Abstract
Background
In spite of treatment advances, HIV infection is associated with cognitive deficits. This is even more important as many persons with HIV infection age and experience age-related cognitive impairments. Both computer-based cognitive training and transcranial direct current stimulation (tDCS) have shown promise as interventions to improve cognitive function. In this study, we investigate the acceptability and efficacy of cognitive training with and without tDCS in older persons with HIV.
Patients and methods
In this single-blind randomized study, participants were 14 individuals of whom 11 completed study procedures (mean age =51.5 years; nine men and two women) with HIV-related mild neurocognitive disorder. Participants completed a battery of neuropsychological and self-report measures and then six 20-minute cognitive training sessions while receiving either active or sham anodal tDCS over the left dorsolateral prefrontal cortex. After training, participants completed the same measures. Success of the blind and participant reactions were assessed during a final interview. Assessments were completed by an assessor blind to treatment assignment. Pre- and post-training changes were evaluated via analysis of covariance yielding estimates of effect size.
Results
All participants believed that they had been assigned to active treatment; nine of the 11 believed that the intervention had improved their cognitive functioning. Both participants who felt the intervention was ineffective were assigned to the sham condition. None of the planned tested interactions of time with treatment was significant, but 12 of 13 favored tDCS (P=0.08). All participants indicated that they would participate in similar studies in the future.
Conclusion
Results show that both cognitive training via computer game playing and tDCS were well accepted by older persons with HIV infection. Results are suggestive that tDCS may improve cognitive function in persons with HIV infection. Further study of tDCS as an intervention for HIV-related cognitive dysfunction is warranted.
Introduction
In spite of advances in the treatment of HIV infection through the development of combination antiretroviral treatments, individuals with HIV infection, even those with nondetectable viral loads, continue to develop HIV-related cognitive deficits.Citation1 These cognitive deficits are significant in light of their impact on patients’ functional status,Citation2–Citation4 medication adherence,Citation4–Citation6 and quality of life.Citation7–Citation10 Cognitive dysfunction may have an even greater impact on those aging with HIV, who face both HIV- and age-related cognitive changes.Citation11,Citation12
Although significant, treatments for HIV-related cognitive deficits are limited. Drug studies have shown that stimulant medications may improve some symptoms of HIV-related cognitive impairment,Citation13 but their usefulness is limited by their abuse potential and side effects.Citation14,Citation15 Other investigators have argued for the utility of computer-delivered cognitive training interventions,Citation16 but the software is not always affordable for indigent patients or those on limited budgets. Further, many programs developed specifically for cognitive training do not have high levels of inherent interest, reducing their uptake outside of compensated research studies.
An alternative strategy to expensive commercial software may be more readily available computer games.Citation17 Developers of cognitive training software programs have often tried to include game elements into their softwareCitation18 to enhance the inherent interest of the training programs, but a vast number of readily available games have already demonstrated their commercial viability. This type of viability stems from the games having high levels of intrinsic interest, play that engages the user, and online social communities. Games such as first-person shooters (in which the participant uses some form of gun to target enemies) have been shown to improve attention and reaction time,Citation18 but their acceptability to the user is limited by the violent nature of their content, which at times includes simulations of violence and gore.Citation18,Citation19
An alternative to first-person shooters would be a car racing game, which also requires attention and cognitive speed but may be more generally acceptable to users. One study showed that a purpose-built car racing game improved cognitive function in older persons.Citation20 Others have also argued for the effectiveness of commercially available games in improving and sustaining cognitive function in older adults.Citation17,Citation21–Citation24
Commercial games are successful precisely because of their ability to engage and sustain users’ interest. A number of games, for example, have millions of engaged users actively involved in online communities. Games may involve team play and some have worldwide tournaments with thousands of users. One group has shown that a commercially available game requiring psychomotor speed was more likely to result in long-term use in users compared to a commercial cognitive training computer program.Citation21,Citation22 Computer games have been shown to have sustained cognitive training effects that may transfer to other domainsCitation25 including cognitive control in older adults.Citation20
In addition to cognitive training, many studies have shown that transcranial direct current stimulation (tDCS) can enhance cognitive function. tDCS involves the application of very small currents (1–2 mA) using a regulated direct current source, usually via sponge electrodes placed over relevant portions of the brain. Studies of tDCS have shown enhancement of specific aspects of cognitive function, including verbal problem solving,Citation26 working memory,Citation27–Citation29 and learning in various contexts including in a computer-based threat detection simulationCitation30 and object location memory in the elderly.Citation31
In addition to effects on cognition, many studies have also shown that tDCS may be an effective adjunct treatment for depression, with individuals receiving both antidepressants and tDCS showing greater improvements than those receiving antidepressant medications alone.Citation32
Although the precise mechanism by which tDCS produces effects on cognition and mood is unclear, one possible mechanism is particularly relevant to the treatment of individuals with HIV infection. Direct current stimulation of neurons in the motor cortex has been shown to stimulate the activity of brain-derived neurotrophic growth factor (BDNF) in that area,Citation33 and it is possible that similar effects occur elsewhere in the brain. BDNF is affected by HIV infectionCitation34,Citation35 and is related to both cognitive decline in older personsCitation36 and depression.Citation37 An intervention that might stimulate its production in older persons with HIV might be an important therapeutic intervention.
To date, no readily identifiable study has evaluated the usefulness of commercial games as a cognitive training strategy among persons with HIV infection nor has any study evaluated their effect combined with tDCS. The purposes of this study were to evaluate the feasibility and acceptability of game-based cognitive training intervention in older persons with HIV infection and to evaluate the acceptability and efficacy of the cognitive training intervention with and without active tDCS. We believed that individuals would find the computer game interesting and that those receiving active tDCS would show improvements in psychomotor speed. As the number of participants is small, analyses focus primarily on description of outcomes and effect size estimation rather than parametric hypothesis testing. We present preliminary data here from our first 11 participants.
Patients and methods
Game
In this study, we chose to use an off-the-shelf computer game that is inexpensive (free to download on some version of Windows™), was of an appropriate level of difficulty, and was widely popular, thus demonstrating its acceptability to potential users. GT Racing 2 (Gameloft SE, Paris, France) requires that individuals steer a simulated car over courses that include city streets, racetracks, and outside courses. Each course requires that the user achieve a basic level of proficiency before moving on to the next level. The initial difficulty level allows the game to provide considerable steering and braking assistance to the person playing, guaranteeing their ability to play the game and achieve at least some success. The game is visually attractive and provides a variety of courses that stimulate user interest. All participants were able to navigate successfully at least the first four courses of the game over six training sessions.
tDCS
Participants
Participants were individuals treated for HIV infection who reported cognitive difficulties and evidenced objective cognitive impairment in two neuropsychological domains. Exclusion criteria included factors that might expose individuals to increased risks if they participated in tDCS, such as history of seizures or bipolar disorder (there have been some reports of mania in studies of tDCS for depressionCitation32,Citation38). Individuals using an extensive list of psychotropic medications were excluded, as drugs in these classes have been shown to modify the effects of tDCS.Citation39 These included medications with serotonergic (many antidepressants), dopaminergic (stimulant medications, antipsychotics), or gamma-amino butyric acid actvitiy (benzodiazepines). Left-handed participants were excluded as well. Participants were also asked about game-playing experience; none reported substantial personal computer (PC) or console gaming experience, although one participant indicated that he played games on his phone occasionally.
Procedures
Recruitment and eligibility determination
Individuals were initially recruited from participants in a previous study during which they completed a battery of cognitive measures, allowing us to identify persons likely to meet entrance criteria. Individuals were also recruited from several local organizations providing services to individuals treated for HIV infection. Participants were first screened by telephone for the presence of subjective cognitive impairments using questions developed by the European AIDS Clinical SocietyCitation40 as well as for medication use, ability to bring laboratory results, and interest in participating in a study of cognitive training and tDCS. All participants were required to be in active treatment for HIV and stable on their current regimen of antiretroviral medications for 1 month. Persons who met initial inclusion and exclusion criteria were scheduled for an in-person visit to determine final eligibility.
At the in-person visit, participants completed a brief battery of neuropsychological measures selected to assess domains commonly affected in persons with HIV infection. These included attention and working memory using the Digit Span subtest of the Wechsler Adult Intelligence Scale, 4th edition, or WAIS-IV,Citation41 which includes assessments of digit span forward, backward, and a number and letter sequencing task. Measures also assessed executive function and mental flexibility with the Trail Making Test, Parts A and B,Citation42 verbal learning and memory with the Hopkins Verbal Learning Test – Revised or HVLT-R,Citation43 and psychomotor speed with the Grooved Pegboard Test.Citation44 Individuals were considered eligible if their performance in two of the domains was one standard deviation (SD) or more below the mean according to normative data. Participants were also required to report subjective cognitive difficulties in at least one of the following areas: 1) memory, 2) cognitive slowing, or 3) problems in attending.Citation40 They thus fulfilled Frascati criteriaCitation45 for mild neurocognitive disorder. All participants were currently in active treatment for HIV, and routine monitoring of treatment response and immune status is part of their care. We required that participants furnish recent laboratory results at study entry, as well as bring all their medications to the eligibility visit, so that we could examine pill bottles and verify that they met protocol eligibility requirements.
Individuals who met entry criteria then completed the Patient’s Assessment of Own Functioning (PAOF),Citation46,Citation47 a more extensive measure of self-reported cognitive difficulties across language, perception, and memory previously used in studies of HIV-related cognitive impairment,Citation47 as well as the Center for Epidemiological Studies Depression (CESD) scale,Citation48 a self-report measure of depressive symptoms. Participants completed these measures using automated computer-assisted self-interview (ACASI) software that only required touching a computer’s screen to record responses. Participants were compensated with US $40 for the first and last sessions and $20 for each training visit. After completing baseline procedures, participants were scheduled for the first training visit.
Computer-based cognitive training
At the first visit, individuals were assigned to treatment condition via a predetermined computer-generated schedule with randomized blocks of four. Individuals were oriented to tDCS procedures and the computer game controller (standard Xbox game controller connected via USB interface to a PC running the Windows® 10 operating system). The investigator sat at another desk behind and to the participant’s left so that the tDCS device and the investigator recording performance were not visible during training. The investigator controlled the computer and the game via a wireless mouse. All individuals participated in tDCS anode placement over the left dorsolateral prefrontal cortex and cathode over the right supraorbital area, with locations determined according to the 10–20 placement system.Citation49 Electrodes were 5×5 cm sponges (Soterix EASY-Pads; Soterix Medical, New York, NY, USA). They were moistened with ~6 cc of sterile saline and held in place with an elastic band. Current was supplied using a Chattanooga Ionto iontophoresis device (DJO International, Surrey, England) with flat carbon electrodes inserted into the dual riveted sponges to improve the uniformity of current density.
Participants were informed that they might feel nothing or minor itching or burning at the onset of tDCS and that the feeling might continue or go away during the training session. This procedure has previously been successfully used to blind research participants to active vs sham tDCS.Citation32 Participants were encouraged to attend to the computer screen as the game and tDCS were initiated. For individuals assigned to active treatment, tDCS was begun and continued for 20 minutes at a current of 1.5 mA. For individuals assigned to sham tDCS, the tDCS device was turned on and the current allowed to ramp up to 1.5 mA over a period of 30 seconds. The device was then turned off out of sight of the participant.
Participants were allowed to work through the game at their own pace, subject to its restrictions. For example, in order to progress through the game, participants had to finish a race in first, second, or third place or finish a course in a specified time prior to accessing the next course. Participants completed at least five trials of each course before progressing. At the conclusion of each training session, participants responded to three questions via ACASI that asked how they would rate their mental abilities, mood, and level of discomfort during that session. All participants completed six training sessions over the next 2 weeks, with most sessions completed with 1-day intervening between sessions (eg, Monday, Wednesday, and Friday).
All activities were completed within a 3-week interval. After the final training session, participants again completed the PAOF, CESD, and the neuropsychological battery administered by an assessor blind to treatment condition. The assessor also completed a final interview during which participants were asked to which group they had been assigned in order to assess the effectiveness of the blinding procedure. They were asked whether they believed the computer training and tDCS were helpful and whether they would participate in the future in a study of tDCS.
Human subjects approval and trial registration
This protocol was approved by the Institutional Review Board of Nova Southeastern University (protocol number 12031424F) and was registered on ClinicalTrials.gov (NCT02647645). All participants provided oral consent for screening procedures and written informed consent prior to randomization and treatment procedures.
Analyses
Data analyses were completed in several steps. Given the small number of participants, it was possible to inspect the data for extreme values, but inspection was supplemented by obtaining frequencies and descriptive statistics. Effects of covariates were evaluated in correlation analyses. Treatment effects were evaluated in analysis of covariance (ANCOVA) models yielding estimates of effect size and plots of estimated marginal means corrected for covariates. Our primary outcome measure was effect size estimates as we anticipated that the current sample size would not provide sufficient power to detect statistical significance.
Treatment effects were also evaluated through inspection of plots derived from ANCOVA models.
As depression can affect self-report of symptoms in persons with HIV infectionCitation2 as well as cognition,Citation2 we assessed the impact of changes in depression on the observed interaction between treatment group and time in post hoc analyses. The change in CESD score over time was calculated and used as a covariate in a subset of analyses to evaluate the effect of changes in depressive symptoms on cognitive measures and self-report of symptoms.
All analyses presented here were completed using the Statistical Package for the Social Sciences, 23rd edition (IBM/SPSS, Armonk, NY, USA).
Results
Demographic and educational data for each participant are presented in . We enrolled 14 individuals, 11 of whom completed all study procedures. Two participants completed the baseline study visit but withdrew prior to randomization as they lived some distance from the study site and felt driving to our research office three times a week for 2 weeks was impractical. Another participant completed baseline assessment and was randomized, but after several training visits was hospitalized for an unrelated health issue and could not complete study procedures in the 3-week period specified in our protocol. The average age of participants was 51.5 years (SD =4.71), and they had completed a wide range of years of education (6–15 years, mean age 11.18 years, SD =2.27). Two women and two Whites were participants so that the majority of participants in this study were African American men.
Table 1 Description of participants
Baseline and follow-up means and SDs for tests and the two self-report measures are presented in . The possible relations of covariates to cognitive variables were explored via parametric (Pearson) and nonparametric (Spearman) correlations. As correlations of age, gender, education, race, and immune status with cognitive variables were often substantial and judged to be potentially meaningful, we included them in ANCOVA models assessing differences in performance before and after training with or without tDCS. Although the number of covariates is substantial, especially in light of the small overall sample size, all showed relations to other variables and might reasonably be expected to be confounders of any evaluation of treatment effect. The complete table of nonparametric correlations is included as a data supplement to this paper ().
Table 2 Test and self-report data
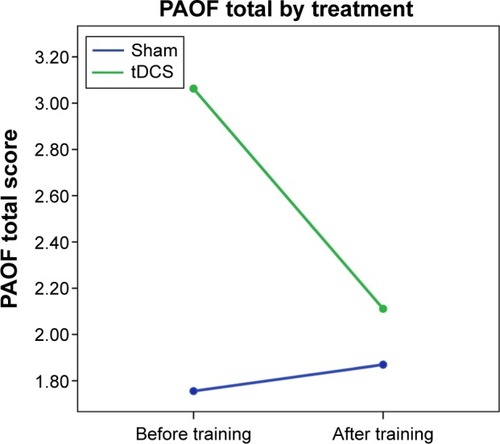
As we were primarily interested in exploration of preliminary results via graphing and effect sizes, repeated measures ANCOVA models for baseline and follow-up were created, with a specific focus on the extent to which the interactions between time and treatment condition might represent an effect of tDCS on cognitive outcomes. Examples of covariate-corrected baseline and follow-up changes for each group are presented in –. (higher scores reflect better performance) presents results for the HVLT total learning score, suggesting that persons in the tDCS score may have improved relatively more over the baseline assessment than did those in the sham group. (higher scores reflect worse performance) shows results for the Grooved Pegboard dominant hand time; in this instance, after taking covariates into account those in the sham group performed more poorly at the second assessment compared to those in the tDCS group. (lower scores indicate fewer complaints) shows changes in the PAOF total score over assessments. While the sham group reported modestly greater overall problems at the follow-up assessment, the figure suggests a substantial decrease in complaints for the tDCS group.
Figure 1 HVLT-R total by group and time.
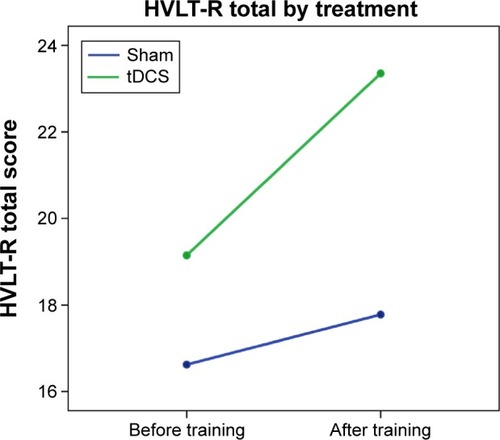
Figure 2 Grooved Pegboard dominant hand by group and time.
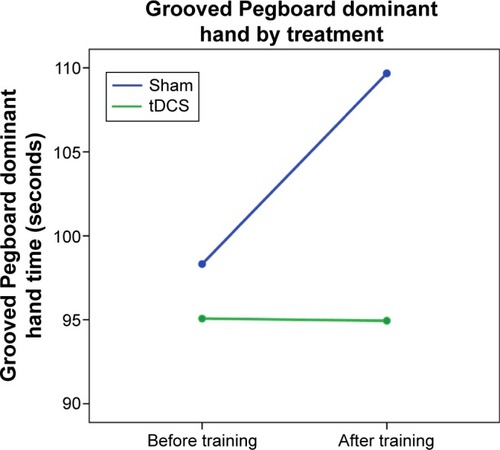
Figure 3 PAOF by group and time.
Effect sizes for the interactions of treatment group by time are presented in . Effect sizes are presented as partial eta squared and converted to the more widely used Cohen’s d. Effect sizes range from moderate to large when interpreted based on the guidelines suggested by Cohen.Citation50 The average of all effect sizes for cognitive measures (not including Trails A and the effects corrected for change in depression) was 1.28. When the negative effect for Trails A is included, the average is 0.99. Of the 13 planned estimates of treatment effect size, 12 were in the positive direction, suggesting a positive effect of tDCS (P=0.08).
Table 3 Effect sizes for the interaction of group by time
also includes effect size estimates for several cognitive measures we believed might be sensitive to changes in depression and the PAOF total score. In these models, change in depressive symptoms was included as a covariate. The inclusion of this variable reduced the effect size for the group by time interaction for the PAOF but increased it for cognitive measures.
Success of blind, and participant reactions
In order to evaluate how successful the blinding procedure was, an interviewer blind to participants’ treatment assignments asked them to which treatment group they believed that had been assigned. All participants indicated that they believed they had been assigned to the active tDCS group. We also asked them whether they believed the intervention had been helpful to them. Nine of the 11 participants stated they felt the intervention had been helpful to them (including several assigned to the sham condition) while two participants stated they were not sure or believed it had not been helpful. Both were assigned to sham treatment. All participants stated they would participate in a similar study in the future. Both men and women indicated that they enjoyed the car racing game, with several participants inquiring about how they could obtain the game in order to continue playing it.
Discussion
The purpose of this study is to explore the acceptability and potential efficacy of computer-delivered cognitive training using commercial gaming software with or without tDCS in persons with HIV infection. Given our small sample size, our analyses evaluated the treatment effects by assessing effect sizes and inspecting graphs of covariate-corrected baseline and follow-up performance. Cognitive testing before and after training suggests the presence of a positive effect of tDCS on learning, memory, and motor speed compared to cognitive training alone. Of the 13 effect sizes presented in , 12 were positive in showing an advantage for the active tDCS group. These findings are illustrated in the figures, which show either relatively greater improvement () or lack of decline () over assessments in those receiving tDCS. In addition, objective findings are mirrored in participants’ self-report of cognitive difficulties ().
Observed changes in performance on working memory tasks such as Digit Span Backward and Sequencing are consistent with other studies that have found improvement in working memory after left dorsolateral prefrontal cortex tDCS stimulation,Citation27 including a study in patients with Parkinson’s disease.Citation51 However, it should be acknowledged that positive treatment effects have not been obtained in all studies.Citation52
Although we did not specifically recruit participants who might be suffering from depression, mean CESD scores for both groups were in a range consistent with clinically significant disturbance of mood. It is thus noteworthy that the interaction of group by time for this measure, while not statistically significant, was small to medium based on Cohen’s interpretive guidelines.Citation50 As tDCS has been successfully used as an adjunctive treatment for depression,Citation53,Citation54 this finding is also consistent with previous literature on tDCS in other patient groups as well as results of a small trial in persons with HIV infection.Citation55
Strengths of this study include the success of the single-blind procedure, as all participants indicated they believed that had been in the active treatment group. All baseline and outcome data were collected either by an assessor blind to participants’ treatment assignment or by way of ACASI, again reducing the likelihood of experimenter bias in these results. We collected information about participants’ subjective experience of the interventions as well; their comments supported objective test results. Our participants were in many respects typical of those who might be expected to benefit from cognitive interventions based on their age, education, and cognitive deficits.
Limitations of this study include the small sample size and single-blind design. The sample size reflects that this is a pilot study targeted at determining whether further study is warranted and whether the interventions would be acceptable to older persons with HIV infection. Lack of a true double-blind design raises the concern about bias induced by the investigators. We did a number of things to decrease the likelihood of experimenter effects, including positioning the experimenter and the tDCS device out of sight of the participant during stimulation, providing neutral information about the likelihood of experiencing physical sensations from stimulation, and collecting rating scale data via ACASI with the investigator out of the room. Follow-up cognitive testing and interviews were completed by an assessor blind to treatment condition. While these procedures reduced the likelihood of experimenter bias, they cannot eliminate it. Participants included small numbers of women and Whites, creating another potential source of bias. As the purpose of this study was to assess the acceptability and possible efficacy of training with and without tDCS, we did not include a no-treatment control condition. Thus, we cannot evaluate the effect of computer training by itself, as all participants receive the same cognitive training intervention. As noted, our sample size is quite small and we controlled for a number of covariates. Since the covariates were all factors that might reasonably be related to performance on outcome measures, such as age, gender, education, and immune status, and thus confound any evaluation of the effect of treatment, we believe this was an appropriate strategy. However, it must be acknowledged that our assessment is based on a small sample.
An anomalous finding was the change in Trails A performance, in which the active the performance of the tDCS group actually declined while that of the sham group improved. Other investigators have evaluated the effect of tDCS on Trail Making Test performance. Fagerlund et alCitation56 assessed the effect of anodal stimulation over M1 (somewhat posterior to the site of stimulation in this study) and found no effect on Trail Making test performance. In a study of tDCS for depression, Brunoni et alCitation57 showed a modest differential in Trails A improvement over time favoring sham treatment (sham improved 8.1 seconds while the active group improved 3.2 seconds). We can speculate that this finding may simply be a random outcome, especially in light of otherwise consistent results favoring tDCS.
Results of this pilot study thus provide suggestive evidence for the efficacy of tDCS combined with computer-delivered cognitive training in improving cognitive function in persons with HIV-related cognitive deficits. Although our small sample size limited the power of this study to detect statistically significant treatment effects, most effect sizes were moderate to large, and all but one were in the direction of a positive effect for tDCS. Objective findings are reflected in participants’ own estimation of the effectiveness of the interventions, although it should be noted that some individuals who received sham tDCS also believed they had benefited from the intervention. This may reflect a nonspecific effect of simply participating in an intervention study or a positive effect of the cognitive training intervention. As we did not include a no-treatment control condition, this possibility cannot be evaluated.
Given the importance of cognitive deficits for affected persons’ functional status and quality of life as well as lack of effective alternative treatments, these results have potential clinical significance. Due to the limited scope of this study, we did not include measures of functional status so that it is not possible to know whether the observed changes in cognitive tasks had an impact on other outcomes directly related to everyday functioning, such as self-care, medication adherence, or driving. Future research should focus on assessing not only laboratory outcome measures but also outcomes with clearer real-world significance, such as instrumental activities of daily living and medication adherence.
Supplementary material
Table S1 Nonparametric correlations (Spearman’s rho) for covariates, treatment, and outcomes
Disclosure
The authors report no conflicts of interest in this work.
References
- HeatonRKCliffordDBFranklinDRJrCHARTER GroupHIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER StudyNeurology201075232087209621135382
- ThamesADBeckerBWMarcotteTDDepression, cognition, and self-appraisal of functional abilities in HIV: an examination of subjective appraisal versus objective performanceClin Neuropsychol201125222424321331979
- ThamesADKimMSBeckerBWMedication and finance management among HIV-infected adults: the impact of age and cognitionJ Clin Exp Neuropsychol201133220020920694873
- ThamesADArentoftARivera-MindtMHinkinCHFunctional disability in medication management and driving among individuals with HIV: a 1-year follow-up studyJ Clin Exp Neuropsychol2013351495823237014
- HinkinCHHardyDJMasonKIMedication adherence in HIV-infected adults: effect of patient age, cognitive status, and substance abuseAIDS200418suppl 1S19S25
- HinkinCHCastellonSADurvasulaRSMedication adherence among HIV+ adults: effects of cognitive dysfunction and regimen complexityNeurology200259121944195012499488
- DegrooteSVogelaersDVandijckDMWhat determines health-related quality of life among people living with HIV: an updated review of the literatureArch Public Health20147214025671112
- DegrooteSVogelaersDPVermeirPSocio-economic, behavioural, (neuro)psychological and clinical determinants of HRQoL in people living with HIV in Belgium: a pilot studyJ Int AIDS Soc2013161864324331754
- MooreRCFazeliPLJesteDVHIV Neurobehavioral Research Program (HNRP) GroupSuccessful cognitive aging and health-related quality of life in younger and older adults infected with HIVAIDS Behav20141861186119724633788
- TozziVBalestraPGalganiSNeurocognitive performance and quality of life in patients with HIV infectionAIDS Res Hum Retroviruses200319864365213678465
- ValcourVShikumaCShiramizuBHigher frequency of dementia in older HIV-1 individuals: the Hawaii aging with HIV-1 cohortNeurology200463582282715365130
- WendelkenLAValcourVImpact of HIV and aging on neuropsychological functionJ Neurovirol201218425626322528478
- HinkinCHCastellonSAHardyDJFarinpourRNewtonTSingerEMethylphenidate improves HIV-1-associated cognitive slowingJ Neuropsychiatry Clin Neurosci200113224825411449032
- WatkinsCCTreismanGJCognitive impairment in patients with AIDS – prevalence and severityHIV AIDS (Auckl)20157354725678819
- CodySLVanceDEThe neurobiology of HIV and its impact on cognitive reserve: a review of cognitive interventions for an aging populationNeurobiol Dis201692Pt B14415626776767
- VanceDEFazeliPLRossLAWadleyVGBallKKSpeed of processing training with middle-age and older adults with HIV: a pilot studyJ Assoc Nurses AIDS Care201223650051022579081
- ZelinskiEMReyesRCognitive benefits of computer games for older adultsGerontechnology20098422023525126043
- GreenCSSeitzARThe impacts of video games on cognition (and how the government can guide the industry)Policy Insights Behav Brain Sci201521101110
- WuSSpenceIPlaying shooter and driving videogames improves top-down guidance in visual searchAtten Percept Psychophys201375467368623460295
- AngueraJABoccanfusoJRintoulJLVideo game training enhances cognitive control in older adultsNature201350174659710124005416
- BelchiorPMarsiskeMSiscoSYamAMannWOlder adults’ engagement with a video game training programAct Adapt Aging201236426927923504652
- BelchiorPMarsiskeMLeiteWLYamAThomasKMannWOlder adults’ engagement during an intervention involving off-the-shelf videogameGames Health J20165315115627310479
- BavelierDGreenCSHanDHRenshawPFMerzenichMMGentileDABrains on video gamesNat Rev Neurosci2011121276376822095065
- BasakCBootWRVossMWKramerAFCan training in a real-time strategy video game attenuate cognitive decline in older adults?Psychol Aging200823476577719140648
- Cardoso-LeitePBavelierDVideo game play, attention, and learning: how to shape the development of attention and influence learning?Curr Opin Neurol201427218519124553464
- CerrutiCSchlaugGAnodal transcranial direct current stimulation of the prefrontal cortex enhances complex verbal associative thoughtJ Cogn Neurosci200921101980198718855556
- FregniFBoggioPSNitscheMAnodal transcranial direct current stimulation of prefrontal cortex enhances working memoryExp Brain Res20051661233015999258
- ZaehleTSandmannPThorneJDJanckeLHerrmannCSTranscranial direct current stimulation of the prefrontal cortex modulates working memory performance: combined behavioural and electrophysiological evidenceBMC Neurosci2011121221211016
- HeimrathKSandmannPBeckeAMullerNGZaehleTBehavioral and electrophysiological effects of transcranial direct current stimulation of the parietal cortex in a visuo-spatial working memory taskFront Psychiatry201235622723784
- ClarkVPCoffmanBAMayerARTDCS guided using fMRI significantly accelerates learning to identify concealed objectsNeuroimage201259111712821094258
- FloelASuttorpWKohlONon-invasive brain stimulation improves object-location learning in the elderlyNeurobiol Aging20123381682168921684040
- BrunoniARValiengoLBaccaroAThe sertraline vs. electrical current therapy for treating depression clinical study: results from a factorial, randomized, controlled trialJAMA Psychiatry201370438339123389323
- FritschBReisJMartinowichKDirect current stimulation promotes BDNF-dependent synaptic plasticity: potential implications for motor learningNeuron201066219820420434997
- AvdoshinaVBachisAMocchettiISynaptic dysfunction in human immunodeficiency virus type-1-positive subjects: inflammation or impaired neuronal plasticity?J Intern Med2013273545446523600400
- BachisAAvdoshinaVZeccaLParsadanianMMocchettiIHuman immunodeficiency virus type 1 alters brain-derived neurotrophic factor processing in neuronsJ Neurosci201232289477948422787033
- BuchmanASYuLBoylePASchneiderJADe JagerPLBennettDAHigher brain BDNF gene expression is associated with slower cognitive decline in older adultsNeurology201686873574126819457
- ShimizuEHashimotoKOkamuraNAlterations of serum levels of brain-derived neurotrophic factor (BDNF) in depressed patients with or without antidepressantsBiol Psychiatry2003541707512842310
- GalvezVAlonzoAMartinDMitchellPBSachdevPLooCKHypomania induction in a patient with bipolar II disorder by transcranial direct current stimulation (tDCS)J ECT201127325625821206371
- MedeirosLFde SouzaICVidorLPNeurobiological effects of transcranial direct current stimulation: a reviewFront Psychiatry2012311023293607
- European AIDS Clinical SocietyGuidelines 8.0BrusselsEuropean AIDS Clinical Society2015
- WechslerDManual for the Wechsler Adult Intelligence Scale – IVSan Antonio TXPearson Assessment2008
- LezakMNeuropsychological Assessment5th edNew YorkOxford University Press2004
- BrandtJBenedictRHHopkins Verbal Learning Test – Revised: Professional ManualOdessa, FLPsychological Assessment Resources2001
- Lafayette InstrumentGrooved Pegboard Test User InstructionsLafayette, INLafayette Instrument2002
- AntinoriAArendtGBeckerJTUpdated research nosology for HIV-associated neurocognitive disordersNeurology200769181789179917914061
- CheluneGJHeatonRKLehmanRAWNeuropsychological and personality correlates of patients’ complaints of disabilityGoldsteinGTarterREAdvances in Clinical Neuropsychology3New YorkSpringer198695126
- RourkeSBHalmanMHBasselCNeurocognitive complaints in HIV-infection and their relationship to depressive symptoms and neuropsychological functioningJ Clin Exp Neuropsychol199921673775610649531
- RadloffLSThe CES-D Scale: a self-report depression scale for research in the general populationAppl Psychol Measurement197713385401
- AcharyaJNHaniACheekJThirumalaPTsuchidaTNAmerican Clinical Neurophysiology Society guideline 2: guidelines for standard electrode position nomenclatureJ Clin Neurophysiol201633430831127482794
- CohenJStatistical Power Analysis for the Behavioral Sciences2nd edNew YorkRoutledge1988
- BoggioPSFerrucciRRigonattiSPEffects of transcranial direct current stimulation on working memory in patients with Parkinson’s diseaseJ Neurol Sci20062491313816843494
- TremblaySLepageJFLatulipe-LoiselleAFregniFPascual-LeoneATheoretHThe uncertain outcome of prefrontal tDCSBrain Stimul20147677378325456566
- NitscheMABoggioPSFregniFPascual-LeoneATreatment of depression with transcranial direct current stimulation (tDCS): a reviewExp Neurol20092191141919348793
- BrunoniARFerrucciRFregniFBoggioPSPrioriATranscranial direct current stimulation for the treatment of major depressive disorder: a summary of preclinical, clinical and translational findingsProg Neuropsychopharmacol Biol Psychiatry201239191622651961
- KnotkovaHRosedaleMStraussSMUsing transcranial direct current stimulation to treat depression in HIV-infected persons: the outcomes of a feasibility studyFront Psychiatry201235922719732
- FagerlundAJFreiliJLDanielsenTLAslaksenPMNo effect of 2 mA anodal tDCS over the M1 on performance and practice effect on Grooved Pegboard Test and Trail Making Test BeNeuro201524
- BrunoniARTortellaGBensenorIMLotufoPACarvalhoAFFregniFCognitive effects of transcranial direct current stimulation in depression: results from the SELECT-TDCS trial and insights for further clinical trialsJ Affect Disord2016202465227253216
