Abstract
The objective of our investigation was to design a self-microemulsifying drug-delivery system (SMEDDS) to improve the bioavailability of probucol. SMEDDS was composed of probucol, olive oil, Lauroglycol FCC, Cremophor EL, Tween-80, and PEG-400. Droplet sizes were determined. In vitro release was investigated. Pharmacokinetics and bioavailability of probucol suspension, oil solution, and SMEDDS were evaluated and compared in rats. Plasma drug concentration was determined by high-performance liquid chromatography. After administration of probucol suspension, plasma drug concentration was very low. Relative bioavailability of SMEDDS was dramatically enhanced in an average of 2.15- and 10.22-fold that of oil solution and suspension, respectively. It was concluded that bioavailability of probucol was enhanced greatly by SMEDDS. Improved solubility and lymphatic transport may contribute to the enhancement of bioavailability.
Introduction
Probucol, a chemical compound with a bisphenol structure, was originally synthesized as an antioxidant by Consolidation Coal Company and screened at Dow Chemical Company for its ability to reduce serum cholesterol.Citation1 Probucol has been used widely in clinical practice for the prevention of the progression of atherosclerosis because this agent acts as a potent antioxidant in addition to having a lipid-lowering action.Citation2 In addition, probucol can stop progression of atherosclerotic plaques in carotid arteries.Citation3
However, probucol is slightly absorbed in the gastrointestinal tract due to its poor solubility in water (the logP was reported to be 10.91).Citation4 The oral bioavailability of probucol is very low. From studies in rats, dogs, and monkeys, it is known that probucol accumulates slowly in adipose tissue. Approximately 90% of probucol administered orally is unabsorbed.Citation5
Research has been focused on enhancing the solubility of poorly water-soluble drugs to improve their oral bioavailability. One of the most popular approaches is lipid-based formulation, such as oils, surfactant dispersions, self-emulsifying formulations, emulsions, and liposomes.Citation6
Self-microemulsifying drug-delivery systems are mixtures of drugs, lipids, surfactants, and cosurfactants, which form a fine oil-in-water (O/W) microemulsion with a droplet size of less than 100 nm when exposed to aqueous media under conditions of gentle agitation or digestive motility that would be encountered in the gastrointestinal (GI) tract. SMEDDS has recently emerged as one of the most interesting approaches to improve oral absorption for poorly water-soluble drugs.Citation7,Citation8 The advantages of SMEDDS include ease of production, enhanced solvent capacity, increased stability, and the potential to administer the final product as an oral soft gelatin cap.Citation9 The commercially available formulation cyclosporine (NeoralTM) is a microemulsion preconcentrate with improved oral bioavailability and reduced inter- and intra-subject variability compared to the original crude emulsion product, SandimmuneTM.Citation10 Similar lipid-based formulations of the human immunodeficiency virus (HIV) protease inhibitors, saquinavir, ritonavir, and amprenavir, have also reached the market.Citation6 To date, probucol formulated in SMEDDS has not yet been evaluated.
The objectives of our present study were to prepare, and characterize a SMEDDS formulation of probucol, and assess its oral absorption in rats.
Materials and methods
Materials
Probucol was purchased from Qihe Jinzhun Technology Co, Ltd, (Shandong, China). Olive oil, PEG-400, Tween-80, and n-hexane were purchased from Sinopharm Chemical Reagent Co, Ltd, (Shanghai, China). Lauroglycol FCC was kindly donated by Gattefosse S.A (Saint-Priest, France). Cremophor EL was kindly provided by BASF Corp. (Washington, NJ). Retinyl acetate was purchased from Sigma Chemical Co. (St Louis, MO). Acetonitrile (high-pressure liquid chromatography [HPLC] grade) was supplied by Burdick and Jackson Corp. (Muskegon, MI). Deionized water was prepared by an Aquapro purification system (Chongqing Yiyang Co Ltd, Chongqin, China). All other chemicals were analytical grade.
Animals
Male Sprague-Dawley (SD) rats (230 ± 20 g) and imprinting control region (ICR) mice (20 ± 2 g), supplied by Department of Experimental Animals, Fudan University (Shanghai, China), were acclimated at 25°C and 55% of humidity under natural light/dark conditions for 1 week before the experiment. All animal experiments were carried out in accordance with guidelines evaluated and approved by the ethics committee of Fudan University.
Preparation of SMEDDS formulations
Probucol SMEDDS was composed of olive oil (13%, w/w), Lauroglycol FCC (27%, w/w), Cremophor EL (20%, w/w), Tween-80 (20%, w/w), and PEG-400 (20%, w/w). Preparation of probucol SMEDDS was simply by mixing the components. Since probucol was difficult to dissolve, it was better to dissolve probucol first in Lauroglycol FCC. Olive oil, Cremophor EL, and PEG-400 were then added slowly with gentle stirring until a homogeneous mixture formed. The mixture was sealed in a glass vial and stored under room temperature.
Droplet size
One gram of SMDROPLET SIZEEDDS was diluted in 250 mL of deionized water, phosphate-buffered saline (PBS; pH 7.0), and 0.1 M hydrochloride solution, respectively. The solution was inverted and shaken gently to mix thoroughly. The droplet size of microemulsion was determined by NicompTM 380 ZLS laser diffraction sizer (PSS Nicomp, Santa Barbara, CA). The measurement conditions were: He–Ne laser; angle, 90°; temperature, 23°C; reflection index, 1.333; wavelength, 632.8 nm; or with adjustment if needed.
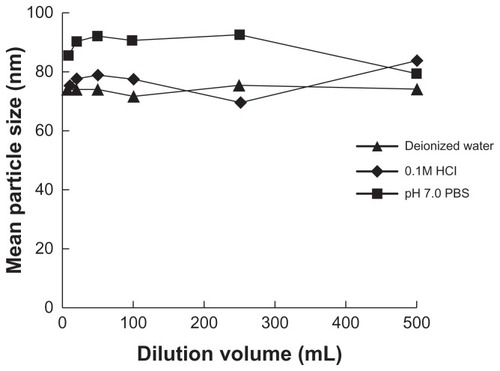
In order to determine the effects of different media and dilution volume on the droplet size of microemulsion, 1 g of SMEDDS was added into 10 mL, 20 mL, 50 mL, 100 mL, 250 mL, and 500 mL of deionized water, PBS (pH 7.0), and 0.1 M hydrochloride solution, respectively, and the resulting solutions were slightly shaken. The droplet sizes of these solutions were analyzed.
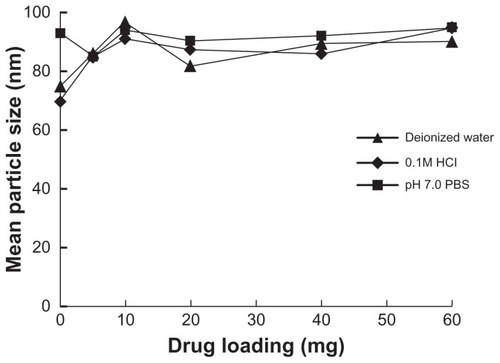
The effect of drug loading on droplet size in different media was studied. Five milligrams, 10 mg, 20 mg, 40 mg, and 60 mg of probucol were added to 1 g of blank SMEDDS. These probucol-loading SMEDDS were diluted to 250 mL deionized water, PBS (pH 7.0), and 0.1 M hydrochloride solution, respectively, and the droplet sizes of resulted solutions were determined.
Morphology
The morphology of SMEDDS was observed using a transmission electron microscope (TEM; CM120; Philips Co, Amsterdam, The Netherlands). SMEDDS was diluted with deionized water at 1:50 and mixed by slight shaking. One drop of diluted samples was deposited on a film-coated copper grid and then stained with one drop of 2% aqueous solution of phosphotungstic acid (PTA), and allowed to dry before observation under the electron microscope.
Dissolution studies
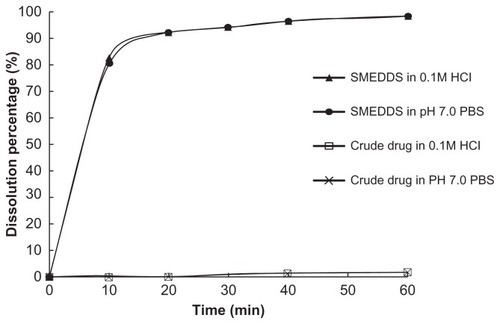
In order to compare the dissolution behaviors of probucol- loaded SMEDDS and crude probucol, PBS (pH 7.0) and 0.1 M hydrochloride solution were chosen as the dissolution media. Dissolution studies were performed according to the method described previously.Citation11
SMEDDS containing 60 mg of probucol or 60 mg of crude probucol was filled in hard gelatin capsules and introduced into 200 mL of a dissolution medium maintained at 37°C. The revolution speed of the paddle was kept constant at 100 rpm. Aliquot of 5 mL was withdrawn at 0, 10, 20, 30, 40 and 60 minutes, and filtered through 0.45 μm membrane filters. The concentration of probucol was determined by HPLC. The removed volume was replaced each time with 5 mL of fresh medium.
Bioavailability study
Bioavailability of probucol SMEDDS was compared with probucol suspension and olive oil solution. Rats were fasted overnight before experiment with free access to water. Probucol suspension (probucol dispersed in 1% [w/v] carboxymethyl cellulose–Na solution), probucol dissolved in olive oil and probucol-loaded SMEDDS (60 mg/kg of body weight) were given to the rats by intragastric administration, respectively. About 0.8 mL of blood sample was collected into heparinized tubes at 0.5, 1, 2, 4, 5, 6, 8, 12, 24, 48, 96, and 120 hours. Blood samples were centrifuged at 10,000 rpm for 10 minutes and plasma samples were collected and stored at −18°C.
Determination of probucol in rat plasma by RP-HPLC
All samples were analyzed by a modified HPLC/UV method. The HPLC system consisted of a LC-10AT pump, a SPD-10A UV detector (Shimadzu, Kyoto, Japan), and a data-processing system (N2000, YingPu Co Ltd, Hangzhou, China). Probucol was separated by a C18 column (5 μm, 4.6 mm × 150 mm; Phenomenex, Torrance, CA) guarded with a refillable pre-column (C18, 2.0 mm × 20 mm; Phenomenex, Torrance, CA) at room temperature. Probucol was detected at 242 nm. The mobile phase was a mixture of acetonitrile: H2O (96:4, v/v) and was delivered at a flow rate of 1 mL · minute−1.Citation12
Liquid–liquid plasma extraction procedure was used as follows: in a 5 mL tube, 300 μL plasma was added, followed by 10 μL of internal standard (50 μg · mL−1 of retinyl acetate) solution, and vortex mixed for 30 seconds. Then 150 μL of ethanol and 200 μL of methanol were added to precipitate the protein. Then 1 mL of hexane was added and vortexed for 3 minutes. After centrifuging at 12,000 rpm for 10 minutes, the upper layer was transferred to another tube and evaporated under a light stream of nitrogen at 40°C. The residue was dissolved by 100 μL of mobile phase and 20 μL was injected for HPLC analysis. Quantification was based on area ratio of probucol and the internal standard.Citation13
The retention times for internal standard and probucol were about 6.7 minutes and 11.0 minutes, respectively. The linearity was obtained in the range from 0.05 to 30 μg · mL−1. The coefficient of variation for intra- and interday assays was less than 5%. The average recovery of probucol from plasma was between 98.6% and 101.4%.
Data analysis
The pharmacokinetics’ parameters were calculated by DAS 2.0 (issued by the State Food and Drug Administration of China for Pharmokinetic Study). Student’s t-tests were performed to evaluate the significant differences. Values are reported as Mean ± standard deviation, and the data were considered statistically significant at P < 0.05.
Results and discussion
SMEDDS formulation
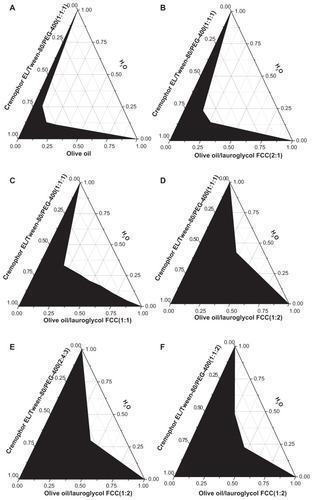
In our pseudoternary phase diagram study (), systems consisting of olive oil and Lauroglycol FCC as oil phase, Cremophor EL and Tween-80 as emulsifiers, and PEG-400 as coemulsifier were titrated with water, and self-emulsifying formulations were selected from regions undergoing infinite dilution. In the diagrams (), with the decrease of olive oil/Lauroglycol FCC ratio, the area of self-microemulsifying region increased. When the Cremophor EL/Tween-80 ratio increased, the area of self-microemulsifying region increased (). Diagrams with higher emulsifier/co-emulsifier ratio had larger self-microemulsifying area (). The optimal formulation of probucol SMEDDS was selected to investigate the self-microemulsifying ability, solubilization ability, and reduced use of emulsifiers, was as follows: olive oil (13%, w/w), Lauroglycol FCC (27%, w/w), Cremophor EL (20%, w/w), Tween-80 (20%, w/w), and PEG-400 (20%, w/w). Sixty milligrams of probucol was dissolved in 1 g of mixture.
Droplet size
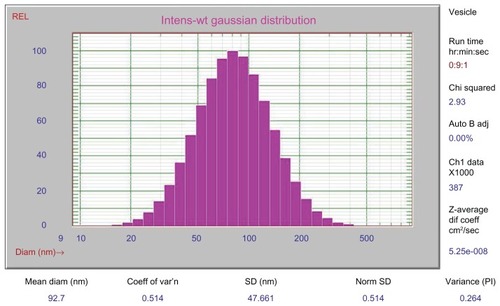
Droplet size after microemulsification was the most important property of SMEDDS. It may affect the release and absorption of drug in GI tract.Citation14,Citation15 The typical size distribution is shown in . It seemed that dilution volume within the investigated range and different dilution media had little effect on droplet size () and self-microemulsifying behavior (P > 0.05). The mean droplet size did not change significantly with increased drug loading (; P > 0.05).
Figure 2 Size distribution of probucol self-microemulsifying drug-delivery system determined by laser diffraction sizer after dilution to deionized water.
Abbreviations: REL, relative; Intens-wt, intensive weight; Auto B adj, automatic B adjust; Ch 1 data, channel 1 data; Z-average diff coeff, Z-average difference coefficient; PI, polydispersity index; Coeff of var’n, coefficient of variance; SD, standard deviation.
Morphology
Morphology of the microemulsions formed from SMEDDS was viewed under a TEM, the microemulsion vesicles appeared as perfect round shapes without aggregation ().
Dissolution studies
Our previous study showed probucol was practically insoluble in water at acidic or neutral pH. As shown in , crude probucol showed negligible release even after 60 minutes in both PBS (pH 7.0) and 0.1 M hydrochloride solution. Whereas, SMEDDS showed rapid dissolution in both solutions, at 10 minute about 80% of probucol from SMEDDS was dissolved in medium, and more than 90% was released after 20 minutes. SMEDDS could form clear and transparent solution in the condition of dissolution quickly.
Bioavailability study
The pharmacokinetic parameters of probucol SMEDDS, oil solution, and suspension were compared in rats. Mean plasma probucol concentration was plotted as a function of time ().
Figure 7 Plasma probucol concentration–time plot after a single oral dose of probucol in three formulations.
Note: Each value represents the mean ± SD (n = 6).
Abbreviations: SD, standard deviation; SMEDDS, self-microemulsifying drug-delivery system.
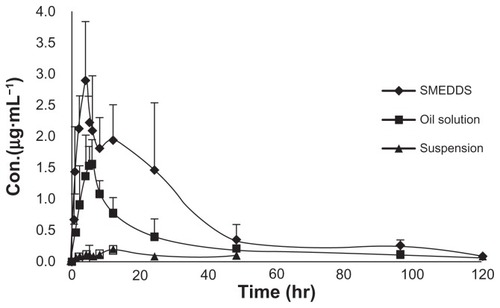
The pharmacokinetic parameters of SMEDDS, oil solution, and suspension are shown in . As can be seen, the maximum concentration (Cmax) of probucol SMEDDS was 3.36 ± 0.84 μg · mL−1, compared with those of oil solution and suspension which were 1.80 ± 0.43 μg · mL−1 and 0.16 ± 0.07 μg · mL−1, respectively. Statistically, the differences in Cmax of probucol SMEDDS were extremely significant (P < 0.01) when compared with Cmax of oil solution (P < 0.01) and suspension (P < 0.001). It was also observed that the area under the curve (AUC0–120 hours) of SMEDDS was 73.59 ± 31.21 μg · mL−1 · hour, and thus the difference was highly significant compared with that of the oil solution (34.16 ± 10.65 μg · mL−1 · hour) (P < 0.02) and suspension (7.20 ± 1.63 μg · mL−1 · hour) (P < 0.001). The bioavailability of SMEDDS was 2.15- and 10.22-fold that of oil solution and suspension, respectively.
Table 1 Pharmacokinetic parameters of probucol in three formulations
Lipid-based formulations such as oil solution and self-microemulsifying drug-delivery systems offer the potential for enhancing the absorption and hence the oral bioavailability of lipophilic drugs. The primary mechanisms are presenting the drug in solubilized form in vivo, delaying gastric emptying, increasing mucosal permeability, and increasing incorporation into lipoproteins, then secreted into the lymphatics, which circumvents the liver, thus reducing the hepatic first-pass metabolism.Citation14,Citation16–Citation18
It was reported that probucol had high lymphatic bioavailability when coadministrated with lipids.Citation19,Citation20 Chylomicrons (CMs) are important in drug delivery since many lipophilic drugs are carried by CMs and transported in lymph.Citation21 Palin and Wilson reported that digestion of oil within the small intestine resulted in the liberation of long-chain fatty acids, which effectively promoted chylomicron formation and consequently the lymphatic transport of DDT or probucol.Citation19
Enhancement of bioavailability of probucol SMEDDS compared with oil solution may be due to effects of a large quantity of surfactants and cosurfactant, including improved mucosal permeability, smaller lipid droplets and greater surface area. In addition, it was reported that Tween-80 can promote CMs secretion and counteracted the inhibitory effects of other surfactants.Citation22
Double peaks of Cmax were observed after administration of SMEDDS. This may be caused by protection effects of surfactants for lipid from digestion, high lymphatic bioavailability of probucol, and the slow flow rate of lymph. It has been proven that the presence of digestion products such as monoglycerides and fatty acids, incorporated in bile salt micelles, increases the solubility of poorly water-soluble drugs and therefore can enhance their absorption. With digestion proceeding and the oil phase breaking down, the surfactant and fatty acids will spread out into the release medium, which causes the drug to release.Citation23,Citation24 Surfactants located at the oil droplet surface can interfere with the attachment of the lipase complex to the oil–water interface and inhibit lipolysis of lipids.Citation25–Citation27
Conclusion
A SMEDDS formulation for probucol was developed, and the optimal formulation was as follows: olive oil (13%, w/w), Lauroglycol FCC (27%, w/w), Cremophor EL (20%, w/w), Tween-80 (20%, w/w), and PEG-400 (20%, w/w). When diluted with water, probucol-loaded SMEDDS could spontaneously form small particles with average droplet size of about 80 nm. Different media, dilution volume and drug loading seemed to have no effect on droplet size and self-microemulsifying behavior. Dissolution percentages of probucol in SMEDDS in 0.1 M HCl or in pH 7.0 PBS were significantly higher than that of crude probucol. Relative bioavailability of probucol SMEDDS was dramatically enhanced, approximately 2.15- and 10.22-fold that of olive oil solution and suspension, respectively. Our studies illustrated the potential use of SMEDDS for the delivery of hydrophobic compounds, such as probucol.
Acknowledgments
The work was sponsored by Shanghai Rising-Star Program (10QA1400800), the Scientific Research Foundation for the Returned Overseas Chinese Scholar. This work was also supported by F Hoffmann-La Roche Ltd (Basel, Switzerland).
Disclosure
No conflicts of interest were reported by the authors of this paper.
Reference
- HeelRCBrogdenRNSpeightTMAveryGSProbucol: a review of its pharmacological properties and therapeutic use in patients with hypercholesterolemiaDrugs197815409428350557
- LiGPYinLLiuTRole of probucol in preventing contrast-induced acute kidney injury after coronary interventional procedureAm J Cardiol200910351251419195512
- SawayamaYShimizuCMaedaNEffects of probucol and pravastatin on common carotid atherosclerosis in patients with asymptomatic hypercholesterolemia. Fukuoka Atherosclerosis Trial (FAST)J Am Coll Cardiol20023961061611849859
- GershkovichPHoffmanAUptake of lipophilic drugs by plasma derived isolated chylomicrons: Linear correlation with intestinal lymphatic bioavailabilityEur J Pharm Sci20052639440416140514
- Physicians’ desk reference: Lorelco Tablets19954914081410
- PoutonCWPorterJHFormulation of lipid-based delivery systems for oral administration: Materials, methods and strategiesAdv Drug Deliv Rev20086062563718068260
- SagarDMShobhonaSSulabhaPVandanaBPDevelopment of SMEDDS using natural lipophile: application to β-artemether deliveryInt J Pharm200836217918318652886
- MetteGAnetteMJeanetLNGittePPBioavailability of seocalcitol II: Development and characterisation of self-microemulsifying drug delivery systems (SMEDDS) for oral administration containing medium and long chain triglyceridesEur J Pharm Sci20062823324216650738
- PoutonCWLipid formulations for oral administration of drugs: non-emulsifying, self-emulsifying and ‘self-microemulsifying’ drug delivery systemsEur J Pharm Sci2000119398
- HoltDWJohnstonAWhich cyclosporin formulationLancet199634811758888200
- PatelARVaviaPRPreparation and in vivo evaluation of SMEDDS (self-microemulsifying drug delivery system) containing fenofibrateAAPS J20079344352
- QiXDCaoDYPreparation and in vivo evaluation of probucol solid dispersionChinese Journal of Pharmaceuticals200536483486
- HenarOJoséLCPatriciaCLiquid chromatographic method for the simultaneous determination of different lipid-soluble antioxidants in human plasma and low-density lipoproteinsJ Chromatogr B Analyt Technol Biomed Life Sci2004803249255
- GershanikTBenitaSSelf-dispersing lipid formulations for improving oral absorption of lipophilic drugsEur J Pharm Biopharm20005017918810840200
- NielsenFSPetersenKBMullertzABioavailability of probucol from lipid and surfactant based formulations in minipigs: Influence of droplet size and dietary stateEur J Pharm Biopharm20086955356218294829
- PorterCJTrevaskisNLCharmanWNLipids and lipid-based formulations: Optimizing the oral delivery of lipophilic drugsNat Rev Drug Discov2007623124817330072
- PoutonCWFormulation of self-emulsifying drug delivery systemsAdv Drug Deliv Rev1997254758
- HumberstoneAJCharmanWNLipid based vehicles for the oral delivery of poorly water soluble drugsAdv Drug Deliv Rev199725103128
- PalinKJWilsonCJThe effect of different oils on the absorption of probucol in the ratJ Pharm Pharmacol1984366416436149296
- YamamotoKFukudaNShiroiSEffects of dietary fat levels on the absorption and tissue accumulation of probucol in the ratArzneimittelforschung199444105910627986244
- BrianKNordskogaCTPhanaDFAn examination of the factors affecting intestinal lymphatic transport of dietary lipidsAdv Drug Deliv Rev200150214411489332
- FergalSStimulation of triglyceride-rich lipoprotein secretion by polysorbate 80: in vitro and in vivo correlation using Caco-2 cells and a cannulated rat intestinal lymphatic modelPharm Res2004212320232615648264
- HernellOStaggersJECareyMCPhysical–chemical behavior of dietary and biliary lipids during intestinal digestion and absorption. 2. Phase behaviour and aggregation states of luminal lipids during duodenal fat digestion in healthy adult human beingsBiochemistry199029204120562328238
- StaggersJEHernellOStaffordRJCareyMCPhysical-chemical behaviour of dietary and biliary lipids during intestinal digestion and absorption. 1. Phase behaviour and aggregation states of model lipid systems patterned after aqueous duodenal contents of healthy adult human beingsBiochemistry199029202820402328237
- SkagerlindPJanssonMHultKSurfactant interference on lipase catalysed reactions in microemulsionsJ Chem Technol Biotechnol1992542772821382460
- CanioniPJulienRRathelotJSardaLInhibition of sheep pancreatic lipase activity against emulsified tributyrin by non-ionic detergentsBiochimie197658751753953065
- ReisPHolmbergKWatzkeHLeserMEMillerRLipases at interfaces: A reviewAdv Coll Interface Sci2009147148