Abstract
Background
Chemotherapy-induced mucositis is inflammation that develops in the oral mucosal due to anticancer treatment. Mucositis has negative consequences that may lead to distress in pediatric patients, resulting in escalated expenses, diminished quality of life, hindrance in cancer therapy, and decreased survival rates. However, despite the numerous methods, oral care protocols are suggested for implementation in the pediatric population despite a lack of high-level evidence studies, particularly regarding which appropriate oral care agents should be administered.
Purpose
This systematic review aimed to identify the effect of oral care intervention in mucositis management among pediatric cancer patients.
Methods
Studies were published between 2014 and 2023 from five databases: PubMed, Embase, Medline, ScienceDirect, and Scopus. They were identified using a search strategy to identify relevant studies that identify oral care interventions for managing mucositis in children with cancer. This study used the Joanna Briggs Institute (JBI) critical appraisal tools to assess the quality of the studies and followed the recommended reporting items for systematic reviews and meta-analyses (PRISMA) guidelines.
Results
Eight studies met the inclusion criteria, including seven RCTs and one quasi-experimental study. Oral care interventions involving tooth brushing, mouthwash, and lip care are performed entirely or partially. The frequency of oral care ranges from two to six times daily, and the duration of intervention is from 5 days to six weeks. Oral care interventions using honey, olive oil, Aloe Vera, Andiroba, and salivary enzyme toothpaste are beneficial to lower the severity of mucositis, reduce pain, minimize mucositis duration, and reduce the use of analgesics, but not significantly improve the child’s quality of life. However, Caphosol mouthwash did not significantly reduce mucositis.
Conclusion
Our study highlights that oral care intervention using effective agents integratively, including honey, olive oil, Aloe vera, Andiroba, and salivary enzyme toothpaste, is essential to manage chemotherapy-induced mucositis among children.
Systematic Review Registration
PROSPERO registration number was CRD42023456278.
Introduction
Cancer is a lethal chronic disease that endangers the health of both adults and children worldwide. World Health Organization estimates that the cancer prevalence in children is around 4%.Citation1 Cancer affects up to 80% of children in developing or low-income countries, and it is estimated that 90,000 children and adolescents die from it each year.Citation2 World Health Organization emphasizes the need to improve effective treatment policies for all children with cancer to increase survival rates, particularly in developing countries.Citation1,Citation3
Children’s cancer treatment is intensive to control the number of cancer cells. Surgery, chemotherapy, and radiotherapy are the three main treatments used. Chemotherapy is the most commonly used treatment for children with cancer because it is highly effective.Citation4 Chemotherapy is performed by administering cytotoxic drugs, which destroy and inhibit the growth of rapidly dividing cells that cannot distinguish between cancerous and normal cells. Chemotherapy is beneficial for systemic cancers that cannot be treated with surgery or radiation. Multiagent chemotherapy has many successes and saves many children from cancer.Citation5
Aside from the numerous benefits of chemotherapy treatment for childhood cancer, there are countless side effects. According to Harris et al, the most common chemotherapy side effects experienced by cancer patients (69.6%) are oral and gastrointestinal. Other chemotherapy-related side effects include fatigue, bone marrow depression, hair loss, and skin problems.Citation3,Citation6 The most common side effect of chemotherapy is oral mucositis (OM), which affects approximately 75% of high-risk patients and 52–80% of children.Citation7 Mucositis symptoms range from mild to severe and may necessitate hospitalization. These symptoms include pain, erythema, edema, ulceration, bleeding, dry mouth, burning sensation, difficulty swallowing and speaking, and can impact all aspects of a child’s life.Citation8
Mucositis-related ulceration and pain can make eating and drinking difficult due to impaired chewing, swallowing, tasting food, and bad breath.Citation9 It can lead to decreased appetite and limited food and drink intake, putting children at risk of dehydration, weight loss, and nutritional changes.Citation10 Severe dietary changes can increase a child’s enteral and parenteral nutrition needs.Citation11 Significant dietary changes have several unintended consequences, including worsening clinical conditions and increasing the risk of infection. It exacerbates the child’s condition and can result in reduced chemotherapy doses, schedule delays, or even treatment discontinuation.Citation12 As a result, the effectiveness of chemotherapy becomes suboptimal, disrupting the remission period and reducing recovery and child survival,Citation8,Citation11,Citation12 thereby increasing the cost and ultimately reducing the quality of life of children.Citation13 These issues can impede cancer treatment and lower survival rates, particularly in immunocompromised children.Citation14
Although numerous chemotherapy-induced mucositis treatment options exist, none can entirely prevent or adequately treat mucositis.Citation13 Elad et al recommend oral care, particularly toothbrushing and gargling, as the primary intervention in treating chemotherapy-induced mucositis. However, the findings of several studies indicate that more research is required to document the expected benefits and risks of various treatment methods. Friend’s systematic reviews suggest oral care is the best mucositis treatment. The oral care intervention includes several Methods: brushing, flossing, gargling, and lip care. Various studies related to oral care interventions use varying methods and show varying results. Apart from the various techniques, the agents used in oral care interventions also vary. Various studies are still being debated regarding using oral care agents to prevent and treat mucositis in the pediatric population.Citation15
The Multinational Association of Supportive Care in Cancer and the International Society of Oral Oncology (MASCC/ISOO) state that implementing basic oral care protocols is recommended in the pediatric population amidst a lack of high-level evidence studies.Citation16 MASCC/ISOO also suggests identifying how effective oral care interventions are in the pediatric population with malignancies. Oral care carried out in managing mucositis in children varies greatly.
Several studies show a variety of oral care interventions to manage mucositis in children, such as teeth brushing only,Citation17 tooth brushing with flossing,Citation18 mouth washing only,Citation19 tooth brushing and mouth washing,Citation12,Citation20 or tooth brushing – mouth washing and lip care.Citation21 The frequency of oral care in cancer children varies according to the literature. According to the earlier study, oral care can be performed 2–3 times daily, every 4–6 hours, or every 2–4 hours.Citation22 Thus, MASCC/ISOO encourages additional research to identify oral care intervention in managing mucositis in children, strengthening the current evidence.Citation16
Information regarding agents used in oral care interventions in the pediatric population is also important, considering that various agents are used with varying effectiveness in preventing and treating mucositis in children. Chlorhexidine is a widely and frequently used agent in oral care interventions as a mouthwash. However, some studies advise against using this agent.Citation23 Other agents, such as povidone-iodine, have limited scientific evidence for preventing mucositis. According to Brown and Gupta, most patients notice an unpleasant taste when using these substances in oral care.Citation24 These substances should not be given over a long period because they may alter the normal bacterial flora in the oral cavity and lead to hyperthyroidism.Citation24 The agents chlorhexidine (0.12–0.2%) and benzydamine (0.15%), according to the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO), have been linked to stinging or burning sensations.Citation25
According to the MASCC/ISOO clinical guidelines, using agents such as honey in oral care interventions has a level of evidence suggested for preventing mucositis in general population patients,Citation25 and special pediatric populations.Citation16 The MASCC/ISOO panel also believes that honey has promising potential in pediatric patients, but more study is needed to strengthen this evidence.Citation16 The MASCC/ISOO pediatric sub-analysis panel cited two studies on honey. The first was a placebo-controlled RCT study that used topical honey to treat mucositis in 90 pediatric patients with hematological malignancies undergoing chemotherapy. The study found that Trifolium alexandrinum-based honey effectively reduced the healing time of ulcerative mucositis. (p=0.0005).Citation26 However, MASCC/ISOO stated the need for further research to identify the effect of honey on mucositis because MASCC also reported a single RCT reporting the ineffectiveness of topical propolis for the treatment of oral mucositis in pediatric patients (n=40) for hematological and solid cancers.Citation27
Several other studies have identified the use of various substances in managing mucositis, including palifermin administration,Citation28 photobiomodulation,Citation19 chewing gum, and ketamine. However, in this systematic review study, the focus is more on identifying what kind of oral care interventions are effective in managing mucositis in children, including identifying the agents used in oral care as embedded, that involved the practice of tooth brushing, mouth washing, or lip care.
It is very important to support and strengthen the evidence regarding the implementation of recommended oral care protocols in the pediatric population, as is the mandate of MASCC/ISOO to strengthen the evidence regarding effective oral care protocols in the management of mucositis in children in light of the lack of high-level evidence studies.Citation16 This systematic review aims to identify oral care interventions used to manage mucositis in children. In addition, the secondary outcome is to identify the effectiveness of the agents used in oral care to reduce the severity of mucositis in children. This systematic review will benefit the field by providing evidence for the efficacy of oral care intervention using specific agents in managing mucositis in children.
Materials and Methods
Study Design
The study design used in this research is a systematic review. The systematic review seeks to inform and assist this process and enable efficient access to evidence for scale-up by synthesizing research from several studies. The framework comprises six fundamental phases, which are as follows: formulating research questions, examining existing literature, filtering studies to identify pertinent ones, evaluating the caliber of investigations, determining each study’s outcome measures, and compiling and presenting findings.Citation29 This systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.Citation30 The study protocol was registered on the PROSPERO database (CRD42023456278).
Search Strategy
From July 30, 2023, to December 30, 2023, two independent reviewers examined research articles published between 2014 and 2023 from five databases: PubMed, Embase, Medline, ScienceDirect, and Scopus. This study used a systematic search strategy based on research questions aligned with medical subject headings, phrases, and subject synonym combinations. Boolean operators (“AND” and “OR”) were applied to each database. The following keywords and Boolean operators were used: (“children OR pediatric”) AND (“cancer OR leukemia OR malignancy”) AND (“oral care OR oral hygiene”) AND (“mucositis score”) AND (“experimental OR randomized trial”).
Inclusion and Exclusion Criteria
The articles were chosen for review based on inclusion and exclusion criteria.
Inclusion Criteria
- Population: studies in which the participants are children with cancer up to 18 years old who are undergoing chemotherapy.
- Intervention: oral care intervention using a specific agent. The researchers use the following definition of oral care: Oral care, or basic oral care, refers to all routine activities performed by patients, caregivers, or healthcare providers to manage mucositis in children with cancer who are undergoing chemotherapy. Oral care procedures involve tooth brushing, flossing, mouth washing, lip care, or topical application to the entire surface of the oral mucosa.Citation25
- Comparison: control group or standard care.
- Outcome: score or severity of mucositis and other secondary outcomes.
- Study type: experimental studies.
- The article’s publication year ranges from 2014 through 2023.
Exclusion Criteria
- Studies on young adults and adult cancer patient.
- Studies for children with other chronic health problems outside cancer.
- Descriptive, cross-sectional, case-control, cohort studies, qualitative studies, feasibility studies, study protocols, conference proceedings, thesis/dissertation, or abstracts.
- Studies in languages other than English.
- Articles with no full text.
Study Selection and Data Extraction
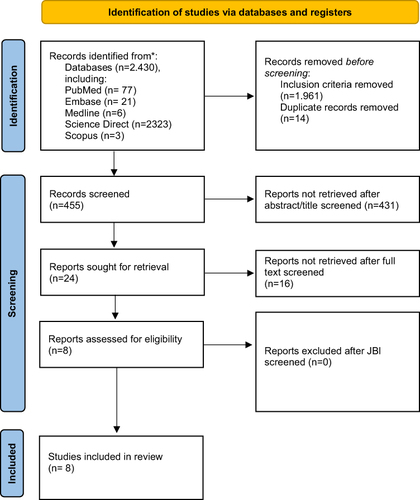
Two reviewers (DG and IN) independently selected the articles from five databases. All search results were imported into the reference management application for data management. Two reviewers independently examined and cross-checked the remaining articles after eliminating any duplicates. A third reviewer (NN) helped resolve disagreements regarding whether to include an article. After the evaluation, the two researchers reached an agreement. The first researcher (IN) who screened the database evaluated the studies based on the relevance of the study title. The reference manager application was used to record studies whose titles and abstracts met the inclusion criteria, and full texts were obtained. Before being transferred to the data extraction table, two independent researchers (IN and AA) evaluated full texts for inclusion criteria and methodological quality. The reviewers (IN, AA, HSM) then extracted data from each article, including the year, study location, study design, sample size and characteristics, the type of oral care-based intervention used, the specific agent used in the oral care intervention, and the findings. The study followed the PRISMA writing guidelines for systematic reviews and meta-analyses ().
Figure 1 PRISMA Flow Diagram. Adapted from Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated :guideline for reporting systematic reviews. BMJ. 2021;372:n71. Creative Commons.Citation30
Quality Assessment
The methodological quality of the included studies was independently assessed by two reviewers (IN, AA) using the Joanna Briggs Institute (JBI) critical appraisal tools for randomized controlled trials and quasi-experiments.Citation31,Citation32 The JBI includes 13 questions for randomized controlled trials and nine for quasi-experimental investigations. Questions have four possible answers: “Yes”, “No”, “Unclear” (if no information about the problem is provided), and “Not applicable (NA)” (if the question is incomplete). Each question was scored as “Yes” (1 point), “No” (0 points), “Unclear” (0 points), or “Not relevant” (0 points). Overall, scores range from 0 to 13 and 0 to 9. If there were a disagreement, NN would be involved in resolving the process using JBI guidelines.Citation31,Citation32
Risk of Bias
The risk of bias for each article in this study is assessed using the cut-off point established by Viswanathan.Citation33 Individual studies using experiment designs had a low risk of bias if 70% of answers were “yes”, a moderate risk if 50–69% of questions were “yes”, and a high risk of bias if “yes” scores were less than 50%. summarizes the appraisal’s conclusion concerning the risk of bias in each included study.
Table 1 The Result of Critical Appraisal for Included Studies
Data Synthesis and Analysis
Two researchers build the data extraction form using the following parameters: (1) author, year, and country; (2) aims; (3) study design; (4) sample characteristics; (5) intervention; (6) outcomes; (7) tools; (8) measurement times; and (9) results of the study (). summarizes the specific oral care intervention carried out in each study, including data extraction: (1) author; (2) study design; (3) cancer type; (4) oral care intervention; (5) oral care agent; (6) doses; (7) sample size; (8) frequency of oral care; (9) length; (10) tools; (11) mucositis score; (12) the effect of the intervention (p-value). The findings are presented as a summary table, followed by a narrative describing the results’ similarities and differences.
Table 2 The Primary Feature of the Systematic Review Included Studies (n=8)
Table 3 Characteristics of Oral Care Intervention
Results
Eight publications explored oral care interventions in the management of mucositis in children with cancer. and highlight the findings of the analysis. Specific characteristics of the implementation of the oral care intervention can be found in . Apart from that, also contains information about the specific agents used in the oral care intervention provided. Based on the findings of this systematic review, the following is an explanation of oral care intervention carried out in managing mucositis symptoms in children with cancer.
Characteristics of Studies
The search yielded 2430 articles. After selection based on inclusion criteria, 455 articles remained: full-text articles in English published between 2014 and 2023, with duplicates removed. Furthermore, 24 articles remained after being eliminated based on title or abstract. The researcher then thoroughly reviewed the full texts of the articles, and eight were ultimately included in this study (). Articles were evaluated using the JBI Critical Appraisal Tool, and good article standards exceeded 60% in terms of criteria and topic relevance.
This review includes studies on West Asia, the Mediterranean, Europe, and South America. depicts two studies from Syria, one from Saudi Arabia, one from Lebanon, one from Turkey, one from Italy, one from the Netherlands, and one from Brazil. Only one study used a quasi-experimental design, while most used an RCT. Two of the studies involved children with cancer in general, while the other six included children with leukemia. This study identified both primary and secondary outcomes. The primary outcome in this study is the degree of mucositis identified using the WHO grading scale, Oral Hygiene Index, and The NCI-CTCAE version 3.0 mucositis scoring system. Meanwhile, secondary outcomes identified include pain scale, length of stay, use of analgesics, and quality of life.
Oral Care Intervention
shows that the oral care intervention involves several different actions. Four studies involved a complete oral care intervention, including toothbrushing, mouth washing, and lip care (topical application).Citation34–36,Citation41 One study involved both toothbrushing and mouth washing,Citation37 one study involved toothbrushing only,Citation38 one study involved mouth washing only,Citation42 and one study carried out with topical application in the mouth area.Citation43 The sample size for each intervention group ranged from 11–32 participants. The frequency with which oral care is carried out varies from 2 times a day,Citation35,Citation39 four times a day,Citation36,Citation37,Citation40,Citation41 and the most frequent is six times a day.Citation34 The length of oral care intervention also varies, mostly being carried out over 5–10 days,Citation34,Citation37–40,Citation36 21 days,Citation41 and the most prolonged intervention was carried out for six weeks.Citation35
Oral Care Agents
shows that the oral care intervention carried out in each study used a specific oral care agent. Three studies used honey as an oral care agent,Citation34,Citation37,Citation41 three studies used olive oil,Citation35–37 one study used toothpaste contains saliva enzymes, essential oils, proteins, and colostrum extract (Bioxtraâ),Citation38 one study used Caphosol mouth rinse,Citation39 one study used Aloe vera,Citation36 and one study used Andiroba.Citation40 This product is used for toothbrushing, mouth washing, and lip care or lubrication in the lip and oral cavity. Based on the findings of this systematic review, the following explanation is provided for the effectiveness of oral care agents on mucositis scores in children with cancer.
Honey
Three of the eight reviewed studies used Honey as an agent in oral care. Several studies have shown that using honey has a positive effect on reducing mucositis scores. Al Jaouni et al found that using local Saudi honey on 20 participants in the intervention group resulted in a 35% reduction in grades III and IV oral mucositis in the treatment group (p=0.02) with an NNT (Number Needed to Treat) of 2. This study also found that oral mucositis patients in the treatment group had a significantly shorter hospital stay than those in the control group. Furthermore, Badr et al used manuka honey as an oral care agent to treat mucositis, applying it to the oral mucosa three times a day by an assigned nurse until healing, which lasted seven days. On day 7, the children who received honey (group 1) had a milder grade of oral mucositis, according to the WHO scale, than the control group (F=5.18, p=0.01).
The other study was conducted by Bulut, who administered natural and standardized flower honey to the experimental group in addition to routine and standard oral care provided in the clinic following each chemotherapy session.Citation41 The study found that children in the honey group experienced a gradual decrease in oral mucositis after the first day of use. There was a significant difference in the degree of OM between follow-up days (p< 0.05). The level of OM in control group children increased gradually on days 4, 8, and 12 but decreased slightly on days 16 and 21. There was a significant difference (p<0.001) between follow-up days and OM grade.
Olive Oil
Three of the nine articles reviewed used olive oil as a treatment for oral mucositis.Citation35–37 Based on these three studies, it can be concluded that using olive oil as a topical agent in oral care effectively lowers mucositis scores in cancer children undergoing chemotherapy.
Andiroba
Carapa guianensis Aubl, commonly known as Andiroba, is a tree belonging to the Meliaceae family. It is large and is generally found in the Amazon region. Andiroba is one of the Amazon region’s most widely used medicinal plants. It has been used by indigenous peoples and local populations for many years in managing upper respiratory tract infections, dermatitis, skin lesions, secondary skin lesions, boils, abrasions, rheumatism, and as a muscle relaxant. Soares et al found that andiroba gel at a concentration of 3% is the only active substance in the gel composition.Citation40 The intervention was performed by administering andiroba oral-based 3% to each patient’s lesion four times daily (every six hours), following oral hygiene procedures. After applying andiroba oral-based, avoid eating or drinking for at least 60 minutes. As a result, andiroba can effectively reduce the severity and symptoms of oral mucositis pain while showing better Results.
Toothpaste
Toothpaste is an oral care ingredient that is used in conjunction with brushing. Special toothpaste containing fluoride, salivary enzymes, essential oils, proteins, and colostrum extract have been proposed to treat oral mucositis, particularly in cases of xerostomia. One study examined two types of toothpaste for oral care in patients with oral mucositis.Citation38 The first toothpaste is Bioxtra Toothpaste, which contains saliva enzymes, essential oils, proteins, and colostrum extract. It’s the second tooth, specifically fluoride toothpaste without menthol. The quality of life and severity of mucositis were evaluated in this study. The findings revealed that using specific toothpastes like Bioxtra in children with grade 1 or 2 oral mucositis had no significant impact on their quality of life, but it significantly impacted their oral hygiene.
Caphosol
Caphosol is a mouthwash that can help prevent or treat mucositis. One study examined the efficacy of Caphosol mouthwash in pediatric patients by measuring the mucositis grade score and the patient’s pain threshold.Citation39 As a result, the number of hospital days with a mucositis score >grade 1 did not differ significantly between the two study groups: the intervention group with Caphosol and the control group with NaCl 0.9%. The Caphosol group had a longer mean duration of mucositis. Despite similar peak pain levels, the Caphosol group had significantly more pain days. Caphosol users took their analgesics for longer. This study shows that Caphosol is ineffective in treating pediatric patients with oral mucositis caused by cancer therapy.
Aloe Vera
Aloe vera is a plant in the Asphodelaceae family that has been used in medicine, cosmetics, and skincare products. The active components of aloe vera gel include enzymes, vitamins, minerals, hormones, and amino acids. Alkhouli et al investigated the efficacy of aloe vera gel in treating mucositis in children with cancer.Citation36 This study used an oral sponge to administer the agents used in the three groups (olive oil, aloe vera, and sodium bicarbonate) four times daily for ten days. All children were instructed not to drink, eat, or gargle for 1.5 hours after receiving the agent. According to the findings of this study, the group that received aloe vera gel saw their mucositis score decrease from grade 3 to grade 2 on the WHO oral mucositis grading scale. This result is inferior to that of olive oil but superior to that of sodium bicarbonate. This study concluded that aloe vera is effective for the management of mucositis.
Primary Outcomes
The primary outcome of oral care-based intervention in the management of mucositis in children is a reduction in the severity of mucositis in children. This decrease in the severity of mucositis is characterized by a reduction in score or grading, a decrease in the degree of pain due to mucositis, and a shorter total duration of mucositis. This study found that oral care interventions containing honey effectively reduced the severity of mucositis,Citation34,Citation41 and the pain associated with mucositis.Citation37 Meanwhile, three studies that used olive oil in oral care procedures showed that olive oil could reduce the severity of mucositis. It was found that children who received the olive oil had less pain than the control group,Citation35,Citation36 although not lower than the honey group.Citation37
The use of Aloe vera in oral care is also beneficial in managing mucositis, as evidenced by findings indicating differences in mucositis grading before and after Aloe vera administration.Citation36 Another oral care agent that statistically shows an effect in reducing the severity of mucositis is fluoride toothpaste (Bioxtraâ), which is effective in improving the oral hygiene grade,Citation38 and Andiroba gel, which can significantly reduce the degree of mucositis and pain caused by mucositis.Citation40 This study also found that the total duration of mucositis, number of days in pain, and use of analgesics were significantly higher in the Caphosol group.Citation39 Caphosol rinse is not recommended for the management of mucositis in children.
Secondary Outcomes
This study identified several secondary outcomes from oral care interventions using specific agents in the management of mucositis in children. The secondary outcomes are the child’s quality of life and the use of analgesics. Bioxtraâ toothpaste may be recommended as clinically effective in improving the oral hygiene grade, but it does not affect the QoL of children undergoing chemotherapy.Citation38 Meanwhile, the use of Caphosol rinse as a mouth washing agent in oral care has not been proven to reduce the degree of mucositis, thereby increasing the number of days in pain and analgesics use.Citation39
Discussion
Oral mucositis is a painful consequence of chemotherapy as a cancer treatment. This disease progresses from redness of the oral mucosa to intractable ulceration that causes pain, discomfort, and the inability to eat or drink.Citation44 Among the oral complications of chemotherapy, oral mucositis (OM) is the most debilitating side effect. Oral mucositis associated with immunosuppression can cause serious and potentially life-threatening consequences that interfere with treatment and the patient’s quality of life.Citation38
Oral mucositis can significantly reduce a patient’s quality of life, emphasizing the importance of managing oral health. Mucositis management consists primarily of pain management with topical and oral analgesics/anesthesia and anti-inflammatory agents, systemic use of antifungal drugs, and, most importantly, oral care (daily oral care).Citation45 Basic oral care refers to all routine actions to reduce bacterial load in the oral cavity, prevent infection, and provide comfort. Oral care practices include mechanical cleaning (brushing and flossing), mouthwash, and lip care.Citation25
Our study found that an oral care intervention program carried out in the management of mucositis has several variations. Several studies have completed basic oral care, including mechanical cleaning (brushing), mouth washing, and lip care. However, most of the oral care programs in the included study were incomplete, and they only included brushing teeth alone, mouth washing alone, topical application, or a combination of the three. In fact, MASCC/ISOO recommends that children’s mucositis be managed with complete basic oral care, which includes brushing teeth, gargling, and lip care. This aims to promote optimal oral health to prevent or alleviate mucositis symptoms. The finding of this study shows that oral care intervention is beneficial for the primary outcome, specifically the degree of mucositis, and is beneficial for secondary outcomes, such as pain score, duration of mucositis, and use of analgesics. This cannot be separated from the frequency, duration, and comprehensiveness of the oral care procedure interventions and the effects of the specific agents used in the oral care procedures.
An important finding from this systematic review is that the interventions provided varied in terms of the time and duration of the intervention. This systematic review revealed an important finding: the interventions varied in time and duration. The frequency of oral care varies from two to six times per day. The study highlights that oral care is performed with only one action (in this case, mouthwash) and at a low frequency (twice per day) despite the specific agent Caphosol not affecting or decreasing the severity of mucositis.Citation39
The main finding of this study concerns the efficacy of agents used in oral care interventions. This study discovered that the following oral care agents are effective in reducing the severity of mucositis in children: honey, olive oil, aloe vera, Andiroba gel, and Fluoride toothpaste with salivary enzymes, essential oils, proteins, and colostrum extract (Biodextra). Meanwhile, Caphosol did not significantly reduce the severity of mucositis.
Another notable finding is that oral care interventions using specific, appropriate agents, such as honey, olive oil, aloe vera, andiroba gel, and salivary enzyme-containing toothpaste, not only improve the main outcome, namely the severity of mucositis but also improve the outcomes. Others include decreased pain intensity, analgesic use, mucositis duration, and the number of days of pain caused by mucositis.
Honey has long been known to have calming properties and is recommended for treating oral mucositis. Honey is a by-product of floral nectar. Honey has excellent antimicrobial properties, low pH, and high osmolarity. The enzyme oxidase converts glucose into non-cytotoxic hydrogen peroxide at high concentrations. Honey lowers prostaglandin levels in lesions, increases nitric oxide concentrations, and has anti-inflammatory and antioxidant properties.Citation46 Studies on using honey in the management of mucositis have yielded positive results. In one study, patients with head or neck cancer were instructed to gargle with 20 mL of honey three times a day and then swallow it. The study concluded that this application relieved oral mucositis pain.Citation47 Other studies have found that honey reduces the severity of oral mucositis.Citation48,Citation49
Three studies have shown that olive oil is effective for treating oral mucositis.Citation35–37 Olive oil reduces the severity of oral mucositis in children and delays its onset. Olive oil’s bioactive components and its role in inhibiting many pathobiological steps of oral mucositis may explain its efficacy in prevention. Olive oil is a natural oil derived from olives that contains monounsaturated fatty acids such as oleic acid, which is the most concentrated, followed by palmitic acid, linoleic acid, and stearic acid. Apart from fatty acids, phenolic compounds account for a significant portion of olive oil’s composition. Monounsaturated fatty acids inhibit inflammation by influencing biomarkers.Citation50 Olive oil can form a layer on the oral mucosa, improving lubrication, reducing microorganism retention, and increasing saliva buffering capacity.Citation51 Ahmad et al found that olive oil leaf extract effectively treated chemotherapy-induced mucositis by decreasing the expression of several pro-inflammatory cytokines.Citation52
Furthermore, this study discovered that andiroba gel is an effective oral care agent for reducing mucositis scores. Carapa guianensis Aubl, also known as andiroba, is a large Meliaceae family tree commonly found in the Amazon region.Citation43 Andiroba is one of the most widely used medicinal plants in the Amazon. Indigenous peoples and residents have used it for many years to treat upper respiratory tract infections, dermatitis, skin lesions, secondary skin lesions, boils, abrasions, rheumatism, and muscle relaxants. Soares et al found that starting treatment with andiroba significantly reduced the severity of oral mucositis on the fourth, fifth, and sixth days, as well as the level of pain on the second, third, and fourth days (p<0.05).Citation40 The andiroba group’s oral mucositis scores were also lower on other days. Furthermore, on the eighth day of follow-up, no patients in the andiroba group reported pain, resulting in an average pain score of zero. Again, on the ninth day of follow-up, no patient in the andiroba group demonstrated symptoms of oral mucositis. Other study reported additional evidence supporting andiroba oil’s therapeutic potential, indicating that its use reduces the severity of oral mucositis and speeds up the healing process.Citation43
Aloe vera is another effective treatment for mucositis. Aloe Vera has numerous beneficial properties. Aloe vera suppresses inflammation by reducing macrophage adhesion, which interferes with the responsible cytokines and aids in wound healing. Topical application of Aloe Vera and olive oil was influential in treating oral mucositis.Citation36 No significant differences were found between Aloe Vera versus olive oil or Aloe Vera versus sodium bicarbonate. However, the findings of this study are consistent with many previous studies, which found that Aloe Vera effectively prevented or treated chemotherapy-induced oral mucositis.Citation53
Some of the agents in this study were used as mouthwash or lip/mouth topical care. However, there is one study that shows that toothpaste contains lysozyme, lactoferrin, lactoperoxidase, colostrum purified stan- dardized extract, sorbitol, xylitol, isoceteth-20, sodium monoflu- orophosphate, hydroxyethylcellulose, sodium benzoate, and titanium dioxide, is beneficial for improving the oral hygiene index in children who experience mucositis.Citation38 The pathogenesis of chemotherapy-induced OM appears to be related to oxidative stress induced by the treatment. The reactive oxygen species (ROS) cause direct and indirect damage through transcription factors, which induce the production of pro-inflammatory cytokines.Citation38 Bio-inspired and biomimetic action in toothpaste containing enzymes is effective for oral health care to avoid disrupting the ecological balance in the oral cavity, particularly in conditions like oral mucositis where the oral mucosa should be protected. Bardellini’s study mentioned that toothpaste containing salivary enzymes was useful for improving the oral hygiene index, but it did not significantly affect quality of life.Citation38 This could be because the oral care intervention using toothpaste only lasted eight days; additionally, the study reported that most patients reported not brushing their teeth for at least two consecutive days due to pain and burning, resulting in poor patient adherence. The lack of impact on the child’s quality of life may be due to attitudes toward oral care interventions.
Strength and Limitation
This systematic review has some limitations. The majority of the studies in this review are from Asia and Eurasia, particularly Turkey, Lebanon, Saudi Arabia, and Syria. Other studies come from Europe, specifically Italy and the Netherlands, as well as South America, specifically Brazil, so it does not include studies that represent several other continents, such as Africa and Australia, so the diversity of respondents’ social and cultural characteristics is limited. This review focuses on oral care interventions and the oral agents that are used within oral care practices. As a result, other studies that used agents administered directly or not integrated into oral care were excluded from this study. This is related to MASCC/ISOO’s recommendation that basic oral care protocols be implemented in the pediatric population despite a lack of high-level evidence studies and that agents or products used to manage mucositis be integrated into oral care practices.
Despite its limitations, this study offers several benefits that should be recognized. The study employed a comprehensive search strategy and a systematic data extraction and quality assessment procedure. Aside from that, the majority of the studies included a homogeneous population, specifically children with leukemia, with the exception of two studies involving general child cancer participants. This study identifies the oral care agents used and the type of oral care program implemented in each included study. Aside from that, the severity of mucositis was assessed in this study, with the majority of participants using the same instrument, the WHO mucositis scale. This study also demonstrates that, in addition to the primary outcome, the severity of mucositis, other outcomes such as duration of mucositis, duration of pain, duration of analgesic use, and quality of life of children were identified as effects of oral care-based intervention. Thus, the study’s findings provide some strong indications of the role of oral care intervention using appropriate specific in managing mucositis in children.
Conclusion
Our study highlights that oral care interventions are beneficial in managing mucositis among cancer children. This study shows that oral care intervention using specific agents, including honey, vitamin E, olive oil, andiroba gel, aloe vera gel, and toothpaste containing saliva enzymes, essential oils, proteins, and colostrum extract, positively impact mucositis management for children with cancer. This study found that, in addition to reducing the severity of mucositis, oral care intervention with the appropriate specific agent has other benefits, such as lowering pain scores, shortening the duration of mucositis, and reducing the use of analgesics. However, it did not significantly improve the child’s quality of life. These findings indicate that an oral care intervention utilizing appropriate specific agents should be implemented to improve cancer care for children with cancer.
Ethics Approval and Content to Participate
This systematic review article involved no subjects; hence, no ethical approval was required or attached.
Disclosure
The authors declare that no significant competing financial, professional, or personal interests might have affected the performance or presentation of the work described in this manuscript.
Acknowledgment
The author would like to thank the Center for Education Financial Services of the Ministry of Education of the Republic of Indonesia in collaboration with Educational Fund Management Institution – Ministry of Finance, Republic of Indonesia, for providing doctoral scholarships to first authors through the Indonesian Education Scholarship. The authors would like to express their gratitude to Universitas Padjadjaran and Universitas Indonesia for their academic support.
Additional information
Funding
References
- World Health Organization. WHO Global Initiative for Childhood Cancer – India Responds. Vol. 5. World Health Organization; 2020. doi:10.1016/j.phoj.2020.06.005
- Union for International Cancer Control. Annual Report Leading the global fight against cancer. 2020:1–60.
- Nurhidayah I, Nurhaeni N, Allenidekania A, Gayatri D. A systematic review of experimental studies on the impact of empowerment-based interventions on child and parent outcomes in the pediatric oncology setting. J Multidiscip Healthc. 2023;16:3717–3735. doi:10.2147/JMDH.S436394
- Volberding PA, Spicer CM, Cartaxo T, Aiuppa L. Childhood Cancer and Functional Impacts Across the Care Continuum. The National Academies Press; 2021. doi:10.17226/25944
- Essawy MA, Abohadida RM, Abd-Elkader WM, Fathy HM, Hassab HM. Comparing the effect of acupressure and ginger on chemotherapy gastrointestinal side-effects in children with leukemia. Complement Ther Med. 2021;60:102730. doi:10.1016/j.ctim.2021.102730
- Li R, Ma J, Chan Y, Yang Q, Zhang C. Symptom Clusters and Influencing Factors in Children with Acute Leukemia during Chemotherapy. Cancer Nurs. 2020;43(5):411–418. doi:10.1097/NCC.0000000000000716
- Feng J, Gao M, Zhao C, et al. Oral administration of probiotics reduces chemotherapy-induced diarrhea and oral mucositis: a systematic review and meta-analysis. Front Nutr. 2022;9:1–17. doi:10.3389/fnut.2022.823288
- Palomo-Colli MÁ, Soltero-Becerril DC, Juárez Villegas LE, et al. Oral mucositis in children with cancer and their relationship with limitation of activities. Gac Mex Oncol. 2019;17(2). doi:10.24875/j.gamo.m19000167
- Mazhari F, Shirazi AS, Shabzendehdar M. Management of oral mucositis in pediatric patients receiving cancer therapy: a systematic review and meta-analysis. Pediatr Blood Cancer. 2019;66(3):1–9. doi:10.1002/pbc.27403
- Cristina L, Damascena L, Nando N, et al. Severe oral mucositis in pediatric cancer patients: survival analysis and predictive factors. Int J Environ Res Public Health. 2020;17(4):1235.
- Al-Rudayni AHM, Gopinath D, Maharajan MK, Menon RK. Impact of oral mucositis on quality of life in patients undergoing oncological treatment: a systematic review. Transl Cancer Res. 2020;9(4):3126–3134. doi:10.21037/tcr.2020.02.77
- Kostak MA, Semerci R, Eren T, Kocaaslan EN, Yildiz F. Effects of oral health care education on the severity of oral mucositis in pediatric oncology patients. Turkish J Oncol. 2020. doi:10.5505/tjo.2020.2366
- Pulito C, Cristaudo A, Porta CL, et al. Oral mucositis: the hidden side of cancer therapy. J Exp Clin Cancer Res. 2020;39(1):1–15. doi:10.1186/s13046-020-01715-7
- Lohakare T, Kumari D, Wanjari MB, Maurya A, Kurian B, Meshram KM. Effectiveness of application of oral regimen, practicing oral health, health education, observation (apho) nursing intervention in preventing and managing oral mucositis in children undergoing chemotherapy: an Interventional Study in Central India. Cureus. 2023;15(6):4–11. doi:10.7759/cureus.40902
- Friend A, Rubagumya F, Cartledge P. Is honey effective as a treatment for chemotherapy-induced mucositis in paediatric oncology patients? J Trop Pediatr. 2018;64(2):162–168. doi:10.1093/tropej/fmx092
- Miranda-Silva W, Gomes-Silva W, Zadik Y, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis: sub-analysis of current interventions for the management of oral mucositis in pediatric cancer patients. Support Care Cancer. 2021;29(7):3539–3562. doi:10.1007/s00520-020-05803-4
- Levin L, Bilder L, Borisov O. Improving oral hygiene skills among children undergoing treatment at the haemato-oncology department - An interventional programme. Int Dent J. 2015;65(4):211–215. doi:10.1111/idj.12171
- Miller MM, Donald DV, Hagemann TM. Prevention and treatment of oral mucositis in children with cancer. J Pediatr Pharmacol Ther. 2012;17(4):340–350. doi:10.5863/1551-6776-17.4.340
- Ribeiro ILA, de Melo ACR, Limão NP, Bonan PRF, Lima Neto ED, Valença AMG. Oral mucositis in pediatric oncology patients: a nested case-control to a prospective cohort. Braz Dent J. 2020;31(1):78–88. doi:10.1590/0103-6440201802881
- Yavuz B, Yılmaz H B. Investigation of the effects of planned mouth care education on the degree of oral mucositis in pediatric oncology patients. J Pediatr Oncol Nurs. 2015;32(1):47–56. doi:10.1177/1043454214554011
- Cheng KKF, Tan LML. A pilot study of the effect of a home-based multimodal symptom-management program in children and adolescents undergoing chemotherapy. Cancer Rep. 2021;4(3):1–9. doi:10.1002/cnr2.1336
- Potter PA, Perry AG, Stockert PA, Hall A. Fundamental of Nursing. 10th ed. Elsevier; 2021.
- Cardona A, Balouch A, Abdul MM, Sedghizadeh PP, Enciso R. Efficacy of chlorhexidine for the prevention and treatment of oral mucositis in cancer patients: a systematic review with meta-analyses. J Oral Pathol Med. 2017;46(9):680–688. doi:10.1111/jop.12549
- Brown TJ, Gupta A. Management of cancer therapy-associated oral mucositis. J Oncol Pract. 2020;16(3):103–109. doi:10.1200/JOP.19.00652
- Elad S, Cheng KKF, Lalla RV, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. 2020;126(19):4423–4431. doi:10.1002/cncr.33100
- Abdulrhman M, Samir El Barbary N, Ahmed Amin D, Saeid Ebrahim R. Honey and a mixture of honey, beeswax, and olive oil-propolis extract in treatment of chemotherapy-induced oral mucositis: a randomized controlled pilot study. Pediatr Hematol Oncol. 2012;29(3):285–292. doi:10.3109/08880018.2012.669026
- Tomaževič T, Jazbec J. A double blind randomised placebo controlled study of propolis (bee glue) effectiveness in the treatment of severe oral mucositis in chemotherapy treated children. Complement Ther Med. 2013;21(4):306–312. doi:10.1016/j.ctim.2013.04.002
- Lucchese A, Matarese G, Manuelli M, et al. Reliability and efficacy of palifermin in prevention and management of oral mucositis in patients with acute lymphoblastic leukemia: a randomized, double-blind controlled clinical trial. Minerva Stomatol. 2016;65(1):45–50.
- Gopalakrishnan S, Ganeshkumar P. Systematic reviews and meta-analysis: understanding the best evidence in primary healthcare. J Fam Med Prim Care. 2013;2(1):9. doi:10.4103/2249-4863.109934
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021:372. doi:10.1136/bmj.n71
- Barker TH, Stone JC, Sears K, et al. The revised JBI critical appraisal tool for the assessment of risk of bias for randomized controlled trials. JBI Evid Synth. 2023;21(3):494–506. doi:10.11124/JBIES-22-00430
- Tufanaru C, Munn Z, Aromataris E, Campbell J, Hopp L. Systematic reviews of effectiveness. In: JBI Manual for Evidence Synthesis. Joanna Briggs Institute; 2020. Available from: https://synthesismanual.jbi.global/. Accessed July 4, 2024.
- Viswanathan M, Ansari MT, Berkman NDN, et al. Assessing the risk of bias of individual studies in systematic reviews of health care interventions - methods guide – chapter | AHRQ effective health care program. Methods Guid Eff Comp Eff Rev. 2012. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22479713.
- Al Jaouni SK, Al Muhayawi MS, Hussein A, et al. Effects of honey on oral mucositis among pediatric cancer patients undergoing chemo/radiotherapy treatment at King Abdulaziz University Hospital in Jeddah, Kingdom of Saudi Arabia. Evid Based Complement Altern Med. 2017;2017:1–7. doi:10.1155/2017/5861024
- Alkhouli M, Laflouf M, Alhaddad M. Evaluation of the effectiveness of olive oil to prevent chemotherapy induced oral mucositis: a randomized controlled clinical trial. Pediatr Dent J. 2019;29(3):123–131. doi:10.1016/j.pdj.2019.08.001
- Alkhouli M, Laflouf M, Comisi JC. Assessing the topical application efficiency of two biological agents in managing chemotherapy-induced oral mucositis in children: a randomized clinical trial. J Oral Biol Craniofacial Res. 2021;11(3):373–378. doi:10.1016/j.jobcr.2021.04.001
- Badr LK, El Asmar R, Hakim S, et al. The efficacy of honey or olive oil on the severity of oral mucositis and pain compared to placebo (standard care) in children with leukemia receiving intensive chemotherapy: a randomized controlled trial (RCT). J Pediatr Nurs. 2023;70:e48–e53. doi:10.1016/j.pedn.2022.12.003
- Bardellini E, Amadori F, Majorana A. Oral hygiene grade and quality of life in children with chemotherapy-related oral mucositis: a randomized study on the impact of a fluoride toothpaste with salivary enzymes, essential oils, proteins and colostrum extract versus a fluoride toothpaste witho. Int J Dent Hyg. 2016;14(4):314–319. doi:10.1111/idh.12226
- Raphael MF, Den Boer AM, Kollen WJW, et al. Caphosol, a therapeutic option in case of cancer therapy-induced oral mucositis in children: results from a prospective multicenter double blind randomized controlled trial. Support Care Cancer. 2014;22(1):3–6. doi:10.1007/s00520-013-2015-0
- Dos Santos Soares A, Wanzeler AM, Cavalcante GH, da Silva Barros EM, Carneiro RD, Tuji FM. Therapeutic effects of andiroba (Carapa guianensis Aubl) oil, compared to low power laser, on oral mucositis in children underwent chemotherapy: a clinical study. J Ethnopharmacol. 2021;264(2020). doi:10.1016/j.jep.2020.113365
- Kobya Bulut H, Güdücü Tüfekci F. Honey prevents oral mocositis in children undergoing chemotherapy: a quasi-experimental study with a control group. Complement Ther Med. 2016;29:132–140. doi:10.1016/j.ctim.2016.09.018
- Schoot RA, van Ommen CH, Stijnen T, et al. Prevention of central venous catheter-associated bloodstream infections in paediatric oncology patients using 70% ethanol locks: a randomised controlled multi-centre trial. Eur J Cancer. 2015;51(14):2031–2038. doi:10.1016/j.ejca.2015.06.126
- Wanzeler AMV, Júnior SMA, Gomes JT, et al. Therapeutic effect of andiroba oil (Carapa guianensis Aubl.) against oral mucositis: an experimental study in golden Syrian hamsters. Clin Oral Investig. 2018;22(5):2069–2079. doi:10.1007/s00784-017-2300-2
- National Cancer Institute. Eating Hints: Before, During, and After Cancer Treatment - NCI. National Cancer Institute; 2014.
- Wong HM. Oral complications and management strategies for patients undergoing cancer therapy. Sci World J. 2014;2014:1–14. doi:10.1155/2014/581795
- Nagi R, Patil DJ, Rakesh N, Jain S, Sahu S. Natural agents in the management of oral mucositis in cancer patients-systematic review. J Oral Biol Craniofacial Res. 2018;8(3):245–254. doi:10.1016/j.jobcr.2017.12.003
- Samdariya S, Lewis S, Kauser H, Ahmed I, Kumar D. A randomized controlled trial evaluating the role of honey in reducing pain due to radiation induced mucositis in head and neck cancer patients. Indian J Palliat Care. 2015;21(3):268–273. doi:10.4103/0973-1075.164892
- Rao S, Hegde SK, Rao P, et al. Honey mitigates radiation-induced oral mucositis in head and neck cancer patients without affecting the tumor response. Foods. 2017;6(9):1–8. doi:10.3390/foods6090077
- Charalambous M, Raftopoulos V, Paikousis L, et al. The effect of the use of thyme honey in minimizing radiation - induced oral mucositis in head and neck cancer patients: a randomized controlled trial. Eur J Oncol Nurs. 2018;34:89–97. doi:10.1016/j.ejon.2018.04.003
- Köseoǧlu O, Sevim D, Kadiroǧlu P. Quality characteristics and antioxidant properties of Turkish monovarietal olive oils regarding stages of olive ripening. Food Chem. 2016;212:628–634. doi:10.1016/j.foodchem.2016.06.027
- Navi K. Olive oil and water - role in oral care. Int J Med Clin Res. 2013;4(1):258–260. doi:10.9735/0976-5530.4.1.258-260
- Ahmed KM. The effect of olive leaf extract in decreasing the expression of two pro-inflammatory cytokines in patients receiving chemotherapy for cancer. A randomized clinical trial. Saudi Dent J. 2013;25(4):141–147. doi:10.1016/j.sdentj.2013.09.001
- Gao Y, Kuok KI, Jin Y, Wang R. Biomedical applications of Aloe vera. Crit Rev Food Sci Nutr. 2019;59:S244–S256. doi:10.1080/10408398.2018.1496320
