 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
The aim of this study was to develop novel biomedicated nanofiber electrospun mats for controlled drug release, especially drug release directly to an injury site to accelerate wound healing. Nanofibers of poly(vinyl alcohol) (PVA), poly(vinyl acetate) (PVAc), and a 50:50 composite blend, loaded with ciprofloxacin HCl (CipHCl), were successfully prepared by an electrospinning technique for the first time. The morphology and average diameter of the electrospun nanofibers were investigated by scanning electron microscopy. X-ray diffraction studies indicated an amorphous distribution of the drug inside the nanofiber blend. Introducing the drug into polymeric solutions significantly decreased solution viscosities as well as nanofiber diameter. In vitro drug release evaluations showed that both the kind of polymer and the amount of drug loaded greatly affected the degree of swelling, weight loss, and initial burst and rate of drug release. Blending PVA and PVAc exhibited a useful and convenient method for electrospinning in order to control the rate and period of drug release in wound healing applications. Also, the thickness of the blend nanofiber mats strongly influenced the initial release and rate of drug release.
Introduction
A wound dressing should ideally provide an optimal healing environment in which healing can occur at a maximum rate compatible with the cosmetic appearance of the healed wound;Citation1,Citation2 however, the infection under dressing caused by burns, split skin graft donor sites, pressure, sores, and diabetic ulcers can lead to prolonged healing time or, in serious conditions, death. Consequently, wounds often require treatment with antibiotics.Citation2–Citation5 Local delivery of drugs sustained for about 1 week is preferred to systemic administration, especially for chronic wounds,Citation5 because of the advantages of these delivery systems such as: a) improving the effectiveness of drug therapy, and selective targeting, b) decreasing the side effects on a tissue, and interference with wound healing, c) reduction of bacterial resistance, and d) reducing the frequency of dressing replacements to increase patient compliance.Citation4,Citation5 Furthermore, a proper wound dressing needs to have wound healing properties, including the essential swelling capability for absorbing excess exudates and oxygen permeability for respiring.Citation5,Citation6 Modern dressings as vehicles for delivering therapeutic agents to wound sites have been applied in the form of hydrogels,Citation2 films,Citation7 sponges,Citation8 foams,Citation9 and recently in the form of nanofibers.Citation6,Citation10 However, little research has been published on the controlled delivery of drugs from polymeric dressing, especially in the form of nanofibers.
In the last decade, nanofibers have attracted increasing attention for their use in biomedical applications such as wound dressings,Citation10–Citation12 tissue engineering,Citation11 and controlled drug delivery.Citation13–Citation21 The very fine diameter and high porous structure of nanofiber mats help drug particles diffuse out of the matrix more efficiently.Citation14,Citation15
The most popular, preferred, and inexpensive technique for fabricating polymeric nanofibers is electrospinning. Unlike common techniques, electrospinning is more convenient for incorporating therapeutic compounds into the polymeric matrix.Citation11 Using electrospun nanofiber mats for wound dressings also has some benefits, such as hemostatic properties, absorbability and semipermeability for respiration of cells, better conformation to the wound surface,Citation11 and the potential for leaving no scar.Citation11,Citation12
Among wound dressings, special attention has been paid to biodegradable polymeric dressings because they can easily be washed off the wound surface.Citation4,Citation5 Poly(vinyl alcohol) (PVA) is a well-known, biologically friendly, biodegradable polymer owing to its desirable properties such as nontoxicity, no carcinogenicity, biocompatibility, and appropriate mechanical properties.Citation1,Citation2,Citation7,Citation18 Because of these properties, PVA is used in some biomedical applications such as soft contact lenses, implants of artificial organs, cartilage skin, and cardiovascular devices.Citation2,Citation18 Because of its flexibility and swelling capability in an aqueous medium, PVA has been much studied as a wound dressing;Citation2 however, its poor stability in water has limited its use in aqueous systems, particularly for drug-delivery applications.Citation18,Citation22 To overcome this problem, PVA has been made insoluble by grafting,Citation18 copolymerizing,Citation22 and cross-linking,Citation23 which require some additional and sometimes complicated and time-consuming processes. In wound dressing applications, PVA has been crosslinked with polyvinyl pyrrolidone,Citation2 sodium alginate,Citation24 and sterculiaCitation25 to provide better characteristics, although a vey quick release burst was reported and a sustained release continued up to 90 hours, 5 hours, and 24 hours, respectively. Poly(vinyl acetate) (PVAc), a biocompatible and biodegradable (owing to the hydrolyzable groups in the side chain) polymer, has also been used in biomedical applications, including drug and cell carriers, and tissue engineering.Citation26,Citation27
PVA is a well-known biocompatible and nontoxic polymer. Biocompatibility of PVA is due to both its compatibility with tissue and blood (plasma proteins), which has been widely reported. For this reason, PVA has been used in numerous devices in contact with blood and tissues such as drug-delivery systems, wound dressings, dialysis membranes, cardiovascular devices, artificial cartilage, tendon, ligament, cornea, lens, skin, and intervertebral disc.Citation4,Citation23,Citation28 A good blood-contact property was observed for pure PVA.Citation29 Moreover, bovine serum albumin (BSA) tends to adsorb to hydrophobic surfaces more than hydrophilic ones.Citation30 PVA has good blood compatibility.Citation24 Ex vivo experiments with canine whole blood have shown that formation of thrombus on PVA is less than on siliconized glass.Citation31 Methods such as blending, grafting, and copolymerization of PVA with other polymers have improved blood compatibility of those polymers, indicating a very high blood compatibility of PVA.
Several reports have indicated blood compatibility of PVAc. PVAc has also been applied in many medical fields because of its biocompatibility. Hydrogels such as PVAc containing functional groups such as COOH usually show good biocompatibility in contact with blood, body fluids, and tissues.Citation32 In another study, PVAc did not adsorb serotonin from platelet-free plasma, and did not cause lysis of erythrocytes.Citation33 PVAc is an inert polymer with the advantage that it does not induce a deleterious reaction in living tissue. Histological study of the embolized rat kidney revealed no detectable damage in the vessel wall and no recanalization for up to 6 months. Also it was shown that PVAc remained inert in blood vessels.Citation34 Several patents are registered for novel compositions suitable for use in embolizing blood vessels using biocompatible polymers including PVAc. The composition of these inventions are particularly useful in embolizing blood vesselsCitation35 or they can be applied to intravascular medical devices.Citation36 The amount of serum albumin adsorbed onto the PVAc latex is considerably low, especially at a pH of 5 to 8, indicating its good compatibility with blood.Citation37 There were no procedural complications, and no thrombus formation on the electrodes of the PVAc catheter.Citation38 Several studies have shown that PVAc improves blood compatibility of some polymers, thus demonstrating the good blood compatibility of this polymer.Citation39
Ciprofloxacin, a fluoroquinolone antibiotic, is one of the most widely used antibiotics in wound healing because of its low minimal inhibitory concentration for both Gram-positive Staphylococcus and Gram-negative Pseudomonas bacteria that cause wound infections.Citation2,Citation3
In the present research, an economical and convenient method of blending was applied to achieve a simultaneous controlled release of CipHCl and high swelling capacity along with coverage conformability for wound healing administration. PVAc, as an inexpensive and hydrophobic polymer, was selected to be blended with PVA solution. While PVAc is insoluble in water (a good solvent of PVA),Citation18 an aqueous solution of 50% acetic acid was used as an appropriate solvent to dissolve both polymers and also CipHCl to attain a stable blend solution. The resulting solution was electrospun into nanofibers.
Materials and methods
Materials
PVA (MW = 72,000 Da, hydrolysis 98%) and acetic acid were purchased from Merck Chemical Co (Darmstadt, Germany), PVAc (MW = 55,000–70,000 Da) was obtained from Carl Roth GmbH (Karlsruhe, Germany), and CipHCl was supplied from TEMAD Co (Tehran, Iran).
Preparation of pure and drug-loaded nanofiber mats
Aqueous acetic acid solution diluted to 50% with distilled water was used as the solvent for PVA and PVAc. The PVA solutions were prepared by dissolving 5% w/v of PVA powder in 50% acetic acid solution at 70°C for 3 hours. The obtained solution was stirred for 17 hours at room temperature. To prepare PVAc solutions, 32% w/v of PVAc granules was dissolved in 50% acetic acid solution and stirred at room temperature for at least 20 hours. PVAc granules were added to the cooled solution of PVA and stirred for 17 hours to prepare the blend solutions of 50:50 by weight (8.5% w/v). CipHCl powder (5 or 10% w/w based on the polymer used) was added to the polymer solutions 2 hours before electrospinning.
The electrospinning solution was transferred to a 1 mL syringe pump with a right angle-shaped needle (gauge 22) attached to it. The resulting fibers were collected on a grounded aluminum plate. All electrospinning processes were carried out at 25°C. The solution flow rate was 0.8 to 1.3 mL/hour, the range of applied positive voltage was 15 to 19 kV, and the distance between the needle tip and the target was 22 cm. The mat thickness was measured using a thickness tester (Model No 674, range 0–10.00 mm; Hans Baer, Zurich, Switzerland). The thickness of the electrospun nanofiber mats ranged from 300 to 360 μm. The electrospun nanofiber mats of PVA:PVAc (50:50 by weight) are hereafter termed blend nanofiber mats.
Characterization of electrospun solutions
Before the electrospinning, the conductivity and viscosity of solutions were measured using a conductivity meter (3540; Jenway, Germany) and a Brookfield digital viscometer (LVDV-II + PRG, Middleboro, MA), respectively. The average value of three determinations was obtained and reported as mean ± SD.
Morphology studies
The morphology of electrospun nanofibers was studied by a scanning electron microscope (XL-30, SEM; Philips, Amsterdam, the Netherlands) after gold coating. The average diameter was determined from the SEM images of each sample. From each image, at least 100 different fiber segments were randomly selected and their diameters were measured using Motic software (v 2, Richmond, BC).
X-ray diffraction studies
X-ray diffraction (XRD) was used to investigate the effect of electrospinning on the degree of crystallization of the drug as well as the influence of the drug on the crystalline structure of polymer nanofibers. The XRD patterns of pure CipHCl and blend nanofiber mats with and without drug were determined using a diffractometer (X pern; Philips, Amsterdam, the Netherlands). The scanning range varied from 2θ = 5° to 40°.
Degree of swelling and weight loss
The degree of swelling and weight loss of nanofiber mats were calculated by EquationEquations 1(1) and Equation2
(2) , respectively. Both tests were carried out in the release medium (phosphate buffered saline [PBS], pH 7.4) at 37°C for 1, 4, and 24 hours.
In vitro drug release studies
Drug release from electrospun nanofiber mats was measured by placing a known mass and approximate dimensions (2.5 × 2.5 cm) of material into 20 mL of PBS (pH 7.4) under constant stirring at 37°C. At certain time points, a nanofiber sample was taken out of the buffer and placed into a fresh buffer solution. The amount of drug released was determined using a UV spectrophotometer at λmax = 271 nm. The release experiments of each sample were performed in triplicate, and average values are reported.
Analysis of drug-release kinetics and modeling
The release kinetics of drugs from a polymer carrier can be described using the Peppas equation:
Statistical analysis
Statistical analysis of data was performed using SPSS software package (v 11; SPSS, Inc, Chicago, IL) by one-way analysis of variance (ANOVA), assuming a confidence level of 95% (P < 0.05) for statistical significance. All data were presented as mean ± standard deviation.
Results and discussion
Morphological structure studies
During electrospinning, as the liquid jet (polymeric solution) is continuously elongated, the solvent of polymeric jet is evaporated quickly, phase separation occurs, the jet solidifies, and nanofibers are formed. In this process, nanofibers without solvent are deposited on the collector; however, to ensure that acetic acid did not remain in the nanofiber mats, the mats were dried in a vacuum dryer for 48 hours (at room temperature) to remove the solvent. This method is also reported in other referencesCitation41–Citation43 for removing DMF, trifluoroacetic acid, and 50% acetic acid from nanofibers. For in vivo experiments, however, some methods such as gas chromatography may be used to verify if solvent remains in the webs after vacuum drying.
As seen in , no beaded nanofibers and no drug crystals were detected by electron microscopy on the surface or outside the fibers loaded with the drug, showing that CipHCl was loaded in the nanofibers, and demonstrating a good compatibility of drug–polymers–solvent.Citation19 Furthermore, indicate that loading CipHCl (10% wt) into the blend nanofibers (50:50 PVA:PVAc) did not influence the appearance of the nanofiber mats.
Figure 1 SEM photographs of electrospun nanofibers without drug and with 10% w/w drug loaded: A, B) PVA; C, D) PVAc; E, F) a 50:50 blend of PVA/PVAc; G, H) effect of drug loading on appearance of PVA/PVAc blend nanofiber mats.
Abbreviations: CipHCl, ciprofloxacin HCl; PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.


The resulting nanofibers also showed that the incorporation of the drug into the nanofibers not only dramatically decreased their average diameter but also reduced the diameter distribution of electrospun nanofibers from 198, 218, and 448 nm to 145, 180, and 405 nm for PVA, PVAc, and a blend of PVA/PVAc, respectively, as shown in . The measurements of viscosity and conductivity of solutions showed that the addition of CipHCl can significantly decrease solution viscosity (); however, it did not affect solution conductivity (data not shown). Solution viscosity influences the morphological structure and average size of resulting fibers.Citation13,Citation16,Citation20
Figure 2 Effect of CipHCl on A) the diameter of nanofibers (n = 100) and B) the solution viscosity (n = 3).
Abbreviations: PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.
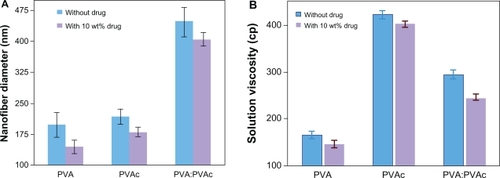
A possible explanation for the decreasing viscosity of the polymeric solution by addition of CipHCl is that the drug could be trapped between polymeric chains, and thus acts as a plasticizer.Citation16
In general, the electrospun polymeric solution with higher viscosity causes more resistance to elongation during electrospinning. In other words, a decrease in solution viscosity causes the spinning jet to be stretched and elongated more easily. Consequently, finer fibers can result.Citation13
XRD studies
CipHCl powder, pure electrospun PVA:PVAc nanofibers, and 10% w/w drug-loaded PVA:PVAc nanofibers were studied by XRD in order to further illustrate the physical structure and distribution of the drug in the nanofibers. As seen in , CipHCl has several characteristic peaks at 2ϑ = 19.04, 24.8, 26.6, and 29.4 because of its regular crystallization.Citation44 XRD pattern of pure PVA:PVAc nanofibers seen in shows typical crystalline peaks at 2ϑ = 13.9, 16.8, 18.4, 26.3, and 28.3. Blend nanofiber mats loaded with 10% w/w CipHCl have the same peaks at 2ϑ = 13.8, 16.9, 18.3, 26.2, and 28.2 (), proving that loading the drug did not change the typical crystallinity of the PVA:PVAc blend nanofibers.
Figure 3 X-ray diffraction of A) CipHCl powder, B) pure 50:50 PVA/PVAc nanofiber mat, and C) 50:50 PVA/PVAc nanofiber mat loaded with 10% w/w drug.
Abbreviations: CipHCl, ciprofloxacin HCl; PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.
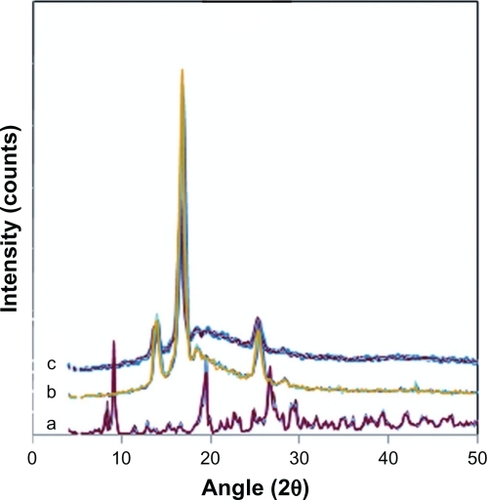
Disappearance of the peaks corresponding to the crystalline CipHCl in PVA:PVAc nanofibers containing 10% w/w drug verified that the drug was dispersed in the polymeric nanofiber matrix in the amorphous phase.
Degree of swelling and weight loss
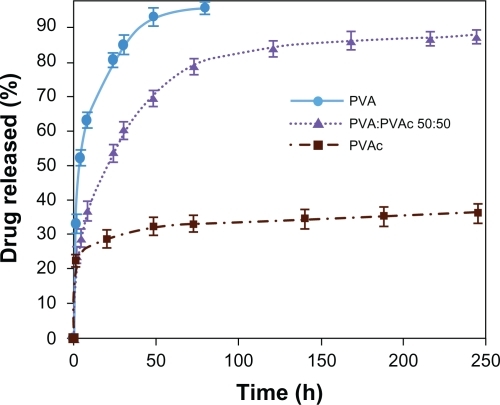
show the degree of swelling and weight loss of PVA, PVAc, and 50:50 blend nanofiber mats in the release medium (PBS, pH 7.4) at 37°C for 1, 4, and 24 hours. As can be seen, both the degree of swelling and weight loss, which are two important factors contributing to drug release, were in the order of PVA > the blend of PVA:PVAc (50:50) >> PVAc nanofiber mats owing to their hydrophilic–hydrophobic properties.
Figure 4 Effect of the type of polymer and/or the drug content A) on the degree of swelling (%) and B) on the weight loss (%) of PVA, PVAc and a 50:50 blend nanofiber mat, and C) effect of the thickness of blend nanofiber mats on the degree of swelling (%) (n = 3).
Abbreviations: PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.
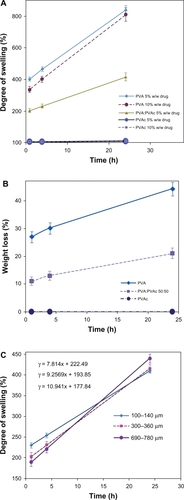
Although the degree of swelling of the blend nanofiber mats leveled off in comparison with PVA nanofibers, it was still high enough to be used in wound dressings to absorb excess exudates even from deep wounds with high amounts of exudates, compared with other results reported.Citation1–Citation3,Citation23–Citation25 The degree of swelling is 203%, 231% and 416% after 1 h, 4 h, and 24 h submersion of the blend nanofibrous mats, respectively.
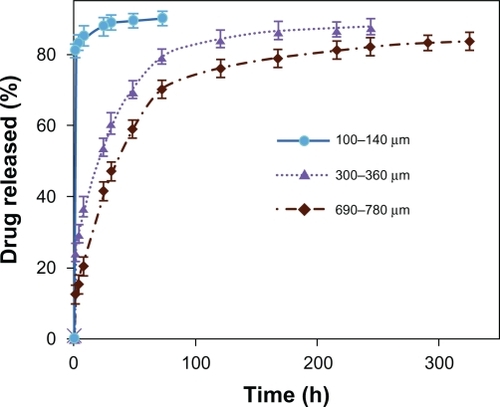
As demonstrated in , at the earlier stages, the degree of swelling and the thickness of the PVA:PVAc blend nanofiber mats are inversely related, ie, the thinner the nanofiber mats, the greater the degree of swelling. However, the degree of swelling after 24 hours of submersion was greater in the thicker nanofibers.
As calculated from the slope of the lines of the changes of swelling vs time (), the swelling rate for the fibers with a thickness of 100 to 140 μm is 7.8% per hour, for 300 to 360 μm 9.2% per hour, and for 690 to 780 μm 10.9% per hour. Nanofiber mats are composed of a great many nanofiber layers: the thicker the mat, the more the layers, and therefore the more compact the interior layers of nanofibers. Over time, the liquid phase has enough time to penetrate into the interior layers and reaches a balance. On the other hand, the more compact areas in thicker nanofibrous mats result in the entrapment of more aqueous molecules in these spaces. As a result, the degree of swelling increases with increased thickness of nanofibrous mats.
Less aqueous molecules at the earlier stage and more at the later stage can cause a greater increase in the rate of swelling in thicker mats than in thinner mats.
In vitro drug release study from nanofiber mats
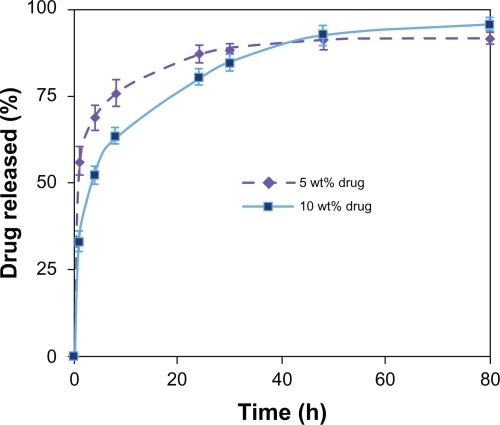
displays the cumulative drug release from PVAc nanofibers loaded with different amounts of CipHCl up to 1920 hours. The results show a relatively quick initial release. Moreover, increasing the drug content from 5% to 10% w/w approximately doubled the initial drug release.
Figure 5 Effect of drug content on release profiles of CipHCl from PVAc (32% w/v) electrospun nanofiber mats vs time (n = 3).
Abbreviations: CipHCl, ciprofloxacin HCl; PVAc, polyvinyl acetate.
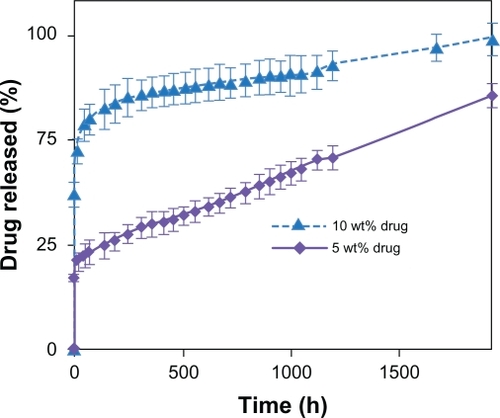
We deduced that although aqueous 50% acetic acid can provide a homogenous solution of PVAc and CipHCl, the incompatibility between hydrophobic PVAc and hydrophilic CipHCl45 would increase the migration of the drug molecules toward the nanofibers surface during the rapid elongation and quick solvent evaporation during electrospinning. These superficial drug molecules can easily diffuse into the aqueous medium and cause an initial quick burst release. As a result, by increasing the drug content, a greater portion of the drug will be located near the surface; therefore, initial release of the drug will be faster.Citation16
After a quick burst release, the profiles of release followed a linear fashion with a very slow rate, ie, about 50% of the drug was released over 80 days of the experiment. This prolonged period of drug release has never been seen in nanofibers before.
The release profiles of CipHCl from the PVA electrospun nanofibers shown in indicate a high initial release as well as a rapid release rate in which total release time was about 80 hours for both amounts of the drug loaded. It has been found that one of the significant factors contributing to drug release in controlled release systems is the behavior of the matrix loaded with the drug in the release medium. As soon as a PVA matrix is exposed to a liquid medium, it begins to swell (), its molecular chains are solvated, and weight loss occurs (). As a result, the high degree of swelling along with the dissolution of the PVA nanofiber mats, which is attributed to its poor stability in aqueous mediums, causes a quick burst release, as reported previously.Citation8,Citation20,Citation18
Figure 6 Effect of drug content on release profiles of CipHCl from PVA (5% w/v) electrospun nanofiber mats vs time (n = 3).
Abbreviations: CipHCl, ciprofloxacin HCl; PVAc, polyvinyl acetate.
According to these profiles, increasing the drug content in PVA nanofiber mats from 5% to 10% w/w lowered initial drug release and improved the control of the release profile. According to , at the initial experiment stages, a higher drug content (10% w/w) in PVA nanofiber mats results in a lower degree of swelling. Consequently, the penetration of the receiving liquid phase through PVA nanofibers decreases and diffusion of the drug from the nanofibers will be retarded. Kumbar and AminabhaviCitation46 and Sanli et alCitation22 studied indomethacine release from polyacrylamide grafted chitosan microspheres and diclofenac sodium release from PVA–sodium alginate beads, respectively. They also observed that increasing the drug content slowed drug release.
compares the effect of polymeric matrix on the initial drug release and the rate of drug release. While PVAc nanofiber mats showed a very slow drug release (regardless of the quick initial release), and PVA nanofiber mats exhibited a rapid rate of drug release and blend nanofiber mats of PVA:PVAc (50:50) a moderate CipHCl release rate (). In other words, incorporation of hydrophobic PVAc into PVA nanofibers decreased the diffusion of release medium into nanofiber mats () and reduced the weight loss (). As a result, it could be possible to control the rate and the burst release of the drug, in comparison with PVA nanofiber mats, to prolong drug release to at least 250 hours.
Figure 7 Effect of polymers blending on the release profiles of CipHCl from medicated electrospun nanofiber mats containing 10% w/w CipHCl vs time (n = 3).
Abbreviations: CipHCl, ciprofloxacin HCl; PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.
illustrates the effect of the thickness of blend nanofiber mats on the release profile if the diameters of the nanofibers were the same. Both the initial release and the rate of drug release from the blend nanofiber mats of PVA:PVAc (50:50) significantly decreased as the thickness of the mats increased, which was related to the swelling behavior of these mats (). Furthermore, the total release time was considerably increased, from 72 hours to 325 hours, by increasing the thickness of the mats from 100 to 140 μm to 680 to 760 μm, respectively. A possible explanation lies in the fact that increasing the thickness of nanofiber mats makes the diffusion passage more compact for the drug to be released out of the matrix, which reduces the amount of drug release.
Figure 8 Effect of thickness on the release behavior from medicated electrospun nanofiber mats of PVA:PVAc (50:50) containing 10% w/w CipHCl vs time (n = 3).
Abbreviations: CipHCl, ciprofloxacin HCl; PVA, polyvinyl alcohol; PVA/PVAc, polyvinyl acetate.
Based on physical evaluations by touch after submersion, PVAc nanofiber mats were too hard to be applied as a wound dressing whereas PVA nanofiber mats and samples of PVA:PVAc (50:50) blend nanofiber mats showed very good flexibility, and could be bent to cover a wound surface. However, for antibiotic delivery systems, a high initial release is necessary to eliminate bacteria before they begin to proliferate.Citation47 Also, a sustained release of antibiotic for about 1 week is needed for infected wounds to prevent further proliferation of bacteria.Citation5 Blend nanofiber mats of PVA:PVAc (50:50) loaded with CipHCl with a thickness of more than 300 to 360 μm showed a relatively quick initial release as well as a sustained release for at least 10 days; moreover, these mats were able to swell substantially and were flexible and were able to conform to the wound, both of which are requirements for wound dressings.
Analysis of drug release kinetics
In order to further assess the mechanism of drug release, the cumulative release profiles were analyzed by Peppas equation,Citation20 zero order, first order, Higuchi, and Hixson–Crowell models.Citation40 The results of regression analysis of data by the Peppas equation and the values of exponent (n) in that equation are shown in .
Table 1 Diffusion constant (n) and regression coefficient of ciprofloxacin HCl release from PVA, PVAc, and blend nanofiber mats calculated by Peppas equation (Mt/M∞ = ktn) (n = 3)
The results indicate the predominance of Fickian diffusion mechanism of release for all formulations, which means that the amount of drug loaded, the type of polymer and the thickness of nanofiber mats did not influence the drug release mechanism.
Generally, drug release from a polymeric matrix is described by a Fickian mechanism when drug diffusion is the main factor in drug release. Diffusion of the drug from a polymeric matrix can occur through two mechanisms: drug diffusion out of the matrix and/or drug diffusion because of matrix degradation.Citation48
The regression coefficient analysis of release data for different kinetic models can be seen in . This table shows that in PVAc nanofibers, the high level of initial release (up to 72 hours) is probably related to the diffusion of the drug located near the nanofibers surfaces, which can be fitted to a Higuchi model. Afterwards, from 72 to 1920 hours, drug release from PVAc nanofibers can be fitted to a Hixson–Crowell model, ie, after 72 hours, the drug was released because of the erosion of the PVAc nanofiber mats. In fact the release behavior can be described by bimodel behavior. Therefore, the erosion mechanism implies diffusion of the drug into the medium. Degradation does not mean complete degradation but involves degradation of nanofibers layer by layer over time, which implies erosion.
Table 2 Regression coefficients of different mathematical models fitted to the release of ciprofloxacin HCl from different nanofibers with the thickness of 300 to 360 μm (n = 3)
For , Higuchi is the best model to fit the drug release mechanism from PVA and the blend PVA:PVAc (50:50) nanofiber mats, because drug diffusion out of these nanofiber mats plays the main role in drug release.
Conclusion
Continuous uniform nanofibers of PVA, PVAc, and a blend of these polymers loaded with CipHCl were successfully electrospun. Addition of the drug reduced the size and narrowed the distribution of electrospun nanofiber diameters, which could be attributed to the decrease in solution viscosity. The amount of drug loaded in nanofibers affected the release behavior and the initial drug release from PVA and PVAc nanofibers. Regardless of the initial rapid release, rate of drug release from PVAc electrospun nanofibrous mats was very slow: about 50% of the drug was released during 80 days in a linear fashion, whereas the PVA nanofiber mats released the drug within 3 days. Using the blend nanofiber mats of PVA:PVAc (50:50) greatly affected drug release behavior; it reduced the amount of the drug released at earlier stages, and sustained the drug release profile for longer times compared with PVA nanofiber mats. However, blending PVAc with PVA solution made blend nanofiber mats more flexible and more comfortable for use as a wound dressing because of a very significant increase in the degree of swelling comparison with PVAc. Increasing the thickness of the blend nanofiber mats controlled both the initial release and the rate of drug release, and increased the degree of swelling, making the mats suitable for healing deep wounds. All formulations showed Fickian drug release kinetics.
Acknowledgements
The authors express their sincere gratitude to the Isfahan University of Technology for the financial support. This project was also supported by the Isfahan Pharmaceutical Sciences Research Center (Research Grant No: 285360).
Disclosure
The authors declare no conflicts of interest in this work.
References
- PalKBanthiaAKMajumdarDKPolyvinyl alcohol-glycine composite membranes: preparation, characterization, drug release and cytocompatibility studiesBiomed Mater20061495518460756
- YuHXuHChenXHaoJJingXMedicated wound dressing on poly (vinyl alcohol)/poly(N-vinyl pyrrolidone)/chitosan hydrogelsJ Appl Polym Sci200610124532463
- TsouTLTangSTHuangYCWuJRYoungJJWangHJPoly (2-hydroxyethyl methacrylate) wound dressing containing ciprofloxacin and its drug release studiesJ Mater Sci Mater Med2005169510015744596
- SuzukiKTaniharaMNishimuraYSuzukiKKakimaruYShimizuYA new drug delivery system with controlled release of antibiotic only in the presence of infectionJ Biomed Mater Res1998421121169740013
- BoatengJSMatthewsKStevensHNEEcclestonGMWound healing dressings and drug delivery systems: a reviewJ Pharm Sci2008972892292317963217
- KangYOYoonISLeeSoChitosan-coated poly(vinyl alcohol) nanofibers for wound dressingsJ Biomed Mater Res Part B201092568576
- SreenivasanKOn the restriction of the release of water-soluble component from polyvinyl alcohol film by blending β-cyclodexterinJ Appl Polym Sci19976518291831
- ÖztürkEAgalarCKeçeciKDenkbasEBPreparation and characterization of ciprofloxacin-loaded alginate/chitosan sponge as wound dressing materialJ Appl Polym Sci200610116021609
- LokeWKLauSKYongLLKhorESumCKWound dressing with sustained anti-microbial capabilityJ Biomed Mater Res20005381710634947
- JunJYuanDYShaoWHShaoZFZhongWYPreparation and characterization of antibacterial silver-containing nanofibers for wound dressing applicationsJ US-China Med Sci200745254
- ZhangYLimCTRamakrishnaSHuangZMRecent development of polymer nanofibers for biomedical and biotechnological applicationsJ Mater Sci Mater Med20051693394616167102
- KhilMSChaDIKimHYKimISBhattaraiNElectrospun nanofibrous polyurethane membrane as wound dressingJ Biomed Mater Res200567675679
- ThompsonCJChaseGGYarinALRenekerDHEffects of parameters on nanofiber diameter determined from electrospinning modelPolymer20074869136922
- HuangZMHeCLYangAEncapsulating drugs in biodegradable ultrafine fibers through co-axial electrospinningJ Biomed Mater Res200677169179
- VerreckGChunIRosenblattJIncorporation of drugs in an amorphous state into electrospun nanofibers composed of a water-insoluble, nonbiodegradable polymerJ Control Rel200392349360
- ZamaniMMorshedMVarshosazJJannesariMControlled release of metronidazole benzoate from poly ɛ-caprolactone electrospun nanofibers for periodontal diseasesEur J Pharm Biopharm20107517918520144711
- XuXYangLXuXUltrafine medicated fibers electrospun from W/O emulsionsJ Control Rel20051083342
- KenawyERHayEANewehyMHEWnekGEControlled release of ketoprofen from electrospun poly(vinyl alcohol) nanofibersMater Sci Eng A2007459390396
- ZengJYangLLiangQInfluence of the drug compatibility with polymer solution on the release kinetics of electrospun fiber formulationJ Control Rel20051054351
- TaepaiboonPRungsardthongUSupapholPDrug-loaded electrospun mats of poly(vinyl alcohol) fibers and their release characteristics of four model drugsNanothechnol20061723172329
- KimKLuuYKChangCIncorporation and controlled release of a hydrophilic antibiotic using poly(lactide-co-glycolide)-based electrospun nanofibrous scaffoldsJ Control Rel2004984756
- SanliOAyNIşiklanNRelease characteristics of diclofenac sodium from poly(vinyl alcohol)/sodium alginate and poly(vinyl alcohol)-grafted-poly(acrylamide)/sodium alginate blend beadsEur J Pharm Biopharm20076520421416996255
- SeabraABOliveiraMGPoly(vinyl alcohol) and poly(vinyl pyrrolidone) blended films for local nitric oxide releaseBiomaterials2004253773378215020153
- KimJOParkJKKimJHDevelopment of polyvinyl alcohol–sodium alginate gel-matrix-based wound dressing system containing nitrofurazoneInt J Pharm2008359798618440737
- SinghBPalLDevelopment of sterculia gum based wound dressings for use in drug deliveryEur Polym J20084432223230
- SilvalingamGChattopadhyaySMadrasGEnzymatic degradation of poly(ɛ-caprolactone), poly(vinyl acetate) and their blends by lipasesChem Eng Sci20035829112919
- NovoaGAGHeinämäkiJMirzaSPhysical solid-state properties and dissolution of sustained-release matrices of polyvinyl acetateEur J Pharm Biopharm20055934335015661507
- SungJHHwangMRKimJOGel characterization and in vivo evaluation of minocycline-loaded wound dressing with enhanced wound healing using polyvinyl alcohol and chitosanInt J Pharm201039223224020230884
- DonTMKingCFChiuWYPengCAPreparation and characterization of chitosan-g-poly(vinyl alcohol)/poly(vinyl alcohol) blends used for the evaluation of blood-contacting compatibilityCarbohyd Polym200663331339
- BonakdarSOrangFRafieniaMImaniRComparison of the effect of hydrophilicity on biocompatibility and platelet adhesion of two different kinds of biomaterialsIranian J Pharm Sci200843744
- IkadaYIwataHHoriiFBlood compatibility of hydrophilic polymersJ Biomed Mater Res198169771812659136
- ParkKRNhoYCPreparation and characterization by radiation of hydrogels of PVA and PVP containing aloe veraJ Appl Polym Sci20049116121618
- AndersenFAAmended final safety assessment of polyvinyl acetateJ Am Coll Toxicol199615166176
- SadatoATakiWIkadaYExperimental study and clinical use of poly(vinyl acetate) emulsion as liquid embolisation materialNeuroradiol199436634641
- EvansSGreffRJWrightJICompositions for use in embolizing blood vessels: US Patent 6342202
- DennisCLKleinGJGebhardtUAndersonKMZillmerGCIntravascular medical device: US Patent 5607463
- SuzawaTShirahamaHFujimotoTAdsorption of bovine serum albumin onto homo- and copolymer laticesJ Colloid Interface Sci19821144150
- BeukemaRPBeukemaWPSmitJJEfficacy of multi-electrode duty-cycled radiofrequency ablation for pulmonary vein disconnection in patients with paroxysmal and persistent atrial fibrillationEuropace20101250250720185490
- HoshiTSawaguchiTKonnoTTakaiMIshiharaKPreparation of molecular dispersed polymer blend composed of polyethylene and poly(vinyl acetate) by in situ polymerization of vinyl acetate using supercritical carbon dioxidePolymer20074815731580
- CostaPLoboJMSModeling and comparison of dissolution profilesEur J Pharm Biopharm200113123133
- WangQDongZDuYKennedyFControlled release of ciprofloxacin hydrochloride from chitosan/polyethylene glycol blend filmsJ Carbohyd Polym200769336343
- LandauORothschildAZussmanEProcessing-microstructure-properties correlation of ultrasensitive gas sensors produced by electrospinningChem Mater200921911
- MengZXZhengWLiLZhengYFFabrication, characterization and in vitro drug release behavior of electrospun PLGA/chitosan nanofibrous scaffoldMater Chem Phys2011125606611
- SteeleJNeufeldREncapsulation of crosslinked gelatin nanofibers to better mimic ECM conditions within alginate microparticlesXVIIth International Conference on BioencapsulationPorto, PortugalOctober 1–2, 2010
- BredaSAJimenez-KairuzAFManzoRHOliveraMESolubility behavior and biopharmaceutical classification of novel high-solubility ciprofloxacin and norfloxacin pharmaceutical derivativesInt J Pharm200937110611319150493
- KumbarSGAminabhaviTMSynthesis and characterization of modified chitosan microspheres: effect of the grafting ratio on the controlled release of nifedipine through microspheresJ Appl Polym Sci20038929402949
- ParkSJKimKSInfluence of hydrophobe on the release behavior of vinyl acetate miniemulsion polymerizationColloids Surf B2005455256
- RosenbergRSevenneyWSeigelSDanNAnomalous release of hydrophilic drugs from poly(ɛ-caprolactone) matricesMol Pharm2007494394817960890