Luo L, Du T, Zhang J, et al. Int J Nanomedicine. 2016;11:501—513.
The authors have advised due to an error at the time of figure assembly, on page 510 is incorrect. The correct is shown below.
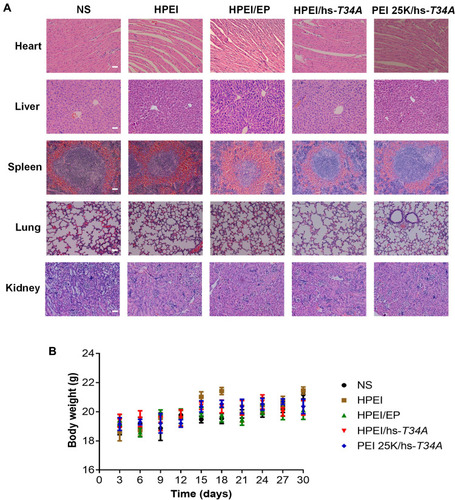
Figure 11 Safety and toxicity evaluation of HPEI/hs-T34A formulation.
Notes: (A) The heart, liver, spleen, lung, and kidney in NS, HPEI alone (50 µg), HPEI/EP, HPEI/hs-T34A, or PEI 25K/hs-T34A group were collected and conducted with HE staining, respectively. Scale bar, 50 µm. (B). Body weight changes in NS, HPEI alone (50 µg), HPEI/EP, HPEI/hs-T34A, or PEI 25K/hs-T34A group, respectively.
Abbreviations: HPEI, heparin–polyethyleneimine; PEI 25K, polyethyleneimine (molecular weight 25,000); HPEI/EP, heparin–polyethyleneimine/empty vector plasmid; NS, normal saline; hs, human survivin.
The authors apologize for this error and advise it does not affect the results of the paper.
