Abstract
Heart failure (HF) has become a major cause of morbidity and mortality with a significant global economic burden. Although well-established clinical tests could provide early diagnosis, access to these tests is limited in developing countries, where a relatively higher incidence of HF is present. This has prompted an urgent need for developing a cost-effective, rapid and robust diagnostic tool for point-of-care (POC) detection of HF. Lateral flow immunoassay (LFIA) has found widespread applications in POC diagnostics. However, the low sensitivity of LFIA limits its ability to detect important HF biomarkers (e.g., brain natriuretic peptide [BNP]) that are normally present in low concentration in blood. To address this issue, we developed an improved LFIA by optimizing the gold nanoparticle (GNP)–antibody conjugate conditions (e.g., the conjugate pH and the amount of added antibody), the diameter of GNP and the concentration of antibody embedded on the test line and modifying the structure of test strip. Through these improvements, the proposed test strip enabled the detection of BNP down to 0.1 ng/mL within 10–15 min, presenting ~15-fold sensitivity enhancement over conventional lateral flow assay. We also successfully applied our LFIA in the analysis of BNP in human serum samples, highlighting its potential use for clinical assessment of HF. The developed LFIA for BNP could rapidly rule out HF with the naked eye, offering tremendous potential for POC test and personalized medicine.
Introduction
Heart failure (HF) is prevalent all over the world, which has become a major cause of mortality and morbidity.Citation1–Citation3 According to a report from the American Heart Association, there were ~5.1 million people diagnosed with HF annually in the United States, of which ~50% of them die within 5 years of diagnosis.Citation4 The prevalence is predicted to increase by 25% in 2030,Citation5,Citation6 and the financial burden is estimated to rise from $32 billion in 2013 to $70 billion in 2030.Citation7 Generally, HF is classified into four stages based on the progression of the disease, where effective interventions can be taken at early stages to prevent or attenuate its progression.Citation8 Therefore, early diagnosis of HF is of paramount importance. The existing clinical diagnosis of HF is mainly based on the electrocardiogram (ECG), electrocardiography, imaging tests (e.g., chest X-ray) and laboratory tests (blood and urine tests), which are equipment dependent, time-consuming and expensive. Thus, they are less applicable in resource-limited settings,Citation9 especially in developing countries where access to health care is limited while higher incidence of HF is present. Therefore, a more feasible tool for early and rapid diagnosis of HF in resource-limited areas is vital to release the local economic pressure and reduce the threat of this disease.
To address the issue, point-of-care (POC) testing of cardiac biomarkers, which is a promising method for improving disease management,Citation10 holds a significant potential.Citation11 Ideal biomarkers for early diagnosis of HF should be highly specific, sensitive and easily accessible. Various biomarkers have been identified for HF, such as renin, norepinephrine, endothelin-1 and natriuretic peptides.Citation12,Citation13 Among these, brain natriuretic peptide (BNP) and the N-terminal fragment of BNP precursor (NT-proBNP) in serum have been known as the gold standard to identify HF and widely utilized in clinical laboratories.Citation14–Citation16 BNP and NT-proBNP are the products of proteolytic processing of the precursor proBNP (pre-proBNP). The removal of signal peptide from pre-proBNP results in proBNP molecule, which is further split into BNP (biologically active molecule) and NT-proBNP (inactive amino terminal fragment). Since small soluble molecules are preferred for early diagnosis due to their fast release kinetics and rapid diffusion from injured tissue to blood,Citation17 BNP is a more desirable cardiac biomarker for detecting HF compared to NT-proBNP.Citation14 Furthermore, BNP possessing a defined cutoff (0.1 ng/mL)Citation9 is easier to make a judgment than NT-proBNP, whose cutoff is susceptible to patient’s age. Therefore, BNP represents a vital blood cardiac biomarker in patients with acute myocardial infarction and advanced HF.Citation12
With advances in POC testing,Citation18–Citation22 lateral flow assays (LFAs) hold tremendous potential to address the above-mentioned challengesCitation23,Citation24 due to the simple, affordable and user-friendly features.Citation25 However, the existing LFAs are associated with the limitation of poor sensitivity.Citation11,Citation26 Several methods have been developed to enhance the sensitivity of LFAs, such as sample concentration,Citation27–Citation30 thermal contrast,Citation31 fluidic control,Citation32–Citation34 temperature–humidity technique,Citation35 probe-based signal enhancement,Citation23,Citation36 enzyme-based signal amplificationCitation37,Citation38 and electrochemical devices-based enhancement.Citation39 However, these approaches require either external equipment,Citation35 high-cost reagentsCitation23,Citation37 or complicated fabrication with multistep procedure,Citation32 limiting their application for POC testing. To date, a low-cost, convenient and equipment-free sensitivity enhancement method has not yet been explored. Hence, there is still an unmet need to develop a simple and sensitive lateral flow immunoassay (LFIA) for the detection of BNP.
In this study, we introduced a highly sensitive and specific LFIA for POC detection of HF through improving the gold nanoparticle (GNP)–antibody conjugate conditions (e.g., the conjugate pH and the amount of added antibody), optimizing the diameter of GNP and the concentration of capture antibody at the test line, and modifying the structure of lateral flow test strip. Through these improvements, we achieved highly sensitive detection of BNP down to 0.1 ng/mL within 10–15 min with a ~15-fold increase in the sensitivity over conventional LFA,Citation30,Citation35,Citation40 as further quantified by using a smartphone. We also successfully applied our LFIA in the analysis of BNP in human serum samples to diagnose HF, demonstrating the potential of the proposed simple sensitivity enhancement strategy for clinical assessment. The prototype would enable rapid, cost-effective and highly sensitive BNP detection, offering great potential for POC and personalized diagnostics especially in the developing world.
Materials and methods
Reagents and instruments
Two kinds of anti-BNP antibodies, 24C5 and 50E1, (Abcam, Cambridge, UK) were used as the capture and conjugate antibodies, respectively. The standard BNP antigen was also obtained from Abcam. Goat anti-mouse IgG polyclonal antibody was obtained from Fapon Biotech Inc. (Shenzhen, China). HAuCl4⋅4H2O was supplied by Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Trisodium citrate was purchased from Amresco, LLC (Solon, OH, USA). Bovine serum albumin (BSA), trehalose, casein and phosphate-buffered saline (PBS; pH7.4, 0.01 M) were obtained from MP Biomaterials, LLC (Solon, OH, USA). Sucrose was obtained from Aladdin Reagent Co., Ltd. (Shanghai, China). All the components for fabricating lateral flow test paper, including a polyvinyl chloride (PVC) backing pad, conjugate pad, absorbent pad, sample pad and nitrocellulose (NC) membrane (HFB13502S25; EMD Millipore, Billerica, MA, USA), were obtained from Jiening Biotech Co., Ltd. (Shanghai, China). All chemicals employed in this study were analytical reagent grade. Other solutions were prepared with ultrapure water (>18 MΩ) from the Barnstead Nanopure ultrapure water purification system (Thermo Fisher Scientific, Waltham, MA, USA).
Preparation of GNPs
GNPs with average diameter of 13±3 and 35±3 nm were prepared according to the previously reported protocol.Citation41,Citation42 Briefly, 4.5 or 1 mL of 1% trisodium citrate and 1 mL of 1% chloroauric acid (HAuCl4) were successively added to three-neck round-bottom flask filled with 100 mL boiled ultrapure water. Then, the color of the solution immediately turned from yellow to purple and finally remained wine red. The prepared GNPsCitation43 were characterized by using transmission electron microscopy (TEM; JEM-2100F) and ultraviolet–visible (UV/vis) spectrophotometry (Figure S1).
Modification of GNPs
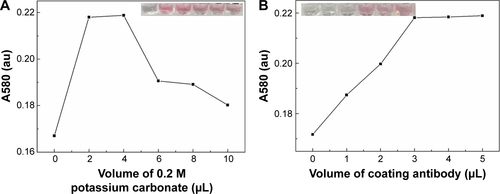
To make a better combination of GNPs and conjugating antibody, we optimized the pH of GNP–antibody conjugation and the amount of the added antibody. Briefly, certain volumes of potassium carbonate (0, 2, 4, 6, 8 and 10 µL) were separately added into the as-prepared 1 mL GNP solution to adjust the pH value; then, the excess antibody was added into the abovementioned solution for 10 min incubation at room temperature (RT). The sodium chloride was subsequently added into each solution, and the optimum pH was determined based on the color change and absorbance value of each solution (Figure S2A). Similarly, different volumes of conjugating antibody (0, 1, 2, 3, 4 and 5 µL) were added into the GNP solution with the optimum pH to optimize their volume based on the color change and absorbance value of each solution (Figure S2B). Then, 10% BSA was added to the abovementioned solution and incubated for 10 min to block nonspecific sites to obtain a final concentration of 0.1%. The supernatant solution was removed after the centrifugation at 12,000× g for 10 min, and the red pellets were suspended in 0.22 mL eluent buffer including 1% BSA, 0.85% Tris, 5% trehalose and 20% sucrose.
Fabrication of lateral flow test strips
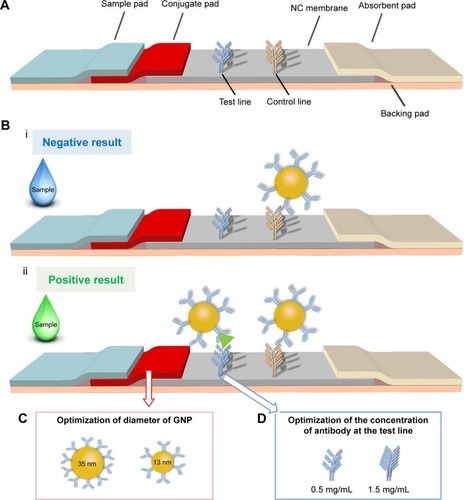
Control and capture antibodies of BNP were diluted with coating buffer (2% trehalose in 0.01 M PBS) to a certain concentration, which were then dispensed on NC membrane as control line and test line and incubated at 37°C for 2 h to immobilize the antibodies. The GNP–Ab conjugates were dispensed on the conjugate pad, and the sample pad was soaked in the treating fluid for half an hour and dried in oven for 2 h. Then, these elements were consecutively mounted on a PVC adhesive backing pad with 2 mm overlap between each two adjacent pads (). The as-assembled pads were cut into strips (2.5 mm) by Rapid Test Cutter ZQ2000 (Shanghai Kinbio Tech Co., Ltd, Shanghai, China).
Figure 1 The schematic of the optimized LFIA for BNP detection.
Notes: (A) The structure of previous LFA is adjusted by adding a conjugate pad to form a new type of LFIA. (B) The negative result (i) and positive result (ii) show the principle of the LFIA. (C) The diameter of GNP is adjusted from 13±3 to 35±3 nm. (D) The concentration of test line is adjusted from 0.5 to 1.5 mg/mL.
Abbreviations: GNP, gold nanoparticle; BNP, brain natriuretic peptide; LFA, lateral flow assay; LFIA, lateral flow immunoassay; NC, nitrocellulose.
BNP detection using LFIAs
A typical LFIA was performed as follows: the sample solution was dispensed onto the sample pad by using a pipette. The color changes at both test and control lines were observed after 10 min. For quantitative analysis, images of the test zones were captured with a smartphone (iPhone 6s) and their optical densities were determined using the Image-Pro Plus software.
Clinical analysis
To prove the sensitivity and specificity of the improved LFIA for BNP detection in clinical serum samples, we tested the LFIA with different concentrations of BNP and four kinds of cardiac biomarkers in spiked human serum. A series of spiked BNP sera (i.e., 1, 0.5, 0.2, 0.1 and 0.01 ng/mL) and other cardiac biomarkers sera were prepared by diluting their antigen stock solution with normal human sera, respectively. The spiked concentration of four interfering cardiac biomarkers was 10 times of their clinical cutoff (i.e., 100 µg/mL of C-reactive protein [CRP], 1 µg/mL of myoglobin [Myo], 30 ng/mL of troponin I [cTnI] and 3 ng/mL of NT-proBNP).Citation44–Citation47 After LFIAs were finished, the results were captured with iPhone 6s and the optical densities of test zones were determined using the Image-Pro Plus software for further quantitative analysis.
Statistical analysis
Student’s t-test was used to compare the data among different groups in optimization assays. Data were expressed as mean ± standard error of the mean (SEM) of three independent experiments (n=3). P<0.05 was reported as statistically significant.
Results and discussion
The work principle of LFIA
The principle of the improved LFIA is based on an on-strip sandwich Ag–Ab reaction, and the protocol is illustrated in . In the presence of target BNP, the target would bind to the GNP–Ab, forming GNP–Ab target complexes, which would in turn interact with the immobilized Ab at the test line to produce a red signal detectable by the naked eye. The excess GNP–Ab complexes would then bind to the immobilized secondary Ab at the control line. To achieve rapid POC analysis, qualitative analysis was performed by observing the color changes, whereas quantitative analysis was achieved with a smartphone (iPhone 6s) based on the optical density of the test zone.
Optimization of LFIAs
Paper material, GNP and detection elements have been shown to have significant effect on the performance of LFA.Citation48,Citation49 However, these three elements are the personal choice of the research group for developing high-sensitive LFACitation50 and have not been optimized simultaneously, although personal choice may satisfy the requirement for detecting some targets. Therefore, we developed an improved LFIA for highly sensitive detection of BNP by simultaneously modifying the structure of test strip and optimizing the GNP–Ab conjugate conditions, the diameter of GNP () and the concentration of antibody at the detect line ().
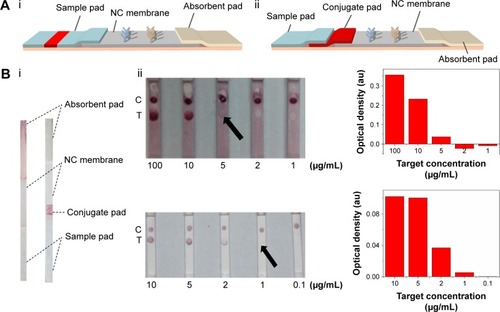
In our previous study,Citation23 the lateral flow test strip was constituted with NC membrane, absorbent pad and sample pad (). However, throughout our experiment on BNP detection, we found that the complex of GNP–Ab was unable to completely wick through the previously used sample pad, resulting in the waste and reduction of the effective tracer and degrade the detection sensitivity. This is because different paper materials are applied for different elements of LFIAs,Citation50–Citation52 where the inert glass fiber is used as the sample pad, which may not be suitable for the release of GNP–Ab. Therefore, in our modified test strip (), we introduced an extra surface-modified polyester layer as the conjugate pad (), which is much thinner than the sample pad to get a complete release of GNP–Ab. Besides, we also adjusted the composition of eluent buffer, where the trehalose was added to protect the activity of conjugate AbCitation53 and the BSA was added to block the nonspecific binding between the GNP–Ab and NC membrane to eliminate the background signal. With the introduction of the conjugate pad and the change of composition of eluent buffer, the GNP–Ab conjugation was completely released, the background color was eliminated and the signal–noise ratio and sensitivity of detection results were enhanced ().
Figure 2 The effect of improved structure of test strip on LFIA performance.
Notes: (A) The schematic diagrams show the structure of test strip before (i) and after (ii) incorporating a piece of conjugate pad into the test strip. (B) The images (i) of comparison of conventional and improved LFAs and the results (ii) which show that the detection limit is improved from 5 µg/mL to 1 µg/mL (the arrows reference the detection limit of the results).
Abbreviations: LFA, lateral flow assay; LFIA, lateral flow immunoassay; NC, nitrocellulose; C, control line; T, test line.
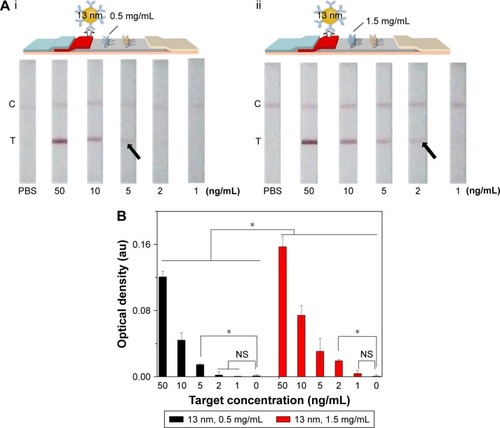
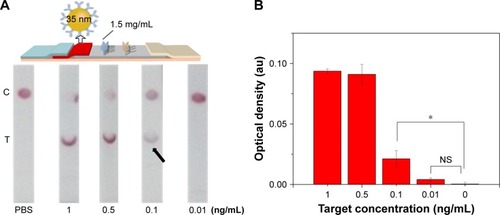
Since the principle of LFIA was based on the interaction of the antibody and GNP through hydrophobic and ionic bonds, the condition of the conjugation of antibody with GNP was one of the most important parameters for this immunoassay.Citation54 To optimize the condition of the conjugation, we adjusted the pH value of GNP solution and the amount of conjugating antibody and found that the best condition of pH value was 8 and the amount of conjugating antibody was ~20 µg (i.e., 6.8 mg/mL). Accordingly, 4 µL of 0.2 M potassium carbonate (to make sure the pH value is 8) and 3 µL conjugating antibody (Figure S2) were successively added into the as-prepared 1 mL GNP solution for 10 min incubation at RT. The resulting GNP solution was used in the following experiments. Besides, we also adjusted the concentration of the immobilized Ab at the test line in the range of 0.5–1.5 mg/mL (). The results indicate that the coating antibody concentration of 1.5 mg/mL showed greater signal of LFIA in BNP detection relative to 0.5 mg/mL (n=3, P<0.05). In fact, the LFIA with 0.5 mg/mL antibody can achieve a detection limit of 5 ng/mL (n=3, P<0.05), and with 1.5 mg/mL antibody can achieve a detection limit of 2 ng/mL (n=3, P<0.05), representing 2.5-fold signal enhancement. The sensitivity enhanced with the increasing concentration of test line. But concomitantly, high concentration of Ab at test line (>1.5 mg/mL) resulted in an observed false-positive result. Therefore, 1.5 mg/mL was selected as the optimized concentration.
Figure 3 The effect of antibody concentration at the test line on LFIA performance.
Notes: (A) The schematic diagram and results of the LFIA with 0.5 mg/mL (i) and 1.5 mg/mL coating antibody (ii). (B) Optical density analysis of (A) (n=3, *P<0.05). (The arrows reference the detection limit of the results).
Abbreviations: LFIA, lateral flow immunoassay; NS, no significance; PBS, phosphate-buffered saline; C, control line; T, test line.
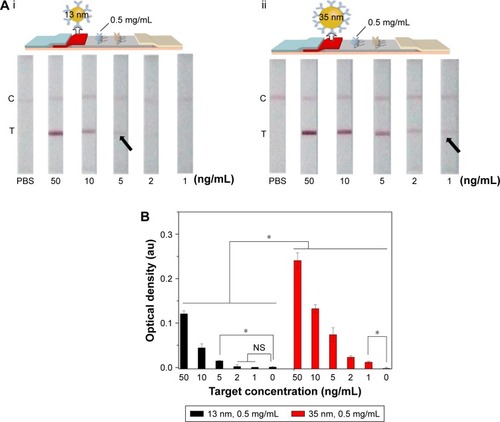
Since the diameter of GNP would affect the optical density of test line detected by the naked eye, we investigated the effect of different diameter of GNP (13±3 and 35±3 nm) on LFIA (). We found that the LFIA with 35±3 nm GNP showed greater signal in BNP detection compared to that with 13±3 nm GNP (n=3, P<0.05). Indeed, the LFIA with 13±3 nm GNP can achieve a detection limit of 5 ng/mL (n=3, P<0.05), and LFIA with 35±3 nm GNP can achieve a detection limit of 1 ng/mL (n=3, P<0.05), representing five-fold signal enhancement. According to the literature,Citation49,Citation55,Citation56 the particles reported (i.e., GNP) can be more easily seen when the size of the particles is larger, but the stability of the colloid will decrease when the diameter is >40 nm,Citation56 which may result in the steric hindrance and hamper the antigen–antibody interaction.Citation50 Thus, we used the diameter of 13±3 and 35±3 nm in this study, and the result showed that the optimum diameter is 35±3 nm.
Figure 4 The effect of GNP diameter on LFIA performance.
Notes: (A) The schematic diagram and results of the LFIA with 13±3 nm GNP (i) and 35±3 nm GNP (ii). (B) Optical density analysis of (A) (n=3, *P<0.05). (The arrows reference the detection limit of the results).
Abbreviations: GNP, gold nanoparticle; LFIA, lateral flow immunoassay; NS, no significance; PBS, phosphate-buffered saline; C, control line; T, test line.
Application of the improved LFIA
First, we used different BNP concentrations to evaluate the performance of optimized LFIAs. With the optimum concentration of antibodies (1.5 mg/mL), GNP diameter (35 nm) and improved structure of test strip (with conjugate pad), the proposed test strip could sensitively detect BNP at 0.1 ng/mL (n=3, P<0.05) with ~15-fold signal enhancement over unmodified LFIA (), which represents the clinical threshold to rule out HF. Moreover, we also assessed the limit of detection of the method. According to the concentration that shows three times of noise detection (signal to noise ratio =0.0955),Citation57,Citation58 the estimated detection limit of our method is 95.5 pg/mL.
Figure 5 Sensitivity assay using the optimized LFIA.
Notes: (A) The images showing LFIA results in the detection of different concentrations of BNP solutions. (B) Optical density analysis of (A) (n=3, *P<0.05). (The arrow references the detection limit of the results).
Abbreviations: BNP, brain natriuretic peptide; LFIA, lateral flow immunoassay; NS, no significance; PBS, phosphate-buffered saline; C, control line; T, test line.
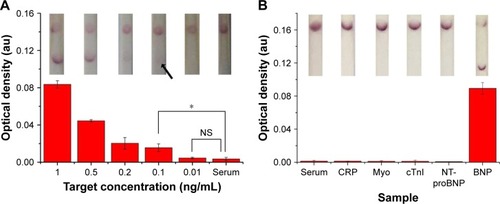
Subsequently, to further show the potential of our proposed LFIA for HF diagnosis at the POC, we tested the improved strip with clinical serum samples. Briefly, we spiked different concentrations of standard BNP antigen (i.e., 1, 0.5, 0.2, 0.1, 0.01 and 0 ng/mL) and four kinds of interfering cardiac biomarkers with 10-fold concentration of their clinical cutoff in normal human serum and then detected them using the proposed test strips. We found that the improved LFIA can detect BNP as low as 0.1 ng/mL (n=3, P<0.05) in spiked serum, which reaches the clinical threshold (). Comparing to the negative detection results of four interfering biomarkers, the test strip showed positive in BNP detection, indicating the improved LFIA with a good specificity ().
Figure 6 Clinical sample testing using the optimized LFIA.
Notes: (A) The sensitivity results of optimized LFIA detect BNP in human sera (the inset shows the images of LFA results in the detection of different concentrations of BNP solutions). (B) The specificity results of optimized LFIA detect BNP in human sera (n=3, *P<0.05) (the inset shows the images of LFA results in the detection of different samples). (The arrow references the detection limit of the results).
Abbreviations: BNP, brain natriuretic peptide; CRP, C-reactive protein; cTnI, troponin I; LFIA, lateral flow immunoassay; Myo, myoglobin; NS, no significance; NT-proBNP, N-terminal fragment of BNP precursor.
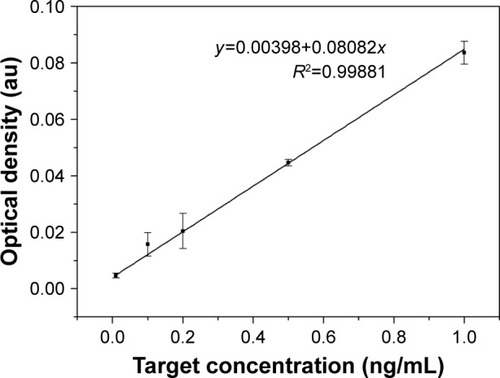
Besides, with the existing target concentration (0.01, 0.1, 0.2, 0.5 and 1 ng/mL) and their corresponding optical densities, we obtained a calibration curve (y =0.00398+0.08082x) with R2=0.99881, showing an excellent linear relationship between target concentration and optical density (). Therefore, when the proposed LFIA detected the BNP sera with 0.4, 0.6 and 0.8 ng/mL, their optical densities were determined using the Image-Pro Plus software. Then, we used the calibration curve to separately convert the optical density to calculate the concentration, and the recovery rates are demonstrated in , with 0.8 ng/mL associated with the largest recovery rates (0.976±0.006).
Table 1 The recovery rates of other three concentrations of BNP
In fact, various methods have been developed for the diagnosis of BNP, such as enzyme-linked immunosorbent assay (ELISA) and chemiluminescent immunoassay (CLIA).Citation59–Citation61 Although these methods have achieved quantitative detection with high sensitivity (2–10 pg/mL),Citation60 they are either time-consuming (~12–36 h), complicated (need tedious procedures), inconvenient (require cumbersome instruments and professional operators) or need large blood volume (~5 mL), which significantly limit their accessibility in resource-limited settings.Citation61 Our proposed test strip is simple, low cost and equipment free, which can be performed by untrained users in resource-limited settings in 10–15 min using only a drop of blood, eliminating the need for large and expensive equipment or laboratory infrastructure. More importantly, compared with the existing methods, our improved LFIA can also meet the clinical demand. It has a clinically relevant BNP level detection limit of 0.1 ng/mL, which is the cutoff point to rule out the HF,Citation9 making it highly suitable for early and rapid diagnosis of HF at the POC.
Conclusion
We developed a simple, cost-effective and highly sensitive LFIA for rapid BNP detection at the POC. We optimized the conjugating condition of GNP–Ab, the diameter of GNP and the concentration of immobilized Ab at test line and adjusted the structure of conventional LFA. Through these improvements, the proposed LFIA could sensitively detect BNP at 0.1 ng/mL in serum samples, representing ~15-fold signal improvement over conventional LFIA. We envision that the proposed test strip may show great potential to rapidly detect HF and facilitate early and appropriate health management at the POC.
Acknowledgments
This work was financially supported by the National Instrumentation Program of China (2013YQ190467). All the data generated during this study are included within the published article and its Supplementary materials.
Disclosure
The authors report no conflicts of interest in this work.
Supplementary materials
Figure S1 Characterization of GNP.
Notes: (A) Characterization of GNP with 13 nm: (i) TEM and size distribution of GNP; (ii) UV–vis absorption spectra of GNP. (B) Characterization of GNP with 35 nm: (i) TEM and size distribution of GNP; (ii) UV–vis absorption spectra of GNP.
Abbreviations: GNP, gold nanoparticle; TEM, transmission electron microscopy; UV–vis, ultraviolet–visible.
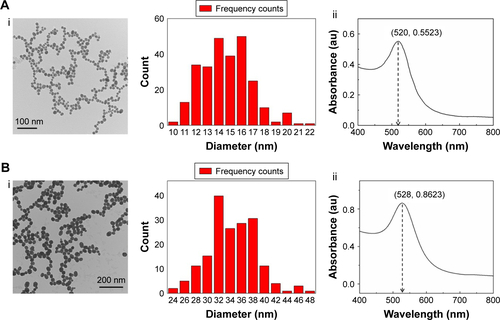
Figure S2 Determination of optimal pH and volume of conjugation.
Notes: (A) Determination of optimal pH for the conjugation of antibody to GNP (The inset shows the images of color change of pH optimization). (B) Determination of optimal volume of coating antibody for conjugation at pH 8 (The inset shows the images of color change of volume optimization).
Abbreviation: GNP, gold nanoparticle.
References
- FleischmannKEBeckmanJABullerCE2009 ACCF/AHA focused update on perioperative beta blockade: a report of the American college of cardiology foundation/American heart association task force on practice guidelinesCirculation2009120212123215119884474
- DicksteinKCohen-SolalAFilippatosGESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the diagnosis and treatment of acute and chronic heart failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM)Eur J Heart Fail2008101093398918826876
- WangC-CChenJ-HYuW-C2012 Guidelines of the Taiwan Society of Cardiology (TSOC) for the diagnosis and treatment of heart failureActa Cardiol Sin201228161195
- GoASMozaffarianDRogerVLHeart disease and stroke statistics – 2013 update: a report from the American Heart AssociationCirculation20131271e6e24523239837
- RogerVLWestonSARedfieldMMTrends in heart failure incidence and survival in a community-based populationJAMA2004292334435015265849
- LevyDKenchaiahSLarsonMGLong-term trends in the incidence of and survival with heart failureN Engl J Med20023473471397140212409541
- HeidenreichPATrogdonJGKhavjouOAForecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart AssociationCirculation2011123893394421262990
- HuntSABakerDWChinMHACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to revise the 1995 Guidelines for the Evaluation and Management of Heart Failure) developed in collaboration with the International Society for Heart and Lung Transplantation endorsed by the Heart Failure Society of AmericaJ Am Coll Cardiol20013872101211311738322
- McCulloughPANowakRMMcCordJB-type natriuretic peptide and clinical judgment in emergency diagnosis of heart failure: analysis from breathing not properly (BNP) multinational studyCirculation2002106441642212135939
- SharmaSZapatero-RodríguezJEstrelaPO’KennedyRPoint-of-care diagnostics in low resource settings: present status and future role of microfluidicsBiosensors20155357760126287254
- McBrideJDCooperMAA high sensitivity assay for the inflammatory marker C-Reactive protein employing acoustic biosensingJ Nanobiotechnology200861118173857
- TsutamotoTWadaAMaedaKPlasma brain natriuretic peptide level as a biochemical marker of morbidity and mortality in patients with asymptomatic or minimally symptomatic left ventricular dysfunction. Comparison with plasma angiotensin II and endothelin-1Eur Heart J199920241799180710581138
- ClericoAPassinoCFranziniMEmdinMCardiac biomarker testing in the clinical laboratory: where do we stand? General overview of the methodology with special emphasis on natriuretic peptidesClin Chim Acta2015443172424937843
- SeferianKRTammNNSemenovAGThe brain natriuretic peptide (BNP) precursor is the major immunoreactive form of BNP in patients with heart failureClin Chem200753586687317384012
- BelenkyASmithAZhangBThe effect of class-specific protease inhibitors on the stabilization of B-type natriuretic peptide in human plasmaClin Chim Acta20043401–216317214734208
- EmdinMPassinoCPronteraCComparison of brain natriuretic peptide (BNP) and amino-terminal ProBNP for early diagnosis of heart failureClin Chem20075371289129717495021
- McDonnellBHeartySLeonardPO’KennedyRCardiac biomarkers and the case for point-of-care testingClin Biochem2009427–854956119318022
- HuaXQianGYangJDevelopment of an immunochromatographic assay for the rapid detection of chlorpyrifos-methyl in water samplesBiosens Bioelectron201026118919420598518
- ChoiJRHuJTangRAn integrated paper-based sample-to-answer biosensor for nucleic acid testing at the point of careLab Chip201616361162126759062
- ChoiJRTangRWangSAbasWABWPingguan-MurphyBXuFPaper-based sample-to-answer molecular diagnostic platform for point-of-care diagnosticsBiosens Bioelectron20157442743926164488
- KolosovaAYSibandaLDumoulinFLateral-flow colloidal gold-based immunoassay for the rapid detection of deoxynivalenol with two indicator rangesAnal Chim Acta2008616223524418482609
- GuoY-RLiuS-YGuiW-JZhuG-NGold immunochromatographic assay for simultaneous detection of carbofuran and triazophos in water samplesAnal Biochem20093891323919303861
- HuJWangLLiFOligonucleotide-linked gold nanoparticle aggregates for enhanced sensitivity in lateral flow assaysLab Chip201313224352435724056409
- ChoiJRHuJWangSPaper-based point-of-care testing for diagnosis of dengue infectionsCrit Rev Biotechnol201737110011126912259
- ChoiJRHuJGongYAn integrated lateral flow assay for effective DNA amplification and detection at the point of careAnalyst2016141102930293927010033
- MaiselASKrishnawswamyPNowakRMRapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failureN Engl J Med2002347316116712124404
- MoghadamBYConnellyKTPosnerJDTwo orders of magnitude improvement in detection limit of lateral flow assays using isotachophoresisAnal Chem20158721009101725495988
- ChiuRYJueEYipATSimultaneous concentration and detection of biomarkers on paperLab Chip201414163021302824950897
- TangRHYangHChoiJRAdvances in paper-based sample pretreatment for point-of-care testingCrit Rev Biotechnol201737441142827075621
- TangRYangHChoiJRImproved sensitivity of lateral flow assay using paper-based sample concentration techniqueTalanta201615226927626992520
- QinZChanWCBoulwareDRAkkinTButlerEKBischofJCSignificantly improved analytical sensitivity of lateral flow immunoassays by using thermal contrastAngew Chem Int Ed Engl20121241844344437
- RivasLMedina-SánchezMde la Escosura-MuñizAMerkoçiAImproving sensitivity of gold nanoparticle-based lateral flow assays by using wax-printed pillars as delay barriers of microfluidicsLab Chip201414224406441425241662
- ChoiJRLiuZHuJPolydimethylsiloxane-paper hybrid lateral flow assay for highly sensitive point-of-care nucleic acid testingAnal Chem201688126254626427012657
- ChoiJRYongKWTangRLateral flow assay based on paper–hydrogel hybrid material for sensitive point-of-care detection of dengue virusAdv Healthc Mater Epub2016118
- ChoiJRHuJFengSWan AbasWAPingguan-MurphyBXuFSensitive biomolecule detection in lateral flow assay with a portable temperature–humidity control deviceBiosens Bioelectron2016799810726700582
- ChoiDHLeeSKOhYKA dual gold nanoparticle conjugate-based lateral flow assay (LFA) method for the analysis of troponin IBiosens Bioelectron20102581999200220167468
- HeYZhangSZhangXUltrasensitive nucleic acid biosensor based on enzyme–gold nanoparticle dual label and lateral flow strip biosensorBiosens Bioelectron20112652018202420875950
- HuJWangSWangLAdvances in paper-based point-of-care diagnosticsBiosens Bioelectron20145458559724333570
- CheungSFChengSKKameiDTPaper-based systems for point-of-care biosensingJ Lab Autom201520431633325787805
- WatanabeTOhkuboYMatsuokaHDevelopment of a simple whole blood panel test for detection of human heart-type fatty acid-binding proteinClin Biochem200134425726311440724
- MaoXMaYZhangAZhangLZengLLiuGDisposable nucleic acid biosensors based on gold nanoparticle probes and lateral flow stripAnal Chem20098141660166819159221
- LiuJLuYPreparation of aptamer-linked gold nanoparticle purple aggregates for colorimetric sensing of analytesNat Protoc20061124625217406240
- BreitnerEKHussainSMComfortKKThe role of biological fluid and dynamic flow in the behavior and cellular interactions of gold nanoparticlesJ Nanobiotechnology2015131125592092
- BlackSKushnerISamolsDC-reactive proteinJ Biol Chem200427947484874849015337754
- De LemosJAAntmanEMGiuglianoRPVery early risk stratification after thrombolytic therapy with a bedside myoglobin assay and the 12-lead electrocardiogramAm Heart J2000140337337810966533
- AdamsJEBodorGSDávilarománVGCardiac troponin I: A marker with high specificity for cardiac injuryCirculation19938811018319322
- WeberMHammCRole of B-type natriuretic peptide (BNP) and NT-proBNP in clinical routineHeart200692684384916698841
- GuptaSHudaSKilpatrickPKVelevODCharacterization and optimization of gold nanoparticle-based silver-enhanced immunoassaysAnal Chem200779103810382017429944
- HendersonKStewartJFactors influencing the measurement of oestrone sulphate by dipstick particle capture immunoassayJ Immunol Methods20022701778412379340
- Posthuma-TrumpieGAKorfJvan AmerongenALateral flow (immuno) assay: its strengths, weaknesses, opportunities and threats. A literature surveyAnal Bioanal Chem2009393256958218696055
- ChanCPSumKWCheungKYDevelopment of a quantitative lateral-flow assay for rapid detection of fatty acid-binding proteinJ Immunol Methods200327919110012969550
- SajidMKawdeA-NDaudMDesigns, formats and applications of lateral flow assay: a literature reviewJ Saudi Chem Soc2015196689705
- LeslieSBIsraeliELighthartBCroweJHCroweLMTrehalose and sucrose protect both membranes and proteins in intact bacteria during dryingAppl Environ Microbiol19956110359235977486995
- ChenZ-PPengZ-FZhangPA sensitive immunosensor using colloidal gold as electrochemical labelTalanta20077251800180419071835
- AveyardJMehrabiMCossinsABravenHWilsonROne step visual detection of PCR products with gold nanoparticles and a nucleic acid lateral flow (NALF) deviceChem Commun20074142514253
- LaitinenMPVuentoMAffinity immunosensor for milk progesterone: identification of critical parametersBiosens Bioelectron19961112120712148896325
- FangCWuSDuanNDaiSWangZHighly sensitive aptasensor for oxytetracycline based on upconversion and magnetic nanoparticlesAnal Methods20157625852593
- MishraRKHayatACatananteGOcañaCMartyJLA label free aptasensor for Ochratoxin A detection in cocoa beans: an application to chocolate industriesAnal Chim Acta201588910626343432
- MairJHammerer-LercherAPuschendorfBThe impact of cardiac natriuretic peptide determination on the diagnosis and management of heart failureClin Chem Lab Med200139757158811522102
- ClericoAEmdinMDiagnostic accuracy and prognostic relevance of the measurement of cardiac natriuretic peptides: a reviewClin Chem2004501335014633912
- ClericoADel RySGiannessiDMeasurement of cardiac natriuretic hormones (atrial natriuretic peptide, brain natriuretic peptide, and related peptides) in clinical practice: the need for a new generation of immunoassay methodsClin Chem200046101529153411017928