Abstract
Background
Schizophrenia is one of the most common devastating psychiatric disorders that negatively affects the quality of life and psychosocial functions. Its etiology involves the interplay of complex polygenic influences and environmental risk factors. Inflammatory markers are well-known etiological factors for psychiatric disorders, including schizophrenia.
Objective
The aim of this study was to investigate the association of proinflammatory cytokine genes, tumor necrosis factor (TNF)-α (−308G/A) and TNF-β (+252A/G) polymorphisms with schizophrenia susceptibility.
Subjects and methods
TNF-α and TNF-β genes were amplified using amplification refractory mutation system primers in 180 schizophrenia patients and 200 healthy matched controls recruited from the Psychiatry Clinic of Prince Sultan Military Medical City, Riyadh. The frequencies of alleles and genotypes of TNF-α (−308G/A) and TNF-β (+252A/G) polymorphisms in patients were compared with those in controls.
Results
The frequencies of TNF-α (−308) allele A and genotype GA were significantly higher, while those of allele G and genotype GG were lower in schizophrenia patients as compared to controls, indicating that genotype GA and allele A of TNF-α (−308G/A) may increase susceptibility to schizophrenia, while genotype GG and allele G may reduce it. On the other hand, the distribution of alleles and genotypes of TNF-β (+252A/G) polymorphism does not differ significantly in patients from controls; however, the frequency of genotype GG of TNF-β (+252A/G) was significantly higher in male patients than in female patients. The distribution of TNF-α (−308G/A) and TNF-β (+252A/G) polymorphisms was almost similar in schizophrenia patients with negative or positive symptoms.
Conclusion
TNF-α (−308G/A) and TNF-β (+252G/A) polymorphisms may increase the susceptibility to schizophrenia in Saudi patients and could be a potential risk factor for its etiopathogenesis. However, further studies are warranted involving a larger sample size to strengthen our findings.
Introduction
Schizophrenia is one of the most common devastating psychiatric disorders with a lifetime morbidity risk of 0.5%–2.7% and heritability estimated at up to 80%.Citation1–Citation3 The genetic, neurodevelopmental, neurotransmitter, and neuroimmunological hypotheses have been suggested to explain the etiopathogenesis of schizophrenia.Citation4,Citation5 It involves the interplay of complex polygenic influences and environmental risk factors operating on brain maturational processes.
Multiple genes have been associated with the development of schizophrenia. Recent studies have confirmed the association of a large number of single-nucleotide polymorphisms in a variety of genes with schizophrenia susceptibility.Citation6–Citation11 Genes encoding cytokines seem to be good candidate genes for schizophrenia. Cytokines play an important role in the central nervous system (CNS) as essential mediators of crosstalk between the brain and immune system and regulate neuroinflammatory processes.Citation12 Some cytokines are normally produced in the healthy brain, where they play critical roles in neurogenesis, migration, differentiation, and synapse formation.Citation13–Citation15 The concentrations of these cytokines have been reported to be increased or decreased in patients with schizophrenia.Citation16 Moreover, the concentrations of the cytokines in the blood serum of schizophrenia patients may vary depending on whether the patient is in active or resting phase of the disease.Citation17 It has also been suggested that schizophrenia may be associated with alterations in the Th1/Th2 cytokine ratios, with a shift toward the Th2 system.Citation4,Citation18,Citation19
Cytokines are the key regulators of immune/inflammatory reactions and influence the dopaminergic, noradrenergic, and serotonergic neurotransmission.Citation20 Tumor necrosis factor (TNF)-α, a proinflammatory cytokine, mediates immune and inflammatory responses and plays a key role in the CNS. It is not only actively transported into the CNS but also released from activated glia cells.
TNF-α (MIM 191160) and TNF-β or lymphotoxin-alpha (LT-α; MIM 153440) are closely related cytokines that share 30% amino acid residues and use the same cell surface.Citation21 Nedwin et alCitation22 reported that the TNF-α and TNF-β genes are located in tandem on chromosome 6 between the Class I and Class II cluster of the major histocompatibility complex chromosome (6p21.1–6p21.3) and show close linkage to the genes for human leukocyte antigen (HLA) classes I (HLA-B) and II (HLA-DR). The production of TNF-α is genetically determined.Citation23 Several polymorphisms in the promoter region of TNF-α and the intron 1 of TNF-β have been associated with changes in the levels of circulating TNF-α.Citation24 TNF-α (−308G/A) promoter polymorphism (rs1800629) is one of the best described single-nucleotide polymorphisms at the nucleotide position −308, which affects a consensus sequence for a binding site of the transcription factor activator protein 2.Citation25 TNF-α (−308G/A) polymorphism leads to a less common allele A (2-allele), which has been associated with increased TNF-α production in vitroCitation26 and a higher rate of TNF-α transcription than that associated with the wild-type GG genotype.Citation27 Conversely, a polymorphism TNF-β (+252A/G) (rs909253) at the nucleotide position +252 within the first intron of TNF-β gene affects a phorbol ester-responsive element. The presence of G at this position defines the mutant allele known as TNF-β * 1 (1-allele), which is associated with higher TNF-α and -β production.Citation28,Citation29 The functional polymorphisms in cytokine genes may result in imbalances in the pro- and anti-inflammatory cytokine production.Citation11 Keeping in view the potential role of these cytokines as the principal mediators of the immune response and the importance of the chromosomal region containing TNF-α and TNF-β genes, the present study was undertaken to examine the association of TNF-α (−308G/A) and TNF-β (+252G/A) polymorphisms with schizophrenia susceptibility in Saudi population. The concomitant analysis of polymorphisms at TNF-α and -β, given that both are involved in the expression of TNF-α, will help in the development of better strategies for the prevention and treatment of schizophrenia.
Subjects and methods
Subjects
The study population consisted of a total of 380 Saudi subjects of either sex. The sample included 180 schizophrenia patients recruited from the Outpatient Psychiatric Clinic of Prince Sultan Military Medical City, Riyadh, Saudi Arabia, and 200 age- and sex-matched healthy subjects as controls. All subjects were biologically unrelated Saudis. To ensure the diagnostic reliability, a systemic search into the case notes of the patients was made. Out of 200 initially selected schizophrenia patients, 20 patients failed to meet the explicit stated criteria and hence were excluded, and only 180 patients were included in this study. Among the confirmed 180 cases of schizophrenia, there were 53 females and 127 males with a mean age of 39±12.5 years and a mean disease duration of 9±4.5 years. Age at the onset of disease ranged from 19 to 64 years (mean 29±2.5 years). Among the confirmed cases of schizophrenia, 94 patients had negative symptoms, while 86 patients had positive symptoms. The female to male ratio of schizophrenia patients in our study was 53:127 (1:2.4). The control group consisted of 60 females and 140 males aged 26–60 years with a mean age of 35±10.5 years. Power was calculated online (http://www.stat.ubc.ca/~rollin/stats/ssize/caco.html).
The diagnosis of schizophrenia was based on the criteria mentioned in the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision version. The patients were further assessed for positive and negative symptoms using Positive and Negative Syndrome Scale as described by Kay et al.Citation30 All subjects in the control group were screened and excluded if they had any history of neurological, psychiatric, or medical disorders or had a past or present involvement in substance abuse. Screening of controls was performed following Johnstone et al.Citation31 None of the control subjects had a first- or second-degree relative with any mental illness. This study was approved by the research and ethical committee of Prince Sultan Military Medical City (PSMMC). Written informed consent was obtained from all subjects in accordance with ethical guidelines set by the ethical committee.
Polymerase chain reaction (PCR) amplification
Genomic DNA was extracted from the peripheral blood of Schizophrenia patients and controls using QIAampR DNA Mini Kit (Qiagen, Valencia, CA, USA). TNF-α and TNF-β genes were amplified using an amplification refractory mutation system-PCR methodology to detect any polymorphism involved at positions −308 of TNF-α and +252 in intron 1 of TNF-β gene. PCR amplification was carried out using PuReTaq Ready-to-Go PCR Beads (GE Healthcare, Buckinghamshire, UK) as described elsewhere.Citation32 The molecular analysis of the samples was performed in the same laboratory and at the same time. The investigator was blind to the phenotype of the subjects at the time of molecular analysis. Later on, the results were separated for patient and control groups and analyzed for the determination of the frequencies of genotypes and alleles.
Statistical analysis
The differences in allele/genotype frequencies between patients and controls were analyzed by the Fisher’s exact test. P-values ≤0.05 were considered significant. Bonferroni correction was applied to minimize error due to multiple comparison test. Therefore, both the P-values, after Bonferroni correction and Fisher’s exact test, are considered in the manuscript and –.
Table 1 Genotype and allele frequencies of TNF-α (−308G/A) variants in schizophrenia patients and matched controls
Table 3 Genotype and allele frequencies of TNF-β (−252A/G) polymorphism in male and female patients
The odd ratio interpreted as relative risk (RR) was calculated following the Woolf’s method as outlined by Schallreuter et al.Citation33 The etiologic fraction (EF) and preventive fraction (PF) were calculated following the formula of Svejgaard et alCitation34 as described in our earlier publication.Citation35
Results
Among Schizophrenia patients, the male to female ratio was 127:53 (2.4:1). The number of controls per case was 1.11, which yielded a power of 95%. In this case–control study, the allele and genotype frequencies of both polymorphisms were determined in schizophrenia patients and unrelated matched controls. The results of the genotype and allele distributions of TNF-α (−308) and TNF-β (+252A/G) polymorphisms in schizophrenia and control groups are summarized in –. The representative gel pictures for different genotypes are shown in and . Allelic frequencies and genotype distributions of both TNF-α and TNF-β gene polymorphisms differ between schizophrenia patients and control subjects. The frequency of GA (−308) genotype was significantly higher (P≤0.0001, P=0.0005, Bonferroni corrected), while the frequency of GG (−308) genotype was lower in patients as compared to controls (P≤0.0001, P=0.0005, Bonferroni corrected). AA genotype was absent in the patients but present in 7% of the controls. The frequency of allele A was found to be significantly increased, while that of allele G was decreased in patients as compared to controls (P≤0.0001). Even after applying Bonferroni correction, the P-values were statistically significant for both genotypes and alleles ().
Figure 1 Amplification of TNF-α (−308G/A) alleles (G and A).
Abbreviation: TNF, tumor necrosis factor.
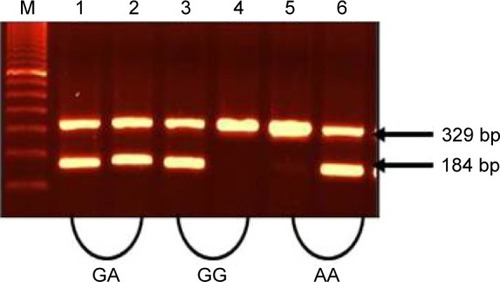
Figure 2 Amplification of TNF-β (+252A/G) alleles (A and G).
Abbreviation: TNF, tumor necrosis factor.
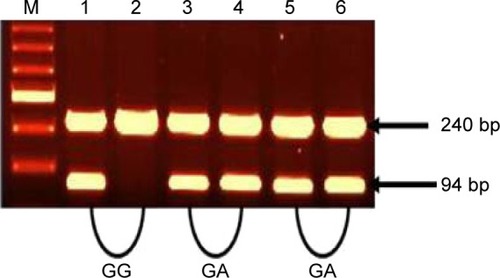
Table 4 Genotype and allele frequencies of TNF-β (LT-α) intron 1 +252 variants in schizophrenia patients and matched controls
The results of TNF-α (−308G/A) polymorphism for the patient group were then stratified into patients with negative or positive symptoms. The distribution of alleles and genotypes of TNF-α (−308G/A) polymorphism was found to be similar in the two groups of schizophrenia patients (data not shown).
Our study on TNF-β gene polymorphism at position +252 of intron 1 showed that although the frequencies of genotypes and alleles differed in patients from controls, the difference was not statistically significant (). Upon stratification of results of schizophrenia into patients with negative symptoms or with positive symptoms, the distribution of genotypes and alleles of TNF-β (+252A/G) polymorphism retained almost the same pattern in the two groups of patients as was in the combined schizophrenia patients except that the frequency of genotype AA was significantly lower in patients with negative symptoms than in those with positive symptoms or controls (, P=0.034). However, the sex difference was clear in the distribution of genotype frequencies of TNF-β (+252A/G) polymorphism. The frequency of genotype GG was significantly higher in male patients than in female patients, while the reverse was found for genotype GA, being higher in female patients than in male patients ().
Table 2 Genotype and allele frequencies of TNF-β (LT-α) intron 1 +252 variants in Schizophrenia patients with negative or positive symptoms and matched controls
Discussion
The higher frequencies of genotype GA and allele A of TNF-α (−308G/A) in schizophrenia patients as compared to controls indicated that the genotype GA and allele A may be susceptible to schizophrenia (RR =71.789, EF =0.688 and RR =2.722, EF =0.397, respectively). The genotype GG and allele G being higher in controls than in patients show their protective nature for schizophrenia (RR =0.042, PF =0.444 and RR =0.367, PF =0.389, respectively). These results suggested that TNF-α (−308G/A) polymorphism may increase the susceptibility to schizophrenia in the Saudi population and support the possible role of the immune response system in the pathogenesis of schizophrenia as suggested earlier by various researchers.Citation3,Citation4
The results of the present study are in accordance with earlier reports indicating the association of allele A of TNF-α (−308G/A) in the etiology of schizophrenia in Italian,Citation36,Citation37 Brazilian,Citation38 Singaporean,Citation39 Pakistani,Citation40 and Polish population.Citation3 On the other hand, some reports indicated the association of allele G of TNF-α (−308G/A) with schizophrenia in German,Citation41 Finnish male,Citation42 Australian,Citation43 and Polish populations.Citation44 Contrarily, several reports indicated no association of TNF-α (−308G/A) polymorphism with the susceptibility or pathogenesis of schizophrenia in the German,Citation45 Korean,Citation46 Australian, Indian Fijian, Indigenous Fijian, and Brahmin populations of IndiaCitation47 and in Taiwanese,Citation48 Japanese,Citation49,Citation50 Chinese Han,Citation51 Finnish,Citation42,Citation52 American Caucasian,Citation53 and Canadian populations.Citation54 These variations in the association of TNF-α (−308G/A) polymorphism with schizophrenia in various ethnic populations may be due to ethnic differences in the distribution of this polymorphism worldwide. The distribution of TNF-α (−308G/A) polymorphism is not uniform in healthy populations, and the frequencies vary from 50 to 98, 1.96 to 40.5, and 0 to 11.9% for GG, GA, and AA genotypes, respectively, in different ethnic populations, showing ethnic variations.Citation55 However, the association of the TNF-α (−308G/A) promoter polymorphism with schizophrenia in the Saudi population and other ethnici-ties has complemented the clinical findings of increased levels of TNF-α in schizophrenic patients, with some support for a functional consequence of the variant as allele A has been associated with increased TNF-α production in vitroCitation26 and a higher rate of TNF-α transcription than the wild-type GG genotype.Citation27 Alterations in the serum TNF-α level of chronic schizophrenia patients have been reported previously,Citation56–Citation58 implicating the role of TNF-α and TNF-α-related signaling pathways in the pathophysiology of schizophrenia. The roles of TNF-α in controlling neuronal excitability and metabolisms of glutamate, dopamine, and serotonin neurotransmitters have made it an outstanding candidate for etiology and pathophysiology of schizophrenia.Citation59 It has been suggested that TNF-α may be a trait marker of schizophrenia and may have an important role in the psychopathology of schizophrenia.Citation60,Citation61 The increased level of TNF-α may increase the production of 3-hydroxykynurenine. The increase in ratio between neurotoxic 3- hydroxykynurenine and neuroinhibitory/neuroprotective kynurenic acid may account for altered neurogenesis and structural abnormalities characteristic of schizophrenia.Citation62 However, Morar et alCitation43 besides supporting the association with the TNF-α (−308G/A) promoter polymorphism emphasized a linkage to the major histocompatibility complex region where the TNF-α gene is located and suggested linkage disequilibrium rather than direct involvement in the disorder. Paul-Samojedny et alCitation3 investigated the combined impact of gene polymorphisms in three proinflammatory cytokines, namely, IL-2, IL-6, and TNF-α, on susceptibility to schizophrenia and hypothesized that IL-6 and TNF-α gene polymorphisms that contribute to changes in the cytokine levels may impair the immune response to infections and also affect the development of the normal brain. The precise neurobiological mechanism explaining these increased risks in relation to infections is not clear, although the role of cytokines and an impaired immune response to these infections during the critical period of brain development are documented.Citation63 Because of the fact that particular schizophrenia subtypes are characterized by different clinical pictures, it is reasonable to perform genetic association studies on homogenous groups of patients. Therefore, we stratified schizophrenia patients into schizophrenia with positive symptoms and schizophrenia with negative symptoms. However, the distribution of alleles and genotypes was similar in the two groups.
On the other hand, our results for another gene TNF-β indicated that TNF-β (+252A/G) polymorphism is not directly associated with schizophrenia in Saudi patients; however, the fact that this polymorphism might be in linkage disequilibrium with TNF-α or other gene located in the same region cannot be ruled out. The TNF-β gene is known to play a central role in neurodevelopment, synaptic plasticity, and the response to neural injury.Citation64 Repeatedly associated with various brain activities and having immunologic, neurochemical, neuroendocrine, and behavioral effects, the TNF-β gene has also been associated with symptoms of schizophrenia.Citation65 In contrast to studies of TNF-α, few reports are available on the association between TNF-β gene polymorphisms and susceptibility to schizophrenia.Citation41,Citation66–Citation68 The immunomodulatory functions of TNF-β is well known and could impact the pathophysiology of schizophrenia. It affects and modulates production of TNF-α, which is supposed to be involved in the development of schizophrenia. In addition, TNF-β is effective in the protection of neuronal cells against glutamate and N-methyl-D-aspartate toxicity, which is considered a neurodevelopmental hypothesis of schizophrenia. Moreover, it is also found to be associated with the regulation of glial cells and stimulation of the synthesis and secretion of nerve growth factors in the CNS.Citation69
Our results also showed significantly higher frequency of genotype GG of TNF-β (+252A/G) in male patients as compared to female patients (). The higher frequency of genotype GG in male patients, which is known to be associated with increased production of TNF-α, might be responsible for the higher prevalence of schizophrenia in male subjects than in female subjects. As far as we know, this is the first study that examines the impact of TNF-α and TNF-β gene polymorphisms among Saudi patients with schizophrenia. The main limitation of this study is the small sample size, while the strengths of this study lie in the fact that it is the first report on Saudi patients with Schizophrenia, showing association of polymorphisms in TNF-α and -β genes. The errors in genotyping both cases and controls have been avoided carefully by using a standard protocol with positive and negative controls. Statistical analysis was performed to obtain P-values, RR, EF and PF, and power.
Conclusion
We suggest that TNF-α (−308G/A) and TNF-β (+252G/A) polymorphisms may be associated with schizophrenia susceptibility in Saudi patients and could be potential risk factors for its etiopathogenesis. However, further studies are required to strengthen these findings.
Acknowledgments
The authors thank the Medical Services Department Administration for providing excellent facilities.
Disclosure
The authors report no conflicts of interest in this work.
References
- SahaSChantDWelhamJMcGrathJA systematic review of the prevalence of schizophreniaPLoS Med200525e14115916472
- McGrathJSahaSChantDWelhamJSchizophrenia: a concise overview of incidence, prevalence, and mortalityEpidemiol Rev200830677618480098
- Paul-SamojednyAOwczarekMKowalczykRAssociation of interleukin 2 (IL-2), interleukin 6 (IL-6), and TNF-alpha (TNF-α) gene polymorphisms with paranoid schizophrenia in a Polish populationJ Neuropsychiatry Clin Neurosci2013251728223487197
- LeonardBEIs there an immunologic basis for schizophrenia?Expert Rev Clin Immunol20051110311220477658
- van OsJKapurSSchizophreniaLancet2009374969063564519700006
- AllenNCBagadeSMcQueenMBSystematic meta-analyses and field synopsis of genetic association studies in schizophrenia: the SzGene databaseNat Genet200840782783418583979
- ShiJGershonESLiuCGenetic associations with schizophrenia: meta-analyses of 12 candidate genesSchizophr Res20081041–39610718715757
- Al-AsmarySKadasahSArfinMTariqMAl-AsmariAGenetic association of catechol-O-methyltransferase val(158)met polymorphism in Saudi schizophrenia patientsGenet Mol Res20141323079308824782165
- HanSAnZLuoXAssociation between CMYA5 gene polymorphisms and risk of schizophrenia in Uygur population and a meta-analysisEarly Interv Psychiatry Epub201592410.1111/eip.12276
- HudsonZDMillerBJMeta-analysis of cytokine and chemokine genes in schizophreniaClin Schizophr Relat Psychoses Epub201672510.3371/CSRP.HUMI.070516
- SrinivasLVellichirammalNNAlexAMNairCNairIVBanerjeeMPro-inflammatory cytokines and their epistatic interactions in genetic susceptibility to schizophreniaJ Neuroinflammation201613110527177030
- CañavateMLGarcía de GaldeanoAArteagaOCross-talk between nervous and immune systems: cytokines modulating morphology and function of both systems under stress conditionsMéndez-VilasAMicroscopy: advances in scientific research and education1Zurbarán, SpainFormatex Research Center2014414421
- SzelényiJViziESThe catecholamine–cytokine balance (interaction between the brain and the immune system)Ann N Y Acad Sci2007111331132417584982
- DevermanBEPattersonPHCytokines and CNS developmentNeuron2009641617819840550
- GarayPAMcAllisterAKNovel roles for immune molecules in neural development: implications for neurodevelopmental disordersFront Synaptic Neurosci2010213621423522
- LiuLJiaFYuanGTyrosine hydroxylase, interleukin-1β and tumor necrosis factor-α are overexpressed in peripheral blood mononuclear cells from schizophrenia patients as determined by semi-quantitative analysisPsychiatry Res201017611720067853
- MaesMBocchio ChiavettoLBignottiSEffects of atypical antipsychotics on the inflammatory response system in schizophrenic patients resistant to treatment with typical neurolepticsEur Neuropsychopharmacol200010211912410706993
- MüllerNRiedelMAckenheilMSchwarzMJCellular and humoral immune system in schizophrenia: a conceptual reevaluationWorld J Biol Psychiatry20001417317912607212
- SchwarzMJMullerNRiedelMAckenheilMThe Th2-hypothesis of schizophrenia: a strategy to identify a subgroup of schizophrenia caused by immune mechanismsMed Hypotheses200156448348611339852
- MüllerNAckenheilMPsychoneuroimmunology, the cytokine network in the CNS, and the implications for psychiatric disordersProgr Neuropsychopharmacol Biol Psychiatry1998221131
- BeutlerBCeramiAThe biology of cachectin/TNF- a primary mediator of the host responseAnnu Rev Immunol198976256552540776
- NedwinGENaylorSLSakaguchiAYHuman lymphotoxin and tumor necrosis factor genes: structure, homology and chromosomal localizationNucleic Acids Res19851317636163732995927
- WestendorpRGLangermansJAHuizingaTWGenetic influence on cytokine production and fatal meningococcal diseaseLancet199734990461701739111542
- SharmaSGhoshBSharmaSKAssociation of TNF polymorphisms with sarcoidosis, its prognosis and tumour necrosis factor (TNF)-α levels in Asian IndiansClin Exp Immunol2008151225125918062795
- AbrahamLJKroegerKMImpact of the -308 TNF promoter polymorphism on the transcriptional regulation of the TNF gene: relevance to diseaseJ Leukoc Biol199966456256610534109
- BraunNMichelUErnstBPGene polymorphism at position-308 of the tumor-necrosis-factor-α (TNF-α) in multiple sclerosis and its influence on the regulation of TNF-α productionNeurosci Lett1996215275788887999
- WilsonAGSymonsJAMcDowellTLMcDevittHODuffGWEffects of a polymorphism in the human tumor necrosis factor a promoter on transcriptional activationProc Natl Acad Sci U S A1997947319531999096369
- MesserGSpenglerUJungMCPolymorphic structure of the tumor necrosis factor (TNF) locus: an NcoI polymorphism in the first intron of the human TNF-beta gene correlates with a variant amino acid in position 26 and a reduced level of TNF-beta productionJ Exp Med199117312092191670638
- AbrahamLJFrenchMADawkinsRLPolymorphic MHC ancestral haplotypes affect the activity of tumour necrosis factor-alphaClin Exp Immunol199392114188096802
- KaySRFiszbeinAOplerLAThe positive and negative syndrome scale (PANSS) for schizophreniaSchizophr Bull19871322612763616518
- JohnstoneECEbmeierKPMillerPOwensDGLawrieSMPredicting schizophrenia: findings from the Edinburgh high-risk studyBr J Psychiatry2005186182515630119
- Al-HarthiFZoumanAArfinMTariqMAl-AsmariATumor necrosis factor-α and -β genetic polymorphisms as a risk factor in Saudi patients with vitiligoGenet Mol Res20131232196220423884763
- SchallreuterKULevenigCKuhnlPLöligerCHohl-TehariMBergerJHistocompatability antigens in vitiligo: Hamburg study on 102 patients from Northern GermanyDermatology199318731861928219421
- SvejgaardAPlatzPRyderLPHLA and disease 1982 – a surveyImmunol Rev1983701932186339368
- KadasahSArfinMTariqMHLA-DRB1 association with schizophrenia in Saudi Arabian patientsInt J Psychiatry Clin Pract201015211211722121859
- BoinFZanardiniRPioliRAltamuraCAMaesMGennarelliMAssociation between -G308A tumor necrosis factor alpha gene polymorphism and schizophreniaMol Psychiatry200161798211244489
- SacchettiEBocchio-ChiavettoLValsecchiP-G308A tumor necrosis factor alpha functional polymorphism and schizophrenia risk: meta-analysis plus association studyBrain Behav Immun200721445045717234379
- Meira-LimaIVPereiraACMotaGFAnalysis of a polymorphism in the promoter region of the tumor necrosis factor alpha gene in schizophrenia and bipolar disorder: further support for an association with schizophreniaMol Psychiatry20038871872012888800
- TanECChongSATanCHTeoYYPengKMahendranRTumor necrosis factor-α gene promoter polymorphisms in chronic schizophreniaBiol Psychiatry200354111205121114643088
- NazMRiazMSaleemMPotential role of Neuregulin 1 and TNF-alpha (-308) polymorphism in schizophrenia patients visiting hospitals in Lahore, PakistanMol Biol Rep20113874709471421127983
- SchwabSGMondabonaSKnappbMAssociation of tumor necrosis factor alpha gene -G308A polymorphism with schizophreniaSchizophr Res2003651192514623370
- HänninenKKatilabHRontucRMattilacKMHurmeMLehtimäkiTTumor necrosis factor-alpha -G308A polymorphism in schizophrenia in a Finnish populationNeurosci Lett20053851768115927374
- MorarBSchwabSGAlbusMMaierWLererBWildenauerDBEvaluation of association of SNPs in the TNF alpha gene region with schizophreniaAm J Med Genet B Neuropsychiatr Genet2007144B331832417171665
- CzerskiPMRybakowskiFKapelskiPAssociation of tumor necrosis factor -308G/A promoter polymorphism with schizophrenia and bipolar affective disorder in a polish populationNeuropsychobiology2008571–2889418515978
- RiedelMKrönigHSchwarzMJNo association between the G308A polymorphism of the tumor necrosis factor-alpha gene and schizophreniaEur Arch Psychiatry Clin Neurosci2002252523223412451465
- PaeCUChaeJHBahkWMTumor necrosis factor-α gene polymorphism at position -308 and schizophrenia in the Korean populationPsychiatry Clin Neurosci200357439940312839521
- HandokoHYNancarrowDJHaywardNKTumor necrosis factor haplotype analysis amongst schizophrenia probands from four distinct populations in the Asia-Pacific regionAm J Med Genet B Neuropsychiatr Genet2003121B11612898567
- TsaiSJHongCJYuYWLinCHLiuLLNo association of tumor necrosis factor alpha gene polymorphisms with schizophrenia or response to clozapineSchizophr Res2003651273214623371
- HashimotoRYoshidaMOzakiNAssociation analysis of the -308G > A promoter polymorphism of the tumor necrosis factor alpha (TNF-α) gene in Japanese patients with schizophreniaJ Neural Transm2004111221722114767724
- WatanabeYMuratakeTKanekoNFukuiNNaraYSomeyaTNo association between the tumor necrosis factor-alpha gene promoter polymorphisms and schizophrenia in a Japanese populationPsychiatry Res200715311617559942
- DuanSXuYChenaWNo association between the promoter variants of tumor necrosis factor alpha (TNF-α) and schizophrenia in Chinese Han populationNeurosci Lett2004366213914315276234
- KampmanOAnttilaSIlliAInteraction of tumor necrosis alpha -G308A and epidermal growth factor gene polymorphisms in early–onset schizophreniaEur Arch Psychiatry Clin Neurosci2005255427928315614591
- ShirtsBHWoodJYolkenRHNimgaonkarVLAssociation study of IL10, IL1β, and IL1RN and schizophrenia using tag SNPs from a comprehensive database: suggestive association with rs16944 at IL1βSchizophr Res2006881–323524416905295
- ZaiGMüllerDJVolavkaJFamily and case-control association study of the tumor necrosis factor-alpha (TNF-alpha) gene with schizophrenia and response to antipsychotic medicationPsychopharmacology (Berl)2006188217118216932925
- Al-RayesHAl-SwailemRAlbelawiMArfinMAl-AsmariATariqMTNF-α and TNF-β gene polymorphism in Saudi rheumatoid arthritis patientsClin Med Insights Arthritis Musculoskelet Disord20114556321792343
- BeumerWDrexhageRCDe WitHVersnelMADrexhageHACohenDIncreased level of serum cytokines, chemokines and adipokines in patients with schizophrenia is associated with disease and metabolic syndromePsychoneuroendocrinology201237121901191122541717
- LuoYHeHZhangMElevated serum levels of TNF-α, IL-6 and IL-18 in chronic schizophrenic patientsSchizophr Res20141592–355655725248940
- LvMHTanYLYanSXDecreased serum TNF-alpha levels in chronic schizophrenia patients on long-term antipsychotics: correlation with psychopathology and cognitionPsychopharmacology (Berl)2015232116517224958229
- TianLTanYChenDReduced serum TNF alpha level in chronic schizophrenia patients with or without tardive dyskinesiaProg Neuropsychopharmacol Biol Psychiatry20145425926424995685
- KubistovaAHoracekJNovakTIncreased interleukin-6 and tumor necrosis factor alpha in first episode schizophrenia patients versus healthy controlsPsychiatr Danub201224suppl 1S153S15622945211
- AjamiAAbedianFHamzeh HosseiniSAkbarianEAlizadeh-NavaeiRTaghipourMSerum TNF-α, IL-10 and IL-2 in schizophrenic patients before and after treatment with risperidone and clozapineIran J Immu-nol2014113200209
- JohanssonAOwe-LarssonBAspLActivation of kynurenine pathway in ex vivo fibroblasts from patients with bipolar disorder or schizophrenia: cytokine challenge increases production of 3-hydroxykynurenineJ Psychiatr Res201347111815182324012176
- AshdownHDumontYNgMPooleSBoksaPLuheshiGNThe role of cytokines in mediating effects of prenatal infection on the fetus: implications for schizophreniaMol Psychiatry2006111475516189509
- StellwagenDMalenkaRCSynaptic scaling mediated by glial TNF-alphaNature200644070871054105916547515
- KronfolZRemickDGCytokines and the brain: implications for clinical psychiatryAm J Psychiatry2000157568369410784457
- JunTYPaeCUChaeJHTNFB polymorphism may be associated with schizophrenia in the Korean populationSchizophr Res2003611394512648734
- DickersonFStallingsCOrigoniABoronowJYolkenRC-reactive protein is associated with the severity of cognitive impairment but not of psychiatric symptoms in individuals with schizophreniaSchizophr Res2007931–326126517490859
- ArabAHElhawaryNAAssociation between ANKK1 (rs1800497) and LTA (rs909253) Genetic Variants and Risk of SchizophreniaBiomed Res Int20152015821827
- PaeCUPotential role of lymphotoxin-alpha (tumor necrosis factor-beta) in the development of schizophreniaMed Hypotheses20076861359136217140746
