Abstract
Introduction
Many stroke patients experience loss of arm function requiring rehabilitation, which is expensive, repetitive, and does not always translate into “real life.” Nintendo Wii Sports™ (Wii™) may offer task-specific training that is repetitive and motivating. The Trial of Wii™ in Stroke (TWIST) is designed to investigate feasibility, efficacy, and acceptability using Wii™ to improve affected arm function for patients after stroke.
Method
This is a randomized controlled trial (RCT), incorporating a qualitative study and health economics analysis that compares playing Wii™ versus arm exercises in patients receiving standard rehabilitation in a home setting within 6 months of stroke with a motor deficit of less than 5 on the MRC (Medical Research Council) scale (arm). In this study, we expect to randomize 240 participants.
Outcome measures
Primary outcome is change in affected arm function at 6 weeks follow-up in intervention and control group using the Action Research Arm Test. Secondary outcomes include occupational performance using the Canadian Occupational Performance Measure, quality of life using the Stroke Impact Scale, cost effectiveness analysis, and a qualitative study investigating factors that influence use of Wii™ for patients and carers.
Conclusion
TWIST is the first UK RCT assessing the feasibility, cost effectiveness, and acceptability of Wii™ in stroke rehabilitation. The trial has been registered with ISRCTN 06807619 and UK CRN 11030. Results of the study will be published after completion of study in August 2014.
Keywords:
Introduction
About 70% of stroke patients experience altered arm function, and about 40% are left with a nonfunctional arm.Citation1 Arm weakness limits the individual’s activities of daily living and impacts negatively on quality of life.Citation2
The greatest recovery in upper limb function is achieved in the 6 months following a stroke, and this period offers the best opportunity for rehabilitation interventions. The main predictor of success in physical therapy is the amount of therapy undertaken, and guidelines for patients recommend high intensity repetitive motions.Citation3 Rehabilitative therapy has focused on practicing “normal” movements to encourage return of relevant function and inhibit compensatory habits that may prevent further recovery. More recently, there has been an emphasis on strengthening, practice of functional tasks, and forcing use of the weak limb to complete everyday activities using constraint-induced movement therapy.Citation4–Citation6 The National Stroke Guidelines endorse 45 minutes of daily physiotherapy in the rehabilitation phase of stroke recovery.Citation1 Physiotherapy, however, is both labor-intensive and expensive, and service provision varies widely. At present, only 50% of people with stroke receive the rehabilitation to meet their needs during the crucial first 6 months.Citation1 Furthermore, although therapists prescribe a home exercise regimen, only about 31% of patients complete the recommended exercise.Citation7
Rehabilitation after stroke using virtual reality therapy is of increasing interest.Citation8–Citation11 A Cochrane review on the effect of virtual reality on arm function found limited evidence that it may be beneficial in improving arm function and activities of daily living when compared with the same dose of conventional therapy. The authors’ conclusion was that present studies were too small and too few to draw conclusions and more randomized controlled trials (RCTs) were needed.Citation12
Nintendo Wii Sports™ (Wii™) is commercially available and offers task specific training that is repetitive and motivatingCitation13,Citation14 and may be a useful adjunct to rehabilitation.Citation15–Citation17 Wii™ games include tennis, golf, boxing, baseball, and bowling activities, which are played using a remote control held in the same way that real world objects such as the handle of a racket are held and manipulated. An accelerometer measures the person’s movements and maps them to the screen. Visual, auditory, and haptic feedbacks contribute to maintaining users’ attention. The motivational and entertainment value of the Wii™ is reflected in its commercial success. The research into its efficacy is limited, although it is being used by stroke therapists.Citation11 Pilot studies have suggested faster task performance following use of Wii™, with no Wii™-related side effects or injuries.Citation16,Citation17
Methods
The Trial of Wii™ in Stroke (TWIST) is a multicenter, pragmatic, parallel group, RCT with blinded outcome assessment, incorporating a qualitative study and cost-effectiveness analysis (). Inclusion and exclusion criteria for study participants are listed in the table.
Table 1 Exclusion and inclusion criteria
Outcome measures
The primary outcome in the RCT is arm function as measured by the Action Research Arm Test (ARAT) at 6 weeks.Citation18 The ARAT assesses ability to handle objects and tests grasp, grip, pinch, and gross movement of elbow and shoulder of the affected arm.Citation19,Citation20
Secondary outcome measures include occupational performance, stroke impact, and severity. The Canadian Occupational Performance Measure (COPM) is used to detect change in self-perception of occupational performance over time.Citation21 The Stroke Impact Scale (SIS) assesses change in impairments, disabilities, and handicaps following a stroke.Citation22 The Modified Rankin Scale (mRankin) measures disability following stroke.Citation23 These outcome measures will be applied at baseline, 6 weeks, and 6 months. Arm function will be assessed at 6 months using the ARAT and Motor Activity Log-14 (MAL14). The MAL14 ascertains how well the participant has performed day to day tasks using their affected arm.Citation24,Citation25 Participants will be asked to complete a diary to capture a daily record of duration of arm exercises and adverse events. The diary is brief and enables the participant or carer to record daily exercise time, adverse events, and visits from the National Health Service (NHS), private carers, and social services staff.
Baseline data collection will include demographic data, type of stroke using the Oxfordshire Community Stroke Project Classification,Citation26 affected arm strength using the Medical Research Council Scale of Muscle Strength,Citation27 and cognitive function using the Montreal Cognitive Assessment.Citation28
Outcome measures will be purchased (ARAT, COPM) or permission to use in study will be obtained from authors (MAL14, EQ-5D™ 3L, mRankin, and SIS).
The qualitative study will explore participants’ and carers’ experiences of using the Wii™ and barriers to its implementation.
The health economic analysis will estimate the incremental cost per quality-adjusted life year (QALY) gained from the cost perspective of health care and personal social services.
Recruitment and flow through study
A total of 240 participants will be recruited to the RCT across ten sites in the UK by the local research practitioners.
Once participants have given written informed consent, outcome measures will be assessed at baseline, 6 weeks, and 6 months. The research practitioner conducting the outcome measures will be blinded to study group allocation.
A research therapist will either install the Wii™ in the participant’s home and instruct them on the use of the Wii™, or will advise the participant on tailored arm exercises to be performed during the 6-week intervention phase. All participants will be instructed to perform warm-up exercises prior to tailored arm exercises or Wii™ exercises to minimize the risk of injury. Written instructions for both groups will be left with the participants. The research therapist will visit the patient prior to the 6-week assessment to collect participant’s diary and Wii™ if applicable. The research therapist will remind participants not to disclose their treatment allocation during the subsequent assessment visits by the research practitioner.
Participants will be telephoned by a non-blinded member of the local research team on a weekly basis to encourage them to complete their diary and enable them to express any concerns they may have had about the study.
All research staff will be trained at site initiation in all outcome measures use of the trial website, case report form (CRF) completion and safety considerations. Training DVDs and telephone conferencing will be made available. Please see for study flow diagram.
Figure 1 Participant flow diagram.
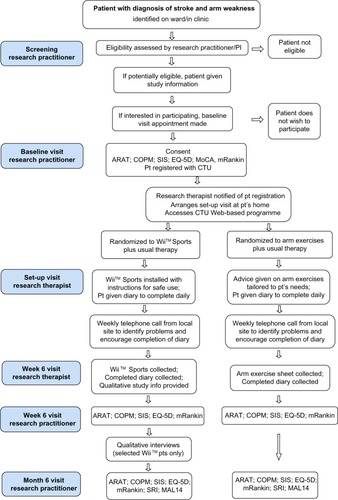
Trial intervention
Eligible participants with arm weakness following stroke will be randomized to one of two groups.
The intervention group will be instructed to undertake up to 45 minutes of exercise, in a seated position, using the Wii™ game daily for 6 weeks at home. The Wii™ console and game will be provided on loan to trial participants as part of the study for the duration of the intervention period.
The control group will be instructed to undertake up to 45 minutes of tailored arm exercises, in a seated position, daily for 6 weeks at home.
Both groups will continue to receive usual care and rehabilitation as provided by their services in the area.
Randomization
Allocation to intervention will be determined by use of a Web-based randomization service, accessed by the research therapist prior to the setup visit. Participants will be randomized using computer-generated randomization codes. Random permuted blocks of varying size will be used to generate the allocation sequence. Allocation will be stratified by center, baseline ARAT score (scores 0–49 and 50–57), and previous use of a Wii™.
Equipment check
The research team will visually inspect the Wii™ and clean with antiseptic wipes prior to allocation to the next participant in the intervention group.
Adverse-event recording
Serious adverse events will be reported by the site’s principal investigator (PI) to the clinical trials unit (CTU) and reported on a monthly basis to the trial steering committee (TSC), chief investigator (CI), and sponsor. Minor adverse events thought not to be related to the trial will not be reported.
Sample size
For the primary outcome of ARAT score at 6 weeks, the literature suggests a standard deviation of about 15 and that a clinically important effect is six points.Citation29 For a two-tailed t-test at the 5% level and with 80% power, completed data are required on 99 patients per group. To allow for attrition, we plan to recruit about 120 patients per group.
Statistical analysis
A statistical analysis plan will be prepared by the study statistician, in advance of any statistical analysis of the study data. Analysis of study data will be on an intention-to-treat basis. Participants choosing to withdraw from either intervention arm will be followed up where possible, and reasons for withdrawal, if elicited, will be reported according to the CONSORT (Consolidated Standards of Reporting Trials) guidelines.Citation30 Baseline characteristics will be summarized using suitable descriptive statistics. The primary outcome will be the ARAT score at 6 weeks. The principal analysis will be analysis of covariance with terms for treatment group and stratification variables. A further analysis will replace baseline ARAT score group (as stratified) by the actual ARAT scores in order to explore the robustness of conclusions. In each case, least-squares mean treatment differences and their 95% confidence intervals will be reported. Residual plots will be produced to check compliance with assumptions. Both analyses will be repeated for the secondary outcome measure of ARAT at 6 months, and other continuous outcome measures. The interactions between treatment group and the stratification variables will be examined and allowed for in the analyses as appropriate.
Analysis of nested qualitative study
All audio recordings will be transcribed verbatim, and the transcribed interviews will be analyzed using thematic analysis.Citation31 The field notes and observation data obtained during the interviews will be included in the analysis process. Themes will be drawn from the data using constant comparison methods; participants’ accounts will be compared and contrasted to enhance interpretation. Deviant cases will be reviewed and accounted for. If data are unclear, findings will be presented to participants to allow further comment in order to aid interpretation. The qualitative researcher will use a computer-aided program to manage the data. For trustworthiness, the lead qualitative researcher will analyze a subset of five transcripts. Any unresolved disagreements will be resolved by discussion with the CI, ensuring that data will be presented in such a way as to maintain blinding of treatment group.
Cost-effectiveness evaluation
The economic evaluation will estimate the incremental cost per QALY gained from the perspective of health and social care utilization. The costs of the Wii™ equipment will be the price paid annuitized over a useful life of 3 years assuming the equipment could be used by eight clients per year. The setup costs of the Wii™ service will be reported separately. The main source of information on resource use will be the participant diaries which will include prompts derived from the Service Receipt Inventory (SRI).Citation32 The SRI will also be used retrospectively at 6 months. When an assessment of need is completed during the trial, details of agreed care package will be recorded. Unit costs of health and social care services will be taken from standard published sources.Citation33,Citation34 Health-related quality of life will be assessed by EQ-5D 3LCitation35 and valued using the York Tariff.Citation36 The economic evaluation will provide within-trial estimates of the incremental cost-effectiveness ratio. If appropriate, modelling beyond the term of the trial will be undertaken. Sampling variation will be analyzed using probabilistic sensitivity analysis, reported as a scatterplot on the cost-effectiveness plane. Sensitivity analysis to estimate the effects of parameter uncertainty will include an exploration of the sensitivity of results to differences in the costs and effects of different provisions of rehabilitation services.
Data collection
Data will be recorded on study-specific data-collection forms (CRFs). Persons authorized to collect and record trial data at each site will be listed on the study site delegation log and authorized by the PI.
Completed CRFs will be checked and signed at the research site by the research practitioner or research therapist (as applicable) before being sent to the CTU for double-data entry onto a password-protected database accessed via the Internet. Forms will be tracked using a Web-based trial-management system. Double-entered data will be compared for discrepancies using a stored procedure. Discrepant data will be verified using the original paper data sheets.
Study oversight
The CI will be responsible for the overall running of the study and will be in regular contact with the principle investigators at the other trial centers. Each trial center will have a PI, a research administrator, a research practitioner, and a research therapist to conduct this study. The CTU will coordinate trial activities and assist with overall trial management and monitoring. A data-monitoring committee will not be convened due to the nature of the intervention and short duration of the trial. The trial-management group (TMG) will include the CI, CTU trial coordinator, trial statistician, and other members of the team and will meet regularly to monitor progress. The TSC will oversee the conduct and safety of the trial. The TSC will include a representative from the funder and a lay person.
Protocol amendments
Early on in the recruitment phase, the TMG reported two emerging issues of concern to the TSC. These were 1) that a proportion of patients were assessed as having the highest possible ARAT score at baseline and 2) the slow recruitment rate. Further to this, the research therapists asked that patients with severe shoulder subluxation be excluded, as pain precluded participants from playing the Wii™ or doing arm exercises.
In response to these concerns, the TSC recommended baseline ARAT stratification for randomization was changed from 30 to 50 points and the MAL14 be added as a secondary outcome measure at 6 months follow-up as a second outcome measure for arm function. The TSC also recommended some changes to the eligibility criteria which were implemented by the TMG: to exclude patients with symptomatic shoulder subluxation, to broaden the inclusion criteria to include patients with non-dominant weakness, and to remove the requirement for participants to have supervision during their exercises.
Finally, the number of participating centers was increased from seven to ten, and the TMG applied to the funder for an extension to the recruitment period in order to maximize the chances that the trial could recruit the full sample size. All changes to the protocol were approved by the Sponsor, the Research Ethics Committee, and the funder.
Conclusion
TWIST is the first UK RCT assessing the feasibility, cost effectiveness, and acceptability of Wii™ in stroke rehabilitation. The trial has been registered with ISRCTN 06807619 and UK CRN 11030. Results of the study will be published after it is completed in August 2014.
Acknowledgments
We would like to thank our participants and caregivers and participating centers. We would also like to thank the Research Design Service South West. We thank Jane Vickery, Senior Clinical Trials Manager (Peninsula Clinical Trials Unit) for her comments on drafts of this paper.
Funding
This paper represents independent research funded under the Research for Patient Benefit Programme (PB-PG-0110-20332). The views expressed in this publication are those of the authors and not necessarily those of the NHS, NIHR (National Institute for Health Research), or Department of Health. The trial has been registered with ISRCTN 06807619 and UK CRN 11030.
Disclosure
The authors report no conflicts of interest in this work.
References
- Royal College of PhysiciansNational Clinical Guideline for StrokeRoyal College of Physicians2012 Available from: https://www.rcplondon.ac.uk/sites/default/files/national-clinical-guidelines-for-stroke-fourth-edition.pdfAccessed August 17, 2014
- Nichols-LarsenDClarkPZeringueAGreenspanABlantonSFactors influencing stroke survivors’ quality of life during subacute recoveryStroke2005361480148415947263
- LanghornPCouparFPollockAMotor recovery after stroke: a systematic reviewLancet Neurol2009874175419608100
- AdaLDorschSCanningCStrengthening interventions increase strength and improve activity after stroke: a systematic reviewAust J Physiother200652424124817132118
- SzturmTPetersJOttoCKapadiaNDesaiATask-specific rehabilitation of finger-hand function using interactive computer gamingArch Phys Med Rehabil200889112213221718996252
- WolfSWinsteinCMillerJEffect of constraint induced movement therapy on upper limb function 3 to 9 months after stroke: the EXCITE randomised clinical trialJAMA20062962095210417077374
- ShaugnessyMResnickBMackoRTesting a model of post-stroke exercise behaviourRehabil Nurs20062111521
- YavuzerGSenelAAtayMStamH“Playstation eyetoy games” improve upper extremity-related motor functioning in subacute stroke: a randomized controlled clinical trialEur J Phys Rehabil Med200844323724418469735
- MeriansAPoiznerHBoianRBurdeaGAdamovichSSensorimotor training in a virtual reality environment: does it improve functional recovery poststroke?Neurorehabil Neural Repair200620225226816679503
- KwakkelGKollenBKrebsHEffects of robot-assisted therapy on upper limb recovery after stroke: a systematic reviewNeurorehabil Neural Repair200822211112217876068
- FlynnSPalmaPBenderAFeasibility of using the Sony PlayStation 2 gaming platform for an individual post stroke: a case reportJ Neurol Phys Ther200731418019018172415
- LaversKEGeorgeSThomasSDeutschJECrottyMVirtual reality for stroke rehabilitationCochrane Database Syst Rev20119CD00834921901720
- ButefischCHummelsheimHDenzlerPMauritzKRepetitive training of isolated movements improves the outcome of motor rehabilitation in the centrally paretic handJ Neurol Sci199513059687650532
- FrenchBLeathleyMSuttonCMcAdamJThomasLForsterAA systematic review of repetitive functional task practice with modelling of resource use, costs and effectivenessHealth Technol Assess200812301177
- Wii-Habilitation [homepage on the Internet]2012 Available from: http://www.wiihabilitation.co.ukAccessed August 17, 2014
- MouawadMDoustCMaxMMcNultyPWii-based movement therapy to promote upper extremity function post-stroke: a pilot studyJ Rehabil Med20114352753321533334
- SaposnikGTeasellRMamdaniMEffectiveness of virtual reality using Wii gaming technology in stroke rehabilitation: a pilot randomised clinical trial and proof of principleStroke2010411477148420508185
- YozbatiranNDer-YeghiaianLCramerSA Standardised approach to performing the action research arm testNeurorehabil Neural Repair200822789017704352
- LangCWagnerJDromerickAEdwardsDMeasurement of upper-extremity function early after stroke: properties of the action research arm testArch Phys Med Rehabil2006871605161017141640
- LinJHsuMSheuCPsychometric comparison of 4 measures for assessing upper-extremity function in people with strokePhys Ther200989884085019556333
- CupEHScholte op ReimerWJThijssenMCvan Kuyk-MinisMAReliability and validity of the Canadian Occupational Performance Measure in stroke patientsClin Rehabil20031740240912785249
- DuncanPWallisDLaiSJohnsonDEmbretsonSLasterLThe Stroke Impact Scale Version 2.0: evaluation of reliability, validity, and sensitivity to changeStroke1999302131214010512918
- BonitaRBeagleholeRModification of Rankin scale: recovery of motor function after strokeStroke19881912149715003201508
- UswattenGTaubEMorrisDVignoloMMcCullochKReliability and validity of the upper extremity Motor Activity Log-14 for measuring real-world arm useStroke2005362493249616224078
- AshfordSSladeMMalapradeFTurner-StokesLEvaluation of functional outcome measures for the hemiparetic upper limb: a systematic reviewJ Rehabil Med20084078779519242614
- BamfordJSandercockPDennisMBurnJWarlowCClassification and natural history of clinically identifiable subtypes of cerebral infarctionLancet199122337152115261675378
- Paternostro-SlugaTGrim-StiegerMPoschMReliability and validity of the Medical Research Council (MRC) scale and a modified scale for testing muscle strength in patients with radial palsyJ Rehabil Med200840866567119020701
- NasreddineZPhillipsNBédirianVThe Montreal Cognitive Assessment (MoCA): a brief screening tool for mild cognitive impairmentJ Am Geriatr Soc20055369569915817019
- DonaldsonCTallisRMillerSSunderlandALemonRPomeroyVEffects of conventional physical therapy and functional strength training on upper limb motor recovery after stroke: a randomized Phase II studyNeurorehabil Neural Repair200923438939719109444
- SchulzKAltmanDMoherDCONSORT 2010 Statement: updated guidelines for reporting parallel group randomized trialsAnn Intern Med2010152111819949136
- GreenJThorogoodNQualitative Health Methods for Health ResearchLondonSage Publications2004
- ChisholmDKnappMRKnudsenHCAmaddeoFGaiteLvan WijngaardenBClient Socio-Demographic and Service Receipt Inventory – European Version: development of an instrument for international research. EPSILON Study 5. European Psychiatric Services: Inputs Linked to Outcome Domains and NeedsBr J Psychiatry Suppl2000177s28s3310945075
- Department of HealthNational Reference CostsDepartment of Health2010 Available from: http://data.gov.uk/dataset/nhs-reference-costs-2010-11Accessed September 5, 2014
- CurtisLUnit Cost of Health and Social CareKentPSSRU, University of Kent2010 Available from: www.pssru.ac.uk/project-pages/unit-costs/2011/Accessed August 17, 2014
- RabinROemarMOppeMEQ-5D-3L User Guide Version 4Rotterdam2011 Available from: http://www.euroqol.org/fileadmin/user_upload/Documenten/PDF/Folders_Flyers/EQ-5D-3L_UserGuide_2013_v5.0_October_2013.pdfAccessed September 5, 2014
- DolanPModeling valuations for EuroQol health statesMed Care19973511109511089366889
