Abstract
Background
There is an increasing commercial demand for nanoparticles due to their wide applicability in various markets, including medicine, catalysis, electronics, chemistry, and energy. In this report, a simple and ecofriendly chemical reaction for the synthesis of gold and silver nanoparticles from Trianthema decandra (Aizoaceae) has been developed.
Methods and results
On treatment of aqueous solutions containing chloroauric acid or silver nitrate with root extract of T. decandra, stable gold or silver nanoparticles were rapidly formed. The kinetics of reduction of gold and silver ions during the reaction was analyzed by ultraviolet-visible spectroscopy. Field emission-scanning electron microscopy showed formation of gold nanoparticles in various shapes, including spherical, cubical, triangular, and hexagonal, while silver nanoparticles were spherical. The size of the gold nanoparticles was 33–65 nm and that of the silver nanoparticles was 36–74 nm. Energy dispersive x-ray and Fourier transform infrared spectroscopy confirmed the presence of metallic gold and metallic silver in the respective nanoparticles. The antimicrobial properties of the synthesized nanoparticles were analyzed using the Kirby-Bauer method. The results show varied susceptibility of microorganisms to the gold and silver nanoparticles.
Conclusion
It is believed that phytochemicals present in T. decandra extract reduce the silver and gold ions into metallic nanoparticles. This strategy reduces the cost of production and the environmental impact. The silver and gold nanoparticles formed showed strong activity against all microorganisms tested.
Introduction
Nanoparticles are a special group of materials with unique features and extensive applications in diverse fields.Citation1 Metal nanoparticles, such as those containing gold and silver have recognized importance in chemistry, physics, and biology because of their unique optical, electrical, and photothermal properties.Citation2 The ease of synthesizing gold and silver nanoparticles and their affinity for binding many biological molecules makes them attractive candidates for study. Various physical and chemical methods have been reported over the last two decades for the synthesis of gold and silver nanoparticles, of which chemical approaches, such as chemical reduction, electrochemical techniques, and photochemical reduction, are most widely used. The green method of nanoparticle synthesis employing plant extracts is a simple and viable alternative to chemical procedures and physical methods.Citation3 Chemical and physical methods are harmful because the chemicals used are toxic, flammable, and are not disposed easily of in the environment.Citation4 In recent years, biosynthesis of nanoparticles has received considerable attention due to the growing need to develop clean and nontoxic chemicals, environmentally friendly solvents, and renewable materials.Citation5 Utilization of biological systems such as plant extracts for the synthesis of nanoparticles has advantages over chemical and physical methods, because these systems are cost-effective, environment friendly, single-step in nature, and easily scaled up for large-scale synthesis.Citation6
Studies have shown that the size, morphology, stability, and physicochemical properties of metal nanoparticles are strongly influenced by the experimental conditions,Citation7–Citation9 and play pivotal roles in controlling the physical, chemical, optical, and electronic properties of these nanoscopic materials. This is particularly important for noble metals, such as gold and silver, which have strong surface plasmon resonance oscillations.Citation10
Several researchers have reported the use of natural products, inactivated plant biomass, and as living plants such as Aloe vera,Citation11 Carica papaya,Citation12 Magnolia kobus, Diospyros kaki,Citation13 Medicago sativa,Citation14,Citation15 Azadirachta indica,Citation16 Pelargonium graveolens,Citation17 Chilopsis linearis,Citation18 and Tamarindus indicaCitation19 for the production of silver and gold nanoparticles (). Large amounts of nanoparticles can be easily synthesized from plants, and the majority of these are nontoxic. They have been used in pharmaceutical drug-based industries for treating lymphocytic leukemia, detecting DNA, inhibiting bacteria and fungi, and preventing burns and open wound infections.Citation20,Citation21
Table 1 Nanoparticles synthesized from different plants and their size range
Trianthema decandra, a prostrate, glabrous, succulent annual belonging to the Aizoaceae family, is found as a weed throughout most of India.Citation22 The plant has various medicinal properties and has been reported to have antioxidant and antimicrobial properties.Citation23 In the present study, we describe the biosynthesis of gold and silver nanoparticles from aqueous root extract of T. decandra for the first time. Chloroauric acid and silver nitrate were reduced to metallic gold and metallic silver respectively, on reaction with root extract of T. decandra, resulting in synthesis of gold and silver nanoparticles. The gold and silver nanoparticles formed were characterized using ultraviolet-visible, scanning electron microscopy, energy dispersive x-ray, and Fourier transform infrared spectroscopy. Antimicrobial assays using gold and silver nanoparticles demonstrated activity against all the microorganisms tested.
Materials and methods
Plant materials
T. decandra L was collected from Salem district, Tamil Nadu, India, during June 2011. The plant was taxonomically identified and authenticated by the Botanical Survey of India, Coimbatore, and voucher specimen BSI/SRC/5/23/10-11/Tech 975 was deposited at the Plant Biotechnology Laboratory, Sri Ramaswamy Memorial University, for future reference.
Chemicals
Gold (III) chloride trihydrate (HAuCl4) and silver nitrate (AgNO3) were purchased from Sigma-Aldrich, St Louis, MO. Double-distilled deionized water was used.
Preparation of plant extracts
Roots of T. decandra were washed with distilled water to remove dirt and soil. The shade-dried materials were powdered and passed through a 40-mesh sieve. Next, 20 g of coarsely powdered material was extracted with 100 mL of double-distilled water. The mixture was filtered through Whatman No 1 filter paper. The extract was stored at 4°C until further use.
Synthesis of gold and silver nanoparticles
For the synthesis of gold and silver nanoparticles, 10 mL of 1 mM aqueous HAuCl4 and AgNO3 solutions were separately added to 5 mL, 10 mL, and 15 mL of aqueous root extract and incubated in the dark. The resulting colloidal solutions of gold and silver were then analyzed using ultraviolet-visible spectroscopy.
Ultraviolet-visible spectroscopy
The colloidal solutions of gold and silver were diluted by adding 2.9 mL of deionized water to 0.1 mL of sample. Bioreduction of HAuCl4 and AgNO3 was recorded as a function of time using water as a reference at 3-hour intervals from zero hour up to 24 hours using an ultraviolet-visible spectrophotometer (Perkin-Elmer, Boston, MA) in a scanning range of 200 nm to 800 nm.
Purification of gold and silver nanoparticles
To remove excess gold and silver ions, the gold and silver colloids were centrifuged at 10,000 rpm for 15 minutes and washed at least three times with deionized water. A dried powder of the gold and silver nanoparticles was obtained by freeze-drying.
Fourier transform infrared spectroscopy
The air-dried powder form of the synthesized gold and silver nanoparticles was ground with KBr pellets and subjected to Fourier transform infrared spectroscopy.
FE-SEM and energy dispersive x-ray spectroscopy
The gold and silver nanoparticles formed were characterized using high resolution field emission scanning electron microscopy (FE-SEM). The samples were prepared by simple drop coating of the suspension of gold and silver nanoparticles separately on a carbon-coated copper grid, by simply dropping a very small amount of the sample on the grid, with excess solution removed using blotting paper. The film on the scanning electron microscopy grid was then allowed to dry under a mercury lamp for 5 minutes. Energy dispersive x-ray spectroscopy was then performed using the Hitachi S-3400 N FE-SEM instrument equipped with a Thermo energy dispersive x-ray spectroscopy attachment.
Antimicrobial activity
The antimicrobial activity of the gold and silver nanoparticles formed was assayed using the standard Kirby-Bauer disc diffusion method.Citation24 Bacterial suspensions of Staphylococcus aureus (MTCC 29213), Streptococcus faecalis (MTCC 0459), Enterococcus faecalis (MTCC 2729), Escherichia coli (MTCC 443), Pseudomonas aeruginosa (MTCC 1035), Proteus vulgaris (MTCC 1771), Bacillus subtilis (MTCC 121), Yersinia enterocolitica (MTCC 840), and the fungus, Candida albicans (MTCC 183), grown overnight were swabbed on separate nutrient agar plates using L-rods. Whatman filter paper (No 1) discs 6 mm in diameter were separately impregnated with 20 μL of a 10 mg/mL solution of gold and silver nanoparticles in dimethylsulfoxide. The discs were evaporated at 37°C for 24 hours and then impregnated on plates. Chloramphenicol and nystatin were used as positive controls for bacteria and the fungus respectively, while dimethylsulfoxide served as a negative control. Triplicates were maintained and the plates were incubated at 37°C for 24–48 hours. Diameters of the zones of inhibition were measured using a meter ruler, and the mean value for each organism was recorded and expressed in millimeters.
Statistical analysis
The data were analyzed by one-way analysis of variance followed by the Duncan’s multiple range test.
Results and discussion
Synthesis and characterization of gold and silver nanoparticles
Nanoparticles of noble metals are characterized by the presence of bright colors produced by oscillations of the surface electron cloud of these particles. The formation of gold and silver nanoparticles was observed by monitoring the change in color, ie, a light yellow solution turning to ruby pink when HAuCl4 was used and a pale yellow solution turning to dark grey when AgNO3 was used. The collective oscillations of electrons in a nanoparticle upon interaction with light of suitable energy causes the nanoparticles to attain the color specific to a particular metal. Particle size, shape, and aggregation depend strongly on the frequency of oscillations.Citation25
The reduction kinetics of aqueous chloroaurate ions and silver ions leading to synthesis of gold and silver nanoparticles was compared using different amounts, ie, 5 mL, 10 mL, and 15 mL of root extract of T. decandra ( and ). Reduction of chloroaurate ions was more rapid than that of silver ions. Gold nanoparticles were synthesized within 3 hours of reaction with the root extract and the components thereof, while more than a 6-hour reaction period was required for synthesis of the silver counterpart. Preliminary phytochemical screening of aqueous extracts of T. decandra showed the presence of various secondary metabolites.Citation26 The bioreduction achieved in the present study is attributed to the metabolites present in the plant. Synthesis of gold nanoparticles was pronounced when 15 mL of root extract was used for bioreduction, while 10 mL of the same root extract was sufficient for production of silver nanoparticles.
Figure 1 Optical photograph of colloidal solution of (A) Trianthema decandra root extract, (B) HAuCl4 solution reduced with 5 mL of T. decandra extract, (C) HAuCl4 solution reduced with 10 mL of T. decandra extract, and (D) HAuCl4 solution reduced with 15 mL of T. decandra extract.

Figure 2 Optical photograph of colloidal solution of (A) Trianthema decandra root extract, (B) AgNO3 solution reduced with 5 mL of T. decandra extract, (C) AgNO3 solution reduced with 10 mL of T. decandra extract, and (D) AgNO3 solution reduced with 15 mL of T. decandra extract.

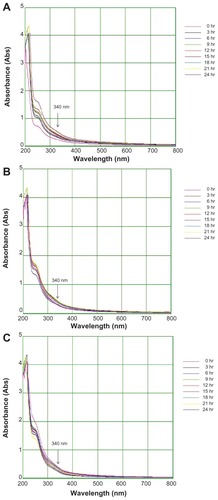
The colloidal gold solution showed strong (0.15 AU), weak (0.04 AU), and moderate (0.08 AU) absorption peaks with 15 mL, 5 mL, and 10 mL of extract respectively, at 575 nm (). The results also indicate a surface plasmon resonance gold band at 530–575 nm. The colloidal silver solution showed strong (0.81 AU), weak (0.23 AU), and moderate (0.31 AU) absorbance peaks with 10 mL, 5 mL, and 15 mL of extract respectively at 340 nm (). Nanoparticles may grow in a process involving rapid bioreduction and strongly influence surface plasmon resonance in the water extract.Citation27 The results of the present study are in accordance with previous reports, including that by Vijayakumar et alCitation27 on Artemisia nilagirica, indicating occurrence of a silver band at 340–400 nm. Further characterization using Fourier transform infrared, scanning electron, and energy dispersive x-ray spectroscopy was carried out for gold nanoparticles formed with 15 mL and silver nanoparticles formed with 10 mL of T. decandra root extract.
Figure 3 Ultraviolet-visible bioreduction kinetics in the 20–800 nm range for colloidal HAuCl4 solution with 5 mL of Trianthema decandra root extract (A), 10 mL of T. decandra root extract (B), and 15 mL of T. decandra root extract (C).
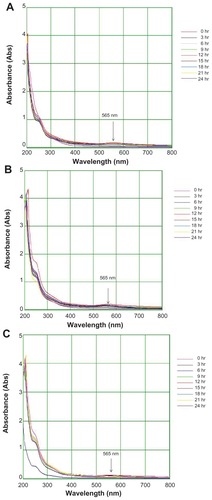
Figure 4 Ultraviolet-visible bioreduction kinetics in the 200–800 nm range for colloidal AgNO3 solution with 5 mL of Trianthema decandra root extract (A), 10 mL of T. decandra root extract (B), and 15 mL of T. decandra root extract (C).
The functional groups present in gold nanoparticles detected using Fourier transform infrared spectroscopy had absorption peaks located at 1649.33 cm−1 and 1020.12 cm−1 in the region of 1000–1800 cm−1, characteristic of gold atoms (). Two absorption peaks located at 1649.33 cm−1 and 1020 cm−1 are indicative of C=C stretch and aromatic C−C bonds, respectively. Distinct peaks in the range of 2344.87 cm−1 and 2364.35 cm−1 correspond to CαN stretch, while peaks at 2925.92 cm−1 and 3422.38 cm−1 correspond to C−H and O−H stretching vibration, respectively.
Figure 5 Fourier transform infrared absorption spectra for nanoparticles synthesized by bioreduction of HAuCl4 ions using Trianthema decandra root extract.
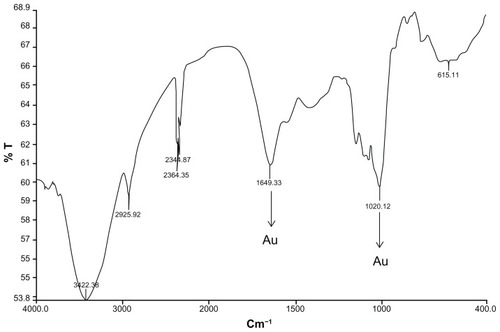
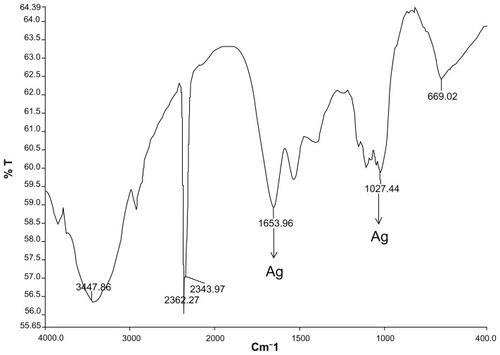
Fourier transform infrared spectroscopy analysis of the silver nanoparticles showed absorption peaks of reduced silver at 1653.96 cm−1 and 1027.44 cm−1 in the region of 1000–1800 cm−1 (). Two absorption peaks located at 1653 cm−1 are associated with the stretch vibration of C=C, and the single absorbance peak located at 1027 cm−1 is assigned to C−N stretching vibrations of amine. Further distinct peaks in the region of 2343.97–2362.27 cm−1 correspond to CαN stretch for nitrile groups, while 3447.86 cm−1 corresponds to O−H stretching vibration.
Figure 6 Fourier transform infrared absorption spectra for nanoparticles synthesized by bioreduction of AgNO3 ions using Trianthema decandra root extract.
Scanning electron micrographs () for the gold nanoparticles reveal numerous spherical, triangular, hexagonal, and cubic structures produced by reduction of goldions with sizes ranging between 33.7 nm and 99.3 nm. An individual gold nanohexagon with an average edge length of 737 nm is also seen in .
Figure 7 Scanning electron micrographs of gold nanoparticles observed from the reaction of Au3+ cations with Trianthema decandra root extract.
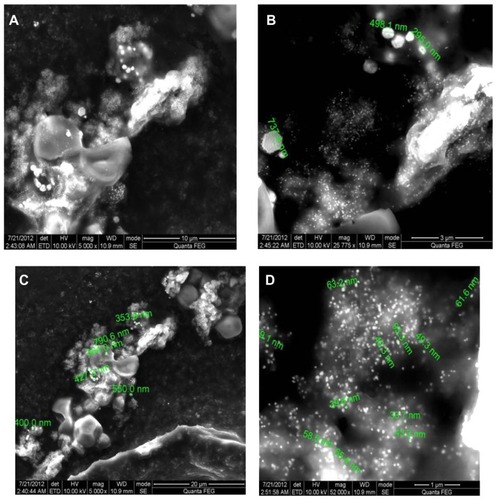
Very recently, Rai et al have demonstrated that the presence of halide ions and modulation of temperature can control the morphology of biologically synthesized gold triangles using lemongrass leaf extract.Citation28 In the present study, nanoparticles resembling triangles with sizes ranging between 33.7 nm and 99.3 nm were produced during bioreduction of HAuCl4, which could be attributed to the chloride ions present in it ().
Spherical silver nanoparticles with diameters of 36–94 nm are depicted in the scanning electron micrographs (). The variation in particle sizes, such as 36, 47, 60, and 94 nm, is possibly due to the fact that the nanoparticles are being formed at different times.
Figure 8 Scanning electron micrographs of silver nanoparticles observed from reaction of Ag+ cations with Trianthema decandra root extract.

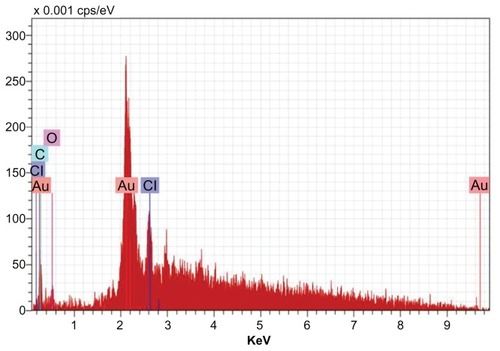
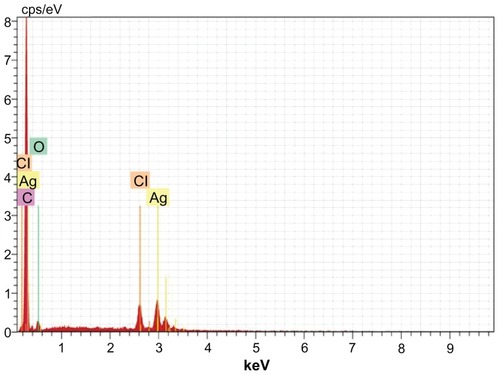
Water-soluble heterocyclic compounds, including saponin, were mainly responsible for the reduction of silver ions or chloroaurate ions to metallic silver and metallic gold, respectively. Further, it has been shown that the presence of chloride ions during nanosynthesis promotes the growth of nanotriangles. In the present study, reduction of chloroaurate ions produced nanotriangles. The presence of gold atoms in the gold nanoparticles was further confirmed by energy dispersive x-ray spectroscopy. The optical absorption peak was observed at approximately 2.2 keV, which is typical of gold nanocrystallite absorption due to surface plasmon resonance. The current energy dispersive x-ray spectroscopic profile of gold nanoparticles of T. decandra () showed strong gold atom signals around 0.30, 2, 2.2, 2.4, and 9.7 keV. Similarly, the presence of silver atoms in the silver nanoparticles was confirmed using energy dispersive x-ray spectroscopy (). The optical absorption peak was observed at 3 keV, which is typical for silver nanocrystallite absorption due to surface plasmon resonance. In an earlier study, individual spherical silver nanoparticles synthesized using alfalfa showed absorption peaks in the range of 2.5–4 keV.Citation29 The current profile of energy dispersive x-ray spectroscopy for silver nanoparticles of T. decandra indicate strong silver atom signals around 0.20, 3, 3.2, and 3.4 keV.
Antimicrobial activity of gold and silver nanoparticles
Antimicrobial activity of the synthesized gold and silver nanoparticles was studied using eight different bacteria, namely S. aureus, S. faecalis, E. faecalis, E. coli, P. aeruginosa, P. vulgaris, B. subtilis, Y. enterocolitica, and a fungus, C. albicans.
Both gold and silver nanoparticles exhibited excellent activity against Y. enterocolitica, P. vulgaris, E. coli, S. aureus, and S. faecalis. Zones of inhibition measuring 13.5–15.5 mm were recorded for the gold nanoparticles, while their silver counterparts recorded zones of 15.5–20.5 mm. Moderate activity was recorded against the other three bacteria tested (, and ). The growth inhibition of C. albicans was double in silver nanoparticles when compared with gold nanoparticles. The antibacterial effect was more pronounced in Gram-negative bacteria than Gram-positive ones. The antimicrobial activity of colloidal silver particles is influenced by the particle dimensions.Citation30 Silver has long been recognized as having an inhibitory effect on microbes present in medical and industrial processes. The most important application of silver and silver nanoparticles is in medical industry, such as in topical ointments to prevent infection against burns and open wounds.Citation31 Gold nanoparticles are harmful to bacteria and fungi. They bind closely to the surface of microorganisms, causing visible damage to cells, with complete destruction of flagella, stimulated production of biofilm, and aggregation within the biofilm.Citation32
Table 2 Mean zone of inhibition of synthesized gold and silver nanoparticles from T. decandra
Figure 11 Activity of gold nanoparticles against different microorganisms depicting zones of inhibition of (A) positive control. (B) gold nanoparticles, and (C) dimethylsulfoxide control.
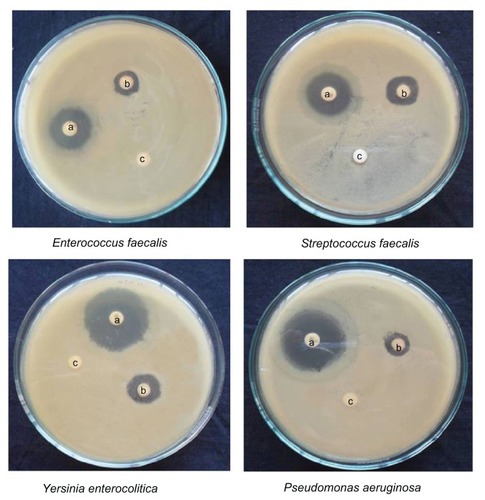
Figure 12 Activity of silver nanoparticles against different micro-organisms depicting zones of inhibition of (A) positive control. (B) silver nanoparticles, and (C) dimethylsulfoxide control.
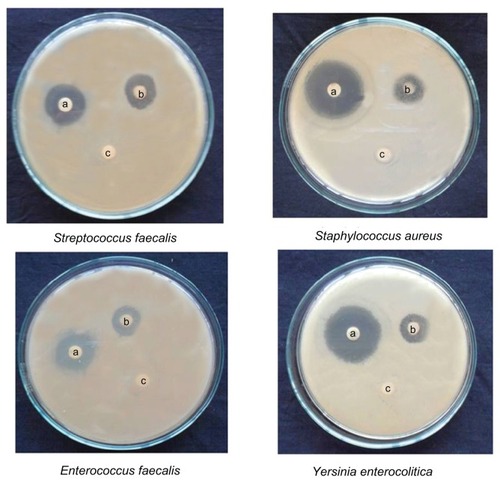
Comparison of gold and silver nanoparticle sizes indicates the latter to be smaller than the former. We attribute the higher activity of silver nanoparticles to their small size. The smaller particles have greater antimicrobial effects.Citation33 Further, the antimicrobial activity in the present study was much higher when compared with previous reports. Silver nanoparticles from Polyalthia longifolia, Balsamodendron mukul, Melia azadirachta, Berberis aristata, Acorus calamus, Embelia ribes, Psoralea corylifolia, Smilax china, Glycyrrhiza glabra, and Andropogon muricatus showed activity against E. coli, P. aeruginosa, S. aureus, and C. albicans, with zones of inhibition measuring less than 8 mm in diameter.Citation30,Citation33 The results of the present study are indicative of pronounced antibacterial effects. They provide an important basis for use of gold and silver nanoparticles synthesized from the root extract of T. decandra in the treatment of human infections associated with the microorganisms used in this study. These might prove to be important leads for the development of new antimicrobial drugs.
Conclusion
To summarize, we achieved successful green synthesis of gold and silver nanoparticles using T. decandra root extract. Silver and gold nanoparticles were synthesized under ambient conditions, and characterization of synthesized nanoparticles was carried out by ultraviolet-visible, Fourier transform infrared, scanning electron and energy dispersive x-ray spectroscopy. It is believed that phytochemicals present in T. decandra extract reduce the silver and gold ions into metallic nanoparticles. This strategy reduces the cost of production and the environmental impact. The silver and gold nanoparticles formed showed strong activity against all microorganisms tested.
Acknowledgments
The authors acknowledge the Botanical Survey of India, TNAU campus, Coimbatore, for identification and authentication of the plant material used in this study. The authors also thank Sri Ramaswamy Memorial University for providing the infrastructure facilities.
Disclosure
The authors declare no conflicts of interest in this work.
References
- MateiACernicaICadarORomanCSchiopuVSynthesis and characterization of ZnO-polymer nanocompositesInt J Mater Form20081767770
- KempMMKumarAMousaSSynthesis of gold and silver nanoparticles stabilized with glycosaminoglycans having distinctive biological activitiesBiomacromolecules20091058959519226107
- RivasLSanchez-CortesSGarcia-RamosJVMorcilloGGrowth of silver colloidal particles obtained by citrate reduction to increase the Raman enhancement factorLangmuir200117574577
- ZhangZPatelRCKothariRJohnsonCPFribergSEAikensPAStable silver clusters and nanoparticles prepared in polyacrylate and inverse micellar solutionsJ Phys Chem B200010411761182
- ChenWCaiWZhangLWangGSonochemical processes and formation of gold nanoparticles within pores of mesoporous silicaJ Colloid Interface Sci200123829129511374924
- FrattiniAPellegriNNicastroDde SanctisOPreparation of amine coated silver nanoparticles using triethylenetetramineMater Chem Phys200594148152
- KnollBKeilmannFNear-field probing of vibrational absorption for chemical microscopyNature1999399134137
- SenguptaSEavaroneDCapilaIZhaoGLWatsonNKiziltepeTTemporal targeting of tumour cells and neovasculature with a nanoscale delivery systemNature200543656857216049491
- WileyBJSunYXiaYSynthesis of silver nanostructures with controlled shapes and propertiesAcc Chem Res2007401067107617616165
- KumarVYadavSKPlant-mediated synthesis of silver and gold nanoparticles and their applicationsJ Chem Technol Biotechnol200984151157
- ChandranSPChaudharyMPasrichaRAhmadASastryMSynthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extractBiotechnol Prog20062257758316599579
- MudeNAvinashIAniketGMahendraRSynthesis of silver nanoparticles using callus extract of Carica papaya – a first reportJ Plant Biochem Biotechnol2009188386
- SongJYJangHKKimBSBiological synthesis of gold nanoparticles using Magnolia kobus and Diopyros kaki leaf extractProcess Biochem20094411331138
- LukmanALGongBMarjoCERoessnerUHarrisATJ Colloid Interface Sci201135343320974473
- SinghAChaudharyMSastryMConstruction of conductive multilayer films of biogenic triangular gold nanoparticles and their application in chemical vapour sensingNanotechnology20061723992405
- ShankarSSRaiAAhmadASastryMRapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using neem (Azadirachta indica) leaf brothJ Colloid Interface Sci20047549650215178278
- SingaraveluGArockiamaryJSGanesh KumarbVGovindarajuKA novel extracellular synthesis of monodisperse gold nanoparticles using marine alga, Sargassumwightii grevilleColloids Surf B Biointerfaces2007579710117350236
- ArmendarizVHerreraIPeralta-VideaJJose-YacamanMTroianiHSantiagoPSize controlled gold nanoparticle formation by Avena sativa biomass: use of plants in nanobiotechnologyJ Nanopart Res20046377382
- ShankerSSRaiAAnkamwarBSinghAAhmedASastryMBiological synthesis of triangular gold nanoprismsNat Mater2004348215208703
- MukherjeePBhattacharyaRBoneNPotential therapeutic application of gold nanoparticles in B-chronic lymphocytic leukemia (BCLL) enhancing apoptosisJ Nanobiotechnology20075417488514
- ChandranSPChaudharyMPasrichaRAhmadASastryMSynthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extractBiotechnol Prog2006257758316599579
- KirtikarKRBasuBDIndian Medicinal PlantsAllahabad, IndiaLalit Mohan Basu Publications1983
- GeethalakshmiRSaradaDVLMarimuthuPEvaluation of antimicrobial and antioxidant potentials of Trianthema decandra LAsian J Biotechnol20102225231
- BauerAWKirbyWMMSherrisJCTurckMAntibiotic susceptibility testing by a standardized single disk methodAm J Clin Pathol1966454934965325707
- LinkSEl-SayedMASize and temperature dependence of the plasmon absorption of colloidal Au nanoparticlesJ Phys Chem1999B10342124217
- GeethalakshmiRSaradaDVLRamasamyKTrianthema decandra L: a review on its phytochemical and pharmacological profileInt J Eng Sci Technol20102976979
- VijayakumarMPriyaKNancyFTNoorlidahAAhmedABABiosynthesis, characterization and anti-bacterial effect of plant-mediated silver nanoparticles using Artemisia nilagiricaIndus Crops Prod201241235240
- RaiASinghAAhmadASastryMRole of halide ions and temperature on the morphology of biologically synthesized gold nanotrianglesLangmuir20062273674116401125
- Gardea-TorresdeyJLGomezEPeralta-VideaJSynthesis of gold nanotriangles and silver nanoparticles using Alfalfa sprouts:a natural source for the synthesis of silver nanoparticlesLangmuir20031913571365
- KaviyaSSanthanalakshmiJViswanathanBGreen synthesis of silver nanoparticles using Polyalthia longifolia leaf extract along with D-sorbitol: study of antibacterial activityJ Nanotechnol201115
- MoronesJRElechiguerraLJCamachoAThe bactericidal effect of silver nanoparticlesNanotechnology2005162346235320818017
- GhoshPHanGDeMKimKCRotelloMVGold nanoparticles in delivery applicationsAdv Drug Deliv Rev2008601307131518555555
- PrashanthSMenakaIMuthezhilanRNavin KumarSSynthesis of plant-mediated silver nano particles using medicinal plant extract and evaluation of its anti microbial activitiesInt J Eng Sci Technol2001362356250