?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
Formation of a dental biofilm by Streptococcus mutans can cause dental caries, and remains a costly health problem worldwide. Recently, there has been a growing interest in the use of peptidic drugs, such as peptide p1025, analogous to the fragments 1025–1044 of S. mutans cellular adhesin, responsible for the adhesion and formation of dental biofilm. However, peptides have physicochemical characteristics that may affect their biological action, limiting their clinical performance. Therefore, drug-delivery systems, such as a bioadhesive liquid-crystalline system (LCS), may be attractive strategies for peptide delivery. Potentiation of the action of LCS can be achieved with the use of bioadhesive polymers to prolong their residence on the teeth. In line with this, three formulations – polyoxypropylene-(5)-polyoxyethylene-(20)-cetyl alcohol, oleic acid, and Carbopol C974P in different combinations (F1C, F2C, and F3C) were developed to observe the influence of water in the LCS, with the aim of achieving in situ gelling in the oral environment. These formulations were assessed by polarized light microscopy, small-angle X-ray scattering, rheological analysis, and in vitro bioadhesion analysis. Then, p1025 and a control (chlorhexidine) were incorporated into the aqueous phase of the formulation (F + p1025 and F + chlorhexidine), to determine their antibiofilm effect and toxicity on epithelial cells. Polarized light microscopy and small-angle X-ray scattering showed that F1C and F2C were LCS, whereas F3C was a microemulsion. F1C and F2C showed pseudoplastic behavior and F3C Newtonian behavior. F1C showed the highest elastic and bioadhesive characteristics compared to other formulations. Antibiofilm effects were observed for F + p1025 when applied in the surface-bound salivary phase. The p1025-loaded nanostructured LCS presented limited cytotoxicity and effectively reduced S. mutans biofilm formation, and could be a promising p1025-delivery strategy to prevent the formation of S. mutans dental biofilm.
Introduction
Streptococcus mutans is closely associated with the etiology of dental caries,Citation1–Citation3 which is still one of the most widespread diseases worldwide, affecting people of all ages throughout their lifetimes.Citation4 It is considered a biofilm-dependent disease, because the pathogenic microbiota, which colonize and proliferate in dental biofilms, are the most important factor, along with a high-carbohydrate diet, for the onset and progression of the disease.Citation5–Citation8 Virulence factors, including the adhesin antigens I/II (Ag I/II), glucosyltransferases, and glucan-binding protein, improve the ability of S. mutans to adhere and accumulate in the dental biofilm.Citation9–Citation12
Numerous studies have demonstrated the importance of immunoresponse against Ag I/II in human protection against S. mutans colonization.Citation13 In vitro studies with Ag I/II-knockout S. mutans strains have shown decreased adhesion to hydroxyapatite on the enamel surface, suggesting that Ag I/II facilitates the adhesion of the bacteria.Citation14 Downregulation of Ag I/II in biofilm cells is critical for initial biofilm formation, but not for established biofilms.Citation15 These findings may provide useful information regarding the importance of Ag I/II as a tool for new strategies to control biofilm-mediated infections.Citation16 In vitro assays of S. mutans adhesion to dental surfaces have identified that the salivary agglutinin present in human saliva is an Ag I/II receptor, mediating the binding between bacteria and teeth.Citation17
Kelly et al identified amino acid residues 1025–1044 in the C-terminal region of Ag I/II as the adhesion epitope of this antigen.Citation18 The synthetic peptide p1025 corresponding to these residues was able to inhibit in vitro binding between S. mutans adhesin and salivary agglutinin. p1025 also reduced the recolonizing of S. mutans in dental biofilm in vivo. Li et al demonstrated that a dentifrice containing p1025 decreased the adhesion of S. mutans hydroxyapatite surfaces covered by saliva.Citation19
Several topical formulations are commonly employed for controlling dental biofilm formation, such as mouthwash, gels, dentifrices, and lozenges. However, these formulations are often characterized by limited drug retention in the oral cavity, and they tend to be rapidly dislodged, diluted, or removed, which can alter the efficacy of the active principles present in their composition, such as antimicrobial agents.Citation20 Therefore, numerous administrations are required to achieve and maintain effective levels of the drug.Citation21–Citation24 Therefore, the development of a drug-delivery nanosystem that provides controlled release of a drug retained for longer in the mucosa would be very advantageous.Citation24
Among all drug-delivery nanosystems, the liquid-crystalline system (LCS) has received considerable attention, because of its excellent potential to control the release of a wide range of biomolecules, including peptides.Citation25–Citation27 The LCS also stands out because it is formed by surfactants that form lamellar, hexagonal, and cubic crystalline liquid mesophases, by the gradual addition of solvents, such as water. The lamellar mesophase is a one-dimensional structure formed by parallel and planar layers of surfactant bilayers separated by layers of solvent. The hexagonal mesophase is formed by layers of surfactant and solvent that are arranged in the form of cylinders forming a two-dimensional structure. Finally, the cubic mesophase is formed by two networks of solvent channels surrounded by surfactant bilayers arranged in a three-dimensional organized structure.Citation28
Therefore, the gradual increase of water in the LCS increases the organization of the system structure, which results in an increase in viscosity of the formulation. As such, LCS are interesting for buccal drug administration, as liquid can be present, facilitating administration of the formulation, eg, by syringe. However, when it comes in contact with the oral environment, the LCS has the capacity to incorporate water from the saliva, becoming a more viscous LC mesophase; therefore, this in situ gelling characteristic allows for the administration of a liquid into the oral cavity via syringe that then becomes a viscous solution that adheres to the teeth for a long period.Citation28,Citation29
An innovative strategy for further increasing residence time of the LCS on the tooth is to incorporate polyacrylic acid polymers in the aqueous phase, once these polymers produce viscous gels with high bioadhesion.Citation30 In this sense, we have investigated the bioadhesion of different polyacrylic acid polymers, such as Carbopol 971P, Carbopol 974P (C974), and polycarbophil, and results showed that the bioadhesion of C974 was highest.Citation31 Jones et al investigated the bioadhesion of different gels for xerostomia treatment, and they also observed that the gel composed of C974 enhanced the residence time of the product.Citation32 Thereby, the objectives of this study were to develop and characterize a nanostructured, bioadhesive LCS composed of C974 dispersion as the aqueous phase, oleic acid (OA) as the oily phase, and polyoxypropylene-(5)-polyoxyethylene-(20)-cetyl alcohol as the surfactant to incorporate the p1025 peptide and to evaluate its in vitro cytotoxicity and effect against the formation of S. mutans biofilm.
Materials and methods
Materials
PPG-5-Ceteth-20 (Procetyl AWS) was purchased from Croda (Campinas, Brazil). OA was purchased from Labsynth (Diadema, São Paulo, Brazil). The carbomer homopolymer type B (C974) was purchased from Lubrizol (Wickliffe, OH, USA). Peptide p1025 with sequence Ac-QLKTADL-PAGRDETTSFVLV-NH2 (molecular weight 2,202.5 Da) was purchased from AminoTech Research and Development (Diadema, SP, Brazil). Chlorhexidine diacetate (Chx) was purchased from Sigma-Aldrich (St Louis, MO, USA). High-purity water was prepared with a Millipore Milli-Q Plus purification system, and its resistivity was 18.2 MΩ⋅cm.
Construction of ternary-phase diagram and preparation of formulations
A ternary-phase diagram was constructed at 25°C±0.5°C by weighing and mixing different amounts of polyoxypropylene- (5)-polyoxyethylene-(20)-cetyl alcohol (PPG-5-Ceteth-20) as the surfactant, OA as the oil phase, polymeric dispersion containing 5% (w:w) C974P, and water as the aqueous phase. The final polymeric concentration in each formulation was 0.5% (w:w). The polymeric dispersion was prepared via dispersing 5% (w/w) C974P in water and homogenizing it at 2,000 rpm in a mechanical stirrer for approximately 10 minutes at 25°C±0.5°C. Following complete dissolution, the pH was adjusted to 6 with triethanolamine (Labsynth) and manual agitation. The pH of each final formulation was 5.5. Then, all formulations were visually classified as a transparent liquid system, translucent liquid system, transparent viscous system, translucent viscous system, opaque viscous system, or phase separation. The different regions in the phase diagram were then delineated. From these data, the formulations F1C, F2C, and F3C were selected for physicochemical characterization.Citation26
Structural features of the formulations
Polarized light microscopy
Polarized light microscopy (PLM) analyses were performed by placing a small amount of each formulation on a glass slide and covering it with a coverslip. Samples were analyzed using a Jenamed (Carl Zeiss Meditec AG, Jena, Germany), evaluating the homogeneity of the dispersion and observing the presence of anisotropy or isotropy at a magnification of 20× and temperature of 25°C.Citation33
Small-angle X-ray scattering
The structural arrangements of the LCSs were analyzed by small-angle X-ray scattering (SAXS) using Brazilian Synchrotron Light Laboratory (Campinas, Brazil) equipment with a type Si(111) monochromator at a wavelength of 1.608 Å that yields a horizontally focused beam. A vertical Pilatus 300K SAXS detector located at 858.45 mm from the sample and a multichannel analyzer 13 were employed to record the intensity of scattering vector q from 0.1 to 3.8 Å at 25°C. Scattering particles in the system without a sample were subtracted from the total intensity of the sample, as a function of the module of the scattering vector:
Rheological analysis
Rheological measurements were performed at 37°C±0.1°C in triplicate using a controlled-stress AR2000 rheometer (TA Instruments, New Castle, DE, USA) with parallel-plate geometry (40 mm diameter) and a sample gap of 200 μm. Formulation samples were carefully applied to the lower plate to minimize sample shearing and were allowed to equilibrate for 3 minutes prior to analysis.Citation30
Determination of flow properties
Flow properties were determined using a controlled shear-rate procedure ranging from 0.01 to 100 seconds and back. Each stage lasted for 120 seconds, with an interval of 10 seconds between the curves. Consistency and flow indices were determined from the power law described in EquationEquation 2(2) for a quantitative analysis of flow behavior:
Oscillatory analyses
Oscillatory analyses were initiated by conducting a stress sweep to determine the viscoelastic region of the formulations. The stress sweep was carried out at a constant frequency of 1 Hz over a stress range of 0.1–10 Pa. A constant shear stress of 1 Pa was selected to perform the frequency sweep over a range of 0.1–10 Hz, which was within the previously determined linear viscoelastic region for all formulations. As such, the storage (G′) and loss (G′′) moduli were recorded. Variations in G′ at low frequencies in a log–log plot of G″ versus ω followed the power law described in EquationEquation 3(3) , given by:
In vitro bioadhesion study
Preparation of disks
Six fresh bovine permanent central incisors were collected, scaled to remove periodontal tissue and other debris, and stored in 1% thymol solution at 4°C for 1 month until use. This research protocol was performed in accordance with the International Guidelines Principles for Biomedical Research involving animals (Council for International Organization of Medical Sciences-CIOMS/International Council for Laboratory Animal Science – ICLAS) and approved by the Ethical Committee on the use of animals (FOA/UNESP, protocol 2014/00618). Teeth with enamel cracks, hypoplasia, and calculus in the middle third of the crown or other morphological alterations were excluded. Enamel blocks were cut transversally from the middle third of the buccal surface of each tooth using a water-cooled, double-faced diamond disk (KG Sorensen, Barueri, Brazil). Specimens were then rounded using a high-speed, water-cooled cylindrical diamond burr (1095; KG Sorensen) to obtain specimens with a diameter of 1 cm containing enamel. Teeth surfaces were polished with wet 200-grit silicon carbide paper (T469-SF Norton; Saint-Gobain Abrasives, Worcester, MA, USA) to normalize the surface.Citation34
Bioadhesion study
A TA-XT Plus texture analyzer (Stable Micro Systems, Godalming, UK) was used for tensile strength measurements. The tooth model was fastened to the upper movable probe with double-sided tape, and the formulation sample was located on the lower platform. Before the test, the tooth was immersed in artificial saliva. The upper probe was lowered until it made contact with the sample, and kept in contact without any additional force applied for 60 seconds. The probe was then raised at a speed of 0.5 mm/second, and the force needed for detachment was registered. The work of bioadhesive force (mN⋅s), which is proportional to the area under the force–time curve, was used to describe bioadhesive characteristics. Seven replicates were analyzed at 37°C±0.5°C.Citation30 The composition of the artificial saliva used was 8 g/L sodium chloride, 0.19 g/L of potassium monobasic phosphate (KH2PO4), and 2.28 g/L of disodium phosphate (Na2HPO4) with pH 6.8.Citation36
Microbiological evaluation
Preparation of the groups
p1025 was solubilized in sterile deionized water with 0.1% acetic acid (CH3COOH) at a concentration of 1,000 μg/mL prior to use and stored in a freezer at −20°C until use. The positive-control group was Chx at 1 mg/mL. Peptide p1025 and Chx were incorporated into the aqueous phase of formulation F3C at a concentration of 1 mg/mL based on a previous study.Citation52 The groups studied were p1025 and Chx solutions, F3C (F), F3C containing p1025 (F + p1025), and F3C containing Chx (F + Chx). F3C was diluted tenfold before incorporating Chx and p1025 to reduce the viscosity for biofilm assays.Citation36
Biofilm biomass assays
The effect of treatments on biofilm formation was studied following the methodology described in Ahn et al,Citation37 with some modifications in biofilm growth in the presence of saliva proposed by Castillo.Citation38 Firstly, stimulated saliva was collected once from two healthy volunteers with good oral health in sterile tubes, after approval by the Human Research Ethics Committee of Araçatuba Dental School, Universidade Estadual Paulista, Brazil (CAAE 13079213.4.0000.5420). All volunteers whose saliva was used in this research provided informed consent. Volunteers were asked not to eat or drink for at least 2 hours prior to collection. The saliva from volunteers was mixed in equal proportions and centrifuged at 4,000× g for 10 minutes at 4°C to remove cell debris. After that, the supernatant was mixed with the buffered solution (0.05 M KCl, 0.02 M KPO4, 0.02 M CaCl2, and 0.02 M MgCl2) at a ratio of 1:1 and 0.1 M phenylmethylsulfonyl fluoride (Sigma-Aldrich) was added. Clarified saliva was used after filter sterilization through a 0.22 μm-filter bottle (Corning, Corning, NY, USA). For the following experiments, each treatment (F + p1025, F + Chx, p1025, and Chx solutions [all at 1 mg/mL]) was diluted 1:3 in saliva.
Overnight cultures of S. mutans UA159 were transferred to prewarmed brain–heart infusion containing 1% glucose and grown at 37°C in 5% CO2 to the mid-exponential phase (OD 0.5). The cultures were then diluted 1:10 and centrifuged at 8,000× g for 5 minutes, washed in 0.9% saline solution, and resuspended in prewarmed buffered tryptone–yeast extract broth 1.25× at pH 7.0 containing 0.5% sucrose (cell suspension 5×105 CFU/mL). Biofilm-formation assays were performed using polystyrene U-bottom cell-culture plates (TPP, Trasadingen, Switzerland) following two different methods: 1) salivary preparation with each treatment was added to each well with the cell culture (salivary fluid phase), or 2) wells were first coated with salivary preparations before being inoculated with cell suspension (salivary surface-bound phase). For experiments with fluid-phase salivary preparation, 120 μL cell suspension was inoculated into the wells concomitantly with 10 μL each treatment + 30 μL saliva, maintaining the proportion of 25% saliva, as proposed by Castillo.Citation38 For experiments with surface-bound salivary preparations, each well was conditioned with each treatment diluted in saliva (1:3) and incubated at 37°C for 1 hour prior to inoculation of cell suspensions into the wells. All plates were incubated at 37°C in 5% CO2 for 24 hours. After that, plates were washed by immersion in sterile distilled water to remove unadhered cells. After brief drying, 150 μL aqueous 1% crystal violet was added to each well, and the plates were incubated at room temperature for 30 minutes. Next, the crystalline violet solution was removed and the plates washed again. The plates were inverted on paper towels and remained for 2 hours at room temperature to dry. The crystalline violet dye (stained biofilm) was then solubilized by incubation with 200 μL ethanol per well for 30 minutes. Then, 100 μL dye in ethanol was transferred to wells of a new microplate, and absorbance was measured at 575 nm with spectrophotometry (Eon Microplate; BioTek Instruments, Winooski, VT, USA) to quantify the biomass of the biofilm.Citation39
Cytotoxicity evaluation
Human epithelial cells from the HaCaT cell line (spontaneous immortalized nontumorigenic human keratinocyte cell line, code 341; BCRJ, Rio de Janeiro, Brazil) were cultured in DMEM (Thermo Fisher Scientific, Waltham, MA, USA) plus 10% fetal calf serum and 100 μg/mL penicillin G–streptomycin. Cells were cultured until reaching a sub-confluent density at 37°C in a wet 5% CO2 atmosphere. Epithelial cells were harvested following 0.25% trypsin–EDTA treatment for 5 minutes (TrypLE Express; Thermo Fisher Scientific) at 37°C. Proteases were then inactivated by adding 0.3 mg/mL trypsin inhibitor, and cells were harvested by centrifugation (500× g for 5 minutes), suspended in fresh medium, seeded in a 96-well microplate (200 μL/well, 106 cells/mL), and incubated overnight at 37°C in a 5% CO2 atmosphere to allow for cell adhesion before stimulation. All treatments (1 mg/mL) were diluted 1:500 in DMEM prior to their use in cell cultures. Dilutions and times of exposure were based on those reported by Guillot et al.Citation42 Cells were then treated with F, p1025, F + p1025, F + Chx, and Chx for 5, 30, and 60 minutes at 37°C in a 5% CO2 atmosphere. After exposure, the culture medium was replaced in the wells and cells again incubated for 24 hours. Then, a colorimetric MTT cell-viability assay (Hoffman-La Roche, Basel, Switzerland) was used to determine the effect of the treatments on cell viability. After 24 hours, the extracts were aspirated and replaced by 90 μL DMEM plus 10 μL MTT solution (5 mg/mL sterile PBS; Sigma-Aldrich). After that, the culture medium with MTT solution was removed and replaced with 100 μL acidified isopropanol solution (0.04 NHCl). Two 50 μL aliquots from each well were transferred to 96-well plates (Costar, Greenwich, CT, USA). Cell viability was evaluated using spectrophotometry as being proportional to the absorbance measured at 570 nm using an enzyme-linked immunosorbent-assay microplate reader (3550-UV; Bio-Rad Laboratories, Hercules, CA, USA). Means were calculated for the groups and transformed into percentages, which represented the inhibitory effect of the extracts on the mitochondrial activity of the cells. The positive control (DMEM) was defined as having 100% cell metabolism.Citation40
Statistical analyses
Characterizations of the formulations are presented using descriptive analysis. Microbiological and cytotoxicity assays were carried out in four replicates over 3 days (n=12). Results for biofilm biomass quantification (OD – Abs550) were analyzed according to the different phases of biofilm, surface-bound salivary phase, and salivary fluid phase, with Kruskal–Wallis and Mann–Whitney tests comparing the different groups of treatments. Data from cytotoxicity assays were analyzed by analysis of variance/Tukey’s tests. All tests were analyzed using SPSS version 17.1, with P<0.05 considered statistically significant.
Results
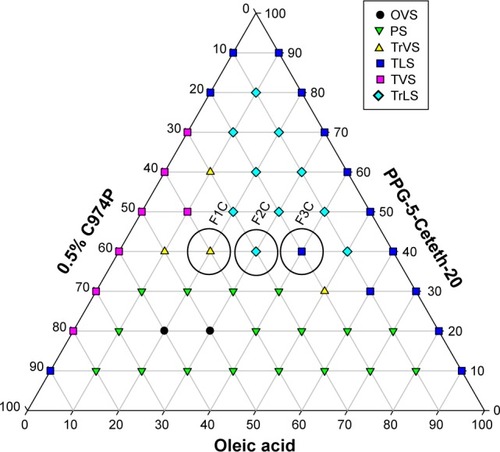
The ternary-phase diagram illustrated in shows that phase separation occurred below 40% of PPG-5-Ceteth-20 in virtually all aqueous- and oily-phase combinations. Furthermore, translucent viscous and transparent viscous systems were formed from 40% PPG-5-Ceteth-20, and the viscosity of the formulations increased starting from 40% surfactant with water addition. Based on these findings, three points named F1C, F2C, and F3C were selected from this diagram region with 40% PPG-5-Ceteth-20, because they were located in a region of viscosity transition. Alternately, with the addition of saliva, F3C showed in situ gelling to the viscous formulation F1C, which may promote higher retention and a longer release of p1025. indicates the composition of the three selected systems.
Table 1 Composition of formulations selected for characterization
Figure 1 Ternary-phase diagram.
Notes: Polyoxypropylene-(5)-polyoxyethylene-(20)-cetyl alcohol (PPG-5-Ceteth-20), oleic acid, and 0.5% C974P dispersion (0.5% C974P). F1C, F2C, and F3C were the formulations selected for structural characterization.
Abbreviations: TLS, transparent liquid system; TrLS, translucent liquid systems; TVS, transparent viscous system; TrVS, translucent viscous system; OVS, opaque viscous system; PS, phase separation.
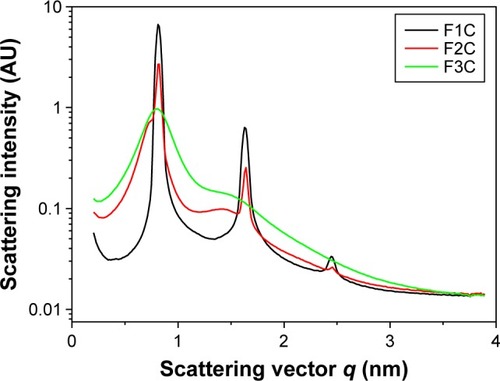
Micrography obtained using PLM shown in demonstrates that F3C is a microemulsion, because it is an isotropic transparent liquid system when visualized in a dark field, while the F2C and F1C formulations are lamellar and hexagonal LC mesophases. They are anisotropic samples identified by malt crosses and striations, respectively. In order to confirm the results obtained by PLM, SAXS measurements were performed, and are shown in . F3C presented a broad and wide peak characteristic of a microemulsion, confirming the PLM results. F1C and F2C indicated that these systems are lamellar LC mesophases, since lamellar mesophases followed the relation 2:1 and hexagonal mesophases 1:1.73:2:2.64.Citation41 PLM showed that only F1C is a hexagonal mesophase; therefore, F1C might be in a transition phase from the lamellar to hexagonal mesophase. These data show that F3C is a precursor of the LCS and that when water is incorporated, it becomes an LCS. Therefore, the findings support our goal to develop a flow formulation to facilitate administration with a syringe, which upon contact with saliva would become a viscous LC mesophase.
Figure 2 Polarized light microscopy of formulations F1C (A), F2C (B), and F3C (C). Magnification 20×.

shows the distance between lamellae:
Table 2 Values of qmax (Å) and distance (d) between lamellae
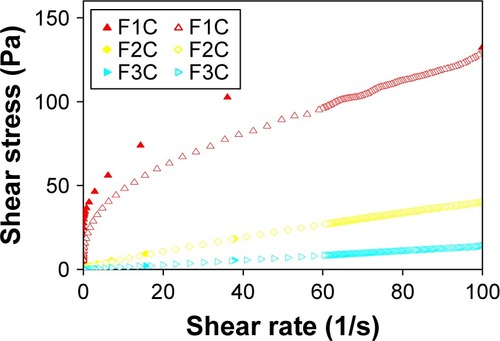
The flow-rheology data for F1C, F2C, and F3C are shown in and . The data showed that both F1C and F2C exhibited non-Newtonian pseudoplastic behavior (n<1), whereas F3C exhibited Newtonian behavior (n=1). Furthermore, the consistency index increased with the addition of water, indicating that F1C is a more viscous formulation. The F1C downward curve did not overlap its upward curve, instead forming an area of hysteresis, and was thus classified as time-dependent thixotropic.
Table 3 Flow behavior (n) and consistency index (κ) of the formulations
Figure 4 Flow properties of formulations F1C, F2C, and F3C.
Notes: The flow properties were determined using a controlled shear rate procedure ranging from 0.01 to 100 s−1 (or ascendent curve – filled symbols) and back (or descendant curve - empty symbols).
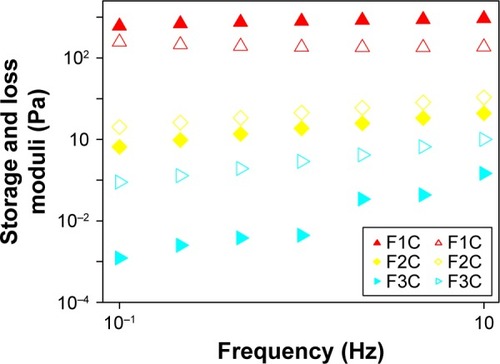
and show the oscillatory rheology data. F1C had a G′ value higher than the G′′ value, a characteristic of elastic formulations. F2C and F3C had higher G′′ values than G′ values, indicating the predominantly viscous behavior characteristic of poorly organized systems. Moreover, the S value of F1C was much higher than that of the other formulations, demonstrating that the bonds among the molecules of F1C are much more resistant.
Table 4 Strength (S) and the viscoelastic exponent (n) of the formulations
Figure 5 Variations in storage moduli (G′; filled symbols) and loss moduli (G′′; empty symbols) as a function of frequency for F1C, F2C, and F3C.
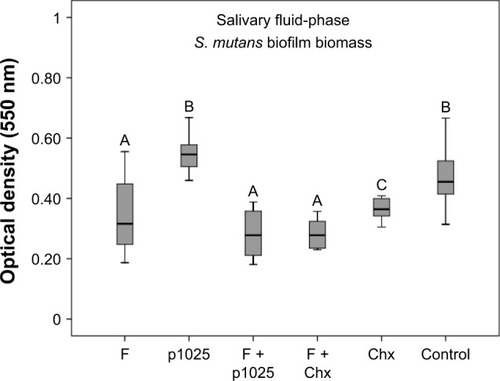
shows the results of the bioadhesion test, indicating that F1C had higher bioadhesion strength than F2C and F3C, suggesting that bioadhesion was strongly influenced by the type of LC mesophase. Results for the quantification of biofilm biomass in the two different phases of biofilm – surface-bound salivary phase and salivary fluid-phase – are shown in and , respectively. All treatments affected biofilm formation when applied in the surface-bound salivary phase. The best antibiofilm effects were observed for F + p1025 and F + Chx, without a statistical difference between them. When applied in the salivary fluid phase, p1025 had no effect on biofilm formation. There were no statistical differences among the F groups (F, F + p1025, and F + Chx). F + Chx had the greatest effect on biofilm formation.
Table 5 Work of bioadhesive force (mN⋅s) of the formulations. Values represent mean ± SD at 37°C
Figure 6 Quantification of Streptococcus mutans biofilm biomass after 24 hours treatment in the surface-bound salivary phase.
Notes: Bars indicate minimum and maximum values. Boxes indicate lower and upper quartiles, respectively. Lines in the middle of boxes are medians (n=12). Different uppercase letters indicate statistical differences among the groups, according to Kruskal–Wallis/Mann–Whitney tests (P<0.05).
Abbreviations: F, liquid-crystalline formulation; p1025, peptide p1025; Chx, chlorhexidine.
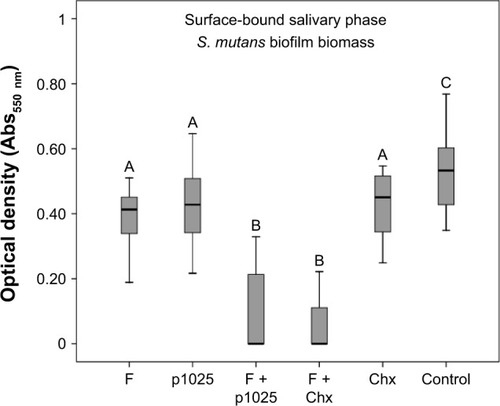
Figure 7 Quantification of Streptococcus mutans biofilm biomass 24 hours after treatment in the salivary fluid phase.
Notes: Bars indicate minimum and maximum values. Boxes indicate lower and upper quartiles, respectively. Lines in the middle of boxes are medians (n=12). Different uppercase letters indicate statistical differences among the groups, according to Kruskal–Wallis/Mann–Whitney tests (P<0.05).
Abbreviations: F, liquid-crystalline formulation; p1025, peptide 1025; Chx, chlo-rhexidine.
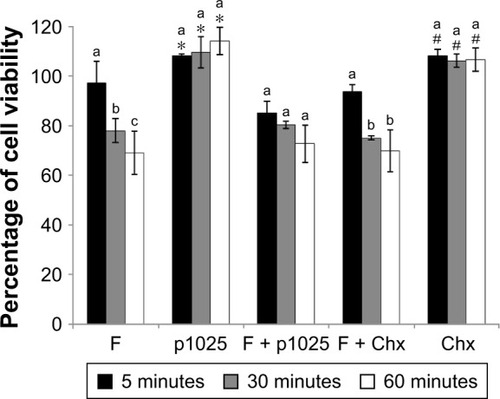
Data from cytotoxicity are presented in . Chx and p1025 solutions were not cytotoxic at any time evaluated. F increased its toxicity when exposure time increased. There was no statistical difference among exposure times for F + p1025. After 30 and 60 minutes, the toxicity of F + Chx increased significantly when compared with toxicity at 5 minutes. Overall, cell viability was 70% or higher for F, F + p1025, and F + Chx, independently of the time of exposure, showing low cytotoxicity for all the treatments.
Figure 8 Epithelial cell viability after 5, 30, and 60 minutes of treatment and growth for 24 hours.
Notes: *Significant difference between p1025 and the other groups (F, F + p1025, F + Chx), except for Chx, considering each time of exposure separately, according to ANOVA/Tukey’s tests; #significant difference between Chx and the other groups (F, F + p1025, F + Chx), except for p1025, considering each time of exposure separately, according to ANOVA/Tukey’s tests. Columns indicate means and bars indicate standard deviations (n=12). Different letters show significant differences among times of exposure (5, 30, or 60 minutes), considering each group separately, according to ANOVA/Tukey’s tests.
Abbreviations: F, liquid-crystalline formulation; p1025, peptide p1025; Chx, chlorhexidine; ANOVA, analysis of variance.
Discussion
We aimed to develop a bioadhesive LCS with in situ gelling properties to prevent the formation of S. mutans biofilm. For this reason, a phase diagram was constructed to determine the percentages of surfactant, oily phase, and aqueous phase required to obtain an LC-precursor system that (with the addition of water) could transform to an LCS for topical administration to the teeth.
After analysis of the phase diagram, it was possible to verify the formation of a fluid system and viscous system beyond phase separation. Specifically, in the region of 40% surfactant, it was possible to obtain a transition phase with the addition of the aqueous phase. In this region, formulations have low concentrations of surfactant, in order to decrease their potential toxicity.Citation33 Then, three formulations (F1C, F2C, and F3C) in this region were structurally analyzed by PLM, SAXS, rheology, and bioadhesion studies. PLM and SAXS showed that combining PPG-5-Ceteth-20, OA, 0.5% C974 dispersion, and water formed an LCS after the addition of water.
The flow-rheology study showed that the LCSs (F1C and F2C) are pseudoplastic fluids and the microemulsion (F3C) is a Newtonian fluid. The microemulsions had Newtonian behavior, since they did not present any type of organized structure that could be disassembled by shear stress.Citation43 However, pseudoplasticity can be due to the LC structure, which causes greater resistance to flow than the microemulsions, which are colloidal systems.Citation44 Such a property is desirable for formulations developed for oral administration, eg, during application at high shear rates, deinterlacing of the polymer chains, and subsequent thinning of the flow will occur, thereby facilitating administration of the formulation. However, upon withdrawal of this tension, the formulation will recover its initial viscosity, remaining longer in the oral environment.
F2C and F3C presented a very low degree of thixotropy. However, the formulation F1C presented a high degree of thixotropy. Thixotropic properties were directly related to the interaction between the components of the formulation. Because of the existence of interaction forces between the more structured regions, the structure can be destroyed by increasing the shear rate and easily recovered when that speed decreases. Therefore, the formulations with the highest degree of thixotropy also presented a higher degree of initial structuring. This increase in hysteresis is closely related to the increase in the microstructure of the LC networks, proving that hysteresis is strongly influenced by the presence of LC.Citation45
Oscillatory rheological analysis evaluates the viscoelastic properties of formulations and provides information about their structural nature, which directly affects their performance.Citation46 F2C and F3C had G′′ value that were higher than G′ values, indicating a predominantly viscous behavior, a characteristic of poorly organized systems. In contrast, F1C showed a highly elastic character. The lamellar phase generally appears as viscous liquid, and the hexagonal phase has a gel-like viscosity. Therefore, the rheological data corroborated the SAXS analysis, showing that when the aqueous phase increased, the system structure became more ordered, as indicated by the increase in lamellae spacing and elasticity.Citation47,Citation48
The main advantage of bioadhesive systems for drug delivery is maintenance of the drug at the application site for longer periods, which allows for enhanced contact of the formulation with the biological barrier, allowing for a reduction in the frequency of product application and increasing patient compliance.Citation49 The bioadhesion results showed that the increase in the aqueous phase in the systems significantly increased the bioadhesion of the formulations, due to the formation of LC. The highest values of bioadhesion for the LCS can be explained by their rheological properties. The increase in viscosity and elastic characteristics contributes to the increase in the residence time of the formulation.Citation44
Furthermore, bioadhesion can be also attributed to presence of the negatively charged polymer C974. Although the literature reports that chitosan hydrogels are very bioadhesive due to the forces of molecular attraction by electrostatic interactions between positive groups of chitosan and negative groups of the biological surface, we were able to demonstrate that the F1C obtained values within the range of bioadhesion for chitosan-based LCS values.Citation33 In this case, bioadhesion was attributable to physicochemical processes, such as hydrophobic interactions, hydrogen, and van der Waals interactions, which are controlled by pH and ionic composition.Citation50 In addition, their chains are flexible enough to diffuse into the saliva and penetrate to form a network. Most of the polyacrylic acid derivatives are not soluble in water, but form viscous gels when hydrated, increasing their ability to adhere to the tooth surface.Citation44
There is no standard formula available for bioadhesive drug-delivery systems for dental applications. However, the LCS formed by the C974 polymer as an aqueous phase was shown to be suitable, because upon hydration there was an increase in bioadhesion strength, pseudoplasticity, and elasticity, resulting in a promising platform for oral p1025 delivery.
In this study, p1025, a peptide that corresponds to residues 1025–1044 in the C-terminal region of SA I/II, was chosen to be incorporated into LCS. Marsh et al (2015) suggested that a delicate balance is needed to control microbiota at levels compatible with health, using substances able to inhibit bacterial traits implicated in disease and retard growth without eliminating beneficial species.Citation51 Thus, in this present study, p1025, a peptide that corresponds to residues 1025–1044 in the C-terminal region of SA I/II, was chosen to be incorporated into LCS, because p1025 is able to inhibit the binding between S. mutans and salivary agglutinin, reducing adhesion and consequent biofilm formation.Citation18
In the present study, when applied in the salivary surface-bound phase, p1025, whether or not it was incorporated into the LCS, showed an inhibitory effect on biofilm formation. It is known that synthetic peptide p1025 is able to bind to salivary agglutinins and interfere with S. mutans adhesion mediated by SA I/II, as confirmed by surface plasmon resonance.Citation18 However, this effect was not observed when p1025 was applied in the salivary fluid-phase. It is possible that p1025 attached to salivary agglutinins is not degraded as easily by salivary proteases as it is in the fluid-phase.
Data from literature reports reveal that the release of agents from the LCS is around 4% at 24 h.Citation52 Although data from the release of antimicrobial agents were not available in the present study, the peptide and Chx release from LCS was probably higher than 4%, since the formulation was diluted previously to reduce viscosity and incorporate antimicrobial agents for biofilm assays.
There was an increase in the anti-biofilm activity of F+p1025 compared to p1025 and F+Chx compared to Chx within 24 h, showing a cumulative effect of these agents when incorporated into F. This result may suggest that p1025 and Chx were released slowly within 24 h, since the mean bacterial reduction was around 20% for p1025/Chx and 80% for F+p1025/F+Chx. Lower bacterial reduction (around 40%) was observed for both F containing p1025 or Chx when applied in the salivary fluid-phase, and this was probably related to dilution in the saliva.
An additional antimicrobial effect of F (around 27%) was observed from the biofilm assays, adding to the effect of F (around 27%) was observed from the biofilm assays, adding to the effect of the incorporated agents. The high concentration of oil in the LCS limits water access, which is one of the main methods of antimicrobial action.Citation53
There are few studies evaluating the inhibitory activity of p1025 on pathogenic microorganisms related to dental caries etiology. Kelly et al demonstrated that topical application of p1025 might selectively prevent recolonization of the tooth surface by S. mutans.Citation18 The relatively long-term resistance to colonization cannot be explained by persistence of the peptide in the oral cavity. The authors suggested that the peptide competitively inhibited initial adhesion of the bacterium to the dental structure, but other bacteria competing for the same ecological niche were able to prevent subsequent colonization. Li et al evaluated the influence of p1025 in solution and two different dentifrices containing p1025 on the adherence of S. mutans, and observed an inhibitory effect in both hydroxyapatite (in vitro) and dental biofilm (in vivo).Citation19 The most significant inhibitory activity in vitro was shown at 50 μmol/L. The clinical efficacy of the dentifrices was confirmed by the significant reduction in plaque scores and S. mutans counts from subjects after 1 month’s treatment with dentifrices containing p1025 in comparison to a control dentifrice.
No study has evaluated the toxicity of p1025 or this formulation of LCS against eukaryotic cells. In the present study, p1025 was not toxic for epithelial cells; however, when it was incorporated into the LCS, there was a significant reduction in cell viability, although viability was maintained at around 70% until 1 hour of exposure. Considering the high capacity of pathogenic bacteria for developing resistance to current antimicrobial agents, the topical administration on teeth of the p1025-loaded LCS, a clinically appropriate and safe formulation, provides a realistic adjunctive approach to prevention of the formation of S. mutans dental biofilm.
Conclusion
An LCS was successfully developed combining polyoxypropylene-(5)-polyoxyethylene-(20)-cetyl alcohol, OA, and C974P dispersion. Characterization tests revealed that the addition of water caused a phase transition consisting of the formation of a hexagonal LC mesophase with pseudo-plasticity, high viscosity, and superior bioadhesion to the teeth. The p1025-loaded LCS showed limited cytotoxicity and presented an effect against the formation of S. mutans biofilm. Taken together, the results suggest that this p1025-loaded LCS offers a promising alternative for the prevention of S. mutans biofilm. Future in vivo studies are needed to evaluate the efficiency of this approach.
Acknowledgments
This study was funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) grants 2012/19235-5, 2013/12285-0, and 2013/01565-1, CNPq, and PADC (Programa de Apoio ao Desenvolvimento Científico-FCF-UNESP).
Disclosure
The authors report no conflicts of interest in this work.
References
- HahnelSMühlbauerGHoffmannJStreptococcus mutans and Streptococcus sobrinus biofilm formation and metabolic activity on dental materialsActa Odontol Scand201270211412121780966
- AlamSBrailsfordSRAdamsSGenotypic heterogeneity of Streptococcus oralis and distinct aciduric subpopulations in human dental plaqueAppl Environ Microbiol20006683330333610919787
- HamadaSSladeHDBiology immunology, and cariogenicity of Streptococcus mutansMicrobiol Rev19804423313846446023
- LagerweijMDvan LoverenCDeclining caries trends: are we satisfied?Curr Oral Health Rep20152421221726523247
- MarshPDRole of the oral microflora in healthMicrob Ecol Health Dis2000123130137
- MarshPDDental plaque as a biofilm and a microbial community: implications for health and diseaseBMC Oral Health20066S1416934115
- LoescheWJMicrobiology of dental decay and periodontal diseaseBaronSMedical Microbiology4th edGalveston (TX)University of Texas1996
- FilocheSWongLSissonsCHOral biofilm: emerging concepts in microbial ecologyJ Dent Res201089181819918089
- RussellMWBergmeierLAZandersEDLehnerTProtein antigens of Streptococcus mutans: purification and properties of a double antigen and its protease-resistant componentInfect Immun19802824864936995311
- SmithRLehnerTBeverleyPCCharacterization of monoclonal antibodies to Streptococcus mutans antigenic determinants III, I, II, and III and their serotype specificitiesInfect Immun19844611681756207107
- LeeSFProgulske-FoxABleiweisASMolecular cloning and expression of a Streptococcus mutans major surface protein antigen, P1 (I/II), in Escherichia coliInfect Immun1988568211421193135272
- SmithDJCaries vaccines for the twenty-first centuryJ Dent Educ200367101130113914587678
- RobinetteRAHeimKPOliMWCrowleyPJMcArthurWPBradyLJAlterations in immunodominance of Streptococcus mutans Ag I/II: lessons learned from immunomodulatory antibodiesVaccine201432337538224252705
- YounsonJKellyCThe rational design of an anti-caries peptide against Streptococcus mutansMol Divers20048212112615209163
- SanuiTGregoryRLAnalysis of Streptococcus mutans biofilm proteins recognized by salivary immunoglobulin AOral Microbiol Immunol200924536136819702948
- PecharkiDPetersenFCAssevSScheieAAInvolvement of antigen I/II surface proteins in Streptococcus mutans and Streptococcus intermedius biofilm formationOral Microbiol Immunol200520636637116238597
- CarlénAOlssonJMonoclonal antibodies against a high-molecular-weight agglutinin block adherence to experimental pellicles on hydroxyapatite and aggregation of Streptococcus mutansJ Dent Res1995744104010477782534
- KellyCGYounsonJSHikmatBYA synthetic peptide adhesion epitope as a novel antimicrobial agentNat Biotechnol199917142479920267
- LiMYWangJLaiGYEffect of a dentifrice containing the peptide of streptococcal antigen I/II on the adherence of mutans streptococcusArch Oral Biol200954111068107319735908
- SouzaCWatanabeEBorgheti-CardosoLNFantiniMCLaraMGMucoadhesive system formed by liquid crystals for buccal administration of poly(hexamethylene biguanide) hydrochlorideJ Pharm Sci1031239143923
- BernegossiJCalixtoGMSanchesPRPeptide KSL-W-loaded mucoadhesive liquid crystalline vehicle as an alternative treatment for multispecies oral biofilmMolecules2015211E3726712726
- BenergossiJCalixtoGFonseca-SantosBHighlights in peptide nanoparticle carriers intended to oral diseasesCurr Top Med Chem201515434535525579347
- GajdziokJBajerováMChalupováZRabiskováMOxycellulose as mucoadhesive polymer in buccal tabletsDrug Dev Ind Pharm20103691115113020545516
- LoftssonTLeevesNBjornsdottirBDuffyLMassonMEffect of cyclodextrins and polymers on triclosan availability and substantivity in toothpastes in vivoJ Pharm Sci199988121254125810585219
- GuoCWangJCaoFLeeRJZhaiGLyotropic liquid crystal systems in drug deliveryDrug Discov Today20101523–241032104020934534
- CalixtoGBernegossiJFonseca-SantosBChorilliMNanotechnology-based drug delivery systems for treatment of oral cancer: a reviewInt J Nanomedicine201593719
- de SouzaALKiillCPdos SantosFKNanotechnology-based drug delivery systems for dermatomycosis treatmentCurr Nanosci201284512519
- CalixtoGMGarciaMHCilliEMChiavacciLAChorilliMDesign and characterization of a novel p1025 peptide-loaded liquid crystalline system for the treatment of dental cariesMolecules201621215826828470
- BruschiMLde FreitasOLaraEHPanzeriHGremiãoMPJonesDSPrecursor system of liquid crystalline phase containing propolis microparticles for the treatment of periodontal disease: development and characterizationDrug Dev Ind Pharm200834326727818363142
- CalixtoGYoshiiACRocha e SilvaHCuryBSChorilliMPolyacrylic acid polymers hydrogels intended to topical drug delivery: preparation and characterizationPharm Dev Technol201520449049625975700
- CarvalhoFCCalixtoGHatakeyamaINLuzGMGremiãoMPChorilliMRheological, mechanical, and bioadhesive behavior of hydrogels to optimize skin delivery systemsDrug Dev Ind Pharm201339111750175723216218
- JonesDSBruschiMLde FreitasOGremiãoMPLaraEHAndrewsGPRheological, mechanical and mucoadhesive properties of thermoresponsive, bioadhesive binary mixtures composed of poloxamer 407 and carbopol 974P designed as platforms for implantable drug delivery systems for use in the oral cavityInt J Pharm20093721495819429268
- SalmaziRCalixtoGBernegossiJRamosMABauabTMChorilliMCurcumin-loaded liquid crystalline precursor mucoadhesive system for the treatment of vaginal candidiasisInt J Nanomedicine2015104815482426257519
- SoaresDGRibeiroAPSaconoNTColdebellaCRHeblingJCostaCATransenamel and transdentinal cytotoxicity of carbamide peroxide bleaching gels on odontoblast-like MDPC-23 cellsInt Endod J201144211612521083572
- MarquesMRLoebenbergRAlmukainziMSimulated biological fluids with possible application in dissolution testingDissolut Technol20111831528
- GawandePVLeungKPMadhyasthaSAntibiofilm and antimicrobial efficacy of Dispersin B KSL-W peptide-based wound gel against chronic wound infection associated bacteriaCurr Microbiol201468563564124445333
- AhnSJWenZTBradyLJBurneRACharacteristics of biofilm formation by Streptococcus mutans in the presence of salivaInfect Immun20087694259426818625741
- CastilloMCFunção de DNA Extracelular e de Acido Lipoteicóico nas Propriedades Estruturais e Funcionais da Matriz Extracelular de Biofilmes Cariogênicos [master’s thesis]São PauloSão Paulo State University2016
- Mattos-GranerRONapimogaMHFukushimaKDuncanMJSmithDJComparative analysis of Gtf isozyme production and diversity in isolates of Streptococcus mutans with different biofilm growth phenotypesJ Clin Microbiol200442104586459215472313
- BedranTBEfeito antimicrobiano e modulador da resposta imune dos peptídeos hBD-3 e LL-37 e dos polifenóis o chá verde e do cranberry2014 Available from: https://repositorio.unesp.br/handle/11449/124090Accessed October 19, 2017
- OyafusoMHCarvalhoFCTakeshitaTMDevelopment and in vitro evaluation of lyotropic liquid crystals for the controlled release of dexamethasonePolymers201798330
- GuillotSMéducinFPoljakKNanostructured monolinolein mini-emulsions as delivery systems: role of the internal mesophase on cytotoxicity and cell internalizationInt J Pharm2017523114215028284919
- PènzesTErősIldikó CsókaIstvánRheological analysis of the structural properties effecting the percutaneous absorption and stability in pharmaceutical organogelsRheol Acta2004435457463
- CarvalhoFCSilvaHRda LuzGMRheological, mechanical and adhesive properties of surfactant-containing systems designed as a potential platform for topical drug deliveryJ Biomed Nanotechnol20128228028922515079
- ChorilliMPrestesPSRigonRBStructural characterization and in vivo evaluation of retinyl palmitate in non-ionic lamellar liquid crystalline systemColloids Surf B Biointerfaces201185218218821411295
- ChorilliMRigonRBCalixtoGRheological characterization and safety evaluation of non-ionic lamellar liquid crystalline systems containing retinyl palmitateJ Biomed Nanotechnol201612239440327305773
- GabbounNHNajibNMIbrahimHGAssafSRelease of salicylic acid and diclofenac acid salts from isotropic and anisotropic nonionic surfactant systems across rat skinInt J Pharm20012121738011165822
- CintraGAPintoLACalixtoGMBioadhesive surfactant systems for methotrexate skin deliveryMolecules2016212E23126901183
- SmartJDRileyRGTsibouklisJThe retention of 14C-labelled poly(acrylic acids) on gastric and oesophageal mucosa: an in vitro studyEur J Pharm Sci2003201839013678796
- WoodleyJBioadhesion: new possibilities for drug administration?Clin Pharmacokinet2001402778411286325
- MarshPDHeadDADevineDAEcological approaches to oral biofilms: control without killingCaries Res201549Suppl 1465425871418
- HuFQHongYYuanHPreparation and characterization of solid lipid nanoparticles containing peptideInt J Pharm20042731–2293515010127
- OliveiraTAPaixãoFGPrestesOSAvaliação da atividade antimicrobiana de sistemas nanoestruturadosLat Am J Pharm2007266878882