Abstract
Transcatheter aortic valve implantation (TAVI) is increasingly performed in patients with severe aortic stenosis. The efficacy and safety have been demonstrated in large randomized trials in patients with high- or intermediate operative risk. With latest-generation transcatheter heart valve (THV) systems, growing operator experience and improved patient selection, clinical outcome has significantly improved with a decline of TAVI-related complications. In this review, the Edwards SAPIEN 3 THV is discussed in terms of technology, procedural advances and complication trends and future developments.
Introduction and current trends
Aortic stenosis is the most common valvular heart disease in the Western world.Citation1 In the last 15 years, since the first-in-human transcatheter aortic valve implantation (TAVI) performed by A Cribier in 2002, the number of TAVI procedures has increased impressively.Citation2 So far, >350,000 procedures have been performed in over 70 countries.Citation3
Over the last decade, the indication for TAVI has gradually shifted from a challenging intervention in inoperable, high-risk patients toward a standardized straightforward procedure in intermediate to even lower-risk patients. Based on evidence from large randomized controlled trials (RCTs), current guidelines recommend TAVI in high-risk patients and also recommend to consider this treatment option for intermediate-risk patients as non-inferiority to surgical aortic valve replacement (SAVR) has been shown.Citation4–Citation6 Currently, there are ongoing trials (ie, Placement of Aortic Transcatheter Valves [PARTNER] 3 and Low-Risk Evolut R), evaluating the safety and efficacy of TAVI even in patients with low operative risk.
SAPIEN 3 transcatheter heart valve
Technical issues
The development of current devices goes back to the first description of a transcatheter heart valve (THV) by HR Andersen et al in 1992.Citation7 Over the last decade, there has been a rapid and impressive evolution in THV, delivery systems and technical approaches. Currently, several competing THVs are available.Citation3
The original Cribier–Edwards THV (Edwards Lifesciences, Irvine, CA, USA) consisted of a stainless-steel frame with equine pericardium valve leaflets and was subsequently modified as the Edwards SAPIEN THV, using among others a higher sealing cuff and bovine pericardium leaflets (). The SAPIEN THV was followed by the SAPIEN XT THV (), which consisted of cobalt chromium alloy frame and bovine pericardium leaflets.
Figure 1 Overview of Edwards Lifesciences balloon-expandable valves.
Note: (A) Edwards SAPIEN, (B) Edwards SAPIEN XT, (C) Edwards SAPIEN 3. Copyright ©2018 Edwards Lifesciences Corporation. Reproduced from https://www.edwards.com [homepage on the Internet]. transcatheter heart valve. Available from: https://www.edwards.com/gb/devices/heart-valves/transcatheter.Citation70
![Figure 1 Overview of Edwards Lifesciences balloon-expandable valves.Note: (A) Edwards SAPIEN, (B) Edwards SAPIEN XT, (C) Edwards SAPIEN 3. Copyright ©2018 Edwards Lifesciences Corporation. Reproduced from https://www.edwards.com [homepage on the Internet]. transcatheter heart valve. Available from: https://www.edwards.com/gb/devices/heart-valves/transcatheter.Citation70](/cms/asset/6167608e-10b2-4658-a099-94699ae3a2be/dmde_a_143897_f0001_c.jpg)
The SAPIEN 3 (S3) THV is the latest generation of Edwards balloon-expandable valves. It features a cobalt chromium alloy frame that provides a high radial strength for circularity and optimal hemodynamics, a low frame height and an open cell geometry, allowing access to coronary vessels for future interventions and an outer polyethylene terephthalate (PET) skirt to minimize paravalvular leakage (PVL). The valve tissue consists of three leaflets manufactured from bovine pericardium (). Four different sizes of the S3 THV are currently available: 20 mm, 23 mm, 26 mm and 29 mm. Selection of the appropriate THV should be made according to multislice computed tomography (MSCT) annulus area-based sizing recommendations provided by the manufacturer.Citation8 The treatable range of aortic annulus diameters is wide and ranges from 18.6 mm to 29.5 mm.
The transfemoral commander delivery catheter (Edwards Lifesciences) allows for accurate positioning of the THV within the native valve. As an aid, a central balloon marker is incorporated as a primary landmark for correct positioning during implantation. The S3 THV is compatible with a 14-French (Fr) (in the case of a 20 mm, 23 mm or 26 mm S3 THV) or 16-Fr (in the case of a 29 mm S3 THV) expandable sheath (eSheath; Edwards Lifesciences). The outer diameter of a 14-Fr sheath is 6 mm and that of a 16-Fr sheath is 6.7 mm, respectively. It should be noted that the outer sheath diameter is 18-Fr (for 20–26 mm S3 THV) and 20-Fr (for 29 mm S3 THV), which increases during THV passage up to 24-Fr (for 20–26 mm S3 THV) and 27-Fr (for 29 mm S3 THV). Taking into consideration that arteries are somewhat compliant, the recommended minimal vessel diameter for a transfemoral approach is 5.5 mm (for 14-Fr eSheath) and 6 mm (for 16-Fr eSheath), respectively. However, due to the expanding nature, some caution needs to be exerted in the case of circular vascular calcifications.
Procedural advances and complication trends
The S3 THV has received CE (Communauté européenne)-mark approval in Europe in January 2014 and US Food and Drug Administration approval in the US in June 2015. The 30-day mortality rate of 2.1% and further important adverse events were among the lowest reported at that time.Citation8 Early clinical results of the PARTNER II trial confirmed the favorable outcome of the S3 THV system with low 30-day mortality rates, which were 2.2% in high-risk or inoperable patients and 1.1% in intermediate-risk patients, respectively.Citation9 Real-world data from large registries have also been promising ().Citation10,Citation11 Further studies with longer follow-up confirmed these excellent results with 1-year mortality rates of 17.7% in inoperable patients and 12.7% in high-risk patients, respectively.Citation12 These results demonstrated a considerable improvement compared with 1-year mortality rates of 31% in inoperable patients and 24% in high-risk patients reported from the first PARTNER trial, which began enrollment in 2007.Citation13,Citation14
Figure 2 Device failure and 30-day mortality rates after TAVI with SAPIEN 3
Abbreviation: TAVI, transcatheter aortic valve implantation.
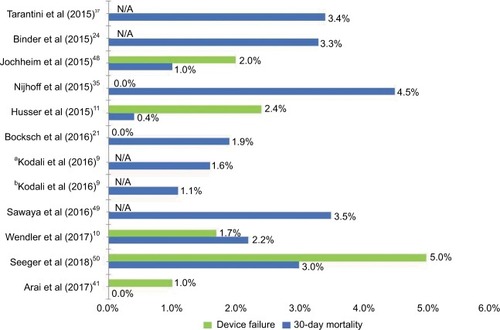
Despite these tremendous advances in survival with new-generation devices, increasing operator experience and improvement in patient selection, there are several TAVI-related complications requiring special attention when evaluating a novel THV. In the following, these important issues, including PVL, conduction disturbances leading to permanent pacemaker implantations (PPIs), vascular complications and cerebrovascular events (CVE), are further discussed with special focus on the S3 THV ().
Table 1 Periprocedural complications and clinical outcomes with SAPIEN 3™ and SAPIEN XT
Paravalvular aortic regurgitation
Historically, PVL has been a frequent complication after TAVI, with much higher rates when compared with SAVR. In a meta-analysis of Généreux et al,Citation15 the incidence of moderate or severe PVL has been reported to be 7.4% after TAVI using first-generation devices. Long-term data of the PARTNER A and B trial with up to 5 years follow-up showed that the presence of PVL negatively impacts prognosis.Citation13,Citation16,Citation17 Severe or asymmetric calcification of the native aortic annulus leading to incomplete apposition, annular eccentricity, malpositioning and undersizing of the device are probable mechanisms contributing to PVL.Citation18–Citation20
As PVL was initially regarded as a barrier for a widespread use of TAVI, the so-called “next”-generation devices were developed to incorporate special features, such as repositionability and retrievability, allowing for a controlled deployment as well as external sealing features to overcome this issue.
In line with this demand, one key modification of the current S3 THV is an outer skirt surrounding the valve frame to provide external sealing and to reduce the rate of PVL. As expected, the number of any kind of PVL was reduced with the S3 THV, and the rate of moderate or severe PVL has decreased from 6.9% with SAPIEN XT to 1.6% with S3.Citation21–Citation25 Besides technical developments of the valve design, more sophisticated sizing algorithms with a routine use of pre-procedural MSCT may also have contributed to a reduction in PVL by an optimized valve deployment.Citation8
New-onset conduction disturbances
Based on the proximity of the cardiac conduction system to the aortic root, conduction disturbances are frequently observed after TAVI and may reach up to 40% of the cases, depending on the implanted THV type.Citation26 The most prevalent conduction disturbances are non-specific intraventricular conduction abnormalities (CA), left bundle branch block (LBBB) and complete atrioventricular block requiring PPI. Development of new-generation devices had the goal to overcome this issue, in particular because it has been shown that new-onset CA, especially new LBBB, and PPI may be negatively associated with recovery of left ventricular function after TAVICitation27–Citation29 and may lead to a higher rate of hospitalizations for worsening heart failure.Citation30 Regarding the impact of PPI on long-term mortality, available data are conflicting.Citation30,Citation31 While some investigations found no effect of PPI on mortality,Citation6,Citation32 recent analyses from the PARTNER trial identified chronic pacing as an independent predictor of 1-year mortality after TAVI.Citation30,Citation33
With the introduction of the new S3 THV, it was of special interest to assess how the new valve design and the outer skirt would affect the rate of new CA and PPI in comparison with its predecessor. Indeed, patients treated with the S3 had a higher rate of new CA compared with SAPIEN XT.Citation34 Nevertheless, the rate of new PPI with S3 compared with SAPIEN XT was comparable, with only a slight, non-significant trend toward a higher rate with S3.Citation24,Citation34,Citation35 In line with other valve types, one of the major baseline predictors for a new PPI with the S3 THV was a pre-existing right bundle branch block (RBBB).Citation34,Citation36 Additionally, it has been postulated that a higher implantation of the THV within the virtual aortic annulus may result in lower pacemaker rates.Citation37 Oversizing has also been identified as an important predictor of PPI,Citation38 and recent data indicate a linear relationship between oversizing and PPI with no ideal sizing range to minimize PPI while maintaining device success.Citation69
Vascular complications
Vascular complications are another major hurdle of the TAVI procedure with initial rates of (major) vascular complications ranging from 1.9% to 17.3%.Citation39 Fortunately, life-threatening complications, such as aortic dissection, annular rupture or left ventricular perforation, have been rare with reported rates of usually <1%.Citation40 Access-site- related vascular complications constitute the most common vascular complications in transfemoral TAVI. Apart from small vessel diameters and severe calcifications, the sheath-to-femoral artery ratio belongs to the main predictors of major vascular complications.Citation39 To address this issue, another key feature of the S3 was the reduction of the delivery system profile. To some extent, this was achieved by the feature that the THV is mounted onto the deployment balloon within the body in the descending aorta instead of an on-balloon delivery. The 14-Fr eSheath can accommodate the 20 mm, 23 mm and 26 mm S3 THV, and the 16-Fr eSheath the 29 mm S3 THV, whereas larger sheaths were necessary for SAPIEN XT, namely 16-Fr (23 mm), 18-Fr (26 mm) and 20-Fr (29 mm).
It has been shown that the modification of the delivery system led to a significant reduction in the mean sheath size with the use of S3 (14.3-Fr) compared with SAPIEN XT (18.1-Fr).Citation41 Consequently, major vascular complications have been reduced from 8.9% using SAPIEN XT to 5.1% with S3.Citation22 This is of high clinical relevance, as vascular complications likely affect clinical outcome with higher mortality and morbidity, longer hospital stays and increased costs.Citation42–Citation44 Accordingly, the average hospital length of stay was significantly longer for patients with major (16 days) and minor (11 days) vascular complications compared with those without (6 days).Citation45 Altogether, the reduction of vascular complications by a reduction in sheath sizes is a major driver enabling the current trend toward a simplification of the procedure.Citation46,Citation47
Cerebrovascular events
CVE belong to the most dreadful complications of TAVI and have a decisive impact on mortality, morbidity and quality of life.Citation51,Citation52 According to a large meta-analysis including >70,000 patients from 64 studies, 3.3% experienced a CVE during or after TAVI. Depending on the timing, several factors have been associated with an increased risk for CVE. The majority (54%) occurs within the first 24 hours of the TAVI procedure. Most likely, embolic mechanisms account for these acute CVE, and balloon post-dilatation and valve dislodgement/embolization belong to the major predictors in this phase. In contrast, new-onset atrial fibrillation (NOAF) seems to be the major predictor of CVE during the subacute phase after TAVI (<30 days).Citation53
With the S3 THV, the rate of CVE has been comparatively low (1.9%).Citation22 This reduced rate of CVE with the S3 THV in line with a general decrease in CVE in TAVI in recent years, most likely reflects not only technical advances in THV technology and delivery systems but also increased operator experience. Potential differences in thrombogenicity as well as platelet activation and coagulation of current THV and different deployment mechanisms are discussed as additional contributors to CVE.Citation54,Citation55 Therefore, due to the devastating character of CVE, optimal periprocedural pharmacotherapy and prevention and optimal medical treatment of NOAF have to be further refined. Furthermore, ongoing trials regarding the optimal medical treatment after TAVI, such as ATLAN-TIS (NCT02664649), ENVISAGE (NCT02943785) and GALILEO (NCT02556203), are awaited with great interest.
Competing devices and procedural aspects
THV systems have usually been categorized according to the deployment mechanism, as either balloon-expandable and self-expanding or mechanically expanding. Although direct randomized comparisons of both technologies are scarce, both have been used in large registries with good clinical outcome.Citation58,Citation59 Historically, mainly the SAPIEN and CoreValve THV families have been compared, whereas nowadays other new THV designs and deployment systems are available, including the self-expanding ACURATE neo™ (Boston Scientific, Marlborough, MA, USA) and Portico™ (Abbott, Saint Paul, MN, USA) valves as well as the mechanically expanded Lotus valve (Boston Scientific). Until data from ongoing RCTs, such as SCOPE I, SCOPE II and SOLVE-TAVI, are available, the CHOICE trial remains the only RCT comparing two THV designs.Citation60,Citation61 In the CHOICE trial, although device success rates were higher with the balloon-expandable SAPIEN XT valve, there was no difference in clinical outcomes after 1 year in a cohort of high-risk patients.Citation61
Hence, with data from RCTs pending, it appears that the majority of patients can be treated safely using both types of THVs with comparable clinical outcome. The MoRENA registry, a large multicenter registry, including 1,121 patients treated with either the balloon-expandable S3 or the self-expanding ACURATE neo (NEO) THV, showed similar procedural and clinical results with both devices,Citation11 which in the case of NEO were in line with data from a large post-market registry.Citation62 Also, in another non-randomized comparison of the S3 THV with the mechanically expanding Lotus THV, comparable results regarding safety were achieved, albeit with a considerably higher PPI rate with the Lotus THV.Citation63
Despite these comparable outcome data, there are certain putative advantages of each deployment mechanism. Compared with S3, NEO was associated with less PPI (9.9% vs 15.5%) and less elevated gradients after TAVI (3.2% vs 6.9%) but showed more moderate or severe PVL (4.8% vs 1.8%).Citation11 Whether certain anatomical features and baseline risk factors, including calcified anatomies, eccentric aortic annuli and pre-existing RBBB, may favor one over the other THV design remains to be addressed by future research.
In line with this notion, a recent study has shown that THV with higher radial force, such as the S3, may have advantages as compared to devices with lower radial force in calcified anatomies.Citation64 Being one of the most used THV in the field, several attempts have been made to simplify and to further reduce the periprocedural risk of the TAVI procedure. With regard to the S3 THV, one important factor is the possibility of direct implantation by omitting prior balloon valvuloplasty. This approach avoids rapid ventricular pacing and may result in a reduction of adverse events, such as annular rupture and CA, and has been successfully applied in a series of patients with no apparent downside in safety.Citation65
Pending evidence from ongoing RCT will further clarify a potential role of patient-tailored THV therapy for individual patients based on anatomical features, baseline risk factors and comorbidities and may further optimize clinical outcome and reduce adverse events after TAVI.
Perspectives
With growing number of TAVI procedures and its widespread application, standardized pre-procedural diagnostic algorithms and intra-procedural steps have been established, resulting in a simplified procedure. With high procedural success rates and reduced complications, an expansion of TAVI to lower-risk patients as well as to specific subgroups, such as patients with degenerated bioprostheses (“valve-in-valve TAVI”) or bicuspid aortic valves, is currently underway.
As the S3 THV has only been in use in Europe for 4 years, data regarding long-term performance are limited so far. In general, long-term data with early-generation devices are encouraging with good bioprosthetic valve function up to 7 years after TAVI.Citation16,Citation66–Citation68 However, due to the relatively high competing risk of death with mortality rates up to 76% after 7 years,Citation68 it is difficult to determine the exact rate of structural valve degeneration. Long-term data in intermediate-risk patients and continuous follow-up of survivors are needed to clarify this issue.
Furthermore, long-term results will shed light into the uncertainty of long-term durability of the S3 THV. Currently, the S3 THV represents one of the most widely used THVs in the field, and accumulating evidence and experience show excellent clinical results. With the successor of the S3 THV, the S3 Ultra, already at the horizon (NCT03471065), we hope to further our understanding of this device in order to offer the best possible care to our patients.
Disclosure
TR reports no conflicts of interest relevant to this work. JB has received proctor fees from Boston Scientific. HM has received proctor fees or speaker honoraria from Abbott, Biotronik, Edwards Lifesciences, Symetis SA and SJM. OH received proctor fees from Boston Scientific and minor congress support from Edwards Lifesciences. The authors report no other conflicts of interest in this work.
References
- IungBBaronGButchartEGA prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart DiseaseEur Heart J200324131231124312831818
- GaedeLBlumensteinJLiebetrauCOutcome after transvascular transcatheter aortic valve implantation in 2016Eur Heart J201839866767529228149
- VahlTPKodaliSKLeonMBTranscatheter Aortic Valve Replacement 2016: A Modern-Day “Through the Looking-Glass” AdventureJ Am Coll Cardiol201667121472148727012409
- BaumgartnerHFalkVBaxJJESC/EACTS Guidelines for the Management of Valvular Heart DiseaseRev Esp Cardiol2017201871110
- NishimuraRAOttoCMBonowROAHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice GuidelinesCirculation201713525e1159e119528298458
- ReardonMJvan MieghemNMPopmaJJSurgical or Transcatheter Aortic-Valve Replacement in Intermediate-Risk PatientsN Engl J Med2017376141321133128304219
- AndersenHRKnudsenLLHasenkamJMTransluminal implantation of artificial heart valves. Description of a new expandable aortic valve and initial results with implantation by catheter technique in closed chest pigsEur Heart J19921357047081618213
- WebbJGerosaGLefèvreTMulticenter evaluation of a next-generation balloon-expandable transcatheter aortic valveJ Am Coll Cardiol201464212235224325456759
- KodaliSThouraniVHWhiteJEarly clinical and echocardiographic outcomes after SAPIEN 3 transcatheter aortic valve replacement in inoperable, high-risk and intermediate-risk patients with aortic stenosisEur Heart J201637282252226227190101
- WendlerOSchymikGTreedeHSOURCE 3 Registry: Design and 30-Day Results of the European Postapproval Registry of the Latest Generation of the SAPIEN 3 Transcatheter Heart ValveCirculation2017135121123113228104716
- HusserOPellegriniCKesslerTOutcomes After Transcatheter Aortic Valve Replacement Using a Novel Balloon-Expandable Trans-catheter Heart Valve: A Single-Center ExperienceJACC Cardiovasc Interv20158141809181626718512
- HerrmannHCThouraniVHKodaliSKOne-Year Clinical Outcomes With SAPIEN 3 Transcatheter Aortic Valve Replacement in High-Risk and Inoperable Patients With Severe Aortic StenosisCirculation2016134213014027400898
- SmithCRLeonMBMackMJTranscatheter versus surgical aortic-valve replacement in high-risk patientsN Engl J Med2011364232187219821639811
- LeonMBSmithCRMackMTranscatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgeryN Engl J Med2010363171597160720961243
- GénéreuxPHeadSJvan MieghemNMClinical outcomes after transcatheter aortic valve replacement using valve academic research consortium definitions: a weighted meta-analysis of 3,519 patients from 16 studiesJ Am Coll Cardiol201259252317232622503058
- MackMJLeonMBSmithCR5-year outcomes of transcatheter aortic valve replacement or surgical aortic valve replacement for high surgical risk patients with aortic stenosis (PARTNER 1): a randomised controlled trialLancet201538599862477248425788234
- KodaliSKWilliamsMRSmithCRTwo-year outcomes after transcatheter or surgical aortic-valve replacementN Engl J Med2012366181686169522443479
- SeiffertMFujitaBAvanesovMDevice landing zone calcification and its impact on residual regurgitation after transcatheter aortic valve implantation with different devicesEur Heart J Cardiovasc Imaging201617557658426160399
- BuzzattiNMaisanoFLatibAComputed tomography-based evaluation of aortic annulus, prosthesis size and impact on early residual aortic regurgitation after transcatheter aortic valve implantationEur J Cardiothorac Surg201343143505151 discussion 50–1
- HusserOHolzamerAReschMProsthesis sizing for transcatheter aortic valve implantation--comparison of three dimensional trans-esophageal echocardiography with multislice computed tomographyInt J Cardiol201316843431343823688431
- BockschWGrossmannBGeislerTClinical outcome and paravalvular leakage of the new balloon-expandable Edwards Sapien 3 valve in comparison to its predecessor model (Edwards Sapien XT) in patients undergoing transfemoral aortic valve replacementCatheter Cardiovasc Interv201688346647527184353
- AndoTBriasoulisAHolmesAATaubCCTakagiHAfonsoLSapien 3 versus Sapien XT prosthetic valves in transcatheter aortic valve implantation: A meta-analysisInt J Cardiol201622047247827390972
- Jerez-ValeroMUrenaMWebbJGClinical impact of aortic regurgitation after transcatheter aortic valve replacement: insights into the degree and acuteness of presentationJACC Cardiovasc Interv2014791022103225234675
- BinderRKStorteckySHegDProcedural Results and Clinical Outcomes of Transcatheter Aortic Valve Implantation in Switzerland: An Observational Cohort Study of Sapien 3 Versus Sapien XT Trans-catheter Heart ValvesCirc Cardiovasc Interv2015810
- YangTHWebbJGBlankePIncidence and severity of paravalvular aortic regurgitation with multidetector computed tomography nominal area oversizing or undersizing after transcatheter heart valve replacement with the Sapien 3: a comparison with the Sapien XTJACC Cardiovasc Interv20158346247125790764
- HouthuizenPvan der BoonRMUrenaMOccurrence, fate and consequences of ventricular conduction abnormalities after transcatheter aortic valve implantationEuroIntervention20149101142115024273252
- HoffmannRHerpertzRLotfipourSImpact of a new conduction defect after transcatheter aortic valve implantation on left ventricular functionJACC Cardiovasc Interv20125121257126323257374
- TzikasAvan DalenBMvan MieghemNMFrequency of conduction abnormalities after transcatheter aortic valve implantation with the Medtronic-CoreValve and the effect on left ventricular ejection fractionAm J Cardiol2011107228528921129715
- NazifTMWilliamsMRHahnRTClinical implications of new-onset left bundle branch block after transcatheter aortic valve replacement: analysis of the PARTNER experienceEur Heart J201435241599160724179072
- NazifTMDizonJMHahnRTPredictors and clinical outcomes of permanent pacemaker implantation after transcatheter aortic valve replacement: the PARTNER (Placement of AoRtic TraNscathetER Valves) trial and registryJACC Cardiovasc Interv201581 Pt A606925616819
- GiustinoGvan der BoonRMMolina-Martin de NicolasJImpact of permanent pacemaker on mortality after transcatheter aortic valve implantation: the PRAGMATIC (Pooled Rotterdam-Milan-Toulouse in Collaboration) Pacemaker substudyEuroIntervention20161291185119327753605
- RegueiroAAbdul-Jawad AltisentOdel TrigoMImpact of New-Onset Left Bundle Branch Block and Periprocedural Permanent Pacemaker Implantation on Clinical Outcomes in Patients Undergoing Transcatheter Aortic Valve Replacement: A Systematic Review and Meta-AnalysisCirc Cardiovasc Interv201695e00363527169577
- DizonJMNazifTMHessPLChronic pacing and adverse outcomes after transcatheter aortic valve implantationHeart2015101201665167126261157
- HusserOKesslerTBurgdorfCConduction Abnormalities and Pacemaker Implantations After SAPIEN 3 Vs SAPIEN XT Prosthesis Aortic Valve ImplantationRev Esp Cardiol201669214114826542175
- NijhoffFAbawiMAgostoniPRamjankhanFZDoevendansPAStellaPRTranscatheter aortic valve implantation with the new balloon-expandable Sapien 3 versus Sapien XT valve system: a propensity score-matched single-center comparisonCirc Cardiovasc Interv201586e00240826033967
- SiontisGCJüniPPilgrimTPredictors of permanent pacemaker implantation in patients with severe aortic stenosis undergoing TAVR: a meta-analysisJ Am Coll Cardiol201464212914025011716
- TarantiniGMojoliMPuritaPUnravelling the (arte)fact of increased pacemaker rate with the Edwards SAPIEN 3 valveEuroIntervention201511334335025405801
- HusserOPellegriniCKesslerTPredictors of Permanent Pacemaker Implantations and New-Onset Conduction Abnormalities With the SAPIEN 3 Balloon-Expandable Transcatheter Heart ValveJACC Cardiovasc Interv20169324425426847116
- ToggweilerSLeipsicJBinderRKManagement of vascular access in transcatheter aortic valve replacement: part 2: Vascular complicationsJACC Cardiovasc Interv20136876777623968697
- BarbantiMWebbJGGilardMCapodannoDTamburinoCTranscatheter aortic valve implantation in 2017: state of the artEuroIntervention201713AAAA11AA2128942382
- AraiTLefèvreTHovasseTComparison of Edwards SAPIEN 3 versus SAPIEN XT in transfemoral transcatheter aortic valve implantation: Difference of valve selection in the real worldJ Cardiol201769356556927288330
- HayashidaKLefèvreTChevalierBTransfemoral aortic valve implantation new criteria to predict vascular complicationsJACC Cardiovasc Interv20114885185821851897
- van KesterenFvan MourikMSVendrikJIncidence, Predictors, and Impact of Vascular Complications After Transfemoral Transcatheter Aortic Valve Implantation With the SAPIEN 3 ProsthesisAm J Cardiol2018121101231123829703437
- DucrocqGFrancisFSerfatyJMVascular complications of transfemoral aortic valve implantation with the Edwards SAPIEN prosthesis: incidence and impact on outcomeEuroIntervention20105666667220142216
- ToggweilerSGurvitchRLeipsicJPercutaneous aortic valve replacement: vascular outcomes with a fully percutaneous procedureJ Am Coll Cardiol201259211311822222073
- BabaliarosVDevireddyCLerakisSComparison of transfemoral transcatheter aortic valve replacement performed in the catheterization laboratory (minimalist approach) versus hybrid operating room (standard approach): outcomes and cost analysisJACC Cardiovasc Interv20147889890425086843
- HusserOFujitaBHengstenbergCConscious Sedation Versus General Anesthesia in Transcatheter Aortic Valve Replacement: The German Aortic Valve RegistryJACC Cardiovasc Interv201811656757829566803
- JochheimDZadroznyMTheissHAortic regurgitation with second versus third-generation balloon-expandable prostheses in patients undergoing transcatheter aortic valve implantationEuroIntervention201511221422026093840
- SawayaFJSpazianoMLefèvreTComparison between the SAPIEN S3 and the SAPIEN XT transcatheter heart valves: A single-center experienceWorld J Cardiol201681273574528070241
- SeegerJGonskaBRottbauerWWöhrleJNew generation devices for transfemoral transcatheter aortic valve replacement are superior compared with last generation devices with respect to VARC-2 outcomeCardiovasc Interv Ther201833324725528643214
- AuffretVRegueiroAdel TrigoMPredictors of Early Cerebrovascular Events in Patients With Aortic Stenosis Undergoing Transcatheter Aortic Valve ReplacementJ Am Coll Cardiol201668767368427515325
- JochheimDZadroznyMRicardIPredictors of cerebrovascular events at mid-term after transcatheter aortic valve implantation - Results from EVERY-TAVI registryInt J Cardiol201724410611128784441
- Nombela-FrancoLWebbJGde JaegerePPTiming, predictive factors, and prognostic value of cerebrovascular events in a large cohort of patients undergoing transcatheter aortic valve implantationCirculation2012126253041305323149669
- Amat-SantosIJRodés-CabauJUrenaMIncidence, predictive factors, and prognostic value of new-onset atrial fibrillation following transcatheter aortic valve implantationJ Am Coll Cardiol201259217818822177537
- StorteckySWindeckerSStroke: an infrequent but devastating complication in cardiovascular interventionsCirculation2012126252921292423248061
- Amat-SantosIJDahouAWebbJComparison of hemodynamic performance of the balloon-expandable SAPIEN 3 versus SAPIEN XT transcatheter valveAm J Cardiol201411471075108225132330
- de Torres-AlbaFKaleschkeGDillerGPChanges in the pacemaker rate after transition from Edwards SAPIEN XT to SAPIEN 3 transcatheter aortic valve implantation: the critical role of valve implantation heightJACC Cardiovasc Interv20169880581327017367
- HammCWMöllmannHHolzheyDThe German Aortic Valve Registry (GARY): in-hospital outcomeEur Heart J201435241588159824022003
- GilardMEltchaninoffHIungBRegistry of transcatheter aortic-valve implantation in high-risk patientsN Engl J Med2012366181705171522551129
- Abdel-WahabMMehilliJFrerkerCComparison of balloon-expandable vs self-expandable valves in patients undergoing transcatheter aortic valve replacement: the CHOICE randomized clinical trialJAMA2014311151503151424682026
- Abdel-WahabMNeumannFJMehilliJ1-Year Outcomes After Transcatheter Aortic Valve Replacement With Balloon-Expandable Versus Self-Expandable Valves: Results From the CHOICE Randomized Clinical TrialJ Am Coll Cardiol201566779180026271061
- MöllmannHWaltherTSiqueiraDTransfemoral TAVI using the self-expanding ACURATE neo prosthesis: one-year outcomes of the multicentre “CE-approval cohort”EuroIntervention2017139e1040e104628804056
- PilgrimTStorteckySNietlispachFRepositionable Versus Balloon-Expandable Devices for Transcatheter Aortic Valve Implantation in Patients With Aortic StenosisJ Am Heart Assoc2016511e00408827856487
- KimWKBlumensteinJLiebetrauCComparison of outcomes using balloon-expandable versus self-expanding transcatheter prostheses according to the extent of aortic valve calcificationClin Res Cardiol201710612995100428795259
- KimWKPrazFBlumensteinJTransfemoral aortic valve implantation of Edwards SAPIEN 3 without predilatationCatheter Cardiovasc Interv2017891E38E4326945934
- ToggweilerSHumphriesKHLeeM5-year outcome after transcatheter aortic valve implantationJ Am Coll Cardiol201361441341923265333
- BarbantiMPetronioASEttoriF5-Year Outcomes After Transcatheter Aortic Valve Implantation With CoreValve ProsthesisJACC Cardiovasc Interv2015881084109126117458
- DeutschMAErlebachMBurriMBeyond the five-year horizon: long-term outcome of high-risk and inoperable patients undergoing TAVR with first-generation devicesEuroIntervention2018141414929581084
- PellegriniCKimWKHolzamerAMulticenter Evaluation of Prosthesis Oversizing of the SAPIEN 3 Transcatheter Heart Valve. Impact on Device Failure and New Pacemaker ImplantationsRev Esp Cardiol Epub2018714
- https://www.edwards.com [homepage on the Internet]. transcatheter heart valve. Available from: https://www.edwards.com/gb/devices/heart-valves/transcatheter.
