?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
Background
Dental implants have become increasingly common for the management of missing teeth. However, peri-implant infection remains a problem, is usually difficult to treat, and may lead eventually to dental implant failure. The aim of this study was to fabricate a novel antibacterial coating containing a halogenated furanone compound, ie, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone (BBF)-loaded poly(L-lactic acid) (PLLA) nanoparticles on microarc-oxidized titanium and to evaluate its release behavior in vitro.
Methods
BBF-loaded PLLA nanoparticles were prepared using the emulsion solvent-evaporation method, and the antibacterial coating was fabricated by cross-linking BBF-loaded PLLA nanoparticles with gelatin on microarc-oxidized titanium.
Results
The BBF-loaded PLLA nanoparticles had a small particle size (408 ± 14 nm), a low polydispersity index (0.140 ± 0.008), a high encapsulation efficiency (72.44% ± 1.27%), and a fine spherical shape with a smooth surface. The morphology of the fabricated antibacterial coating showed that the BBF-loaded PLLA nanoparticles were well distributed in the pores of the microarc oxidation coating, and were cross-linked with each other and the wall pores by gelatin. The release study indicated that the antibacterial coating could achieve sustained release of BBF for 60 days, with a slight initial burst release during the first 4 hours.
Conclusion
The novel antibacterial coating fabricated in this study is a potentially promising method for prevention of early peri-implant infection.
Introduction
Dental implants are the most innovative and superior treatment used in dentistry, and are becoming increasingly one of the major treatment modalities for replacement of missing teeth.Citation1,Citation2 The success rate for dental implants has been frequently reported to be as high as 90%–95% for both partially and completely edentulous patients in longitudinal studies.Citation3,Citation4 Despite this high success rate, many studies have demonstrated that dental implants do sometimes fail.Citation5,Citation6 Peri-implant infection, which may be categorized as early or late, is reported to be one of the most important causes of dental implant failure.Citation7–Citation9 Early infection occurs before osseointegration, while late infection occurs after prosthetic rehabilitation. Surgical trauma and bacterial invasion during and after implant insertion are the primary causes of early infection.Citation10 Late infection seems to be related to peri-implantitis.Citation11 Bacteria adhering to the surface of the implant and forming biofilm play a pivotal role in early peri-implant infection.Citation12,Citation13 Although various measures, such as thorough disinfection and stringent aseptic operation, have been proposed to mitigate bacterial contamination, there is still evidence that bacterial invasion occurs after dental implant surgery.Citation9 Additionally, over 300 species of bacteria are present in the oral cavity, each region of which is home to a characteristic assemblage of microbial flora.Citation14 Bacteria on the mucosa and adjacent teeth can invade the peri-implant soft tissue, and may potentially progress deeper into the bone and undermine the osseointegration process.Citation10 Because there is still no good means available to eliminate peri-implant infection completely after it has occurred, it is imperative to explore effective ways to prevent bacterial invasion. Fabricating antibacterial coatings on titanium implants is an important strategy to prevent early peri-implant infection, which inhibits initial bacterial adhesion.Citation15 Therefore, various antibacterial coatings containing many kinds of antibacterial have been designed. However, concerns about the susceptibility of bacteria in the oral cavity to antibacterial and drug resistance of bacteriaCitation16,Citation17 have encouraged the search for new effective antibacterial strategies. Another matter of concern has been the antibacterial release time.Citation15
In recent years, halogenated furanones, a class of secondary metabolites originally extracted from the red algae, Delisea pulchra, have been demonstrated to have antimicrobial properties that inhibit microbial colonization on the algal surface.Citation18,Citation19 Subsequently, a variety of analogs of the natural halogenated furanones have been synthesized and evaluated for their efficacy. These compounds have been demonstrated to have potent antimicrobial activity against a great number of Gram-positive and Gram-negative bacteria and to inhibit biofilm formation.Citation20,Citation21 Halogenated furanone compounds can control multicellular behavior in various microbial species induced by autoinducer-1 and autoinducer-2,Citation22 which are used for cell-to-cell communication to control production and secretion of virulence factors, proteolytic activity, carbohydrate metabolism, and biofilm formation.Citation23 The novel nature of their mechanisms of action would allow these new antimicrobials to be effective against bacteria that are currently antibiotic-resistant.Citation24 In spite of the inhibitory effects on microbial biofilm formation, reports of the effects on eukaryotic cell viability and toxicity of halogenated furanone compounds are scarce, and reports on genotoxic effects seem to be lacking.Citation25,Citation26 Owing to their advantages, many researchers have suggested that halogenated furanone compounds would potentially be ideal antibacterial agents on implants for preventing biofilm infection.Citation27,Citation28 However, there have been few relevant reports on how to use halogenated furanone compounds in implants until now.
During the past decade, considerable research efforts have aimed at developing controlled-release systems for several drugs (eg, anesthetics, antibiotics, antitumoral drugs, proteins) from biodegradable polymers such as poly(D, L-lactic acid) (PLA), poly(D, L-lactide-co-glycolide), and poly(-caprolactone).Citation29,Citation30 Among them, PLA has been widely developed as a microsphere/nanoparticle carrier due to its desirable biocompatible and biodegradable properties. PLA is also approved by the US Food and Drug Administration for clinic use. The simplest and most commonly employed method for preparing microspheres/nanoparticles is oil-in-water emulsion-solvent evaporation, which shows good encapsulation efficiency of water-insoluble compounds.
However, being bioinert, the integration of titanium implants in bone is not in good condition. Previous studies have suggested that the rate and extent of osseointegration is mainly determined by the properties of the implant surface.Citation31 Many efforts have been made to modify the composition and topography of the implant surface and thereby achieve improved osseointegration.Citation31,Citation32 Current trends include use of endosseous dental implant surfaces embellished with nanostructured topography.Citation33,Citation34 Microarc oxidation is a commonly used surface modification technique for producing an inorganic glass-ceramic-like coating on the titanium surface, for the purpose of providing corrosion-resistance and wear-resistance or various functional properties.Citation35,Citation36 Previous studies have showed that microarc oxidation helps to form a biologically active bone-like apatite layer on titanium, which may improve the biological response.Citation37 Moreover, the microarc oxidation coating has a porous surface, with pores 1–5 μm in diameter at regular intervals, providing space for adhesion of nanoparticles.
The purpose of the current study was to prepare poly(L-lactic acid) (PLLA) nanoparticles containing a halogenated furanone compound, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone (BBF), and then fabricate a new antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc-oxidized titanium and evaluate the release behavior in vitro, in order to provide a new method for prevention of early peri-implant infection.
Materials and methods
Materials
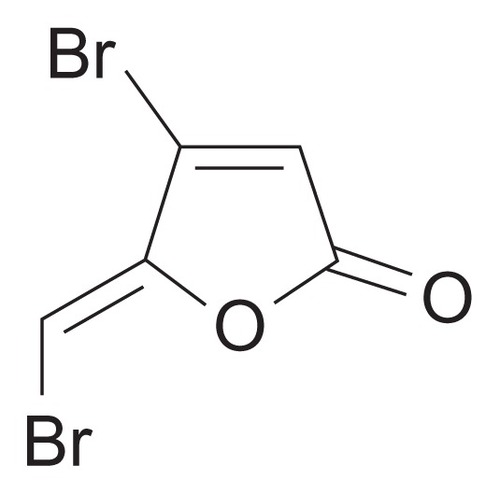
BBF (≥97.0%), PLLA (number average 99,000, average molecular weight 152,000), poly(vinyl alcohol) (PVA, molecular weight 89,000–98,000) and gelatin powder (type A) were purchased from Sigma-Aldrich Chemicals (St Louis, MO). The chemical structure of BBF is shown in . Commercially pure titanium (grade 2) was provided by Xi’an Aerospace New Material Co, Ltd (Xi’an, China). Methanol (high-performance liquid chromatography grade) and other chemicals and solvents (analytical grade) were supplied by Tianjin Fuchen Chemical Factory (Tianjin, China).
Preparation of BBF-loaded PLLA nanoparticles
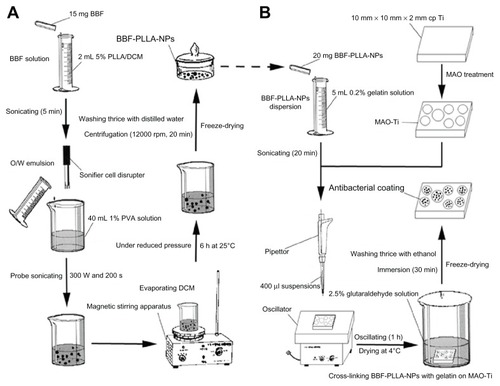
BBF-loaded PLLA nanoparticles were prepared using the classical oil-in-water emulsion solvent-evaporation method. The procedure is schematically shown in . Briefly, the oil phase containing 15 mg of BBF was added to 2 mL of dichloromethane in which was dissolved 100 mg of PLLA (5% w/v) under sonication for 5 minutes. This oil phase was added to 40 mL of 1% (w/v) PVA aqueous solution, which was prepared by heating and stirring during addition of PVA. The mixed solution was then probe-sonicated using a Sonifier Cell Disrupter (JY98-IIIN, Ningbo Xingzhi Biological Technology Co, Ltd, China) in an ice bath to produce an oil-in-water emulsion. The intensity and duration of the sonication process were 300 W and 200 seconds, respectively. The resulting emulsion was stirred using a magnetic stirring device (90-1, Zhengzhou KaiPeng Experiment Instrument Co, Ltd, China) for 6 hours at 25°C under reduced pressure to evaporate the dichloromethane completely. After preparation, the nanoparticles were collected by centrifugation at 12,000 rpm for 20 minutes. The collected nanoparticles were then washed three times with distilled water in an ultrasonic bath at 37°C, isolated by centrifugation, and finally freeze-dried.
Figure 2 Schematic presentation of the preparation of BBF-loaded PLLA nanoparticles and the fabrication of antibacterial coating containing BBF-loaded PLLA nanoparticles on MAO-Ti. (A) Preparation of BBF-loaded PLLA nanoparticles. (B) Fabrication of antibacterial coating containing BBF-loaded PLLA nanoparticles on MAO-Ti.
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; DCM, dichloroethane; PLLA, poly(L-lactic acid); NPs, nanoparticles; MAO-Ti, microarc-oxidized titanium; PVA, poly (vinyl alcohol); O/W, oil/water.
Characterization of BBF-loaded PLLA nanoparticles
Size, size distribution, and surface morphology
The mean particle size, size distribution, and polydispersity index of the nanoparticles were assessed by dynamic light scattering using a particle size analyzer (SALD-7101, Shimadzu Corporation, Tokyo, Japan) at 25°C. For a typical experiment, 10 mg of BBF-loaded PLLA nanoparticles were sonicated to suspend in 5 mL of distilled water and then analyzed using the particle size analyzer. Each value reported was the mean ± standard deviation for three replicate samples. The shape and surface morphology of the nanoparticles were observed by scanning electron microscopy (SEM, S-4800, Hitachi Co, Japan).
Determination of drug incorporation
The amount of drug incorporated into the nanoparticles was determined by high-performance liquid chromatography (Waters, Milford, MA), using a reverse phase Lichrospher C18 column (pore size, 5 μm, 250 mm × 4.6 mm, Diamonsil, Dikma Technologies Inc, Lake Forest, CA). Next, 5 mg of freeze-dried BBF-loaded PLLA nanoparticles was dissolved in 1 mL of dichloromethane and stirred after addition of 10 mL of methanol to precipitate the polymer preferentially. The precipitate was removed using a membrane filter. The solvent was then evaporated until dry with a nitrogen stream, followed by addition of 10 mL of methanol/water mixture (75/25, v/v). The clear solution obtained was put into a vial for detection of the BBF concentration using high-performance liquid chromatography. The mobile phase consisted of a mixture of methanol/water (75/25, v/v) and the flow rate was 1.0 mL per minute. The column effluent was detected at 286.0 nm using a variable wavelength detector with a column temperature of 30°C. The experiments were performed in triplicate.
Drug incorporation was expressed both as actual drug loading (w/w) and encapsulation efficiency (%), represented by Equationequations (1)(1) and Equation(2)
(2) , respectively, as:
In vitro BBF release study
The release study for BBF-loaded PLLA nanoparticles was performed using the dialysis bag diffusion method. First, 20 mg of freeze-dried BBF-loaded PLLA nanoparticles (containing about 2.04 mg BBF) was suspended in 5 mL of phosphate-buffered saline (containing NaCl 8 g/L, KCl 0.2 g/L, Na2HPO4 1.44 g/L, and KH2PO4 0.24 g/L, stabilized with sodium azide 0.2 g/L, pH 7.4) and transferred to a dialysis bag (molecular weight cutoff 3500 g/mol, Shanghai Green Bird Science and Technology Development Co, Ltd, Shanghai, China). This bag was introduced into 100 mL of phosphate-buffered saline, with continuous gentle magnetic stirring at 37°C under sink conditions. At predetermined time intervals, 1 mL aliquots were withdrawn and replaced with the same volume of fresh buffer. All samples were taken for analysis of BBF concentration, which was first extracted with 1 mL of dichloromethane, followed by evaporating until dry using a nitrogen stream, and 10 mL of a methanol/water (75/25, v/v) mixture was then added. High-performance liquid chromatography analysis was carried out as described in the previous section. Next, 2.04 mg of BBF powder was examined under the same conditions for comparison. The experiments were performed in triplicate. Additionally, the morphology of the BBF-loaded PLLA nanoparticles in the dialysis bags was observed by SEM at predetermined time intervals to investigate the process of degradation.
Preparation of commercially pure titanium specimens and microarc oxidation
Plate specimens (10 mm × 10 mm × 2 mm) were used in this study. All the specimens were mechanically ground using various grades of silicon carbide paper (80–1200 grit), and then ultrasonically cleaned in ethanol, rinsed with water, and air-dried. Microarc oxidation was carried out in an aqueous electrolytic solution containing 0.04 M β-glycerophosphate sodium and 0.2 M calcium acetate, using a pulsed direct current power supply (microarc oxidation equipment was designed and manufactured by Xi’an University of Technology, Xi’an, China). The applied voltage, frequency, duty cycle, and oxidizing time were 300 V, 600 Hz, 8.0%, and 5 minutes, respectively.Citation38 The microarc-oxidized titanium specimens were then rinsed with distilled water and air-dried immediately. The surface morphology of the specimens before and after microarc oxidation was observed by SEM. The surface phase composition of the oxide layer formed by the microarc oxidation process was analyzed by x-ray diffraction (XRD-7000S, Shimadzu Corporation, Tokyo, Japan).
Fabrication of antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc-oxidized titanium
The antibacterial coating was achieved by cross-linking the BBF-loaded PLLA nanoparticles with gelatin on microarc-oxidized titanium.Citation39 The procedure is schematically shown in . In brief, 20 mg of BBF-loaded PLLA nanoparticles were sonicated for 20 minutes to disperse them in 5 mL of 0.2% gelatin solution (w/v), from which 400 μL suspensions were dropped onto the microarc-oxidized titanium. The microarc-oxidized titanium specimen was then oscillated on an oscillator (HY-2, Hangzhou Huier Experiment Instrument Co, Ltd, China) for one hour to enable the nanoparticles to penetrate the pores on the surface of the microarc-oxidized titanium. After oscillating, the microarc-oxidized titanium specimen was dried at 4°C, followed by immersion in 2.5% (w/v) glutaraldehyde solution for 30 minutes to cross-link the gelatin. Finally, the microarc-oxidized titanium specimen was washed with ethanol three times to remove the remaining glutaraldehyde and freeze-dried. The surface morphology of the antibacterial coating containing BBF-PLLA nanoparticles on microarc microarc-oxidized titanium was observed by SEM.
In vitro study of BBF release from the antibacterial coating
Release of BBF from the antibacterial coating was performed by placing one BBF-PLLA nanoparticle-coated microarc-oxidized titanium specimen with 5 mL of phosphate-buffered saline in a dialysis bag (molecular weight cutoff 3500 g/mol). This bag was then introduced into 20 mL of phosphate-buffered saline with continuous gentle magnetic stirring at 37°C. At predetermined time intervals, 1 mL aliquots were withdrawn and replaced with the same volume of fresh buffer. The BBF concentration in phosphate-buffered saline was measured according to the methods previously described. The experiments were performed in triplicate. The surface morphology of the antibacterial coating immersed in phosphate-buffered saline was also observed by SEM at predetermined time intervals to investigate the process of degradation.
Results
Preparation and characterization of BBF-loaded PLLA nanoparticles
Size, size distribution, and surface morphology
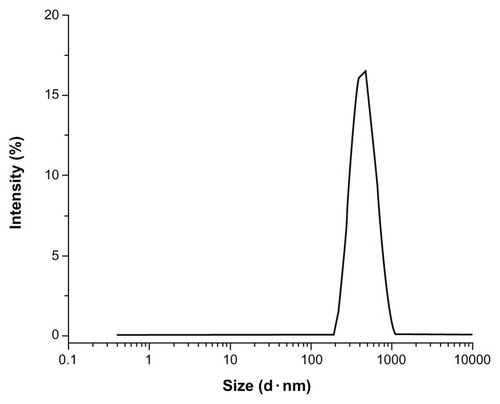
BBF-loaded PLLA nanoparticles were successfully prepared using the oil-in-water emulsion solvent-evaporation method. Particle size measured by dynamic light scattering revealed that the BBF-loaded PLLA nanoparticles had a mean particle size of 408 ± 14 nm. shows a narrow size distribution with a polydispersity index of 0.140 ± 0.008. A fine dispersion with long-term nanoparticle stability should have a narrow size distribution and a low polydispersity index, which is defined as the log normal distribution width of the particle diameter. Particle size can be controlled by altering several experimental parameters, such as polymer concentration, intensity and duration of sonication, and the volume ratio of the aqueous and organic phases.
Figure 3 Size distribution of BBF-loaded PLLA nanoparticles.
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; PLLA, poly(L-lactic acid).
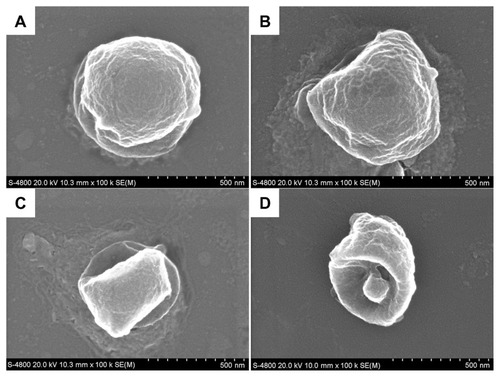
The surface morphology of BBF-loaded PLLA nanoparticles is shown in . All nanoparticles were spherical in shape without any aggregation or adhesion (). The particle size distribution was well proportioned and in good agreement with the result measured by dynamic light scattering. Moreover, the BBF-loaded PLLA nanoparticles had a smooth surface, on which no pores and cracks were observed at high magnification (). Surface morphology is a crucial property for microspheres/nanoparticles, given that in vitro release experiments have shown that the initial burst from a smooth surface is less than that from a rough surface.Citation40
Drug incorporation
Drug loading (w/w) and encapsulation efficiency (%) of BBF determined by high-performance liquid chromatography analysis of the prepared PLLA nanospheres were 0.102 ± 0.002 mg/mg and 72.44% ± 1.27%, respectively. The high encapsulation efficiency was probably due to the high affinity and solubility of BBF in the organic solvent and low solubility in the aqueous phase, which would consequently reduce the possibility of drug loss by diffusion during preparation of the nanospheres. Therefore, a large amount of BBF was successfully encapsulated in the PLLA nanospheres. Encapsulation efficiency can be affected by many factors, including the molecular weight of the polymer, particle size, and the mass ratio of drug and polymer.
In vitro release study of BBF from BBF-loaded PLLA nanoparticles
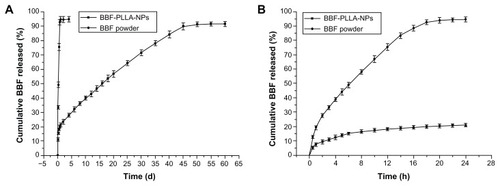
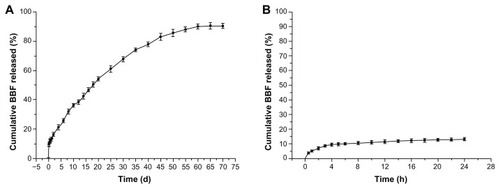
showed the in vitro release profiles for BBF from BBF-loaded PLLA nanoparticles and BBF powder. Experimental points over the complete time assay are shown in , and the first 24 hours in the release study are shown in . The in vitro release profiles for BBF from BBF-loaded PLLA nanoparticles showed a biphasic release pattern, namely an initial burst release and a more continuous slow release. The initial burst release showed that 15.4% of the accumulated BBF was released from the nanoparticles at the first sampling time of 6 hours. The burst effect is normally attributable to release of drug that is adsorbed or close to the surface of the nanoparticles, which is known to be permeable to water.Citation41 In the second stage, release of BBF from nanoparticles was typically sustained and dependent on degradation of the polymer matrix and drug diffusion, which is a slower process. As time progressed, the nanoparticles were degraded, resulting in diffusion of water into their core and BBF diffusing into that water. The results obtained in the sustained release period show that 74.5% of the accumulated BBF was released slowly during the next 45 days. In the control group, 92.7% of BBF was released from the BBF powder in the first 18 hours.
Figure 5 In vitro release curves for BBF from BBF-loaded PLLA nanoparticles and BBF powder in phosphate-buffered saline. (A) Experimental points over the complete time assay. (B) First 24 hours of the release study (n = 3).
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; PLLA, poly(L-lactic acid); NPs, nanoparticles.
The degradation process for BBF-loaded PLLA nanoparticles in phosphate-buffered saline is shown in . After a 10-day degradation process, a rough irregular degraded surface was observed by SEM (). The surface of BBF-loaded PLLA nanoparticles became rougher and the spherical shape deformed after 20 days of degradation in phosphate-buffered saline (). As degradation proceeded, the BBF-loaded PLLA nanoparticles showed gradual loss of their spherical shape. The BBF-loaded PLLA nanoparticles showed only partial retention of their shapes due to degradation of the polymer after 30 days (). Surface degradation of the polymer was accompanied with a reduction in particle size. After 45 days of degradation in phosphate-buffered saline, the bulk of the BBF-loaded PLLA nanoparticles was degraded, leaving behind a shell-like polymer (). Degradation proceeded faster internally than externally in phosphate-buffered saline buffer due to internal build up of acidic byproducts that self-catalyze the degradation process, resulting in formation of hollow cores.Citation42 Our morphological study results agreed with the BBF release curve from PLLA nanoparticles.
Figure 6 Scanning electron microscopic images of the degradation process of BBF-loaded PLLA nanoparticles in phosphate-buffered saline. (A) 10 days. (B) 20 days. (C) 30 days. (D) 45 days.
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; PLLA, poly(L-lactic acid).
The PLLA nanoparticle degradation rate was related to environmental factors, including temperature and pH. The pH of the phosphate-buffered saline in the release studies was checked after 2 months had elapsed, and was still within the range of 7.0–7.4. Therefore, the volume and buffering capacity of the phosphate-buffered saline were sufficient to diminish any effect of PLLA degradation on the pH of the degradation medium.
Microarc oxidation
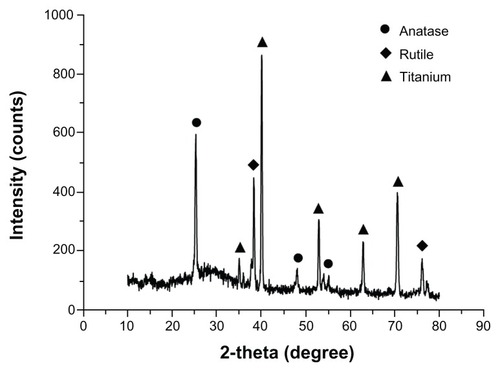
Microarc oxidation was carried out using a pulsed direct current power supply. shows SEM images of the surface morphology of commercially pure titanium before and after microarc oxidation. The surface of the commercially pure titanium before microarc oxidation shows some parallel grooves oriented along the machined direction (). After microarc oxidation, a porous coating with pores 1–3 μm in diameter distributed at regular intervals was observed (). The pores formed by oxygen bubbles in the coating growth process, could provide space for adhesion of nanoparticles in the next procedure. The surface phase composition of the microarc oxidation coating is shown in . Peaks for anatase, rutile, and titanium were detected on the microarc-oxidized titanium.
Figure 7 Scanning electron microscopic images of the surface morphology of commercially pure titanium (A) before and (B) after microarc oxidation.

Figure 8 X-ray diffraction spectra of the surface phase composition of the microarc oxidation coatings.
Microarc oxidation coatings usually exhibit good adhesion to substrates, and the wear resistance, corrosion resistance, and microhardness of the coating have greatly improved.Citation35,Citation36 Previous studies have confirmed that microarc oxidation coatings containing Ca and P show significantly increased hydrophilicity and roughness, which would be beneficial for cell attachment, propagation, and bone growth.Citation43 Therefore, microarc oxidation is considered to be one of the best methods for modifying the titanium implant surface and improving its bioactive capability.
Fabrication and surface morphology of antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc-oxidized titanium
The antibacterial coating was fabricated by cross-linking BBF-loaded PLLA nanoparticles successfully with gelatin on microarc-oxidized titanium. shows the surface morphology of an antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc-oxidized titanium. The pores on the surface of microarc-oxidized titanium were almost overlaid by the cross-linked nanospheres/gelatin composite coating, and there were only a few nanospheres adherent to the flat areas between the pores due to the oscillating procedure during fabrication (), which would effectively prevent the antibacterial nanospheres from falling off during insertion of the dental implant. The BBF-loaded PLLA nanoparticles were well distributed in the pores of the microarc oxidation coating, and cross-linked with each other and the walls of the pores by gelatin (). This antibacterial coating had relatively high stability and binding capacity because it was integrated with the microarc oxidation coating by physical interlocking.Citation39
Figure 9 Scanning electron microscopic images of the antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc-oxidized titanium. Antibacterial coating at low (A) and high (B) magnification.
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; PLLA, poly(L-lactic acid).

In vitro release study of BBF from the antibacterial coating
shows the in vitro release profile for BBF from the antibacterial coating. It also shows a biphasic release phenomenon, which is similar to the release profile for BBF from the BBF-loaded PLLA nanoparticles. The initial burst release showed that 9.4% of the accumulated amount of BBF was slowly released from the antibacterial coating at the first sampling time of 4 hours. At the second stage of release, 80.8% of BBF was slowly released from the antibacterial coating during the next 60 days. However, compared with the release curves for BBF from BBF-loaded PLLA nanoparticles, the cross-linked antibacterial coating showed a slightly lower initial burst release and a longer sustained release period. The results of the current study agree with previous research findings.Citation39 It is thought that the presence of the cross-linked gelatin created a barrier for the surface-associated drug to diffuse into the phosphate-buffered saline, resulting in a lower initial burst release.
Figure 10 In vitro release curves for BBF from antibacterial coating containing BBF-loaded poly(L-lactic acid) nanoparticles on microarc-oxidized titanium. (A) Experimental points over the complete time assay. (B) First 24 hours for the release study (n = 3).
Abbreviation: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone.
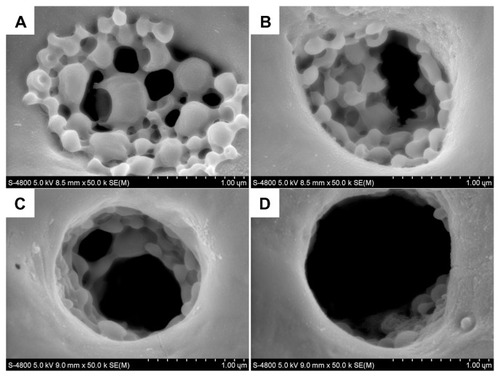
The morphological study of the degradation process for antibacterial coating in phosphate-buffered saline is shown in . After a 15-day degradation process, some of the BBF-loaded PLLA nanoparticles in the fabricated antibacterial coating deformed from their spherical shape (). As degradation proceeded, some of the nanoparticles separated from the cross-linked gelatin due to external degradation after 30 days of immersion in the stirred phosphate-buffered saline (). Subsequently, the number of nanoparticles separating out increased (). After 60 days of degradation, there were only a few degraded nanoparticles and fragments remaining in the pores on the microarc-oxidized titanium (). These findings are consistent with the release curves for BBF from the antibacterial coating.
Figure 11 Scanning electron microscopic images of the degradation process for antibacterial coating containing BBF-loaded PLLA nanoparticles on microarc oxidation-treated titanium in phosphate-buffered saline. (A) 15 days. (B) 30 days. (C) 45 days. (D) 60 days.
Abbreviations: BBF, (Z-)-4-bromo-5-(bromomethylene)-2(5H)-furanone; PLLA, poly(L-lactic acid).
Discussion
Researchers in the past have used various methods to try and prevent bacterial adhesion on dental implants, including physical and chemical modification of the implant surface. Physical surface treatments to alter the surface properties are a relatively simple way to repel bacterial colonization. Annunziata et al demonstrated decreased bacterial (Streptococcus pyogenes and Streptococcus sanguinis) adhesion and proliferation on a titanium nitride coating.Citation44 However, more attention has been paid to the improved osseointegration effect of physical surface treatments. Antibacterial coatings have been traditionally designed to prevent initial adhesion of bacteria onto the implant surface. Many surface coatings containing or releasing antimicrobial agents, such as cephalothin, amoxicillin, gentamicin, tobramycin, and vancomycin, have been incorporated into implants with some success.Citation45 Nevertheless, with the emergence of bacterial drug resistance,Citation16,Citation17 this form of therapy may be short-lived and may soon become ineffective. Additionally, several reports have pointed out that some antimicrobial agents may cause cell damage.Citation46 Chlorhexidine has been widely used in dentistry as a treatment for periodontal infection because of its broad spectrum of antimicrobial action and low risk of drug resistance. Studies have shown that chlorhexidine can be absorbed onto the TiO2 layer on the titanium surface, but the gradual release period lasts only a few days.Citation47 Silver has also been introduced onto titanium to enhance bactericidal ability, but the mechanism of bactericidal action needs further clarification. Therefore, using new effective antibacterial agents on implants and taking measures to ensure slow antibacterial release are essential in preventing early peri-implant infection.
Halogenated furanone compounds have been considered to be potentially ideal antibacterial agents on implants for preventing biofilm infection due to their broad spectrum antimicrobial activity,Citation20,Citation21 novel mechanisms of antimicrobial action,Citation22 and lack of toxicity and genotoxic effects.Citation25,Citation26 The BBF used in this study is a member of the synthetic halogenated furanone family of compounds. Previous studies have indicated that BBF is one of the most active halogenated furanone compounds for inhibiting biofilm formation at low concentrations (30–40 μM).Citation20 In the current study, a novel antibacterial coating containing BBF-loaded PLLA nanoparticles was fabricated on microarc-oxidized titanium. The results of the BBF release study showed that the antibacterial coating could sustain release of BBF for 60 days, with a slight initial burst release during the first 4 hours. Quirynen et al suggested that initial colonization of peri-implant pockets with bacteria occurred within the first 2 weeks after dental endosseous implantation, and that the number of bacteria in peri-implant pockets increased afterwards (2–13 weeks).Citation48 Therefore, the novel antibacterial coating fabricated in this study (sustaining release of BBF for 60 days) is a potentially promising method for prevention of early peri-implant infection and retrograde peri-implantitis,Citation49 which would be necessary for long-term success.
Furthermore, our research group has evaluated the biocompatibility and antimicrobial activity of the fabricated antibacterial coating in vitro. Our results indicate that the antibacterial coating has excellent biocompatibility and potent antimicrobial activity against various conventional periodontal pathogens, such as Porphyromonas gingivalis, Actinobacillus actinomycetemcomitans, Prevotella intermedia, and Fusobacterium. These results will be reported in an upcoming publication. Our next step will be to investigate the biocompatibility and antimicrobial activity of the fabricated antibacterial coating in vivo.
Conclusion
In this study, a novel antibacterial coating containing BBF-loaded PLLA nanoparticles was fabricated on microarc-oxidized titanium. The morphology of the antibacterial coating showed that the BBF-loaded PLLA nanoparticles were well distributed and cross-linked with each other and the pore walls by gelatin. The release study indicates that the antibacterial coating could sustain release of BBF for 60 days, with a slight initial burst release during the first 4 hours. The novel antibacterial coating fabricated in this study is a potentially promising method for the prevention of early peri-implant infection.
Acknowledgment
This project was supported by a grant from the National Natural Science Foundation of China (51001116).
Disclosure
The authors report no conflicts of interest in this work.
References
- BuserDMericske-SternRBernardJPLong-term evaluation of non-submerged ITI implants. Part 1: 8-year life table analysis of a prospective multi-center study with 2359 implantsClin Oral Implants Res199781611729586460
- EkelundJALindquistLWCarlssonGEJemtTImplant treatment in the edentulous mandible: a prospective study on Branemark system implants over more than 20 yearsInt J Prosthodont20031660260814714838
- FerrignoNLauretiMFanaliSGrippaudoGA long-term follow-up study of non-submerged ITI implants in the treatment of totally edentulous jaws. Part I: Ten-year life table analysis of a prospective multicenter study with 1286 implantsClin Oral Implants Res20021326027312010156
- SimonisPDufourTTenenbaumHLong-term implant survival and success: a 10–16-year follow-up of non-submerged dental implantsClin Oral Implants Res20102177277720636731
- Palma-CarrioCMaestre-FerrinLPenarrocha-OltraDPenarrocha-DiagoMAPenarrocha-DiagoMRisk factors associated with early failure of dental implants. A literature reviewMed Oral Patol Oral Cir Bucal201116e514e51721196891
- Schwartz-AradDLavivALevinLFailure causes, timing, and cluster behavior: an 8-year study of dental implantsImplant Dent20081720020718545052
- LindheJMeyleJPeri-implant diseases: consensus report of the sixth european workshop on periodontologyJ Clin Periodontol20083528228518724855
- Heitz-MayfieldLJPeri-implant diseases: diagnosis and risk indicatorsJ Clin Periodontol20083529230418724857
- QuirynenMDe SoeteMvan SteenbergheDInfectious risks for oral implants: a review of the literatureClin Oral Implants Res20021311912005139
- PyeADLockhartDEDawsonMPMurrayCASmithAJA review of dental implants and infectionJ Hosp Infect20097210411019329223
- QuirynenMTeughelsWMicrobiologically compromised patients and impact on oral implantsPeriodontol 200020033311912812950846
- MombelliAvan OostenMASchurchEJLandNPThe microbiota associated with successful or failing osseointegrated titanium implantsOral Microbiol Immunol198721451513507627
- LeonhardtARenvertSDahlenGMicrobial findings at failing implantsClin Oral Implants Res19991033934510551058
- PasterBJBochesSKGalvinJLBacterial diversity in human subgingival plaqueJ Bacteriol20011833770378311371542
- ZhaoLChuPKZhangYWuZAntibacterial coatings on titanium implantsJ Biomed Mater Res B Appl Biomater20099147048019637369
- FeresMHaffajeeADAllardKSomSGoodsonJMSocranskySSAntibiotic resistance of subgingival species during and after antibiotic therapyJ Clin Periodontol20022972473512390569
- Prieto-PrietoJCalvoAMicrobiological basis of oral infections and sensitivity to antibioticsMed Oral Patol Oral Cir Bucal20049Suppl1118
- de NysRGivskovMKumarNKjellebergSSteinbergPDFuranonesProg Mol Subcell Biol200642558616805438
- RenDBedzykLAYeRWThomasSMWoodTKDifferential gene expression shows natural brominated furanones interfere with the autoinducer-2 bacterial signaling system of Escherichia coliBiotechnol Bioeng20048863064215470704
- JanssensJCSteenackersHRobijnsSBrominated furanones inhibit biofilm formation by Salmonella enterica serovar TyphimuriumAppl Environ Microbiol2008746639664818791004
- KuehlRAl-BatainehSGordonOFuranone at subinhibitory concentrations enhances staphylococcal biofilm formation by luxS repressionAntimicrob Agents Chemother2009534159416619620329
- RenDBedzykLAYeRWThomasSMWoodTKDifferential gene expression shows natural brominated furanones interfere with the autoinducer-2 bacterial signaling system of Escherichia coliBiotechnol Bioeng20048863064215470704
- DanielsRVanderleydenJMichielsJQuorum sensing and swarming migration in bacteriaFEMS Microbiol Rev20042826128915449604
- RaffaRBIannuzzoJRLevineDRBacterial communication (“quorum sensing”) via ligands and receptors: a novel pharmacologic target for the design of antibiotic drugsJ Pharmacol Exp Ther200531241742315528454
- BavejaJKLiGNordonREBiological performance of a novel synthetic furanone-based antimicrobialBiomaterials2004255013502115109863
- HumeEBBavejaJMuirBThe control of Staphylococcus epidermidis biofilm formation and in vivo infection rates by covalently bound furanonesBiomaterials2004255023503015109864
- BavejaJKWillcoxMDHumeEBKumarNOdellRPoole-WarrenLAFuranones as potential anti-bacterial coatings on biomaterialsBiomaterials2004255003501215109862
- Lonn-StensrudJLandinMABennecheTPetersenFCScheieAAFuranones, potential agents for preventing Staphylococcus epidermidis biofilm infections?J Antimicrob Chemother20096330931619098295
- PancholiKA review of imaging methods for measuring drug release at nanometre scale: a case for drug delivery systemsExpert Opin Drug Deliv2012920321822235965
- KashiTSEskandarionSEsfandyari-ManeshMImproved drug loading and antibacterial activity of minocycline-loaded PLGA nanoparticles prepared by solid/oil/water ion pairing methodInt J Nanomedicine2012722123422275837
- Le GuehennecLSoueidanALayrollePAmouriqYSurface treatments of titanium dental implants for rapid osseointegrationDent Mater20072384485416904738
- GuidaLAnnunziataMRocciAContaldoMRulloROlivaABiological response of human bone marrow mesenchymal stem cells to fluoride-modified titanium surfacesClin Oral Implants Res2010211234124120497444
- MendoncaGMendoncaDBAragaoFJCooperLFAdvancing dental implant surface technology – from micron- to nanotopographyBiomaterials2008293822383518617258
- AnnunziataMOlivaABuoscioloAGiordanoMGuidaAGuidaLBone marrow mesenchymal stem cell response to nano-structured oxidized and turned titanium surfacesClin Oral Implants Res20122373374021679255
- WangYMJiangBLLeiTQGuoLXMicroarc oxidation coatings formed on Ti6Al4V in Na2SiO3 system solution: microstructure, mechanical and tribological propertiesSurf Coat Technol20062018289
- YaoZPJiangZHWuXHSunXTWuZDEffects of ceramic coating by micro-plasma oxidation on the corrosion resistance of Ti-6Al-4V alloySurf Coat Technol200520024452450
- WuJLiuZMZhaoXHGaoYHuJGaoBImproved biological performance of microarc-oxidized low-modulus Ti-24 Nb-4Zr-7.9Sn alloyJ Biomed Mater Res B Appl Biomater20109229830619904739
- WeiDZhouYJiaDWangYCharacteristic and in vitro bioactivity of a microarc-oxidized TiO(2)-based coating after chemical treatmentActa Biomater2007381782717478133
- XuXHLuPGuoMQFangMZCross-linked gelatin/nanoparticles composite coating on micro-arc oxidation film for corrosion and drug releaseAppl Surf Sci201025623672371
- YoshizawaHNishinoSShiomoriKNatsugoeSAikoTKitamuraYSurface morphology control of polylactide microspheres enclosing irinotecan hydrochlorideInt J Pharm200529611211615885462
- LeeJWGardellaJJHicksWJHardRBrightFVAnalysis of the initial burst of drug release coupled with polymer surface degradationPharm Res20032014915212636151
- FuKPackDWKlibanovAMLangerRVisual evidence of acidic environment within degrading poly(lactic-co-glycolic acid) (PLGA) microspheresPharm Res20001710010610714616
- LimYWKwonSYSunDHKimHEKimYSEnhanced cell integration to titanium alloy by surface treatment with microarc oxidation: a pilot studyClin Orthop Relat Res20094672251225819434468
- AnnunziataMOlivaABasileMAThe effects of titanium nitride-coating on the topographic and biological features of TPS implant surfacesJ Dent20113972072821856369
- StigterMBezemerJde GrootKLayrollePIncorporation of different antibiotics into carbonated hydroxyapatite coatings on titanium implants, release and antibiotic efficacyJ Control Release20049912713715342186
- InceASchutzeNHendrichCJakobFEulertJLohrJFEffect of polyhexanide and gentamycin on human osteoblasts and endothelial cellsSwiss Med Wkly200713713914517370154
- KozlovskyAArtziZMosesOKamin-BelskyNGreensteinRBInteraction of chlorhexidine with smooth and rough types of titanium surfacesJ Periodontol2006771194120016805682
- QuirynenMVogelsRPeetersWvan SteenbergheDNaertIHaffajeeADynamics of initial subgingival colonization of ‘pristine’ peri-implant pocketsClin Oral Implants Res200617253716441782
- QuirynenMVogelsRAlsaadiGNaertIJacobsRvan SteenbergheDPredisposing conditions for retrograde peri-implantitis, and treatment suggestionsClin Oral Implants Res20051659960816164468