Abstract
As the prevalence of chronic obstructive pulmonary disease (COPD) continues to grow, management of the disease still faces considerable challenges. Despite the existence of effective pharmacological treatments, patient adherence is often poor. Side effects of medications and patients’ concerns about potential side effects may contribute to poor adherence. Situated as they are at the frontline of patient care in the clinic, nurse practitioners play an important role in the management of COPD. This review discusses the current literature on medications available for management of COPD, focusing primarily on their safety and tolerability. This information can be particularly important for nurse practitioners, who can be invaluable in identifying side effects, and providing education to patients with COPD on the available treatments and the associated side effects. By helping patients to understand the balance of benefits and risks of treatment, nurse practitioners may be able to help improve adherence and thereby improve patient outcomes.
Introduction
Chronic obstructive pulmonary disease (COPD) is the third leading cause of death and the fastest growing of the major causes of mortality in the United States.Citation1 More than 12 million adults were diagnosed with COPD in 2007,Citation2 and it has been estimated that a similar number of people with the disease remain undiagnosed.Citation3 Nurse practitioners are ideally placed to play an important role in the early diagnosis and management of COPD. As the prevalence of COPD rises, these patients are likely to make up an increasing proportion of the nurse practitioner’s caseload.
Management of COPD involves both pharmacological and nonpharmacological aspects. Pharmacological management can be complex because COPD is a chronic, progressive condition in patients who often suffer from comorbidities.Citation4 As with many chronic conditions, patients with COPD may require long-term treatment, which may increase the risk of adverse events and raise concerns about adherence to prescribed medication. Chronic disease management increasingly involves a collaborative team approach, with nurse practitioners central to the team, although distribution of tasks between team members varies. The purpose of this review is to discuss the role of nurse practitioners in the management of patients with COPD, with a particular focus on issues concerning the safety and tolerability of pharmacological therapy.
COPD: an overview
The chronic airway limitation characteristic of COPD is due to inflammation that results in obstruction of the small airways and destruction of lung parenchyma.Citation4 Common symptoms include chronic cough, sputum production, and progressive, persistent dyspnea that worsens with exertion. COPD can be prevented and existing COPD can be treated, but the disease is incurable and the airway limitation is not fully reversible.Citation4 As COPD progresses, patients are forced to limit their activities and may experience depression and a decline in their quality of life.Citation5 Patients with COPD also commonly experience comorbid conditions related to aging and/or smoking, including cardiovascular disease, osteoporosis, and depression.Citation4
Smoking is by far the most common risk factor for development of COPD, but other factors such as outdoor, occupational, and indoor air pollution may also cause COPD in the absence of smoking.Citation4 Factors such as genetics, infections, nutrition, and oxidative stress may also have a role in the development and/or progression of COPD. The prevalence of COPD rises sharply with age;Citation6 a meta-analysis of studies from 28 countries estimated the prevalence of COPD in individuals aged ≥ 40 years at 9.7% and in individuals aged ≥ 65 years at 15.0%.Citation7 Furthermore, even if smoking rates were to decline dramatically, the prevalence of COPD may continue to rise over the coming years due to aging of the population and the history of smoking.Citation8 Historically, the prevalence of COPD has been higher among men than women, although recent trends suggest that this gender difference may be declining in regions where smoking rates are similar between the genders.Citation9 Despite the existence of risk factors and development of symptoms, many patients may be unaware of existing COPD. For example, a meta-analysis on the global prevalence of the disease estimated that 9.2% of the population met spirometric criteria for COPD but only 4.9% reported a doctor’s diagnosis of the disease.Citation7
National and international guidelines, including those prepared by the Global Initiative for Chronic Obstructive Lung Disease (GOLD),Citation4 American Thoracic Society/European Respiratory Society,Citation10 and Canadian Thoracic Society,Citation11 provide recommendations for the diagnosis, management, and treatment of COPD. According to the GOLD guidelines updated in December 2011, spirometry is now required to make a confident diagnosis of COPD, whereas previously it was used to support the diagnosis.Citation4 The fixed ratio of postbronchodilator forced expiratory volume in one second (FEV1) to the forced vital capacity (FVC), or FEV1/FVC, needs to be < 70%Citation4 and the staging system of spirometric classification, which has now been replaced with grading, uses: FEV1 ≥ 80% predicted (GOLD 1, mild); 50% ≤ FEV1 < 80% predicted (GOLD 2, moderate); 30% ≤ FEV1 < 50% predicted (GOLD 3, severe); and FEV1 < 30% predicted (GOLD 4, very severe). In order to assess symptoms in patients, the GOLD guidelines suggest the use of the Modified British Medical Research Council questionnaire or the COPD Assessment Test.Citation4 Canadian Thoracic Society guidelines also suggest incorporating measures of dyspnea and disability into clinical assessment of disease severity to individualize management decisions.Citation11 It is not always possible to distinguish COPD from chronic asthma in some patients, so it is assumed that the two exist together.Citation4
One of the most important initial steps for management of COPD is to reduce exposure to risk factors, including cigarette smoke and/or occupational dusts, fumes, and gases. Given the leading role of smoking as a cause of COPD, it may not be surprising that smoking cessation is the only intervention shown to reduce the rate of disease progression and related mortality.Citation12 Patient education is a particularly important component of smoking cessation intervention, and education about some aspects of COPD may help patients to cope.Citation4
Pharmacological therapies are useful to manage symptoms, and some are indicated to reduce exacerbations, which become increasingly important goals as the disease progresses. Various classes of drugs may be appropriate based on the patient’s symptoms, risk of exacerbation, and grade of airflow limitation ().Citation4 In general, short-acting bronchodilators are recommended as needed at all stages of the disease, and long-acting bronchodilators may be appropriate as regular maintenance therapy for patients with moderate to severe COPD. The long-acting anticholinergic, tiotropium, was also recently approved for reduction of exacerbations in patients with COPD.
Table 1 Therapy at each grade of chronic obstructive pulmonary disease as recommended by GOLD (Global Initiative for Chronic Obstructive Lung Disease)
An inhaled corticosteroid (ICS) may be appropriate as add-on therapy to long-acting bronchodilators in patients with severe and very severe COPD. For instance, a combination of the long-acting β-adrenergic, salmeterol, and fluticasone, an ICS, is indicated to reduce exacerbations in patients with COPD with a history of exacerbations.Citation4 Systemic corticosteroids are recommended in the treatment of acute exacerbations of COPD, but their long-term use is not recommended due to lack of evidence of benefit and side effects.Citation13
The 3-year TOwards a Revolution in COPD Health (TORCH)Citation14 and 4-year Understanding Potential Long-term Impacts on Function with Tiotropium (UPLIFT)Citation15 trials both included mortality as an endpoint. In TORCH, the difference in mortality between the salmeterol/fluticasone and placebo treatment groups did not reach statistical significance (salmeterol/fluticasone 12.6%, placebo 15.2%; hazard ratio [HR] 0.825; 95% confidence interval [CI]: 0.681–1.002; P = 0.052).Citation14 In UPLIFT, all-cause mortality was significantly lower in the tiotropium group compared with placebo while patients were receiving treatment (up to day 1440, tiotropium 14.4%, placebo 16.3%; HR 0.87; 95% CI: 0.76–0.99) but not significantly different when data from the following 30 days were included (up to day 1470, tiotropium 14.9%, placebo 16.5%; HR 0.89; 95% CI: 0.79–1.02).Citation15,Citation16 In both TORCH and UPLIFT, mean FEV1 was improved.Citation14,Citation15 A post hoc analysis of TORCH suggested that salmeterol/fluticasone reduced the rate of FEV1 decline compared with placebo.Citation17 These treatments and the associated side effects are considered in more detail in the following section.
Safety and tolerability of COPD medications
summarizes the side effects of COPD treatments observed in clinical practice, and outlines alerts for nurse practitioners, derived from cautions described in the product prescribing information. Most side effects experienced with medications for COPD are relatively minor, and are bothersome to the patient rather than actually harmful; however, rare side effects that are potentially very serious may occur, and associations with other side effects have been reported, although a definite link to treatment is not proven in all cases (). Both the experience of unpleasant side effects and a lack of patient knowledge about the disease and treatments can increase the risk of nonadherence.Citation18 The discussion below addresses side effects frequently encountered in clinical practice, either as actual events experienced by patients or as potential concerns expressed by the patient.
Table 2 Most common potential side effects reported with COPD medications
Short-acting bronchodilators
Short-acting bronchodilators include short-acting β2-agonists and the short-acting anticholinergic agent, ipratropium.Citation4
Short-acting β2-agonists
Short-acting β2-agonists, such as albuterol and levalbuterol, are usually available in a pressurized metered-dose inhaler or as a solution for nebulization, and are recommended for as-needed use to relieve symptoms at all stages of COPD. These agents are generally well tolerated, but patients may suffer from occasional troublesome class side effects, such as hypokalemia, hyperglycemia, elevated heart rate, tremors, headache, and palpitation, which are well established, predictable, and dose related ().Citation41,Citation42
Potential concerns about possible cardiovascular side effects and increased mortality associated with β2-agonists (particularly fenoterol) used to treat asthma were raised in the late 1980s. Although further studies and meta-analyses have provided conflicting evidence,Citation43,Citation44 the GOLD guidelines indicate that no association has been found between β2-agonist use and increased mortality in COPD ().Citation4 However, adverse effects may occur more frequently in patients with underlying cardiac comorbidities; therefore, short-acting β2-agonists should be used with care in patients with cardiovascular disease.
Table 3 Conflicting evidence for increased risk of cardiovascular events and mortality with bronchodilators
Ipratropium bromide
Ipratropium is well established as a short-acting bronchodilator in COPD, available in a pressurized metered-dose inhaler or as a solution for nebulization, and indicated for regular use four times daily as maintenance therapy for COPD. As with other anticholinergics, dry mouth is the main side effect of this drug.Citation54 Other side effects include increased intraocular pressure and urinary outflow obstruction (), so care should be taken with ipratropium in patients with glaucoma or prostatic hyperplasia.Citation54–Citation56 Some patients complain of a bitter taste, though this may be considered a difference from other treatments rather than a side effect.Citation55 Some studies have also suggested a possible association between ipratropium and an increase in cardiovascular events and mortality ().Citation12,Citation48,Citation49 Evidence from a meta-analysis by Singh et al that considered ipratropium and tiotropium together suggested a possible association between anticholinergic treatment and increased mortality and cardiovascular events ().Citation50 However, the United States Food and Drug Administration recently concluded that the available evidence demonstrates that tiotropium does not increase the risk of stroke, heart attack, or death.Citation57
Long-acting bronchodilators
Long-acting bronchodilators are recommended as regular maintenance therapy for patients with moderate to severe COPD ().Citation4 They include long-acting β2-agonists (LABAs) and the long-acting anticholinergic agent, tiotropium.
Long-acting β2-agonists
LABAs are available in the United States as dry powder inhalers (salmeterol and formoterol) or as a solution for nebulization (formoterol and arformoterol), taken twice daily for maintenance therapy. The most common adverse events highlighted in the prescribing information for LABAs are similar to those for short-acting β2-agonists ().Citation58–Citation60 Concerns about rare cardiovascular adverse effects and increased mortality have also been raised for LABAs, especially by studies in patients with asthma, but there is conflicting evidence on these side effects in patients with COPD (). The Salmeterol Multicenter Asthma Research Trial found a small but significant increased risk of respiratory/asthma-related mortality compared with placebo in patients with asthmaCitation61 and a meta-analysis of studies of LABAs in patients with COPD and asthma found an increased risk of treatment-related cardiovascular events.Citation43 In contrast, no increased risk compared with placebo was found in two other studies,Citation14,Citation45 a systematic review,Citation46 and a pooled analysisCitation47 that included only patients with COPD. The Food and Drug Administration has now recommended that LABAs should not be used without an asthma controller medication for treatment of patients with asthma,Citation62 but LABAs are still approved as monotherapy for management of COPD.
Tiotropium bromide
Tiotropium bromide is the only long-acting anticholinergic on the market, and is available in a dry powder inhaler. It is taken once daily as a maintenance bronchodilator therapy and is indicated for reduction of exacerbations in COPD.Citation63 Adverse effects associated with tiotropium are similar to those observed with ipratropium, with the most common side effect being dry mouth ().Citation52,Citation63 Again, tiotropium should be used with caution in patients with glaucoma or prostatic hyperplasia.
Although the meta-analysis by Singh et al that considered ipratropium and tiotropium together suggested a possible association between anticholinergic treatment and increased mortality and cardiovascular events (),Citation50 another meta-analysisCitation51 and two pooled analysesCitation27,Citation53 showed no increased risk with tiotropium treatment. Furthermore, results from the UPLIFT study, a large-scale, 4-year, randomized controlled trial, indicated that tiotropium reduced cardiac morbidity.Citation15 In light of these data, the Food and Drug Administration recently concluded that tiotropium does not increase the risk of stroke, heart attack, or death.Citation57
Methylxanthines
Theophylline is the most widely used methylxanthine in the treatment of COPD, but owing to toxicity at higher doses, other bronchodilators are usually preferred.Citation4 Dose-related adverse effects include nausea, vomiting, seizures, and arrhythmias ().Citation64
Corticosteroids
Inhaled corticosteroids
ICS are recommended as add-on therapy in combination with long-acting bronchodilators for patients with severe and very severe COPD ().Citation4 The most commonly prescribed ICS are fluticasoneCitation65 and budesonide.Citation66 They are available as pressurized metered-dose inhalers or dry powder inhalers, and are usually administered twice daily. ICS are not recommended for monotherapy in COPD.
Common side effects of ICS include bruising of the skin,Citation31,Citation32 oral candidiasis, and hoarseness ().Citation67 More serious side effects that may occur in a small number of patients include pneumonia and a reduction in bone mineral density (a precursor to osteoporosis). An increased risk of pneumonia with ICS in patients with COPD has been shown in several randomized trialsCitation14,Citation68–Citation70 and a meta-analysis.Citation71 There is evidence both supportingCitation72–Citation74 and refutingCitation75,Citation76 the effect of ICS on bone mineral density and/or bone fractures (). The results from these studies may be confounded by disease severity,Citation77 but it is still recommended that bone mineral density is assessed prior to prescribing any ICS in COPD and that it is carried out periodically thereafter. There is also a small risk of a dose-dependent increase in cataracts and glaucoma with ICSCitation67 and it is recommended that regular eye examinations be performed on patients receiving these drugs.
Table 4 Conflicting evidence for increased risk of bone mineral density loss with inhaled corticosteroids
Oral corticosteroids
Oral corticosteroids, prednisone and prednisolone, may be administered in patients with very severe COPD and acute exacerbations, but long-term use is not recommended, especially at the high doses generally required, due to potentially harmful adverse events that may contribute to the development of diabetes, hypertension, osteoporosis, and steroid-induced myopathy ().Citation4,Citation13
Phosphodiesterase-4-inhibitors
The phosphodiesterase-4-inhibitor, roflumilast, was approved by the Food and Drug Administration in 2011 to reduce the frequency of exacerbations in patients with severe COPD. Adverse events associated with roflumilast include diarrhea, weight loss, nausea, insomnia, headache, and back pain.Citation78 In two clinical trials comparing the effect of salmeterol or tiotropium in the presence or absence of roflumilast, it was evident that weight loss was greater in patients also experiencing headache and/or gastrointestinal adverse events, indicating a causal relationship between these adverse events.Citation79 As a consequence of the greater potential for weight loss with roflumilast, regular monitoring of weight should be encouraged in patients treated with this drug.
Combination products
Combination products, including ipratropium plus albuterol,Citation80 salmeterol plus fluticasone,Citation81 and formoterol plus budesonide,Citation82 are commonly prescribed as maintenance therapy for patients with COPD. Salmeterol plus fluticasone is indicated to reduce the risk of exacerbations in patients with a history of exacerbations.Citation81 Generally, the side effect profiles of the combination products reflect those of the individual components (),Citation80–Citation82 and no additional safety concerns have been reported.
Role of the nurse practitioner in COPD
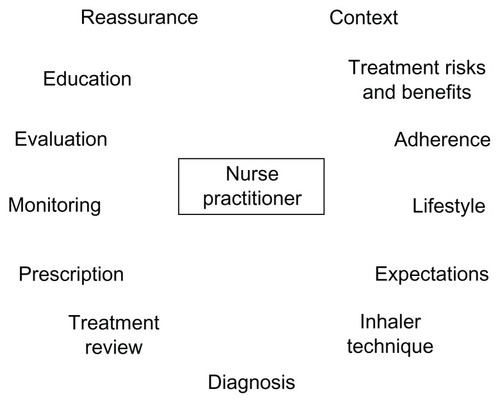
COPD is a chronic disease, often with gradual onset, and is often seen in older patients who may have comorbidities that also require treatment, complicating its diagnosis and management. Limited physician time may lead to inadequacies in the diagnosis and management of COPD.Citation83 Awareness of COPD and comorbid conditions among patients is low,Citation84 and communication between patients and health care providers may be poor.Citation85 Strengthening the role of the nurse practitioner in management of COPD could be an important strategy to improve patient education and communication. The scope of the nurse practitioner’s role within the disease management team in COPD may vary between clinics, but could include diagnosis, prescription, patient monitoring, ongoing evaluation of treatment success, and modification of treatment where required (). In some clinics, some of these roles may be carried out by other staff, while other tasks, such as writing and modifying prescriptions, are restricted to physicians and nurse practitioners.
As with other chronic diseases, adherence to therapy is often poor among patients with COPD.Citation18,Citation86 Patients may fail to adhere to treatment because they have inadequate knowledge of the disease and its treatments,Citation18,Citation86,Citation87 which could potentially result in unrealistic expectations of treatment outcomes. Alternatively, patients may be discouraged by unpleasant side effectsCitation18 or reports of potential side effects. Even if the patient attempts to adhere to therapy, they may underuse, overuse, or improperly use the inhaler devices.Citation18 Furthermore, patients may be prescribed several COPD medications as well as medications for other comorbidities, thus complicating the treatment regimen.Citation18,Citation87 Because COPD is a chronic disease, patients may receive long-term pharmacologic treatment. Therefore, nurse practitioners should be alert to the possibility of patients developing diseases for which COPD medications can be a risk factor, such as osteoporosis, diabetes, or cataracts. Nurse practitioners can play an important role in addressing and resolving these issues.
Communication between clinic staff and patients is critical to improving adherence and outcomes.Citation88 The nurse practitioner is ideally situated to educate the patient on the nature of COPD, the lifestyle changes required, and realistic expectations of potential therapeutic benefits. Nurse practitioners can also provide instruction regarding proper use of devices and help patients to understand better the potential benefits and risks of treatments. The nurse practitioner may offer practical advice on the management of multiple therapies, such as creating a medication administration schedule tailored to suit individual lifestyles. In addition, the nurse practitioner can help to educate the patients about self-monitoring of symptoms and can help to determine whether any dosage adjustments or other changes in treatment are required.
Practical considerations for nurse practitioners
Nurse practitioners play a key role in core teams managing the ongoing treatment of COPD. Handling patient concerns about safety and tolerability is an important aspect of the nurse practitioner’s responsibilities, and may help to improve adherence to treatment. Therefore, it is important that the nurse practitioner establishes an ongoing dialog with patients, so as to understand their issues and concerns, and to provide practical education about their illness and its management and treatment.
Nurse practitioners should take a proactive approach to addressing safety and tolerability issues with patients when they are first prescribed a particular medication. Patients should leave the clinic with a good understanding of what side effects may occur, how serious the side effects may be, and what action they should take if a particular side effect occurs. The nurse practitioner should ascertain whether the patient already has any concerns that may prevent him or her from filling the prescription, and try to address them.
When a patient expresses a concern, nurse practitioners must recognize the need for an individual approach to that patient. They need to assess whether the concern stems from a side effect that the patient has actually experienced or a hypothetical concern based on something they have read or heard about. Some patients may be particularly bothered by minor side effects, and adherence is more likely to be a problem in these patients. Some side effects may present a more serious safety concern for certain patients (eg, those with existing cardiovascular disease). The nurse practitioner must assess the best approach, such as continuing with treatment and providing reassurance, continuing with treatment and taking steps to avoid or minimize the side effect, considering other medications or treatment options, or considering whether referral to a specialist is required. Patient concerns should be reassessed regularly. Importantly, the nurse practitioner can help patients to put the side effects they experience or fear into context, helping them to understand that appropriate treatment may be expected to reduce symptoms and exacerbations, and improve their quality of life, whereas they risk continued symptoms and possibly COPD exacerbations in the absence of adherence to treatment.
Conclusion
Despite the growing prevalence of COPD, a lack of knowledge and/or concerns about the side effects of medications can contribute to poor adherence with treatment in these patients. Nurse practitioners can play an important role in the management of the disease by providing patients with education and helping to monitor and individualize their treatments. In particular, nurse practitioners can help to address patients’ concerns by providing them with information about the possible occurrence of side effects, advising them on appropriate action, and ensuring that they understand the balance between the potential benefits of treatment and the potential risks of side effects.
Disclosure
This article was developed on the basis of presentations and discussions at the Long-term Considerations in the Course and Treatment of COPD Taskforce meeting in Miami, FL, United States December 9–10, 2008, which was supported by Boehringer Ingelheim Pharmaceuticals Inc, and Pfizer Inc. Medical writing assistance, supported financially by Boehringer Ingelheim and Pfizer, was provided by Andrew Cooper of Envision Scientific Solutions, during the preparation of this review. Boehringer Ingelheim and Pfizer were given the opportunity to check the data used in the manuscript for factual accuracy only. PS has received editorial support funded by Boehringer Ingelheim and Pfizer. NH has received research grant support from GlaxoSmithKline, Boehringer Ingelheim, Novartis, Sepracor, AstraZeneca, and Dey Inc. He is on the speakers’ bureaus of GlaxoSmithKline, Boehringer Ingelheim and AstraZeneca, and is a consultant to GlaxoSmithKline, Novartis, Boehringer Ingelheim, Sepracor, and Dey Inc.
References
- JemalAWardEHaoYThunMTrends in the leading causes of death in the United States, 1970–2002JAMA2005294101255125916160134
- National Institutes of HealthMorbidity and mortality: 2009 chart book on cardiovascular, lung, and blood diseases Available from: http://www.nhlbi.nih.gov/resources/docs/2009_ChartBook.pdfAccessed July 5, 2012
- ManninoDMHomaDMAkinbamiLJFordESReddSCChronic obstructive pulmonary disease surveillance – United States, 1971–2000MMWR Surveill Summ2002516116
- Global Initiative for Chronic Obstructive Lung DiseaseGlobal strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease Updated Dec 2011. Available from: http://www.goldcopd.orgAccessed July 5, 2012
- BelferMHReardonJZImproving exercise tolerance and quality of life in patients with chronic obstructive pulmonary diseaseJ Am Osteopath Assoc2009109526827819451260
- LopezADShibuyaKRaoCChronic obstructive pulmonary disease: current burden and future projectionsEur Respir J200627239741216452599
- HalbertRJNatoliJLGanoABadamgaravEBuistASManninoDMGlobal burden of COPD: systematic review and meta-analysisEur Respir J200628352353216611654
- FeenstraTLvan GenugtenMLHoogenveenRTWoutersEFRutten-van MölkenMPThe impact of aging and smoking on the future burden of chronic obstructive pulmonary disease: a model analysis in The NetherlandsAm J Respir Crit Care Med2001164459059611520721
- ManninoDMBuistASGlobal burden of COPD: risk factors, prevalence, and future trendsLancet2007370958976577317765526
- CelliBRMacNeeWAgustiAStandards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paperEur Respir J200423693294615219010
- O’DonnellDEHernandezPKaplanACanadian Thoracic Society recommendations for management of chronic obstructive pulmonary disease – 2008 update – highlights for primary careCan Respir J200815 Suppl A1A8A
- AnthonisenNRConnettJEEnrightPLManfredaJHospitalizations and mortality in the Lung Health StudyAm J Respir Crit Care Med2002166333333912153966
- WaltersJAWaltersEHWood-BakerROral corticosteroids for stable chronic obstructive pulmonary diseaseCochrane Database Syst Rev20053CD00537416034972
- CalverleyPMAndersonJACelliBSalmeterol and fluticasone propionate and survival in chronic obstructive pulmonary diseaseN Engl J Med2007356877578917314337
- TashkinDPCelliBSennSA 4-year trial of tiotropium in chronic obstructive pulmonary diseaseN Engl J Med2008359151543155418836213
- CelliBDecramerMKestenSLiuDMehraSTashkinDPMortality in the 4-year trial of tiotropium (UPLIFT) in patients with chronic obstructive pulmonary diseaseAm J Respir Crit Care Med20091801094895519729663
- CelliBRThomasNEAndersonJAEffect of pharmacotherapy on rate of decline of lung function in chronic obstructive pulmonary disease: results from the TORCH studyAm J Respir Crit Care Med2008178433233818511702
- RestrepoRDAlvarezMTWittnebelLDMedication adherence issues in patients treated for COPDInt J Chron Obstruct Pulmon Dis20083337138418990964
- DahlRGreefhorstLANowakDInhaled formoterol dry powder versus ipratropium bromide in chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2001164577878411549532
- HananiaNADarkenPHorstmanDThe efficacy and safety of fluticasone propionate (250 microg)/salmeterol (50 microg) combined in the Diskus inhaler for the treatment of COPDChest2003124383484312970006
- BoydGMoriceAHPounsfordJCSiebertMPeslisNCrawfordCAn evaluation of salmeterol in the treatment of chronic obstructive pulmonary disease (COPD)Eur Respir J19971048158219150318
- DonohueJFParseyMVAndrewsCEvaluation of the efficacy and safety of levalbuterol in subjects with COPDCOPD20063312513217240614
- RennardSIAndersonWZuWallackRUse of a long-acting inhaled beta2-adrenergic agonist, salmeterol xinafoate, in patients with chronic obstructive pulmonary diseaseAm J Respir Crit Care Med200116351087109211316640
- BaumgartnerRAHananiaNACalhounWJSahnSASciarappaKHanrahanJPNebulized arformoterol in patients with COPD: a 12-week, multicenter, randomized, double-blind, double-dummy, placebo- and active-controlled trialClin Ther200729226127817472819
- HanrahanJPHananiaNACalhounWJSahnSASciarappaKBaumgartnerRAEffect of nebulized arformoterol on airway function in COPD: results from two randomized trialsCOPD200851253418259972
- CasaburiRMahlerDAJonesPWA long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary diseaseEur Respir J200219221722411866001
- KestenSJaraMWentworthCLanesSPooled clinical trial analysis of tiotropium safetyChest200613061695170317166984
- TaylorJKotchARiceKIpratropium bromide hydrofluoroalkane inhalation aerosol is safe and effective in patients with COPDChest200112041253126111591569
- RossiAKristufekPLevineBEComparison of the efficacy, tolerability, and safety of formoterol dry powder and oral, slow-release theophylline in the treatment of COPDChest200212141058106911948033
- OhtaKFukuchiYGrouseLA prospective clinical study of theophylline safety in 3810 elderly with asthma or COPDRespir Med200498101016102415481279
- BurgePSCalverleyPMJonesPWSpencerSAndersonJAMaslenTKRandomised, double blind, placebo controlled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trialBMJ200032072451297130310807619
- PauwelsRALöfdahlCGLaitinenLALong-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European Respiratory Society Study on Chronic Obstructive Pulmonary DiseaseN Engl J Med1999340251948195310379018
- RennardSITashkinDPMcElhattanJEfficacy and tolerability of budesonide/formoterol in one hydrofluoroalkane pressurized metered-dose inhaler in patients with chronic obstructive pulmonary disease: results from a 1-year randomized controlled clinical trialDrugs200969554956519368417
- TashkinDPRennardSIMartinPEfficacy and safety of budesonide and formoterol in one pressurized metered-dose inhaler in patients with moderate to very severe chronic obstructive pulmonary disease: results of a 6-month randomized clinical trialDrugs200868141975200018778120
- AaronSDVandemheenKLHebertPOutpatient oral prednisone after emergency treatment of chronic obstructive pulmonary diseaseN Engl J Med2003348262618262512826636
- AliaIde la CalMAEstebanAEfficacy of corticosteroid therapy in patients with an acute exacerbation of chronic obstructive pulmonary disease receiving ventilatory supportArch Intern Med2011171211939194622123804
- DaviesLAngusRMCalverleyPMOral corticosteroids in patients admitted to hospital with exacerbations of chronic obstructive pulmonary disease: a prospective randomised controlled trialLancet1999354917745646010465169
- MaltaisFOstinelliJBourbeauJComparison of nebulized budesonide and oral prednisolone with placebo in the treatment of acute exacerbations of chronic obstructive pulmonary disease: a randomized controlled trialAm J Respir Crit Care Med2002165569870311874817
- NiewoehnerDEErblandMLDeupreeRHEffect of systemic glucocorticoids on exacerbations of chronic obstructive pulmonary disease. Department of Veterans Affairs Cooperative Study GroupN Engl J Med1999340251941194710379017
- WaltersJAGibsonPGWood-BakerRHannayMWaltersEHSystemic corticosteroids for acute exacerbations of chronic obstructive pulmonary diseaseCochrane Database Syst Rev20091CD00128819160195
- AbramsonMJWaltersJWaltersEHAdverse effects of beta-agonists: are they clinically relevant?Am J Respir Med20032428729714719995
- KellyHWRisk versus benefit considerations for the β2-agonistsPharmacotherapy2006269 Pt 2164S174S16945063
- SalpeterSROrmistonTMSalpeterEECardiovascular effects of β-agonists in patients with asthma and COPD: a meta-analysisChest200412562309232115189956
- SuissaSAssimesTErnstPInhaled short acting β agonist use in COPD and the risk of acute myocardial infarctionThorax2003581434612511719
- CampbellSCCrinerGJLevineBECardiac safety of formoterol 12 μg twice daily in patients with chronic obstructive pulmonary diseasePulm Pharmacol Ther200720557157916911869
- RodrigoGJNanniniLJRodríguez-RoisinRSafety of long-acting β-agonists in stable COPD: a systematic reviewChest200813351079108718460518
- FergusonGTFunck-BrentanoCFischerTDarkenPReisnerCCardiovascular safety of salmeterol in COPDChest200312361817182412796155
- OgaleSSLeeTAAuDHBoudreauDMSullivanSDCardiovascular events associated with ipratropium bromide in COPDChest20101371131919363211
- LeeTAPickardASAuDHBartleBWeissKBRisk for death associated with medications for recently diagnosed chronic obstructive pulmonary diseaseAnn Intern Med2008149638039018794557
- SinghSLokeYKFurbergCDInhaled anticholinergics and risk of major adverse cardiovascular events in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysisJAMA2008300121439145018812535
- BarrRGBourbeauJCamargoCARamFSTiotropium for stable chronic obstructive pulmonary disease: a meta-analysisThorax2006611085486216844726
- ObaYZazaTThameemDMSafety, tolerability and risk benefit analysis of tiotropium in COPDInt J Chron Obstruct Pulmon Dis20083457558419281075
- CelliBDecramerMLeimerIVogelUKestenSTashkinDPCardiovascular safety of tiotropium in patients with COPDChest20101371203019592475
- Atrovent® HFA [package insert]102011 Available from: http://bidocs.boehringer-ingelheim.com/BIWebAccess/ViewServlet.ser?docBase=renetnt&folderPath=/Prescribing+Information/PIs/Atrovent+HFA/10003001_US_1.pdfAccessed July 5, 2012
- GrossNJAnticholinergic agents in asthma and COPDEur J Pharmacol20065331–3363916488410
- ScullionJEThe development of anticholinergics in the management of COPDInt J Chron Obstruct Pulmon Dis200721334018044064
- MicheleTMPinheiroSIyasuSThe safety of tiotropium – the FDA’s conclusionsN Engl J Med2010363121097109920843240
- Brovana® [package insert]22012 Available from: http://www.brovana.com/BrovanaApprovedLabelingText.pdfAccessed July 5, 2012
- Foradil® Aerolizer® [package insert]32012 Available from: http://www.spfiles.com/piforadil.pdfAccessed July 5, 2012
- Serevent® Diskus® [package insert]12012 Available from: http://us.gsk.com/products/assets/us_serevent_diskus.pdfAccessed July 5, 2012
- NelsonHSWeissSTBleeckerERYanceySWDorinskyPMThe Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterolChest20061291152616424409
- ChowdhuryBADal PanGThe FDA and safe use of long-acting beta-agonists in the treatment of asthmaN Engl J Med2010362131169117120181964
- Spiriva® HandiHaler® [package insert]32012 Available from: http://bidocs.boehringer-ingelheim.com/BIWebAccess/ViewServlet.ser?docBase=renetnt&folderPath=/Prescribing+Information/PIs/Spiriva/Spiriva.pdfAccessed July 5, 2012
- MedlinePlusTheophylline82010 Available from: http://www.nlm.nih.gov/medlineplus/druginfo/meds/a681006.htmlAccessed July 5, 2012
- Flovent-HFA® [package insert]12012 Available from: http://us.gsk.com/products/assets/us_flovent_hfa.pdfAccessed July 5, 2012
- Pulmicort Flexhaler® [package insert]72010 Available from: http://www1.astrazeneca-us.com/pi/pulmicortfh.pdfAccessed July 5, 2012
- GartlehnerGHansenRACarsonSSLohrKNEfficacy and safety of inhaled corticosteroids in patients with COPD: a systematic review and meta-analysis of health outcomesAnn Fam Med20064325326216735528
- CrimCCalverleyPMAndersonJAPneumonia risk in COPD patients receiving inhaled corticosteroids alone or in combination: TORCH study resultsEur Respir J200934364164719443528
- KardosPWenckerMGlaabTVogelmeierCImpact of salmeterol/fluticasone propionate versus salmeterol on exacerbations in severe chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2007175214414917053207
- WedzichaJACalverleyPMSeemungalTAHaganGAnsariZStockleyRAThe prevention of chronic obstructive pulmonary disease exacerbations by salmeterol/fluticasone propionate or tiotropium bromideAm J Respir Crit Care Med20081771192617916806
- DrummondMBDasenbrookECPitzMWMurphyDJFanEInhaled corticosteroids in patients with stable chronic obstructive pulmonary disease: a systematic review and meta-analysisJAMA2008300202407241619033591
- Lung Health Research GroupEffect of inhaled triamcinolone on the decline in pulmonary function in chronic obstructive pulmonary diseaseN Engl J Med2000343261902190911136260
- HubbardRBSmithCJSmeethLHarrisonTWTattersfieldAEInhaled corticosteroids and hip fracture: a population-based case-control studyAm J Respir Crit Care Med200216612 Pt 11563156612406825
- LeeTAWeissKBFracture risk associated with inhaled corticosteroid use in chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2004169785585914711795
- JohnellOPauwelsRLöfdahlCGBone mineral density in patients with chronic obstructive pulmonary disease treated with budesonide TurbuhalerEur Respir J20021961058106312108857
- FergusonGTCalverleyPMAndersonJAPrevalence and progression of osteoporosis in patients with COPD: results from the TOwards a Revolution in COPD Health studyChest200913661456146519581353
- LanghammerAForsmoSSyversenULong-term therapy in COPD: any evidence of adverse effect on bone?Int J Chron Obstruct Pulmon Dis2009436538019888355
- CalverleyPMRabeKFGoehringUMKristiansenSFabbriLMMartinezFJRoflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trialsLancet2009374969168569419716960
- FabbriLMCalverleyPMIzquierdo-AlonsoJLRoflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with longacting bronchodilators: two randomised clinical trialsLancet2009374969169570319716961
- Combivent® [package insert]92011 Available from: http://bidocs.boehringer-ingelheim.com/BIWebAccess/ViewServlet.ser?docBase=renetnt&folderPath=/Prescribing+Information/PIs/Combivent+IA/combivent.pdfAccessed July 5, 2012
- Advair® Diskus® [package insert]92011 Available from: http://us.gsk.com/products/assets/us_advair.pdfAccessed July 5, 2012
- Symbicort® [package insert]62010 Available from: http://www1.astrazeneca-us.com/pi/symbicort.pdfAccessed July 5, 2012
- MoorePLPractice management and chronic obstructive pulmonary disease in primary careAm J Med20071208 Suppl 1S23S2717678940
- BarrRGCelliBRManninoDMComorbidities, patient knowledge, and disease management in a national sample of patients with COPDAm J Med2009122434835519332230
- NelsonMHamiltonHEImproving in-office discussion of chronic obstructive pulmonary disease: results and recommendations from an in-office linguistic study in chronic obstructive pulmonary diseaseAm J Med20071208 Suppl 1S28S3217678941
- CramerJABradley-KennedyCScaleraATreatment persistence and compliance with medications for chronic obstructive pulmonary diseaseCan Respir J2007141252917315055
- LaforestLDenisFVan GanseECorrelates of adherence to respiratory drugs in COPD patientsPrim Care Respir J201019214815420094689
- BourbeauJBartlettSJPatient adherence in COPDThorax200863983183818728206