AGEs are positivity associated with insulin resistance, obesity, and age.Citation61,Citation62 Currently it is unclear how AGEs contribute to poor muscle health; however, it has been proposed that AGE-associated muscle weakness arises through increased protein cross-linking within muscle, thereby interfering with contractility, and also increasing inflammation and oxidative stress.Citation14,61Citation61 Indeed, AGE concentrations within skeletal muscle increase across the lifespan.Citation61 Skin autofluorescence is a common, surrogate measure of AGEs, which is representative of long-term glycemic control.Citation63 Skin autofluorescence was higher (indicating higher AGEs) in a cross-sectional study of 166 Japanese adults (mean age of 63 years) with T2DM and sarcopenia when compared with non-sarcopenic T2DM controls, and was associated with low muscle strength and total body lean mass.Citation64 Another study in 232 Japanese adult men reported that higher skin autofluorescence was associated with lower hand grip strength and leg extension power.Citation65 In 9,203 Japanese men and women with a mean age of 58 years, SMI was inversely correlated with skin autofluorescence.Citation66 Furthermore, in a subpopulation of this study consisting of 1,934 participants aged >60 years, skin autofluorescence was negatively associated with grip and hip flexion and abduction strength.Citation66 Currently, reliability of skin autofluorescence as a proxy for AGE concentrations is questionable due to measurement variability associated with factors including skin pigmentation, presence of body creams and changes in blood flow.Citation67,Citation68 Nevertheless, skin autofluorescence is a quick, non-invasive measurement, and there is some evidence of a link between poor metabolic and muscle health through AGE accumulation within skeletal muscle. However, longitudinal studies assessing the effect of AGEs on components of sarcopenia using more robust measures of AGE concentrations (tissue biopsy and serum/urinary samples) are required.
Vascular complications
Micro- and macrovascular complications of T2DM result from chronic hyperglycemia, AGE accumulation, inflammation, and oxidative stress and have the capacity in their own right to affect skeletal muscle.Citation69,Citation70 Healthy vasculature is crucial for the transfer of nutrients, removal of waste products and oxygenation of tissues, including skeletal muscle. Micro- and macrovascular complications are strongly interconnected in T2DM.
Microvascular complications
Neuropathy, retinopathy, and nephropathy are common microvascular complications in T2DM. Damaged nerve cells innervating skeletal muscle can impair muscle contractility, resulting in reduced muscle strength. A recent study reported that in men and women aged >50 years with T2DM, individuals with neuropathy had lower knee extension strength relative to body weight compared to individuals without neuropathy.Citation71 In addition to impairing muscle strength, nerve damage also causes muscle atrophy.Citation72 Both of these muscle impairments contribute to poor physical performance. In support, a cross-sectional analysis of the Health ABC study including 2,364 older adults (aged 73–82 years) demonstrated that poor peripheral nerve function was independently associated with poor physical performance outcomes including Short Physical Performance Battery (SPPB) scores, gait speed and standing balance ratio.Citation73 In the same study, individuals with T2DM also had poorer physical performance when compared with controls, and although adjusting for peripheral nerve function attenuated the relationship between T2DM and physical performance measures, it still remained significant.Citation73 Thus, neuropathy is one of the various T2DM complications that contributes to poor muscle health and ultimately, sarcopenia.
Although diabetic retinopathy does not appear to inherently affect muscle function, it can affect vision, which is a key component of balance. Balance is important for both locomotion and physical performance. Unsurprisingly, impaired vision in T2DM contributes to increased falls risk.Citation74 In a cross-sectional analysis of the Singapore Epidemiology of Eye Diseases Study (n=9,481, aged 40–80 years), adults with T2DM and diabetic retinopathy had higher odds (OR:1.31 [95%CI:1.07–1.60]) of falling compared to individuals without diabetes in a fully adjusted model.Citation74 Additionally, individuals with diabetes and mild (OR:1.81 [95%CI:1.23–2.67]) and moderate (OR:1.89 [95%CI:1.16–3.07]) non-proliferative diabetic retinopathy had increased falls risk when compared to individuals that had diabetes without diabetic retinopathy.Citation74 Falls have an enormous economic burdenCitation75 and they contribute to a large proportion of global injury-related deaths.Citation76 There are very few studies that have explored the relationship between diabetic retinopathy and sarcopenia; however, a small cross-sectional study in 316 Japanese adults with T2DM (aged >20 years) reported that diabetic retinopathy increased odds for prevalent sarcopenia (OR:7.78 [95%CI:1.52–39.81]) and low muscle strength (OR:6.25 [95%CI:1.15–33.96]) and was negatively associated with muscle quality defined by hand grip strength relative to SMI (ß=−0.136; P=0.005).Citation77 Due to the small sample size of this study, there is insufficient evidence to conclude that there is a definitive relationship between diabetic retinopathy and sarcopenia. Further studies in large T2DM and sarcopenic populations that include measures of diabetic retinopathy are required.
Diabetic nephropathy causes chronic kidney disease (CKD),Citation78 which contributes to muscle wasting.Citation79 In 387 older Asians (mean age 68 years) with T2DM, diabetic nephropathy was associated with 2.5-fold (95%CI:1.35–5.13) higher odds of prevalent sarcopenia defined by AWGS criteria.Citation80 Sarcopenia prevalence has been reported to increase with CKD severityCitation81 and depending on the definition utilized, also predicts mortality in CKD patients.Citation82 Pathophysiological mechanisms that are thought to contribute to sarcopenia in CKD include increased inflammation and protein loss, reduced vitamin D synthesis, mitochondrial dysfunction, and metabolic acidosis.Citation79 In 100 CKD patients not on dialysis, high sensitivity CRP was associated with ALM/BMI (r=−0.268).Citation81 In another study of 148 adult CKD patients (aged ≥18 years) not on dialysis, glomerular filtration rate was positively associated with lean mass, appendicular SMI and ALM.Citation83 In addition to losses in muscle mass, respiratory capacity of muscle also appears to be lower in CKD, which could further contribute to development and/or progression of sarcopenia. Muscle biopsies taken in a cohort of adults with stage 5 CKD patients demonstrated that mitochondrial volume density is lower in individuals with CKD compared with non-CKD controls.Citation84 Targeting muscle mass and mitochondrial biogenesis may improve mortality rates in this population.
Macrovascular complications
A key macrovascular complication in T2DM is atherosclerosis. Atherosclerosis appears to progress faster in individuals with T2DM, as it is exacerbated by poor metabolic health.Citation85 Additionally, chronic hyperglycemia inhibits nitric oxide-mediated vasodilation through increased oxidative stress, AGE accumulation, and direct actions on endothelial nitric oxide synthase.Citation86 Both of these complications increase the risk for hypertension, which can lead to end organ damage.Citation87 In 365 hypertensive Turkish adults aged >18 years without T2DM, skeletal muscle mass was negatively associated with hypertensive retinopathy, a measure of end organ damage, in both adults and older adults.Citation88 However, in this same study, SMI was not associated with hypertensive end organ damage. It is plausible that hypertension can damage myocytes; however, there is also evidence that suggests muscle mass can influence blood pressure. A recent study demonstrated that increased muscle mass induced via myostatin knockout leads to reduced systolic and diastolic blood pressure and mean arterial pressure in an obese mouse model (db/db), independent of adiposity and body weight.Citation89 The positive effects of increased muscle mass on blood pressure were thought to be due to improved renal function resulting from metabolic improvements attributed to increased muscle mass. It is possible that increased muscle mass would confer similar vascular benefits in humans; however, this is difficult to determine given that muscle mass is currently increased in humans primarily through exercise. Hence, it is unclear whether increases in muscle mass, or other beneficial effects of exercise are improving vascular health, although both are likely to contribute. Nevertheless, interventions targeting muscle hypertrophy may have the additional benefit of improving vascular health.
Peripheral artery disease (PAD) is another macrovascular complication associated with T2DM that affects up to a quarter of diabetes patients.Citation90 PAD measured using ankle-brachial index (an indirect, non-invasive measure of PAD) has been associated with lower hip extension and knee and hip flexion strength in older adults.Citation70 It has also been reported that older adults with PAD have slower gait speed compared to controls without PAD.Citation91 Reduced blood flow in PAD can result in ischemia, leading to poorer muscle strength, mass and performance outcomes.Citation92 Also, PAD can lead to reduced physical activity and exercise due to the pain associated with this complication, which may further contribute to poor muscle health outcomes. Further studies investigating the relationship between micro- and macrovascular complications and sarcopenia outcomes including muscle composition and function are required in T2DM populations.
IMAT and T2DM
Muscle quality is an underappreciated component of sarcopenia.Citation9 Parallel to muscle loss with aging, numerous neuromuscular changes contribute significantly to reduced force-production capacity, including inter- and intra-muscular adipose tissue (IMAT). IMAT is an ectopic fat depot associated with poor metabolicCitation93 and muscle health outcomes.Citation94,Citation95 Obese and T2DM individuals have greater amounts of thigh IMAT compared with lean controls, and IMAT is associated with insulin sensitivity.Citation96 In overweight and obese non-diabetic adults, calf IMAT was negatively associated with insulin sensitivity in both men (r = −0.40) and women (r = −0.43) using gold-standard MRI and glucose clamp techniques.Citation93 In comparison, subcutaneous adipose tissue, a considerably larger fat depot, does not appear to predict insulin sensitivity.Citation96 In a cross-sectional analysis of the Look AHEAD study (n=183), overweight and obese men and women with T2DM had greater IMAT at higher levels of total-body adiposity when compared with non-diabetic overweight and obese controls.Citation97 In addition to influencing insulin sensitivity, various studies have reported that IMAT is an independent predictor of physical functionCitation94,Citation95 and falls risk.Citation98,Citation99 In fact, IMAT is a stronger predictor of mobility function in older adults than lean mass.Citation95 In a cross-sectional study (n=79), obese adults with T2DM and peripheral neuropathy had higher amounts of calf muscle IMAT and lower subcutaneous fat when compared with obese age-matched controls both with and without T2DM.Citation100 The authors suggested that IMAT may exacerbate peripheral neuropathy through increased concentrations of localized cytokines, including TNF-α and IL-6,Citation100 which have been implicated in disrupted nerve cell homeostasis and neuropathy.Citation101 IL-6 is associated with an increased loss of muscle mass and strength in T2DM,Citation46,Citation47 and this is likely due to direct effects on myocytes, alongside indirect effects on neurons and vasculature. Given that IMAT consists of non-contractile tissue, fat infiltration into skeletal muscle affects elastic properties of skeletal muscle.Citation102 It is clear that IMAT makes an important contribution to the relationship between sarcopenia and T2DM in older individuals, and should be considered as a key outcome measure in future interventional studies.
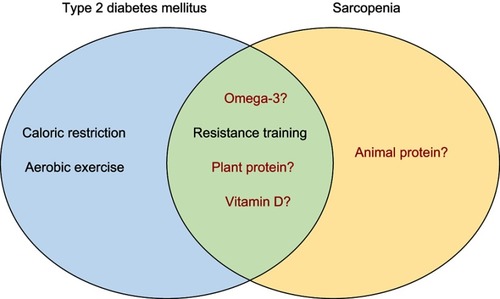
Impaired muscle function may contribute to high levels of sedentary behavior in T2DM patients and contribute to a positive feedback loop where functional impairments lead to metabolic impairments, and vice versa. This positive feedback loop can be broken using lifestyle interventions with proven effectiveness for improving metabolic and musculoskeletal health.
Treatment and prevention of T2DM and sarcopenia
T2DM is commonly treated using glucose-lowering medications. Some of these may influence muscle mass (), but for the majority, their effects on sarcopenia components are unclear. Metformin increases AMP-activated protein kinase activity,Citation103 which inhibits the mechanistic target of rapamycin (mTOR), a key factor in muscle growth.Citation104 However, metformin is being investigated as an adjunct therapy to resistance training in older adults,Citation105 as it may assist in the mobilization of M2 macrophages, which could have anti-inflammatory properties in skeletal muscle.Citation105 In the MrOS study, men with diabetes treated with metformin alone and metformin plus thiazolidinediones (insulin sensitizers) over 3.5±0.7 years follow-up had an attenuated loss of total lean mass and ALM compared to men with diabetes that were not treated with insulin sensitizers, and men with untreated diabetes.Citation106 Thus, it is unclear whether metformin has positive, negative, or negligible effects on muscle mass and strength in humans. Insulin has anabolic effects on muscle mass and protein metabolism in young and non-insulin resistant men and women;Citation32,Citation107 however, this does not seem to occur for individuals with T2DM.Citation32,Citation108 Indirectly, insulin therapy may improve muscle health by reducing blood glucose concentrations, which, as described above, can have negative effects on muscle through inflammation, oxidative stress and increased AGEs if chronically elevated. Sulfonylureas have been reported to cause muscle atrophy in a very small proportion of users;Citation109 however, to the best of our knowledge there are no clinical trials investigating this relationship in a controlled setting. Overall, there is a paucity of data pertaining to the relationship between glucose-lowering medication and muscle health in humans, but purported effects on body weight and muscle mass in T2DM are described in .
Table 2 Direct effect of glucose-lowering medications on body weight and muscle mass in type 2 diabetes
There are no approved pharmacological treatments for sarcopenia. Possible therapeutic targets include hormonal interventions such as a transdermal testosterone gelCitation116 and selective androgen receptor modulators.Citation117,Citation118 Transdermal testosterone gels increase serum concentrations of androgens, which have well-documented anabolic effects on skeletal muscle.Citation119 However, these have undesirable side effects in regions such as the prostate gland and seminal vesicles, and have virializing side effects in women.Citation120 Thus, selective androgen receptor modulators (SARMs) that only bind with androgen receptors within skeletal muscle are an attractive therapeutic.Citation121 SARMs have acceptable safety profiles, and positive effects on body composition and physical function in clinical trials.Citation117,Citation118 Other novel treatments for sarcopenia undergoing clinical trials in humans include activin type IIB receptor antagonistsCitation121 and myostatin blockades (NCT01963598), which inhibit myostatin action. Myostatin is a negative regulator of myogenesis, and its inhibition significantly increases skeletal muscle mass in various animal knockouts.Citation89,Citation122 Improving muscle mass, quality and function using these treatments could also lead to indirect improvements in metabolic health; however, further studies are required to establish their safety and efficacy. Even if pharmacological agents are approved for the treatment of sarcopenia in the future, lifestyle modification will likely remain the primary therapy for the management of T2DM and sarcopenia.
Physical activity and exercise
Higher levels of physical activity are protective against the development of T2DM and sarcopenia. In the US Diabetes Prevention Program which included 3,234 non-diabetic adults, a lifestyle modification program (low-calorie, diet-induced weight loss of 7% of initial body weight and ≥150 min moderate-intensity physical activity per week) resulted in 58% reduced incidence of T2DM over three years, which was significantly more effective than metformin, a first-line glucose-lowering drug.Citation123 In addition to reducing the incidence of T2DM, lifestyle interventions also achieve remission of T2DM.Citation124 In the Look AHEAD study, approximately 12% of individuals completing a lifestyle intervention (aerobic activity for ≥50 min/week in the first month and ≥175 min/week after 6 months, alongside a weight loss goal of ≥10%) experienced partial or complete remission of T2DM compared with diabetes support and education (control).Citation124 Similarly, a meta-analysis has reported that any engagement in physical activity reduces the likelihood of incident sarcopenia (OR:0.45 [95%CI:0.37–0.55]) in adults older than 40 years.Citation125 Recent findings from the Look AHEAD study demonstrated that the lifestyle intervention resulted in better lower-limb performance, and lower likelihood (adjusted OR: 0.84 [95%CI:0.71–0.99]) of having poor gait speed (<0.8 m/s) in individuals with T2DM after 11 years. However, lifestyle intervention did not improve hand grip strength when compared with controls.Citation126 This is unsurprising given aerobic exercise is generally not effective for improving muscle strength, despite its ability to improve metabolic health.Citation127
Indeed, a recent network meta-analysis including 2,208 individuals ≥18 years of age with T2DM reported that supervised aerobic and strength training were both effective at reducing HbA1c compared with no exercise.Citation127 However, the greatest reductions in HbA1c were experienced by individuals undertaking combined resistance and aerobic exercise.Citation127 Furthermore, a recent randomized control trial (RCT) in 137 pre-diabetic adults (mean age 59 years) reported that two-year supervised resistance training alone (HR:0.35 [95%CI:0.15‐0.79]) and in combination with aerobic training (HR:0.26 [95%CI:0.11‐0.62]) were as effective as aerobic training alone (HR:0.28 [95%CI:0.13‐0.64]) in preventing progression to T2DM.Citation128 Resistance training is the most effective strategy for improving both muscle mass and function in sarcopenic individuals,Citation129 and is similarly effective for improving muscle strength, size, and quality and metabolic health in older adults with T2DM.Citation130 Resistance training may therefore reduce the risk of developing sarcopenia in T2DM patients.
Combining resistance and aerobic training with diet-induced weight loss may be the most effective strategy for improving metabolic and musculoskeletal health in obese older adults. Resistance training may prevent or minimize muscle mass losses commonly associated with diet-induced weight loss,Citation131 which needs to be avoided in older adults at risk of sarcopenia, including those with T2DM. In dieting obese older adults, combined resistance and aerobic training led to a 21% improvement in physical performance,Citation132 and improved strength and preserved muscle mass more than aerobic training alone.Citation132 Similarly, in obese older adults intentionally losing weight over 18 months, resistance training combined with caloric restriction resulted in less loss of lean mass (−1.5%) than caloric restriction combined with aerobic training (−3.1%) and caloric restriction alone (−2.0%).Citation133 Nevertheless, the effect of exercise and diet-induced weight loss interventions on metabolic and muscle health in older adults with T2DM and sarcopenia is largely unexplored.
Diet
Caloric restriction is an effective strategy for weight loss in overweight and obese individuals.Citation134 While weight loss can lead to remission of T2DM,Citation135 as described above, it can lead to significant losses in muscle mass.Citation131 To an extent, muscle mass losses may be commensurate to decreased loading as a result of losses in fat mass; however, muscle mass losses during caloric restriction may also result from inadequate protein intake.
Protein is important for the maintenance and growth of skeletal muscle. In older adults, there is evidence to suggest that recommended daily intake (RDI) of protein should be between ~1.2–1.6 g/kg/day,Citation136 despite the RDI in many countries being ~0.8 g/kg/day.Citation137,Citation138 In the Health ABC study, older adults in the highest quintile of energy-adjusted protein intake (1.2±0.4 g/kg/day) lost approximately 40% less ALM and total lean mass over three years when compared with individuals in the lowest quintile for protein intake (0.8±0.3 g/kg/day).Citation139 Different protein sources may also affect the risk for incident T2DM. Findings from the Melbourne Collaborative Cohort Study (MCCS) and a meta-analysis showed that individuals in the highest animal and total protein intake category had increased pooled risk ratios for incident T2DM (RR:1.19 [95%CI:1.11–1.28] and RR:1.09 [95%CI:1.06–1.13], respectively), compared with the lowest intake category. Interestingly, individuals in the highest plant protein intake category had reduced, albeit not significantly, risk for incident T2DM (RR:0.95 [95%CI:0.89–1.02]) compared with individuals in the lowest intake category.Citation140 It was also reported that in the MCCS cohort alone (n=21,523), plant protein was negatively associated with incident T2DM in women (OR:0.60 [95%CI:0.37–0.99]), but not men (OR:1.26 [95%CI:0.75–2.13]). Thus, to ensure older adults with, or at risk of, T2DM are meeting protein requirements, increases in protein intake from primarily plant sources may be most appropriate. Plant protein appears to have a lower skeletal muscle anabolic response when compared with animal protein; however, proposed strategies to augment the anabolic properties of plant protein include fortification of plant-based protein sources with amino acids, selective breeding of plants to improve amino acid profiles, and consuming a combination of plant protein sources.Citation141
The efficacy of combining protein supplementation with resistance training to enhance exercise-related muscle outcomes in older adults with and without sarcopenia is still unclear. A recent meta-analysis by Morton et al demonstrated that protein supplementation was effective for improving lean mass in resistance-trained adults, but appears to have very little effect on muscle outcomes in older adults.Citation142 This meta-analysis also reported that protein intake beyond ~1.6 g/kg/day did not further increase changes in lean mass.Citation142 Interestingly, Beaudart et al recently reported that an additional effect of protein supplementation combined with exercise on muscle mass and strength in older adults with sarcopenia was only evident in 3/12 RCTs included in their systematic review.Citation143 Nevertheless, ensuring adequate protein intake is important for muscle maintenance and, potentially, growth, which in turn is beneficial for metabolic health.
Vitamin D may also affect metabolic and muscle health. Low vitamin D has been linked to poor physical functionCitation144 and glycemic control.Citation145 Vitamin D receptors are located within the pancreas and skeletal muscle, suggesting a role in both glucose and muscle homeostasis. A recent randomized controlled trial in older adults with low vitamin D concentrations (≥8 and ≤20 ng/mL) reported that vitamin D supplementation (800 IU/d) for 12 months did not affect lower-extremity power, strength or lean mass.Citation146 On the contrary, meta-analyses by Stockton et alCitation147 and Rabenda et alCitation148 suggest that vitamin D supplementation can improve muscle strength, but only in individuals with baseline 25-hydroxyvitamin D [25(OH)D] <25 and <30 nmol/L, respectively. Interestingly, vitamin D also shows potential as an adjunct therapy during exercise. In a meta-analysis by Antoniak and Greig, minor improvements in lower limb muscle strength (standard mean difference: 0.98 [95%CI: 0.73–1.24]) were evident with combined vitamin D and exercise versus exercise alone; however, for the timed up and go test (TUG), there were no additive effects of vitamin D combined with exercise compared with exercise alone.Citation149 Only three studies were included in the analysis of lower limb muscle strength, and two studies were included in the analysis of TUG, highlighting the need for further research into the effects of combined vitamin D supplementation and exercise. There is also a need for more RCTs investigating the effect of vitamin D on muscle in vitamin D deficient populations, both with and without T2DM and sarcopenia.
Regarding direct effects of vitamin D on metabolic health, a recent RCT utilizing gold-standard measures of insulin sensitivity and high-dose vitamin D supplementation (100,000 IU initial bolus; 4,000 IU/d) reported no effects on insulin sensitivity in 54 non-diabetic, vitamin D deficient ([25(OH)D] ≤50 nmol/L) overweight and obese adults.Citation150 Furthermore, a subgroup analysis (n=23) was performed in this study in participants with baseline 25(OH)D concentrations <30 nmol/L; however, there was no effect of vitamin D supplementation on insulin sensitivity.Citation150 It should be noted that this sub-analysis was likely underpowered. Also, vitamin D supplementation has been reported to have no effect on insulin sensitivity in vitamin D deficient individuals ([25(OH)D] ≤50 nmol/L) with T2DM.Citation151 Meta-analyses have reported conflicting findings; with vitamin D supplementation influencing glucose regulation in prediabetes and diabetesCitation145,Citation152–Citation154 but not in other populations.Citation155 Future studies in larger T2DM and non-T2DM populations with moderate to severe vitamin D deficiency ([25(OH)D] <30 or 25 nmol/L) may help establish whether there is an effect of vitamin D supplementation on various aspects of glucose metabolism, and whether this effect is limited to individuals with poorer metabolic health.
Lastly, omega-3 supplementation alone and in combination with exercise may improve metabolic and muscle health. Omega-3 supplementation can affect muscle directly by increasing muscle protein synthesisCitation156 and indirectly by decreasing systemic inflammation.Citation157 In 118 postmenopausal women, 6 months of fish oil-based supplementation (rich in omega-3 fatty acids) was associated with improved gait speed compared with placebo (3±16 vs -3.5±14%.Citation158 Furthermore, in 60 healthy older adults (aged 60–85 years), 6 months of fish oil-based omega-3 supplementation increased thigh muscle volume (mean change: 3.6% [95%CI:0.2–7.0%]), handgrip strength (mean change: 2.3 kg [95%CI:0.8–3.7 kg]) and composite 1 repetition maximum muscle strength scores (mean change: 4.0% [95%CI:0.8–7.3%]).Citation159 However, RCTs have also reported that omega-3 supplementation alone has no effect on muscle health in older populations.Citation160,Citation161 Similar to vitamin D supplementation, omega-3 supplementation has shown some potential as an adjunct therapy during exercise. In 51 older adults (mean age 64 years) that completed a 12-week resistance training program combined with flaxseed oil-derived alpha-linolenic acid (ALA) supplementation (rich in omega-3 fatty acids) or placebo, ALA supplementation resulted in a small increase in knee flexor muscle thickness, and a decrease in IL-6 concentrations in men.Citation162 Although, it is still unclear what type and dosage of omega-3 supplement is most effective for preventing and treating sarcopenia. Regarding the effect of omega-3 supplementation on metabolic health, studies assessing the effect of omega-3 supplementation on fasting glucose,Citation159 insulin resistance,Citation163 and T2DMCitation164 have reported little, or no effect. There are also limited data on nutritional interventions in individuals with comorbid T2DM and sarcopenia. Further studies in well-defined populations, particularly those with nutritional deficiencies, will help clarify the effect of the aforementioned nutritional interventions on metabolic and muscle health.
Conclusion
There are numerous bidirectional links between T2DM and sarcopenia, and the existence of one condition may increase the risk of developing the other. Factors such as insulin resistance, inflammation, AGE accumulation, increased oxidative stress and vascular complications can all affect various components of muscle health; and impaired muscle health can also contribute to development and progression of T2DM. The use of different operational definitions of sarcopenia clouds our understanding of this relationship, and therefore, development of a consensus definition is paramount. Evidence suggests that several lifestyle interventions (identified in ) can improve and maintain functional and metabolic health in individuals with T2DM and sarcopenia, but evidence for the most effective and feasible interventions in those with both conditions is lacking. Further research is needed to better understand, treat and prevent comorbid sarcopenia and T2DM, the beneficial outcomes of which would include increased independence and quality of life in these individuals.
Author contributions
All authors contributed to conception and design, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
JM is supported by a Research Training Program Scholarship. BDC is supported by a National Heart Foundation Future Leader Fellowship (100864). DS is supported by a National Health and Medical Research Council R.D. Wright Biomedical Career Development Fellowship (GNT1123014). The authors report no other conflicts of interest in this work.
References
- Cho NH , Shaw JE , Karuranga S , et al. IDF diabetes atlas: global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract . 2018;138:271–281. doi:10.1016/j.diabres.2018.02.023 29496507
- American Diabetes A. Diagnosis and classification of diabetes mellitus. Diabetes Care . 2009;32 Suppl 1(Suppl1):S62–S67. doi:10.2337/dc09-S062 19118289
- Harreiter J , Kautzky-Willer A , Pacini G . Sex and gender differences in risk, pathophysiology and complications of type 2 diabetes mellitus. Endocr Rev . 2016;37(3):278–316. doi:10.1210/er.2015-1137 27159875
- Briggs AM , Woolf AD , Dreinhöfer K , et al. Reducing the global burden of musculoskeletal conditions. Bull World Health Organ . 2018;96(5):366–368. doi:10.2471/BLT.17.204891 29875522
- St-Onge M-P , Gallagher D . Body composition changes with aging: the cause or the result of alterations in metabolic rate and macronutrient oxidation? Nutrition . 2010;26(2):152–155. doi:10.1016/j.nut.2009.07.004 20004080
- Hong S , Chang Y , Jung HS , Yun KE , Shin H , Ryu S . Relative muscle mass and the risk of incident type 2 diabetes: a cohort study. PLoS One . 2017;12(11):e0188650. doi:10.1371/journal.pone.0188650 29190709
- Wang Y , Rimm EB , Stampfer MJ , Willett WC , Hu FB . Comparison of abdominal adiposity and overall obesity in predicting risk of type 2 diabetes among men. Am J Clin Nutr . 2005;81(3):555–563.15755822
- Cruz-Jentoft AJ , Baeyens JP , Bauer JM , et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing . 2010;39(4):412–423.20392703
- Cruz-Jentoft AJ , Bahat G , Bauer J , et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing . 2018;48(1):16–31. doi:10.1093/ageing/afy169
- Studenski SA , Peters KW , Alley DE , et al. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci . 2014;69(5):547–558. doi:10.1093/gerona/glu010 24737557
- Fielding RA , Vellas B , Evans WJ , et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc . 2011;12(4):249–256. doi:10.1016/j.jamda.2011.01.003 21527165
- Chen LK , Liu LK , Woo J , et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc . 2014;15(2):95–101. doi:10.1016/j.jamda.2013.11.025 24461239
- Scott D , de Courten B , Ebeling PR . Sarcopenia: a potential cause and consequence of type 2 diabetes in Australia’s ageing population? Med J Aust . 2016;205(7):329–333.27681976
- Nowotny K , Jung T , Höhn A , Weber D , Grune T . Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules . 2015;5:194–222. doi:10.3390/biom5010194 25786107
- Amog K , Raina P , Mayhew AJ , et al. The prevalence of sarcopenia in community-dwelling older adults, an exploration of differences between studies and within definitions: a systematic review and meta-analyses. Age Ageing . 2018;48(1):48–56.
- Shafiee G , Keshtkar A , Soltani A , Ahadi Z , Larijani B , Heshmat R . Prevalence of sarcopenia in the world: a systematic review and meta- analysis of general population studies. J Diabetes Metab Disord . 2017;16:21. doi:10.1186/s40200-017-0302-x 28523252
- Murata Y , Kadoya Y , Yamada S , Sanke T . Sarcopenia in elderly patients with type 2 diabetes mellitus: prevalence and related clinical factors. Diabetol Int . 2018;9(2):136–142. doi:10.1007/s13340-017-0339-6 30603361
- Wang T , Feng X , Zhou J , et al. Type 2 diabetes mellitus is associated with increased risks of sarcopenia and pre-sarcopenia in Chinese elderly. Sci Rep . 2016;6:38937. doi:10.1038/srep38937 27958337
- Kim TN , Park MS , Yang SJ , et al. Prevalence and determinant factors of sarcopenia in patients with type 2 diabetes. Diabetes Care . 2010;33(7):1497. doi:10.2337/dc09-2310 20413515
- Park SW , Goodpaster BH , Lee JS , et al. Excessive loss of skeletal muscle mass in older adults with type 2 diabetes. Diabetes Care . 2009;32(11):1993–1997. doi:10.2337/dc09-0264 19549734
- Yang L , Smith L , Hamer M . Gender-specific risk factors for incident sarcopenia: 8-year follow-up of the English longitudinal study of ageing. J Epidemiol Community Health . 2019;73(1):86. doi:10.1136/jech-2018-211258 30368480
- Son JW , Lee SS , Kim SR , et al. Low muscle mass and risk of type 2 diabetes in middle-aged and older adults: findings from the KoGES. Diabetologia . 2017;60(5):865–872. doi:10.1007/s00125-016-4196-9 28102434
- Wu H , Liu M , Chi VTQ , et al. Handgrip strength is inversely associated with metabolic syndrome and its separate components in middle aged and older adults: a large-scale population-based study. Metabolism . 2019;93:61–67. doi:10.1016/j.metabol.2019.01.011 30690038
- Thiebaud D , Jacot E , DeFronzo RA , Maeder E , Jequier E , Felber JP . The effect of graded doses of insulin on total glucose uptake, glucose oxidation, and glucose storage in man. Diabetes . 1982;31(11):957–963.6757014
- Lee CG , Boyko EJ , Strotmeyer ES , et al. Association between insulin resistance and lean mass loss and fat mass gain in older men without diabetes mellitus. J Am Geriatr Soc . 2011;59(7):1217–1224. doi:10.1111/j.1532-5415.2011.03472.x 21718263
- Kuo CK , Lin LY , Yu YH , Wu KH , Kuo HK . Inverse association between insulin resistance and gait speed in nondiabetic older men: results from the U.S. National Health and Nutrition Examination Survey (NHANES) 1999-2002. BMC Geriatr . 2009;9:49. doi:10.1186/1471-2318-9-49 19922671
- Abbatecola AM , Ferrucci L , Ceda G , et al. Insulin resistance and muscle strength in older persons. J Gerontol A Biol Sci Med Sci . 2005;60(10):1278–1282.16282559
- Sherwani SI , Khan HA , Ekhzaimy A , Masood A , Sakharkar MK . Significance of HbA1c test in diagnosis and prognosis of diabetic patients. Biomark Insights . 2016;11:95–104. doi:10.4137/BMI.S38440 27398023
- Yoon JW , Ha Y-C , Kim KM , et al. Hyperglycemia is associated with impaired muscle quality in older men with diabetes: the Korean Longitudinal Study on Health and Aging. Diabetes Metab J . 2016;40(2):140–146. doi:10.4093/dmj.2016.40.2.140 27126884
- Kalyani RR , Metter EJ , Egan J , Golden SH , Ferrucci L . Hyperglycemia predicts persistently lower muscle strength with aging. Diabetes Care . 2015;38(1):82–90. doi:10.2337/dc14-1166 25392294
- Russell ST , Rajani S , Dhadda RS , Tisdale MJ . Mechanism of induction of muscle protein loss by hyperglycaemia. Exp Cell Res . 2009;315(1):16–25. doi:10.1016/j.yexcr.2008.10.002 18973755
- Pereira S , Marliss EB , Morais JA , Chevalier S , Gougeon R . Insulin resistance of protein metabolism in type 2 diabetes. Diabetes . 2008;57(1):56. doi:10.2337/db07-0887 17940118
- de Rekeneire N , Peila R , Ding J , et al. Diabetes, hyperglycemia, and inflammation in older individuals. Diabetes Care . 2006;29(8):1902. doi:10.2337/dc05-2327 16873800
- Vozarova B , Weyer C , Lindsay RS , Pratley RE , Bogardus C , Tataranni PA . High white blood cell count is associated with a worsening of insulin sensitivity and predicts the development of type 2 diabetes. Diabetes . 2002;51(2):455. doi:10.2337/diabetes.51.2.455 11812755
- Haddad F , Zaldivar F , Cooper DM , Adams GR . IL-6-induced skeletal muscle atrophy. J Appl Physiol . 2005;98(3):911–917. doi:10.1152/japplphysiol.01026.2004 15542570
- Goyal R , Faizy AF , Siddiqui SS , Singhai M . Evaluation of TNF-α and IL-6 levels in obese and non-obese diabetics: pre- and postinsulin effects. N Am J Med Sci . 2012;4(4):180–184. doi:10.4103/1947-2714.94944 22536561
- King DE , Mainous AG , Buchanan TA , Pearson WS . C-reactive protein and glycemic control in adults with diabetes. Diabetes Care . 2003;26(5):1535. doi:10.2337/diacare.26.5.1535 12716818
- Vozarova B , Weyer C , Hanson K , Tataranni PA , Bogardus C , Pratley RE . Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes Res . 2001;9(7):414–417. doi:10.1038/oby.2001.54 11445664
- Makki K , Froguel P , Wolowczuk I . Adipose tissue in obesity-related inflammation and insulin resistance: cells, cytokines, and chemokines. ISRN Inflamm . 2013;2013:139239. doi:10.1155/2013/139239 24455420
- Mesinovic J , McMillan BL , Shore-Lorenti C , De Courten B , Ebeling RP , Scott D . Metabolic syndrome and its associations with components of sarcopenia in overweight and obese older adults. J Clin Med . 2019;8:2. doi:10.3390/jcm8020145
- Bach E , Nielsen RR , Vendelbo MH , et al. Direct effects of TNF-α on local fuel metabolism and cytokine levels in the placebo-controlled, bilaterally infused human leg. Diabetes . 2013;62(12):4023. doi:10.2337/db12-0863 23835341
- Carey AL , Steinberg GR , Macaulay SL , et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes . 2006;55(10):2688–2697. doi:10.2337/db05-1404 17003332
- Serrano AL , Baeza-Raja B , Perdiguero E , Jardi M , Munoz-Canoves P . Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab . 2008;7(1):33–44. doi:10.1016/j.cmet.2007.11.011 18177723
- Payette H , Roubenoff R , Jacques PF , et al. Insulin-like growth factor-1 and interleukin 6 predict sarcopenia in very old community-living men and women: the Framingham Heart Study. J Am Geriatr Soc . 2003;51(9):1237–1243.12919235
- Alemán H , Esparza J , Ramirez FA , Astiazaran H , Payette H . Longitudinal evidence on the association between interleukin-6 and C-reactive protein with the loss of total appendicular skeletal muscle in free-living older men and women. Age Ageing . 2011;40(4):469–475. doi:10.1093/ageing/afr040 21565862
- Park SW , Goodpaster BH , Strotmeyer ES , et al. Accelerated loss of skeletal muscle strength in older adults with type 2 diabetes: the health, aging, and body composition study. Diabetes Care . 2007;30(6):1507–1512. doi:10.2337/dc06-2537 17363749
- Visser M , Pahor M , Taaffe DR , et al. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: the Health ABC Study. J Gerontol A Biol Sci Med Sci . 2002;57(5):M326–M332.11983728
- Hamer M , Molloy GJ . Association of C-reactive protein and muscle strength in the English Longitudinal Study of Ageing. Age (Dordr) . 2009;31(3):171–177. doi:10.1007/s11357-009-9097-0 19466582
- Gomes MJ , Martinez PF , Pagan LU , et al. Skeletal muscle aging: influence of oxidative stress and physical exercise. Oncotarget . 2017;8(12):20428–20440. doi:10.18632/oncotarget.14670 28099900
- Asmat U , Abad K , Ismail K . Diabetes mellitus and oxidative stress—A concise review. Saudi Pharm J . 2016;24(5):547–553. doi:10.1016/j.jsps.2015.03.013 27752226
- Aragno M , Mastrocola R , Catalano MG , Brignardello E , Danni O , Boccuzzi G . Oxidative stress impairs skeletal muscle repair in diabetic rats. Diabetes . 2004;53(4):1082.15047625
- Zacarias-Flores M , Sanchez-Rodriguez MA , Garcia-Anaya OD , Correa-Munoz E , Mendoza-Nunez VM . Relationship between oxidative stress and muscle mass loss in early postmenopause: an exploratory study. Endocrinol Diabetes Nutr . 2018;65(6):328–334. doi:10.1016/j.endinu.2018.01.009 29650435
- Scicchitano BM , Pelosi L , Sica G , Musarò A . The physiopathologic role of oxidative stress in skeletal muscle. Mech Ageing Dev . 2018;170:37–44. doi:10.1016/j.mad.2017.08.009 28851603
- Short KR , Bigelow ML , Kahl J , et al. Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A . 2005;102(15):5618–5623. doi:10.1073/pnas.0501559102 15800038
- Abbatecola AM , Paolisso G , Fattoretti P , et al. Discovering pathways of sarcopenia in older adults: a role for insulin resistance on mitochondria dysfunction. J Nutr Health Aging . 2011;15(10):890–895. doi:10.1007/s12603-011-0366-0 22159778
- Conley KE , Jubrias SA , Esselman PC . Oxidative capacity and ageing in human muscle. J Physiol . 2000;526 Pt 1(Pt1):203–210.10878112
- Bua E , Johnson J , Herbst A , et al. Mitochondrial DNA-deletion mutations accumulate intracellularly to detrimental levels in aged human skeletal muscle fibers. Am J Hum Genet . 2006;79(3):469–480. doi:10.1086/507132 16909385
- Schrauwen-Hinderling VB , Kooi ME , Hesselink MK , et al. Impaired in vivo mitochondrial function but similar intramyocellular lipid content in patients with type 2 diabetes mellitus and BMI-matched control subjects. Diabetologia . 2007;50(1):113–120. doi:10.1007/s00125-006-0475-1 17093944
- Morino K , Petersen KF , Dufour S , et al. Reduced mitochondrial density and increased IRS-1 serine phosphorylation in muscle of insulin-resistant offspring of type 2 diabetic parents. J Clin Invest . 2005;115(12):3587–3593. doi:10.1172/JCI25151 16284649
- Singh R , Barden A , Mori T , Beilin L . Advanced glycation end-products: a review. Diabetologia . 2001;44(2):129–146. doi:10.1007/s001250051591 11270668
- Haus JM , Carrithers JA , Trappe SW , Trappe TA . Collagen, cross-linking, and advanced glycation end products in aging human skeletal muscle. J Appl Physiol . 2007;103(6):2068–2076. doi:10.1152/japplphysiol.00670.2007 17901242
- Forbes JM , Sourris KC , de Courten MPJ , et al. Advanced glycation end products (AGEs) are cross-sectionally associated with insulin secretion in healthy subjects. Amino Acids . 2014;46(2):321–326. doi:10.1007/s00726-013-1542-9 23832534
- Mulder DJ , Water TV , Lutgers HL , et al. Skin autofluorescence, a novel marker for glycemic and oxidative stress-derived advanced glycation endproducts: an overview of current clinical studies, evidence, and limitations. Diabetes Technol Ther . 2006;8(5):523–535. doi:10.1089/dia.2006.8.523 17037967
- Mori H , Kuroda A , Ishizu M , et al. Association of accumulated advanced glycation end products with a high prevalence of sarcopenia and dynapenia in patients with type 2 diabetes. J Diabetes Investig . 2019;35:731–737. doi:10.1111/jdi.13014
- Momma H , Niu K , Kobayashi Y , et al. Skin advanced glycation end product accumulation and muscle strength among adult men. Eur J Appl Physiol . 2011;111(7):1545–1552. doi:10.1007/s00421-010-1779-x 21188413
- Tabara Y , Ikezoe T , Yamanaka M , et al. Advanced Glycation End Product Accumulation Is Associated with Low Skeletal Muscle Mass, Weak Muscle Strength, and Reduced Bone Density: The Nagahama Study. J Gerontol A Biol Sci Med Sci 2018. doi:10.1093/gerona/gly233
- Chaudhri S , Fan S , Davenport A . Pitfalls in the measurement of skin autofluorescence to determine tissue advanced glycosylation content in haemodialysis patients. Nephrology . 2013;18(10):671–675. doi:10.1111/nep.12118 23815495
- Noordzij MJ , Lefrandt JD , Graaff R , Smit AJ . Dermal factors influencing measurement of skin autofluorescence. Diabetes Technol Ther . 2011;13(2):165–170.21284484
- Fowler MJ . Microvascular and macrovascular complications of diabetes. Clin Diabetes . 2008;26(2):77.
- McDermott MM , Criqui MH , Greenland P , et al. Leg strength in peripheral arterial disease: associations with disease severity and lower-extremity performance. J Vasc Surg . 2004;39(3):523–530.14981443
- Nomura T , Ishiguro T , Ohira M , Ikeda Y . Diabetic polyneuropathy is a risk factor for decline of lower extremity strength in patients with type 2 diabetes. J Diabetes Investig . 2018;9(1):186–192.
- Andersen H , Gjerstad MD , Jakobsen J . Atrophy of foot muscles. Diabetes Care . 2004;27(10):2382.15451904
- Strotmeyer ES , de Rekeneire N , Schwartz AV , et al. The relationship of reduced peripheral nerve function and diabetes with physical performance in older white and black adults. Diabetes Care . 2008;31(9):1767.18535192
- Gupta P , Aravindhan A , Gand ATL , et al. Association between the severity of diabetic retinopathy and falls in an asian population with diabetes: the Singapore Epidemiology of Eye Diseases Study. JAMA Ophthalmol . 2017;135(12):1410–1416.29145583
- Haddad YK , Bergen G , Florence CS . Estimating the economic burden related to older adult falls by state. J Public Health Manag Pract . 2019;25(2):E17–E24.
- Haagsma JA , Graetz N , Bolliger I , et al. The global burden of injury: incidence, mortality, disability-adjusted life years and time trends from the Global Burden of Disease study 2013. Injury Prevention . 2016;22(1):3.26635210
- Fukuda T , Bouchi R , Takeuchi T , et al. Association of diabetic retinopathy with both sarcopenia and muscle quality in patients with type 2 diabetes: a cross-sectional study. BMJ Open Diabetes Res Care . 2017;5(1):e000404.
- Collins AJ , Foley RN , Gilbertson DT , Chen S-C . United States renal data system public health surveillance of chronic kidney disease and end-stage renal disease. Kidney Int Suppl . 2015;5(1):2–7.
- Wang XH , Mitch WE . Mechanisms of muscle wasting in chronic kidney disease. Nat Rev Nephrol . 2014;10(9):504–516.24981816
- Fung FY , Koh YLE , Malhotra R , et al. Prevalence of and factors associated with sarcopenia among multi-ethnic ambulatory older Asians with type 2 diabetes mellitus in a primary care setting. BMC Geriatr . 2019;19(1):122.31035928
- VAd S , Oliveira D , Barbosa SR , et al. Sarcopenia in patients with chronic kidney disease not yet on dialysis: analysis of the prevalence and associated factors. PLoS One . 2017;12(4):e0176230–e0176230.28448584
- Pereira RA , Cordeiro AC , Avesani CM , et al. Sarcopenia in chronic kidney disease on conservative therapy: prevalence and association with mortality. Nephrol Dial Transplant . 2015;30(10):1718–1725.25999376
- Zhou Y , Hellberg M , Svensson P , Hoglund P , Clyne N . Sarcopenia and relationships between muscle mass, measured glomerular filtration rate and physical function in patients with chronic kidney disease stages 3-5. Nephrol Dial Transplant . 2018;33(2):342–348.28340152
- Gamboa JL , Billings F , Bojanowski MT , et al. Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiol Rep . 2016;4(9):e12780.27162261
- Martín-Timón I , Sevillano-Collantes C , Segura-Galindo A , Del Cañizo-Gómez FJ . Type 2 diabetes and cardiovascular disease: have all risk factors the same strength? World J Diabetes . 2014;5(4):444–470.25126392
- Singleton JR , Smith AG , Russell JW , Feldman EL . Microvascular complications of impaired glucose tolerance. Diabetes . 2003;52(12):2867.14633845
- McMaster William G , Kirabo A , Madhur Meena S , Harrison David G . Inflammation, immunity, and hypertensive end-organ damage. Circ Res . 2015;116(6):1022–1033.25767287
- Doğan MH , Karadag B , Ozyigit T , Kayaoglu S , Ozturk AO , Altuntas Y . Correlations between sarcopenia and hypertensive target organ damage in a Turkish cohort. Acta Clin Belg . 2012;67(5):328–332.23189539
- Butcher Joshua T , Mintz James D , Larion S , et al. Increased muscle mass protects against hypertension and renal injury in obesity. J Am Heart Assoc . 2018;7(16):e009358.30369309
- Lange S , Diehm C , Darius H , et al. High prevalence of peripheral arterial disease and low treatment rates in elderly primary care patients with diabetes. Exp Clin Endocrinol Diabetes . 2004;112(10):566–573.15578331
- Dziubek W , Bulińska K , Stefańska M , et al. Peripheral arterial disease decreases muscle torque and functional walking capacity in elderly. Maturitas . 2015;81(4):480–486.26119244
- McDermott Mary M . Lower extremity manifestations of peripheral artery disease. Circ Res . 2015;116(9):1540–1550.25908727
- Boettcher M , Machann J , Stefan N , et al. Intermuscular adipose tissue (IMAT): association with other adipose tissue compartments and insulin sensitivity. J Magn Reson Imaging . 2009;29(6):1340–1345.19422021
- Scott D , Shore-Lorenti C , McMillan LB , et al. Calf muscle density is independently associated with physical function in overweight and obese older adults. J Musculoskelet Neuronal Interact . 2018;18(1):9–17.29504574
- Marcus RL , Addison O , Dibble LE , Foreman KB , Morrell G , LaStayo P . Intramuscular adipose tissue, sarcopenia, and mobility function in older individuals. J Aging Res . 2012. doi:10.1155/2012/629637
- Goodpaster BH , Thaete FL , Kelley DE . Thigh adipose tissue distribution is associated with insulin resistance in obesity and in type 2 diabetes mellitus. Am J Clin Nutr . 2000;71(4):885–892.10731493
- Gallagher D , Kelley DE , Yim J-E , et al. Adipose tissue distribution is different in type 2 diabetes. Am J Clin Nutr . 2009;89(3):807–814.19158213
- Scott D , Johansson J , McMillan LB , Ebeling PR , Nordstrom A , Nordstrom P . Mid-calf skeletal muscle density and its associations with physical activity, bone health and incident 12-month falls in older adults: the Healthy Ageing Initiative. Bone . 2018;120:446–451. doi:10.1016/j.bone.2018.12.004
- Frank-Wilson AW , Farthing JP , Chilibeck PD , et al. Lower leg muscle density is independently associated with fall status in community-dwelling older adults. Osteoporos Int . 2016;27(7):2231–2240.26879201
- Bittel DC , Bittel AJ , Tuttle LJ , et al. Adipose tissue content, muscle performance and physical function in obese adults with type 2 diabetes mellitus and peripheral neuropathy. J Diabetes Complications . 2015;29(2):250–257. doi:10.1016/j.jdiacomp.2014.11.003 25547717
- Skundric DS , Lisak RP . Role of neuropoietic cytokines in development and progression of diabetic polyneuropathy: from glucose metabolism to neurodegeneration. Exp Diabesity Res . 2003;4(4):303–312. doi:10.1155/EDR.2003.303 14668051
- Csapo R , Malis V , Sinha U , Du J , Sinha S . Age-associated differences in triceps surae muscle composition and strength – an MRI-based cross-sectional comparison of contractile, adipose and connective tissue. BMC Musculoskelet Disord . 2014;15(1):209. doi:10.1186/1471-2474-15-209 24939372
- Musi N , Hirshman MF , Nygren J , et al. Metformin increases AMP-activated protein kinase activity in skeletal muscle of subjects with type 2 diabetes. Diabetes . 2002;51(7):2074–2081. doi:10.2337/diabetes.51.7.2074 12086935
- Mihaylova MM , Shaw RJ . The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol . 2011;13(9):1016–1023. doi:10.1038/ncb2329 21892142
- Long DE , Peck BD , Martz JL , et al. Metformin to Augment Strength Training Effective Response in Seniors (MASTERS): study protocol for a randomized controlled trial. Trials . 2017;18(1):192. doi:10.1186/s13063-017-1932-5 28441958
- Lee CG , Boyko EJ , Barrett-Connor E , et al. Insulin sensitizers may attenuate lean mass loss in older men with diabetes. Diabetes Care . 2011;34(11):2381. doi:10.2337/dc11-1032 21926282
- Cetrone M , Mele A , Tricarico D . Effects of the antidiabetic drugs on the age-related atrophy and sarcopenia associated with diabetes type II. Curr Diabetes Rev . 2014;10(4):231–237.25245021
- Fanzani A , Conraads VM , Penna F , Martinet W . Molecular and cellular mechanisms of skeletal muscle atrophy: an update. J Cachexia Sarcopenia Muscle . 2012;3(3):163–179. doi:10.1007/s13539-012-0074-6 22673968
- Mele A , Calzolaro S , Cannone G , Cetrone M , Conte D , Tricarico D . Database search of spontaneous reports and pharmacological investigations on the sulfonylureas and glinides-induced atrophy in skeletal muscle. Pharmacol Res Perspect . 2014;2(1):e00028–e00028. doi:10.1002/prp2.28 25505577
- Diabetes Prevention Program Research G. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care . 2012;35(4):731–737. doi:10.2337/dc11-1299 22442396
- van Dieren S , Czernichow S , Chalmers J , et al. Weight changes and their predictors amongst 11 140 patients with type 2 diabetes in the ADVANCE trial. Diabetes Obes Metab . 2012;14(5):464–469. doi:10.1111/j.1463-1326.2012.01556.x 22226008
- Fujita S , Rasmussen BB , Cadenas JG , Grady JJ , Volpi E . Effect of insulin on human skeletal muscle protein synthesis is modulated by insulin-induced changes in muscle blood flow and amino acid availability. Am J Physiol Endocrinol Metab . 2006;291(4):E745–E754. doi:10.1152/ajpendo.00271.2005 16705054
- Raz I , Chen Y , Wu M , et al. Efficacy and safety of sitagliptin added to ongoing metformin therapy in patients with type 2 diabetes. Curr Med Res Opin . 2008;24(2):537–550. doi:10.1185/030079908X260925 18194595
- Scheen AJ . Pharmacodynamics, efficacy and Safety of Sodium–glucose Co-Transporter Type 2 (SGLT2) inhibitors for the treatment of type 2 diabetes mellitus. Drugs . 2015;75(1):33–59. doi:10.1007/s40265-014-0337-y 25488697
- Zinman B , Hoogwerf BJ , Duran Garcia S , et al. The effect of adding exenatide to a thiazolidinedione in suboptimally controlled type 2 diabetes: a randomized trial. Ann Intern Med . 2007;146(7):477–485.17404349
- Atkinson RA , Srinivas-Shankar U , Roberts SA , et al. Effects of testosterone on skeletal muscle architecture in intermediate-frail and frail elderly men. J Gerontol A Biol Sci Med Sci . 2010;65(11):1215–1219. doi:10.1093/gerona/glq118 20601412
- Dalton JT , Barnette KG , Bohl CE , et al. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled phase II trial. J Cachexia Sarcopenia Muscle . 2011;2(3):153–161. doi:10.1007/s13539-011-0034-6 22031847
- Papanicolaou DA , Ather SN , Zhu H , et al. A phase IIA randomized, placebo-controlled clinical trial to study the efficacy and safety of the selective androgen receptor modulator (SARM), MK-0773 in female participants with sarcopenia. J Nutr Health Aging . 2013;17(6):533–543. doi:10.1007/s12603-013-0335-x 23732550
- Neto WK , Gama EF , Rocha LY , et al. Effects of testosterone on lean mass gain in elderly men: systematic review with meta-analysis of controlled and randomized studies. Age (Dordr) . 2015;37(1):9742. doi:10.1007/s11357-014-9742-0 25637335
- Narayanan R , Coss CC , Dalton JT . Development of selective androgen receptor modulators (SARMs). Mol Cell Endocrinol . 2018;465:134–142. doi:10.1016/j.mce.2017.06.013 28624515
- Becker C , Lord SR , Studenski SA , et al. Myostatin antibody (LY2495655) in older weak fallers: a proof-of-concept, randomised, phase 2 trial. Lancet Diabetes Endocrinol . 2015;3(12):948–957. doi:10.1016/S2213-8587(15)00298-3 26516121
- Lv Q , Yuan L , Deng J , et al. Efficient generation of myostatin gene mutated rabbit by CRISPR/Cas9. Sci Rep . 2016;6:25029. doi:10.1038/srep25029 27113799
- Knowler WC , Barrett-Connor E , Fowler SE , et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med . 2002;346(6):393–403. doi:10.1056/NEJMoa012512 11832527
- Gregg EW , Chen H , Wagenknecht LE , et al. Association of an intensive lifestyle intervention with remission of type 2 diabetes. JAMA . 2012;308(23):2489–2496. doi:10.1001/jama.2012.67929 23288372
- Steffl M , Bohannon RW , Sontakova L , Tufano JJ , Shiells K , Holmerova I . Relationship between sarcopenia and physical activity in older people: a systematic review and meta-analysis. Clin Interv Aging . 2017;12:835–845. doi:10.2147/CIA.S132940 28553092
- Houston DK , Neiberg RH , Miller ME , et al. Physical function following a long-term lifestyle intervention among middle aged and older adults with type 2 diabetes: the look AHEAD study. J Gerontol Series A . 2018;73(11):1552–1559. doi:10.1093/gerona/glx204
- Pan B , Ge L , Xun Y-Q , et al. Exercise training modalities in patients with type 2 diabetes mellitus: a systematic review and network meta-analysis. Int J Behav Nutr Phys Act . 2018;15(1):72. doi:10.1186/s12966-018-0703-3 30045740
- Dai X , Zhai L , Chen Q , et al. Two-year-supervised resistance training prevented diabetes incidence in people with prediabetes: a randomised control trial. Diabetes Metab Res Rev . 2019;e3143. doi:10.1002/dmrr.3143 30768758
- Vlietstra L , Hendrickx W , Waters DL . Exercise interventions in healthy older adults with sarcopenia: a systematic review and meta-analysis. Australas J Ageing . 2018;37(3):169–183. doi:10.1111/ajag.12521 29638028
- Hovanec N , Sawant A , Overend TJ , Petrella RJ , Vandervoort AA . Resistance training and older adults with type 2 diabetes mellitus: strength of the evidence. J Aging Res . 2012;2012:284635. doi:10.1155/2012/284635 22988507
- Sardeli A , Komatsu T , Mori M , Gáspari A , Chacon-Mikahil M . Resistance training prevents muscle loss induced by caloric restriction in obese elderly individuals: a systematic review and meta-analysis. Nutrients . 2018;10(4):423. doi:10.3390/nu10040423
- Villareal DT , Aguirre L , Gurney AB , et al. Aerobic or resistance exercise, or both, in dieting obese older adults. N Engl J Med . 2017;376(20):1943–1955. doi:10.1056/NEJMoa1616338 28514618
- Beavers KM , Ambrosius WT , Rejeski WJ , et al. Effect of exercise type during intentional weight loss on body composition in older adults with obesity. Obesity (Silver Spring) . 2017;25(11):1823–1829. doi:10.1002/oby.21977 29086504
- Greco M , Chiefari E , Montalcini T , et al. Early effects of a hypocaloric, mediterranean diet on laboratory parameters in obese individuals. Mediators Inflamm . 2014;2014:750860. doi:10.1155/2014/750860 24729662
- Taylor R , Al-Mrabeh A , Zhyzhneuskaya S , et al. Remission of human type 2 diabetes requires decrease in liver and pancreas fat content but is dependent upon capacity for β cell recovery. Cell Metab . 2018;28(4):547–556. doi:10.1016/j.cmet.2018.07.003 30078554
- Phillips S , Chevalier S , Leidy H . Protein “Requirements” beyond the RDA: Implications for Optimizing Health. Appl Physiol Nutr Metab 2016;41(5): 565–572. doi:10.1139/apnm-2015-0550
- Trumbo P , Schlicker S , Yates AA , Poos M . Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J Am Diet Assoc . 2002;102(11):1621–1630.12449285
- Nowson C , O’Connell S . Protein requirements and recommendations for older people: a review. Nutrients . 2015;7(8):6874–6899. doi:10.3390/nu7085311 26287239
- Health ABCS , Newman AB , Nicklas BJ , et al. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: the Health, Aging, and Body Composition (Health ABC) Study. Am J Clin Nutr . 2008;87(1):150–155. doi:10.1093/ajcn/87.1.150 18175749
- Shang X , Scott D , Sanders KM , et al. Dietary protein intake and risk of type 2 diabetes: results from the Melbourne Collaborative Cohort Study and a meta-analysis of prospective studies. Am J Clin Nutr . 2016;104(5):1352–1365. doi:10.3945/ajcn.116.140954 27629053
- van Vliet S , Burd NA , van Loon LJC . The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J Nutr . 2015;145(9):1981–1991. doi:10.3945/jn.114.204305 26224750
- Morton RW , Murphy KT , McKellar SR , et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br J Sports Med . 2018;52(6):376.28698222
- Beaudart C , Dawson A , Shaw SC , et al. Nutrition and physical activity in the prevention and treatment of sarcopenia: systematic review. Osteoporos Int . 2017;28(6):1817–1833. doi:10.1007/s00198-017-3980-9 28251287
- Dang M , Shore-Lorenti C , McMillan BL , et al. Associations of serum 25-hydroxyvitamin D with physical performance and bone health in overweight and obese older adults. Int J Environ Res Public Health . 2019;16(3). doi:10.3390/ijerph16030509.
- Mirhosseini N , Vatanparast H , Mazidi M , Kimball SM . Vitamin D supplementation, glycemic control, and insulin resistance in prediabetics: a meta-analysis. J Endocr Soc . 2018;2(7):687–709. doi:10.1210/js.2017-00472 29951596
- Shea MK , Fielding RA , Dawson-Hughes B . The effect of vitamin D supplementation on lower-extremity power and function in older adults: a randomized controlled trial. Am J Clin Nutr . 2019;109(2):369–379. doi:10.1093/ajcn/nqy290 30715090
- Stockton KA , Mengersen K , Paratz JD , Kandiah D , Bennell KL . Effect of vitamin D supplementation on muscle strength: a systematic review and meta-analysis. Osteoporos Int . 2011;22(3):859–871. doi:10.1007/s00198-010-1407-y 20924748
- Rabenda V , Buckinx F , Slomian J , et al. The effects of vitamin D on skeletal muscle strength, muscle mass, and muscle power: a systematic review and meta-analysis of randomized controlled trials. J Clin Endocrinol Metab . 2014;99(11):4336–4345. doi:10.1210/jc.2014-1742 25033068
- Antoniak AE , Greig CA . The effect of combined resistance exercise training and vitamin D(3) supplementation on musculoskeletal health and function in older adults: a systematic review and meta-analysis. BMJ Open . 2017;7(7):e014619–e014619. doi:10.1136/bmjopen-2016-014619
- Mousa A , Naderpoor N , de Courten MP , et al. Vitamin D supplementation has no effect on insulin sensitivity or secretion in vitamin D-deficient, overweight or obese adults: a randomized placebo-controlled trial. Am J Clin Nutr . 2017;105(6):1372–1381. doi:10.3945/ajcn.117.152736 28490514
- Gulseth HL , Wium C , Angel K , Eriksen EF , Birkeland KI . Effects of vitamin D supplementation on insulin sensitivity and insulin secretion in subjects with type 2 diabetes and vitamin D deficiency: a randomized controlled trial. Diabetes Care . 2017;40(7):872–878. doi:10.2337/dc16-2302 28468770
- Lee CJ , Iyer G , Liu Y , et al. The effect of vitamin D supplementation on glucose metabolism in type 2 diabetes mellitus: a systematic review and meta-analysis of intervention studies. J Diabetes Complications . 2017;31(7):1115–1126. doi:10.1016/j.jdiacomp.2017.04.019 28483335
- Wu C , Qiu S , Zhu X , Li L . Vitamin D supplementation and glycemic control in type 2 diabetes patients: a systematic review and meta-analysis. Metabolism . 2017;73:67–76. doi:10.1016/j.metabol.2017.05.006 28732572
- Hu Z , Chen J , Sun X , Wang L , Wang A . Efficacy of vitamin D supplementation on glycemic control in type 2 diabetes patients: a meta-analysis of interventional studies. Medicine (Baltimore) . 2019;98(14):e14970. doi:10.1097/MD.0000000000014970 30946322
- Jamka M , Woźniewicz M , Jeszka J , Mardas M , Bogdański P , Stelmach-Mardas M . The effect of vitamin D supplementation on insulin and glucose metabolism in overweight and obese individuals: systematic review with meta-analysis. Sci Rep . 2015;5:16142. doi:10.1038/srep16142 26543018
- Smith GI , Atherton P , Reeds DN , et al. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: a randomized controlled trial. Am J Clin Nutr . 2011;93(2):402–412. doi:10.3945/ajcn.110.005611 21159787
- Kiecolt-Glaser JK , Belury MA , Andridge R , Malarkey WB , Hwang BS , Glaser R . Omega-3 supplementation lowers inflammation in healthy middle-aged and older adults: A randomized controlled trial. Brain Behav Immun . 2012;26(6):988–995. doi:10.1016/j.bbi.2012.05.011 22640930
- Hutchins-Wiese HL , Kleppinger A , Annis K , et al. The impact of supplemental n-3 long chain polyunsaturated fatty acids and dietary antioxidants on physical performance in postmenopausal women. J Nutr Health Aging . 2013;17(1):76–80. doi:10.1007/s12603-012-0415-3 23299384
- Smith GI , Julliand S , Reeds DN , Sinacore DR , Klein S , Mittendorfer B . Fish oil-derived n-3 PUFA therapy increases muscle mass and function in healthy older adults. Am J Clin Nutr . 2015;102(1):115–122. doi:10.3945/ajcn.114.105833 25994567
- Krzyminska-Siemaszko R , Czepulis N , Lewandowicz M , et al. The effect of a 12-week omega-3 supplementation on body composition, muscle strength and physical performance in elderly individuals with decreased muscle mass. Int J Environ Res Public Health . 2015;12(9):10558–10574. doi:10.3390/ijerph120910558 26343698
- Sneddon AA , Tsofliou F , Fyfe CL , et al. Effect of a conjugated linoleic acid and omega-3 fatty acid mixture on body composition and adiponectin. Obesity (Silver Spring) . 2008;16(5):1019–1024. doi:10.1038/oby.2008.41 18356842
- Cornish SM , Chilibeck PD . Alpha-linolenic acid supplementation and resistance training in older adults. Appl Physiol Nutr Metab . 2009;34(1):49–59. doi:10.1139/H08-136 19234585
- Gharekhani A , Dashti-Khavidaki S , Lessan-Pezeshki M , Khatami MR . Potential effects of omega-3 fatty acids on insulin resistance and lipid profile in maintenance hemodialysis patients: a randomized placebo-controlled trial. Iran J Kidney Dis . 2016;10(5):310–318.27721230
- Wu JHY , Micha R , Imamura F , et al. Omega-3 fatty acids and incident type 2 diabetes: a systematic review and meta-analysis. Br J Nutr . 2012;107 Suppl 2(0 2):S214–S227. doi:10.1017/S0007114512001602 22591895