Abstract
Introduction
Based on many published reports, African American patients with cancer experience higher pain severity scores and lower pain relief than White patients. This disparity results from undertreatment of pain and is compounded by low adherence to prescribed non-opioid and opioid analgesics among African American patients with cancer. While nearly one in four patients use cannabis to manage cancer-related symptoms, less is known about how cannabis use influences pain relief in this patient population.
Methods
This study is based on preliminary data from an ongoing study of longitudinal outcomes of opioid therapy among African American and White patients with cancer. Linear mixed-effects models were utilized to assess the interaction of race and cannabis use on pain relief using “least pain” item scores from the Brief Pain Inventory (BPI) averaged across three time points. Models were adjusted for sociodemographic and clinical variables.
Results
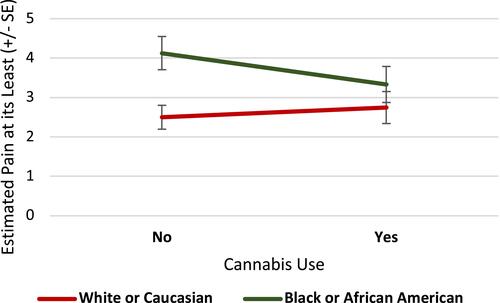
This analysis included 136 patients (49 African American, 87 White). Overall, 30.1% of the sample reported cannabis use for cancer pain. The mean “least pain” score on BPI was 3.3 (SD=2.42) on a scale of 0–10. African American patients had a mean “least pain” score 1.32±0.48 units higher (indicating lower pain relief) than White patients (p=0.006). Cannabis use did not have a significant main effect (p=0.28). However, cannabis use was a significant moderator of the relationship between race and “least pain” (p=0.03). In the absence of cannabis use, African Americans reported higher “least pain” scores compared to Whites (mean difference=1.631±0.5, p=0.001). However, this disparity was no longer observed in African American patients reporting cannabis use (mean “least pain” difference=0.587±0.59, p=0.32).
Conclusion
These findings point to the possible role of cannabis in cancer pain management and its potential to reduce racial disparities. These findings are preliminary and further research into the role of cannabis in cancer pain outcomes is needed.
Introduction
Unrelieved pain is an issue of significant magnitude across the trajectory of cancer; however, the burden is disproportionate for some groups. One of the most consistent findings on pain treatment disparities pertain to African Americans as identified in key national reportsCitation1 and other systematic reviews.Citation2,Citation3 A meta-analysis synthesizing 20 years of published research in the United States suggests that African American patients have the highest risk of pain undertreatment than any other racial and ethnic subgroup, regardless of the diagnosis.Citation4 While both Whites and African American patients report significant cancer-related pain across a number of studies,Citation5–Citation8 African American patients report a clinically meaningfulCitation9 higher level of pain scores—an indicator of inadequate pain relief. Stark disparities also have been found between African Americans and Whites in their use of prescribed non-opioid and opioid analgesics for cancer pain.Citation10 In one study, adherence to scheduled opioids (ie, percentage of the total number of prescribed doses taken) was only 57% for African Americans compared to 77% for Whites.Citation8 Of note, African Americans were 3 times more likely than Whites to show inconsistent patterns of opioid adherenceCitation11 and appear to have unique concerns related to use of analgesics. For instance, lower household income, greater need for opioid information, more severe side-effects, and poor communication with providers predicted lower analgesic adherence for African Americans, but not for Whites.Citation8
Recent surveys report that nearly one in four cancer patients use cannabis,Citation12,Citation13 and among those who use cannabis, 75% use it to manage symptoms such as pain, anxiety, and insomnia.Citation14,Citation15 Importantly, patients with cancer also report using cannabis to avoid opioids, which patients consider “harder medications” due to intolerable side effects, stigma, and addiction concerns.Citation16,Citation17 The purpose of this brief report is to describe cannabis use in a diverse sample of patients with cancer and to evaluate whether cannabis use is associated with addressing racial disparities in reported cancer pain relief.
Methods
This study concerns the analysis of preliminary data from a larger, ongoing parent study (1R01NR017853) to elucidate longitudinal outcomes of opioid therapy in patients with cancer and how patterns of opioid use over time relate to patient-reported outcomes and healthcare utilization. The data are based on a repeated measures survey with patient-reported outcomes collected at 5 time points (ie, T1= baseline; T2= 1 month; T3 = 2 months; T4= 3 months, T5 =5 months) combined with daily patient-reported data through a smartphone-based application, mobile Ecological Momentary Assessment (mEMA, Ilumivu.com). Patients are recruited from outpatient medical oncology clinics of the University of Pennsylvania Health System. Patients are included if they are ≥18 years of age, self-identify as African Americans or Whites; are ambulatory patients with non-skin malignancies; and are prescribed opioids for pain (patients with neuropathic pain are included). Patients are excluded if they are receiving opioids only for treatment of an opioid use disorder (Medication for Opioid Use Disorder); are prescribed opioids through non-oral routes (eg, intravenous, subcutaneous, rectal, transdermal, and transmucosal routes); have any cognitive impairment or medical condition that interfere with informed consent; or are residing in a nursing home or receiving hospice care.
Potentially eligible patients are contacted by phone by trained research staff who review the purpose of the study using a standardized script. If a participant is interested, an eligibility checklist is reviewed with the participant and the trained staff answers any questions related to the study. If the patient is eligible and remains interested, an appointment is made based on patient’s convenience. Initially, study staff made home visits to collect data to reduce participant burden. However, due to the COVID-19 pandemic, data collection has been transitioned to phone-based data collection, which has not posed any significant difficulty with maintaining study procedures and rigor. The informed consent form is reviewed prior to data collection and all participants receive a copy of the form for their records with the contact information of the study principal investigator and study project manager. The study was approved by the University of Pennsylvania’s Institutional Review Board (IRB protocol # 833009). This study was conducted in accordance with the Declaration of Helsinki.
Study Measures
The cannabis use variable is obtained from two sources: patients’ self-report during baseline study interview (T1), as well as extracted from a review of patients’ electronic medical records. In the self-reported interview, patients are asked,
in the past week, have you used any of the following to manage your pain? Please check all that apply. If you did not use any of these therapies, then select none of the above.
The response choices include medical cannabis, recreational cannabis, and hemp-based cannabidiol (CBD) products. Patients who reported the use of CBD products were not included in the current analysis as “cannabis” users because most CBD products are available as supplements only, which have limited evidence for pain management and there are currently no approved pharmaceutical grade CBD-exclusive products for pain management.Citation18
Pain is assessed using the Brief Pain Inventory (BPI)-Short Form at each data collection time point. The BPI is comprised of two subscales assessing pain severity and pain-related interference.Citation19–Citation21 The pain severity subscale includes four items (“worst pain”, “least pain”, “average pain”, and “pain now”) assessing different dimensions of pain severity. The BPI “least pain” item was used due to its clinical meaningfulness in assessing pain at its lowest or degree of pain relief in an index period. The item asks, “please rate your pain by circling the one number that best describes your pain “at its least” in the last 24 hours,” and is scored on a scale of 0–10 (0=no pain and 10=pain as bad as you can imagine). A higher “least pain” score indicates lower pain relief. The psychometric properties of the BPI are well established with cancer patients with a Cronbach’s alpha that ranges from 0.77 to 0.91.Citation19,Citation20 The validity and reliability of single recall-based “least pain” items for pain intensity have been demonstrated in the cancer population.Citation22
Analgesic side-effects are assessed using the Medication Side-effects Checklist (MSEC). MSEC elicits information on the presence and severity of eight common analgesic side-effects (ie, constipation, drowsiness, nausea, vomiting, confusion, dry mouth, stomach irritation, itching) on a scale of 0–10 (no severity-extreme severity). The internal consistency reliability is 0.80.Citation23
The Pain Self-Efficacy Questionnaire (PSEQ)Citation24 is used to assess confidence in performing activities while in pain (household chores, socializing, work, coping with pain without medications). The internal consistency reliability for this tool ranges 0.81–0.92.Citation24
We also used the Current Opioid Misuse Measure (COMM),Citation25 a 17-item tool, to assess behaviors that contextualize current opioid misuse events with a cut-off of ≥9 suggesting opioid misuse. The internal consistency reliability for this tool ranges 0.81–0.86.Citation25
Data Analysis
The present analysis includes the first 136 patients enrolled in the ongoing study. The data presented here are from three time points (T1-T3) from the ongoing parent study. Summary statistics were computed for the 136 patients included in this analysis and stratified by race for participants’ baseline clinical and demographic characteristics using means and standard deviations for continuous measures, and frequencies and percentages for categorical measures. Clinical and demographic characteristics were compared between race groups using chi-squared and one-way ANOVA for categorical and continuous measures, respectively. Distribution of the outcome measure was assessed using histograms and tested for normality using Shapiro–Wilk tests and Kolmogorov–Smirnov tests.
Linear mixed-effects models were produced to assess the effects of race and cannabis use on participants’ least pain (ie, time-averaged BPI “least pain” scores at three time points). Cannabis use was then assessed as a moderator of the relationship between race and “least pain” by testing the cannabis use by race interaction term. An unstructured covariance matrix was used to account for within-subject variance in pain outcomes. All models were adjusted for the effects of participant age, gender, income, insurance status, cancer stage, cancer treatment status (active treatment or completed cancer treatment), prescribed morphine milligram equivalent per day (MME/day), opioid prescription type (long-acting only, immediate-release opioid only; or both), current opioid misuse measure (COMM score), medication side-effects (MSCE score), pain self-efficacy (PSEQ score). Statistical analysis was conducted using SAS 9.4 for Windows.
Results
and summarize the sociodemographic and illness-related characteristics of the sample by their self-identified race. The present analysis included 136 patients (n=87 Whites [64%]; n=49 African Americans [36%]), who were largely older adult (M=60.9 years; SD=11.79), and females (75%) with low annual household incomes of less than $30,000 (43%). Cancers were typically (46.3%) advanced (Stage III/IV), and breast (27.9%) and lung (10.3%) cancer were most common. Most opioid prescriptions (50.4%) included combined long- and short-acting formulations, with high MME/day: MME/day (30.9%) and ≥90 /day (30.9%) and >90 MME/day (29.4%). The mean “least pain” score was 3.3 (SD=2.42) overall, where the average “least pain” score by group was lower among Whites (M=2.5, SD=2.1) than African Americans (M=4.7, SD=2.32). At baseline, 41 patients (30.1%) reported cannabis use, with the majority of these patients reporting medical cannabis ().
Table 1 Sociodemographic Characteristics by Participant Race
Table 2 Illness and Pain Characteristics by Participant Race
Table 3 Reported Cannabis Use by Participant Race
Race was found to be a significant predictor of “least pain” in the adjusted longitudinal analysis. Specifically, African American participants were estimated to have a mean “least pain” score 1.32±0.48 units higher (ie, indicating lower pain relief) than White participants (p=0.006). Cannabis use was not found to exhibit a significant main effect on “least pain” (p=0.28).
However, in assessing cannabis use as a moderator of the relationship between race and “least pain”, the interaction term between race and cannabis was found to be statistically significant (p=0.03). Pairwise (across time and including baseline) comparisons of time-averaged model-based means () indicated that a significant racial disparity in pain relief exists among those who did not use cannabis such that African American patients reported higher “least pain” scores (indicating lower pain relief) compared to White patients (mean difference=1.631±0.5, p=0.001; ). However, this disparity was no longer evident among those who used cannabis (mean difference=0.587±0.59, p=0.32). We also conducted a sensitivity analysis in which we adjusted for other substance use. We did not find other substance use to significantly predict pain relief (p=0.3397) and found the interaction between race and cannabis use to remain statistically significant (p=0.0347).
Table 4 Model-Based Least Pain Score Means for the Interaction Model
Discussion
In this preliminary analysis of a larger, ongoing parent study of the longitudinal outcomes of opioid therapy among patients with cancer, we evaluated whether cannabis moderates the relationship between race and pain outcomes. Our primary finding is that cannabis may reduce racial disparities in pain relief in this population. Although this finding should be considered preliminary, it highlights the growing need to better understand the role of cannabis in cancer pain management, particularly as it relates to racial disparities.
Indeed, we found that among patients with cancer who did not report cannabis use, there was a significant disparity in adequate pain relief between African American and White patients, with African American patients reporting significantly higher “least pain” scores compared to White patients. This finding is consistent with a large body of evidence demonstrating that African Americans experience inadequate pain relief, relative to White patients.Citation4,Citation7,Citation26,Citation27 These relatively high “least pain” scores are also particularly striking considering that 60% of the sample were prescribed over 50 MME/day of opioids. Adding to the literature, our data suggest that this disparity is significantly reduced among patients who report cannabis use. Given the well-established disparities in the prescribing of analgesics and in particular opioids for cancer pain among African American and White patients, this finding warrants additional research to better understand the influences of cannabis use on opioid access, opioid use, pain relief, and racial differences.
Consistent with recent surveys,Citation12,Citation13,Citation28 approximately 30% of the current sample reported using cannabis. Although the majority of these patients reported using medical cannabis, several patients reported using recreational cannabis or a combination of medical and recreational cannabis. Patients concerned about stigmaCitation29 may be reluctant to disclose cannabis use and thus, these findings may be an underrepresentation of cannabis use. Potential racial differences in perceived risk of cannabis use such that African Americans report higher perceived risk than WhitesCitation30 may have contributed to underreporting of use in this sample. Although the prior study did not differentiate between perceived risk associated with recreational purposes versus medical reasons, the current data provide insight into the role of cannabis in cancer pain management when considered in the context of race.
In contrast to some previous studies,Citation14,Citation15 we did not find an overall main effect of cannabis use on pain. This could be due in part to the fact that the current study was focused on opioid adherence profiles and future studies would benefit from a more granular and longitudinal assessment of cannabis use as it relates to self-reported pain and opioid use. Additionally, increased focus on form (inhaled, oral, topical) and frequency of cannabis use in real time is necessary to better clarify and understand the interaction between cancer pain, opioid use, and pain outcomes. While cannabis use is common, there continues to be a lack of comfort in conversation between patient and clinician around use, often leading to illicit (ie, recreational) rather than state-regulated access to medical cannabis.Citation31 This is important as medical cannabis is purer and there are formulations that provide greater analgesia which is not necessarily the case in recreational cannabis. Also, vaping or smoking cannabis can be carcinogenic. An open, nonjudgmental discussion between the clinician and the patient can maximize the potential efficacy of cannabis and mitigate risks. For us to understand the true breadth and impact of cannabis use on patients with cancer, knowing the limitations in communication and lack of standard legal approach and availability of cannabis state by state, we need to examine both licit and illicit cannabis use to tell the full story.
Limitations
Our study is based on data from a single health system. The aims presented here do not reflect the original aims of the funded grant and represent emergent findings. Thus, these findings are preliminary, hypothesis-generating, and subject to change based on additional data that are being collected or studies designed specifically to investigate these aims. The cannabis data are based on patient’s self-report, which currently represents the best source of data, as the use of cannabis is not reliably collected in patients’ medical records. The analysis does not account for frequency, route, and source (medical vs recreational cannabis) as the data are not reliably available. Patients may use recreational cannabis for medical reasons and frequently transition between medical and recreational sources based on cost and availability. Also, we did not include patients receiving CBD in this analysis given the significant variation in over-the-counter (ie, supplemental) CBD products, which have limited evidence for pain management.Citation17,Citation32 Further, while the developers of the BPI survey recommend that all four pain severity items be employed,Citation20 they also acknowledge use of single BPI items,Citation20 which is supported by the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) recommendations for assessing pain in pain studies and clinical trialsCitation33–Citation35 and by other industry standards.Citation36 However, we acknowledge that despite the validity and reliability of the BPI “least pain” item,Citation21 it is a unidimensional report of pain severity limiting our understanding of the other dimensions of pain severity and whether or not the reported “least pain” scores were actually tolerable pain levels for the patients.Citation37
We did not intentionally analyze least pain scores, but it was an incidental finding that we tested further. While we did not detect statistical significance (alpha set at 0.05) of the race by cannabis interaction for other BPI pain items (pain worst, pain now, pain average), we did observe consistent trends in the direction of effects for BPI worst pain, BPI pain severity subscale, and BPI pain interference subscale, which strengthens confidence in the preliminary results (). The effect size estimates presented may be useful to researchers who may need effect size data to compute sample sizes for future studies.
Table 5 Effect Size Estimates for Race×Cannabis Use Interaction for BPI Outcomesa
While our study adjusted for daily morphine milligram equivalent as a covariate in the analysis presented, due to limitations of our current sample size, we did not assess complex interactions that included daily or oral morphine equivalent as an additional moderator of associations between race, cannabis use, and pain outcomes and recommend that future studies investigate the relationship with opioid daily dose.
Conclusions
These preliminary findings point to an important role of cannabis use in cancer pain and its potential to reduce racial disparities in cancer pain management, which is a significant clinical issue. Future studies are needed to study the role of cannabis more rigorously in cancer pain outcomes, including investigating its role in closing racial disparities. Studies are also needed to understand the role of medical vs recreational sources of cannabis in these outcomes.
Disclosure
Dr Rebecca Ashare report grants from NIH, grants from Novo Nordisk, Inc. during the conduct of the study. The authors report no other conflicts of interest in this work.
Additional information
Funding
References
- Institute of Medicine. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, D.C.: National Academies Press; 2011.
- Anderson KO, Green CR, Payne R. Racial and ethnic disparities in pain: causes and consequences of unequal care. J Pain. 2009;10(12):1187–1204. doi:10.1016/j.jpain.2009.10.002
- Cintron A, Morrison RS. Pain and ethnicity in the United States: a systematic review. J Palliat Med. 2006;9(6):1454–1473. doi:10.1089/jpm.2006.9.1454
- Meghani SH, Byun E, Gallagher RM. Time to take stock: a meta-analysis and systematic review of analgesic treatment disparities for pain in the United States. Pain Med. 2012;13(2):150–174. doi:10.1111/j.1526-4637.2011.01310.x
- Castel LD, Saville BR, Depuy V, Godley PA, Hartmann KE, Abernethy AP. Racial differences in pain during 1 year among women with metastatic breast cancer: a hazards analysis of interval-censored data. Cancer. 2008;112(1):162–170. doi:10.1002/cncr.23133
- Jayadevappa R, Johnson JC, Chhatre S, Wein AJ, Malkowicz SB. Ethnic variation in return to baseline values of patient-reported outcomes in older prostate cancer patients. Cancer. 2007;109(11):2229–2238. doi:10.1002/cncr.22675
- Meghani SH, Kang Y, Chittams J, McMenamin E, Mao JJ, Fudin J. African Americans with cancer pain are more likely to receive an analgesic with toxic metabolite despite clinical risks: a mediation analysis study. J Clin Oncol. 2014;32(25):2773–2779. doi:10.1200/JCO.2013.54.7992
- Meghani SH, Thompson AM, Chittams J, Bruner DW, Riegel B. Adherence to analgesics for cancer pain: a comparative study of African Americans and whites using an electronic monitoring device. J Pain. 2015;16(9):825–835.
- Farrar JT, Young JP Jr, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain. 2001;94(2):149–158. doi:10.1016/S0304-3959(01)00349-9
- Meghani SH, Hanlon A, Bubanj J, Riegel B, Bruner D. Do self-reported analgesic barriers translate into objective analgesic adherence for cancer pain? J Pain. 2013;14(4):S38. doi:10.1016/j.jpain.2013.01.447
- Meghani SH, Knafl GJ. Patterns of analgesic adherence predict health care utilization among outpatients with cancer pain. Patient Prefer Adherence. 2016;10:81–98. doi:10.2147/PPA.S93726
- Bar-Lev Schleider L, Mechoulam R, Lederman V, et al. Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med. 2018;49:37–43. doi:10.1016/j.ejim.2018.01.023
- Pergam SA, Woodfield MC, Lee CM, et al. Cannabis use among patients at a comprehensive cancer center in a state with legalized medicinal and recreational use. Cancer. 2017;123(22):4488–4497. doi:10.1002/cncr.30879
- Turgeman I, Bar-Sela G. Cannabis use in palliative oncology: a review of the evidence for popular indications. Isr Med Assoc J. 2017;19(2):85–88.
- Martell K, Fairchild A, LeGerrier B, et al. Rates of cannabis use in patients with cancer. Curr Oncol. 2018;25(3):219–225. doi:10.3747/co.25.3983
- Lucas P, Walsh Z. Medical cannabis access, use, and substitution for prescription opioids and other substances: a survey of authorized medical cannabis patients. Int J Drug Policy. 2017;42:30–35. doi:10.1016/j.drugpo.2017.01.011
- Meghani SH, Wool J, Davis J, Yeager KA, Mao JJ, Barg FK. When Patients Take Charge of Opioids: Self-Management Concerns and Practices Among Cancer Outpatients in the Context of Opioid Crisis. J Pain Symptom Manage. 2020;59(3):618–625.
- Boyaji S, Merkow J, Elman RNM, Kaye AD, Yong RJ, Urman RD. The role of cannabidiol (CBD) in chronic pain management: an assessment of current evidence. Curr Pain Headache Rep. 2020;24(2):4. doi:10.1007/s11916-020-0835-4
- Anderson KO, Richman SP, Hurley J, et al. Cancer pain management among underserved minority outpatients: perceived needs and barriers to optimal control. Cancer. 2002;94(8):2295–2304. doi:10.1002/cncr.10414
- Cleeland CS, Gonin R, Hatfield AK, et al. Pain and its treatment in outpatients with metastatic cancer. N Engl J Med. 1994;330(9):592–596. doi:10.1056/NEJM199403033300902
- Cleeland CS. The brief pain inventory: user guide; 2009. Available from: https://www.mdanderson.org/documents/Departments-and-Divisions/Symptom-Research/BPI_UserGuide.pdf. Accessed July 28, 2021.
- Jensen MP, Wang W, Potts SL, Gould EM. Reliability and validity of individual and composite recall pain measures in patients with cancer. Pain Med. 2012;13(10):1284–1291. doi:10.1111/j.1526-4637.2012.01470.x
- Ward SE, Carlson-Dakes K, Hughes SH, Kwekkeboom KL, Donovan HS. The impact on quality of life of patient-related barriers to pain management. Res Nurs Health. 1998;21(5):405–413. doi:10.1002/(SICI)1098-240X(199810)21:5<405::AID-NUR4>3.0.CO;2-R
- Nicholas MK. The pain self-efficacy questionnaire: taking pain into account. Eur J Pain. 2007;11(2):153–163. doi:10.1016/j.ejpain.2005.12.008
- Butler SF, Budman SH, Fernandez KC, et al. Development and validation of the current opioid misuse measure. Pain. 2007;130(1–2):144–156. doi:10.1016/j.pain.2007.01.014
- Belcher SM, Watkins Bruner D, Hofmeister CC, Kweon J, Meghani SH, Yeager KA. Characterizing pain experiences: African American patients with multiple myeloma taking around-the-clock opioids. Clin J Oncol Nurs. 2020;24(5):538–546. doi:10.1188/20.CJON.538-546
- Meghani SH, Rosa WE, Chittams J, Vallerand AH, Bao T, Mao JJ. Both race and insurance type independently predict the selection of oral opioids prescribed to cancer outpatients. Pain Manag Nurs. 2020;21(1):65–71. doi:10.1016/j.pmn.2019.07.004
- Tringale KR, Huynh-le MP, Salans M, Marshall DC, Shi Y, Hattangadi-Gluth JA. The role of cancer in marijuana and prescription opioid use in the United States: a population-based analysis from 2005 to 2014. Cancer. 2019;125(13):2242–2251.
- Satterlund TD, Lee JP, Moore RS. Stigma among California’s medical marijuana patients. J Psychoactive Drugs. 2015;47(1):10–17. doi:10.1080/02791072.2014.991858
- Pacek LR, Mauro PM, Martins SS. Perceived risk of regular cannabis use in the United States from 2002 to 2012: differences by sex, age, and race/ethnicity. Drug Alcohol Depend. 2015;149:232–244. doi:10.1016/j.drugalcdep.2015.02.009
- Hasin DS, Shmulewitz D, Sarvet AL. Time trends in US cannabis use and cannabis use disorders overall and by sociodemographic subgroups: a narrative review and new findings. Am J Drug Alcohol Abuse. 2019;45(6):623–643. doi:10.1080/00952990.2019.1569668
- Chesney E, McGuire P, Freeman TP, Strang J, Englund A. Lack of evidence for the effectiveness or safety of over-the-counter cannabidiol products. Ther Adv Psychopharmacol. 2020;10:2045125320954992. doi:10.1177/2045125320954992
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113(1–2):9–19. doi:10.1016/j.pain.2004.09.012
- Dworkin RH, Turk DC, Wyrwich KW, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain. 2008;9(2):105–121. doi:10.1016/j.jpain.2007.09.005
- Turk DC, Dworkin RH, Burke LB, et al. Developing patient-reported outcome measures for pain clinical trials: IMMPACT recommendations. Pain. 2006;125(3):208–215. doi:10.1016/j.pain.2006.09.028
- Administration FaD. Guidance for industry patient-reported outcome measures: use in medical product development to support labeling claims; 2009. Available from: https://www.fda.gov/media/77832/download. Accessed July 28, 2021.
- Markman JD, Gewandter JS, Frazer ME. Comparison of a pain tolerability question with the numeric rating scale for assessment of self-reported chronic pain. JAMA Netw Open. 2020;3(4):e203155. doi:10.1001/jamanetworkopen.2020.3155