Abstract
Background
The majority of previous neuroimaging studies have demonstrated both structural and functional abnormalities in obstructive sleep apnea (OSA). However, few studies have focused on the regional intensity of spontaneous fluctuations during the resting state and the relationship between the abnormal properties and the behavioral performances. In the present study, we employed the amplitude of low-frequency fluctuation (ALFF) method to explore the local features of spontaneous brain activity in OSA patients (OSAs).
Methods
Twenty-five untreated male severe OSAs and 25 age-matched and years-of-education-matched male good sleepers (GSs) were included in this study. The ALFF method was used to assess the local features of spontaneous brain activity. The mean signal values of the altered ALFF areas were analyzed with receiver operating characteristic curve. Partial correlation analysis was used to explore the relationship between the observed mean ALFF values of the different areas and the behavioral performances.
Results
Compared with GSs, OSAs had significantly higher scores for body mass index, apnea–hypopnea index, arterial oxygen saturation <90%, arousal index, and Epworth Sleepiness Scale (ESS) score; furthermore, OSAs had significantly lower scores for rapid eye movement sleep and in the Montreal Cognitive Assessment (MoCA). Compared with GSs, OSAs showed significant lower-ALFF areas in the cluster of the right precuneus and bilateral posterior cingulate gyrus, as well as a higher-ALFF area in the left inferior frontal gyrus. The area under the curve values of the lower- and higher-ALFF areas were 0.90 and 0.93, respectively. Further diagnostic analysis exhibited that the sensibility and specificity of the two clusters were 80% and 92%, respectively. The mean signal value of the lower-ALFF cluster displayed significant positive correlations with lowest oxygen saturation (r=0.447, P=0.025) and MoCA score (r =0.405, P=0.045).
Conclusion
OSAs may involve in a dysfunction in the default mode network and an adaptive compensatory response in the frontal lobe, which reflect the underlying pathophysiology of cognitive impairment.
Introduction
Obstructive sleep apnea (OSA), characterized by repeated obstructions of upper airway with intermittent hypoxic exposure, is associated with multiple detrimental physiological and psychological consequences, in addition to being associated with cardiorespiratory diseases, including pulmonary hypertension,Citation1 systemic hypertension,Citation2 cardiac arrhythmias,Citation3 cardiac ischemiaCitation4 and cerebral ischemia.Citation5 OSA may also cause daytime sleepiness,Citation6 increase the risk of a traffic accident,Citation7 and diminish both quality of lifeCitation8 and work performance,Citation9 as well as being accompanied by impairment in several cognitive domains, including attention and vigilance decrements, memory gaps, psychomotor dysfunction, and abnormalities in executive functions.Citation10–Citation12 On the basis of available population-based studies, OSA affects 3%–7% of adult men, 2%–5% of adult women,Citation13–Citation15 and up to 4% of children.Citation16,Citation17 The main pathophysiologic mechanism of cognitive deficits from OSA, including intermittent hypoxia, intermittent hypercapnia, and sleep fragmentation,Citation18 are still unclear.
Previous neuroimaging studies have investigated 1) diminutions in gray matter concentration in the left hippocampus, left rectus gyrus, bilateral superior frontal gyrus, left precentral gyrus, bilateral frontomarginal gyrus, bilateral anterior cingulate gyrus, right insular gyrus, bilateral caudate nucleus, bilateral thalamus, bilateral amygdalo-hippocampus, bilateral inferior temporal gyrus, and cerebellum, and 2) gray matter volume deficits in bilateral hippocampus, bilateral lateral temporal areas, right cuneus, right middle temporal gyrus, left dorsolateral prefrontal cortex, right middle temporal gyrus, and left cerebellum using voxel-based morphometry techniques in OSA patients (OSAs).Citation19–Citation23 Moreover, diffusion tensor imaging studies showed a wide range of changes in white matter integrity within the corpus callosum, frontal cortex, temporal cortex, parietal cortexes, cingulate bundle, and cerebellum.Citation24–Citation26 A single-photon emission computed tomography study found that severe OSAs showed reduced cerebral blood flow in bilateral parahippocampal gyrus, right lingual gyrus, pericentral gyrus, and cuneus.Citation27 Decreased neural activations associated with cognitive impairment have been found in multiple brain regions, including the cingulate gyrus, dorsolateral prefrontal gyrus, inferior frontal gyrus, left postcentral gyrus, inferior and posterior parietal lobes, insula, and right putamen using task-state functional magnetic resonance imaging (fMRI) in OSAs.Citation28–Citation30 Recently, use of resting-state fMRI (rs-fMRI) has been increasing to investigate the ongoing neuronal processes in OSA. Although the majority of previous neuroimaging studies have focused on the brain structural and task-state functional changes of OSAs, few studies have evaluated the changes in blood oxygen-level-dependent (BOLD) signals of regional spontaneous activity of OSAs during resting state and their relationships with behavioral performances.
Amplitude of low-frequency fluctuation (ALFF), a newly developed rs-fMRI approach, calculates the square root of the power spectrum in a low-frequency range (0.01–0.08 Hz), for detecting the regional intensity of spontaneous fluctuations in BOLD signals.Citation31–Citation33 Furthermore, the ALFF has been proven to have test–retest reliabilityCitation34 and has already been applied to patient studies investigating attention deficit hyperactivity disorder,Citation31 sleep deprivation,Citation35 schizophrenia,Citation36 and early Alzheimer’s disease.Citation37 However, it has not yet been used to explore the pathophysiological changes in OSAs. This study is the first to utilize ALFF as an index to investigate the intrinsic brain activity traits of OSAs and its potential mechanisms.
Materials and methods
Subjects
Twenty-five untreated male severe OSAs and 25 age-matched and years-of-education-matched male good sleepers (GSs) were included in this study from the Sleep Monitoring Room of the Respiratory Department of The First Affiliated Hospital of Nanchang University. Each subject was assessed by a detailed clinical interview and physical examination; in addition, the subjects completed a sleep questionnaire and underwent overnight polysomnography. The inclusion and exclusion criteria for the OSAs and GSs were as the same as in our previous study.Citation38,Citation39 The inclusion criteria for the OSAs were as follows: male sex; age >22 years but <60 years; and an apnea–hypopnea index (AHI) >30 events per hour. The exclusion criteria for both OSAs and GSs were as follows: 1) other sleep disorders, such as insomnia and sleep-related eating disorders; 2) history of cardiovascular disease, hypertension, or diabetes mellitus; 3) central nervous system disorders (neurodegenerative diseases, epilepsy, head injury, psychosis, hypothyroidism, or current depression); 4) left-handedness; 5) alcohol or illicit drug abuse; 6) current intake of psychoactive medications; and 7) contraindications to MRI, such as claustrophobia, metallic implants, or devices in the body. This study was approved by The Human Research Ethics Committee at The First Affiliated Hospital of Nanchang University, and all participants provided written informed consent forms.
Polysomnography
The day before the sleep studies, all OSAs and GSs were asked to refrain from drinking alcohol or caffeinated beverages. Full nocturnal polysomnography monitoring was performed on OSAs and GSs using the Respironics LE-series physiological monitoring system (Alice 5 LE; Respironics, Orlando, FL, USA) in the Sleep Center of our hospital. Standard electroencephalogram, electrooculogram, chin electromyogram, electrocardiogram, thoracic and abdominal movements, and snoring were recorded. Arterial oxygen saturation (SaO2) was measured transcutaneously by fingertip pulse oximetry. In accordance with the American Academy of Sleep Medicine guidelines, apnea was defined as the continuous cessation of airflow for more than 10 seconds and hypopnea was defined as a decrease in airflow by >30% with arousal or oxygen desaturation >4%.Citation40,Citation41 The AHI was calculated as the average of the total number of apnea and hypopnea events experienced per hour of sleep. Subjects’ performances were recorded on videotape and were continuously observed by a polysomnography technician. Polysomnography was performed from 10 pm to 6 am next morning.
Neuropsychological evaluation
All subjects filled in a sleep questionnaire to assess their daytime sleepiness by the Epworth Sleepiness Scale (ESS), with scores between 0 and 24.Citation42 A score higher than 6 was considered somnolence. All subjects underwent a cognitive assessment using the Montreal Cognitive Assessment (MoCA)Citation43 tool, administered by two independent neuropsychologists, to evaluate their executive function, naming, attention, calculation, language, abstraction, memory, and orientation. The total MoCA score is 30. A total MoCA score <26 indicates cognitive impairment, whereas a score ≥26 indicates normal cognitive function. If the length of schooling was <12 years, one point was added to the total score, so as to adjust for educational bias.Citation44
MRI parameters
MRI scanning was performed on a 3-Tesla MRI scanner (Siemens, Erlangen, Germany). High-resolution T1-weighted images were acquired with a three-dimensional spoiled gradient-recalled echo sequence in a sagittal orientation: 176 images (repetition time =1,900 ms; echo time =2.26 ms; thickness =1.0 mm; gap =0.5 mm; acquisition matrix =256×256; field of view =250×250 mm2, flip angle =9°) were obtained. Finally, 240 functional images (repetition time =2,000 ms; echo time =30 ms; thickness =4.0 mm; gap =1.2 mm; acquisition matrix =64×64; flip angle =90°; field of view =230×230 mm2; 30 axial slices with gradient-recalled echo-planar imaging [EPI] pulse sequence) covering the whole brain were obtained.
fMRI data analysis
Functional data were checked using MRIcro software (www.MRIcro.com) to exclude defective data. The first ten time points of the functional images were discarded due to the possible instability of the initial MRI signals and the participants’ adaptation time to the scanning environment. On the basis of MATLAB2010a (Mathworks, Natick, MA, USA), the remainder of the data preprocessing was performed by DPARSFA (http://rfmri.org/DPARSF) software, including DICOM form transformation, slice timing, head motion correction, spatial normalization, smoothing with a Gaussian kernel of 6×6×6 mm3 full width at half maximum (FWHM). Motion time courses were obtained by estimating the values for translation (mm) and rotation (degrees) for each subject. Participants who had >1.5 mm maximum displacement in x, y, or z planes and 1.5° of angular motion during the whole fMRI scans were rejected. The Friston six head motion parameters were used to regress out head motion effects based on recent work showing that higher-order models were more effective in removing head motion effects.Citation45,Citation46 Linear regression was also applied to remove other sources of spurious covariates along with their temporal derivatives, including the signal from a ventricular region of interest (ROI) and the signal from a region centered in the white matter.Citation47 Of note, the global signal was not regressed out in the present data, as in the study by Guo et alCitation48 for the reason that there is still a controversy around the removal of the global signal in the preprocessing step of resting-state data.Citation47,Citation49 After head-motion correction, the fMRI images were spatially normalized to the Montreal Neurological Institute (MNI) space using the standard EPI template and resampling the images at a resolution of 3×3×3 mm3. After preprocessing, the time series for each voxel were linearly detrended to reduce low-frequency drift, physiological high-frequency respiratory and cardiac noise, and time series linear detrending. The time series for each voxel were transformed to the frequency domain, and the power spectrum was then obtained. Because the power of a given frequency is proportional to the square of the amplitude of this frequency component, the square root was calculated at each frequency of the power spectrum, and the averaged square root was obtained across 0.01–0.08 Hz at each voxel. This averaged square root was taken as the ALFF. The details of ALFF calculation are as described in a previous study.Citation31 To reduce the global effects of variability across the participants, the ALFF of each voxel was divided by the global mean ALFF value for each participant.
Statistical analysis
Subject characteristics, including age, body mass index (BMI), education, ESS score, MoCA score, and sleep-disordered breathing parameters, were tested using independent sample t-tests to compare OSAs with GSs using IBM Statistical Package for the Social Sciences version 19.0 (SPSS 19.0), and a P-value <0.05 was deemed significant. For functional data, two-sample Student’s t-test was used to analyze the difference between the two groups, with age and years of education as nuisance covariates of no interest. A corrected significance level of individual voxel P<0.001 and a cluster volume (V) ≥270 mm3 (a minimum continuous V of 270 mm3), using a false discovery rate (FDR)-corrected cluster threshold of P<0.05, was used to determine statistical significance.
On the basis of the ALFF findings, the brain regions that demonstrated significant level of difference between groups were indentified. These regions were classified as ROIs and saved as masks using the REST Version1.8 software (http://www.resting-fmri.Sourceforge.net). For each ROI, the mean ALFF value was extracted by averaging the ALFF values over all voxels for each OSA. Finally, the mean ALFF values were entered into IBM SPSS 19.0 to calculate their correlations with the behavioral performances.
Results
Demographic and clinical results
Demographic and clinical characteristics of each group are summarized in . The OSAs had significantly higher scores for BMI (t =6.25, P<0.001), AHI (t =15.51, P<0.001), SaO2 <90% (t =6.66, P<0.001), arousal index (t =6.85, P<0.001), and ESS score (t =7.64, P<0.001) and had significantly lower scores for rapid eye movement (REM) sleep (t =-5.4, P<0.001) and MoCA (t =-2.16, P=0.036) than the GSs.
Table 1 Demographic and clinical characteristics of OSAs and GSs
ALFF results
Compared with GSs, OSAs showed significant lower-ALFF areas in the cluster of right precuneus and bilateral posterior cingulate gyrus and a higher-ALFF area in the left inferior frontal gyrus. The details are presented in and . The mean ALFF values of these altered areas were extracted ().
Figure 1 Compared to GSs, OSAs showed altered ALFF areas.
Abbreviations: GSs, good sleepers; OSAs, patients with obstructive sleep apnea; ALFF, amplitude of low-frequency fluctuation.
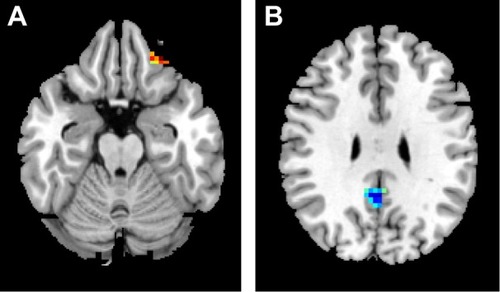
Figure 2 Mean ALFF signal values for altered regional brain areas.
Abbreviations: GSs, good sleepers; OSAs, patients with obstructive sleep apnea; ALFF, amplitude of low-frequency fluctuation; Prc, precuneus; Pcc, posterior cingulate gyrus.
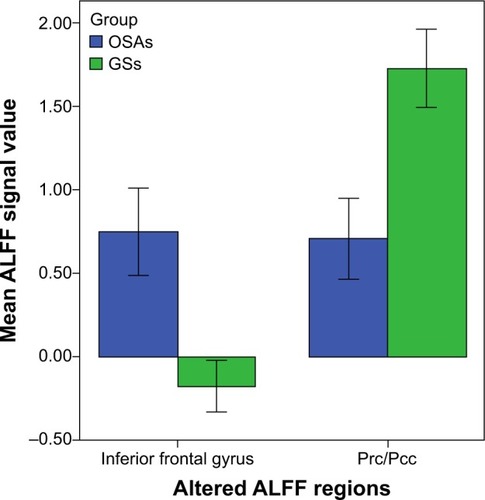
Table 2 Two-sample t-test differences between OSAs and GSs using ALFF method
Correlation results
In the OSAs, the AHI score displayed a significant positive correlation with the arousal index (r=0.642, P=0.001) and negative correlations with REM% (r=−0.429, P=0.032) and MoCA score (r=−0.405, P=0.045). N2% (percentage of total sleep time at diagnostic polysomnography spent in the relevant stage) displayed negative correlations with REM% (r=−0.584, P=0.002) and ESS score (r=−0.531, P=0.006). BMI displayed significant positive correlation with arousal index (r=0.582, P=0.002) and negative correlation with the lowest oxygen saturation (r=−0.647, P<0.001).
The mean signal value of the observed lower-ALFF area displayed significant positive correlations with the lowest oxygen saturation (r=0.447, P=0.025) and MoCA score (r=0.405, P=0.045).
Receiver operating characteristic curve
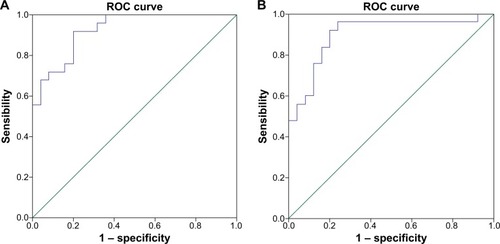
Because different ALFF areas were found between OSAs and GSs, they might be utilized as markers to separate the OSAs from the GSs. To test this possibility, the mean ALFF values of the different brain regions were extracted and used for analysis of the receiver operating characteristic curves. In the present study, the values for the areas under the curves of the left inferior frontal gyrus and the cluster of right precuneus and bilateral posterior cingulate gyrus were 0.93 and 0.90, respectively. Further diagnostic analysis showed that the sensibility and specificity of the two clusters were 92% and 80%, respectively. The details are presented in .
Figure 3 ROC curve analysis of mean ALFF signal values for altered regional brain areas.
Abbreviations: AUC, area under the curve; ALFF, amplitude of low-frequency fluctuation; ROC, receiver operating characteristic.
Discussion
Our previous studyCitation50 had demonstrated that many brain areas have obvious sex differences after normal sleep status and during sleep loss status. In this study, so as to avoid the influence of the sex differences or a lopsided sex ratio, only male OSAs were recruited. Our study is the first to investigate the effect of OSA on resting-state brain activity using the ALFF method. In our current study, we found that OSAs showed higher ALFF in the left inferior frontal lobe and lower ALFF in the cluster of right precuneus and bilateral posterior cingulate gyrus, compared with GSs, and showed lower MoCA score than GSs. Moreover, the observed lower-ALFF area displayed significant positive correlations with the lowest oxygen saturation and MoCA score.
As known, the lower-ALFF area in the cluster of right precuneus and bilateral posterior cingulate gyrus is largely included in the default mode networks (DMNs).Citation51 Similarly, Prilipko et alCitation52 found that OSAs showed abnormal deactivation in the DMN during working-memory tasks and a significantly positive correlation between the deactivation of DMN regions and behavioral performance,Citation52 suggesting that suppression of activity in the DMN plays a role in cognitive impairment. Beebe et alCitation53 also found, using sophisticated meta-analytic models, that OSA had a substantial impact on vigilance and executive functions but a negligible impact on intellectual and verbal function. Yaouhi et alCitation54 found that the glucose metabolic activity of precuneus and cingulate gyrus, which were not atrophic, was reduced in OSAs, compared with that in GSs, using a 18F-fluoro-2-deoxy-D-glucose positron emission tomography method,Citation54 and that this functional impairment may have been caused partly by remote effects originating from morphologically impaired areas with decreased connectivity.Citation55 Furthermore, our study found a positive correlation between cerebral deactivation in parts of regions of the DMN and the lowest oxygen saturation, suggesting that intermittent hypoxia may be an important factor for the DMN dysfunction in OSAs.
The cingulate area is activated by dyspnea,Citation56 breathlessness,Citation57 and emotion related to the need for airCitation58 and is involved in autonomic functions, including maintenance of blood pressure and salivary secretion, which suggests that the cingulate cortex has a complex and indirect relationship with the central networks that control respiration.Citation59,Citation60 Previous structural neuroimaging studies have found gray matter loss and white matter integrity reduction in the cingulate in OSAs.Citation25,Citation26 Joo et alCitation20 also found that OSAs had decreased gray matter concentrations in the cingulate cortex, which may explain the clinical manifestations such as respiratory, affective, and cardiovascular disturbances. Ayalon et alCitation61 found that OSAs showed decreased brain activation in left precentral gyrus, left anterior, and posterior cingulate, compared with control subjects, during an attention task. Similar results have been reported during valsalva maneuvers.Citation62 These studies suggest that the cingulate gyrus may be particularly vulnerable to OSA-related impairment, independent of the specific cognitive challenge measured. In support of these functional and structural findings, our study found lower-ALFF areas in the posterior cingulate in OSAs compared with GSs. Moreover, a significant positive correlation between the lower-ALFF area and the MoCA score was observed in the OSAs. This brain–behavior relationship demonstrated that the abnormal properties of the posterior cingulate were associated with impaired cognitive function in OSAs.
The precuneus plays an important role in fundamental cognitive functioning, including episodic memory retrieval, visual–spatial imagery, self-processing, and consciousness.Citation63 Our previous studyCitation38 found lower regional homogeneity area in the precuneus, which showed a significant negative correlation with sleep time, suggesting that decreased sleep time may be an important factor for dysfunction in the precuneus. In the current study, the lower-ALFF area in the right precuneus displayed significant positive correlation with MoCA score, suggesting that the abnormalities of the precuneus may be associated with a cognitive dysfunction.
Ayalon et alCitation64 found that compared with GSs, OSAs showed increased activity in the bilateral inferior frontal gyrus, and a significant relationship between increased activation in the left inferior frontal gyrus and better immediate recall during a verbal learning task was found, suggesting an adaptive compensatory response. In support of this finding, in our study, OSAs showed higher ALFF in the left inferior frontal lobe, compared with GSs, which was consistent with previous studies on sleep deprivationCitation65 and healthy aging.Citation66
Conclusion
In conclusion, our findings suggest that the ALFF method may be a useful noninvasive imaging tool and a symbolic early biomarker for the detection of cerebral changes and for indexing of the changes of cognitive function, which may be helpful in the development of imaging biomarkers for the detection of cerebral changes. Furthermore, the abnormal spontaneous activity of the DMN region and the frontal lobe may be involved in the underlying pathophysiology of OSAs. However, there are several limitations that should be paid attention to. First, a larger sample size, as well as female and children populations, should be studied. Second, another group comparison, both before and after the treatment, will yield significant insight.
Acknowledgments
This work was supported by the Jiangxi Provincial Department of Science and Technology Support Program (grant numbers 20132BBG70061, 20121BBG70056, and 20141BBG70026), Jiangxi Provincial Department of Natural Science Foundation Project (grant 20132BAB205100), Jiangxi Provincial Department of Graduate Innovation Foundation (grant YC2013-S007), Chinese Department of National Innovation Experiment Program for University Students (grants 201210403052 and 2012181).
Disclosure
The authors report no conflicts of interest in this work. This was not an industry-supported study. None of the authors have any personal or financial involvement with organizations that hold financial interest in the subject matter.
References
- SajkovDMcEvoyRDObstructive sleep apnea and pulmonary hypertensionProg Cardiovasc Dis200951536337019249442
- StradlingJRSleep apnoea and systemic hypertensionThorax198944129849892694420
- FlemonsWWRemmersJEGillisAMSleep apnea and cardiac arrhythmias: Is there a relationship?Am Rev Respir Dis199314836186218368632
- KoskenvuoMKaprioJTelakiviTSnoring as a risk factor for ischaemic heart disease and stroke in menBr Med J198729416193101779
- MunozRDuran-CantollaJMartínez-VilaESevere sleep apnea and risk of ischemic stroke in the elderlyStroke20063792317232116888274
- MalhotraAWhiteDPObstructive sleep apnoeaLancet2002360932823724512133673
- GeorgeCFPSleep. 5: driving and automobile crashes in patients with obstructive sleep apnoea/hypopnoea syndromeThorax200459980480715333860
- EnglemanHMDouglasNJSleep. 4: sleepiness, cognitive function, and quality of life in obstructive sleep apnoea/hypopnoea syndromeThorax200459761862215223874
- CarotenutoMEspositoMParisiLDepressive symptoms and childhood sleep apnea syndromeNeuropsychiatr Dis Treat2012836937322977304
- KimHCYoungTMatthewsCGWeberSMWoodwardARPaltaMSleep-disordered breathing and neuropsychological deficits: a population-based studyAm J Respir Crit Care Med19971566181318199412560
- Ferini-StrambiLBaiettoCDi GioiaMRCognitive dysfunction in patients with obstructive sleep apnea (OSA): partial reversibility after continuous positive airway pressure (CPAP)Brain Res Bull2003611879212788211
- SaunamäkiTJehkonenMA review of executive functions in obstructive sleep apnea syndromeActa Neurol Scand2007115111117156260
- FengJChenBYPrevalence and incidence of hypertension in obstructive sleep apnea patients and the relationship between obstructive sleep apnea and its confoundersChin Med J (Engl)2009122121464146819567173
- YoungTPeppardPEGottliebDJEpidemiology of obstructive sleep apnea: a population health perspectiveAm J Respir Crit Care Med200216591217123911991871
- [No authors listed]Sleep apnea: is your patient at risk?. National heart, lung, and blood institute working group on sleep apneaAm Fam Physician19965312472538546051
- LumengJCChervinRDEpidemiology of pediatric obstructive sleep apneaProc Am Thorac Soc20085224225218250218
- FengJWuQZhangDChenBYHippocampal impairments are associated with intermittent hypoxia of obstructive sleep apneaChin Med J (Engl)2012125469670122490498
- SuzukiYJJainVParkAMDayRMOxidative stress and oxidant signaling in obstructive sleep apnea and associated cardiovascular diseasesFree Radic Biol Med200640101683169216678006
- MorrellMJMcRobbieDWQuestRACumminARGhiassiRCorfieldDRChanges in brain morphology associated with obstructive sleep apneaSleep Med20034545145414592287
- JooEYTaeWSLeeMJReduced brain gray matter concentration in patients with obstructive sleep apnea syndromeSleep201033223524120175407
- TorelliFMoscufoNGarreffaGCognitive profile and brain morphological changes in obstructive sleep apneaNeuroimage201154278779320888921
- ZhangQWangDQinWAltered resting-state brain activity in obstructive sleep apneaSleep201336565165923633747
- MorrellMJJacksonMLTwiggGLChanges in brain morphology in patients with obstructive sleep apnoeaThorax2010651090891420861295
- KumarRChavezASMaceyPMWooMAYan-GoFLHarperRMAltered global and regional brain mean diffusivity in patients with obstructive sleep apneaJ Neurosci Res201290102043205222715089
- MaceyPMHendersonLAMaceyKEBrain morphology associated with obstructive sleep apneaAm J Respir Crit Care Med2002166101382138712421746
- MaceyPMKumarRWooMABrain structural changes in obstructive sleep apneaSleep200831796797718652092
- JooEYTaeWSHanSJChoJWHongSBReduced cerebral blood flow during wakefulness in obstructive sleep apnea-hypopnea syndromeSleep200730111515152018041484
- ThomasRJRosenBRSternCEWeissJWKwongKKFunctional imaging of working memory in obstructive sleep-disordered breathingJ Appl Physiol20059862226223415677733
- AyalonLAncoli-IsraelSDrummondSPAltered brain activation during response inhibition in obstructive sleep apneaJ Sleep Res200918220420819302344
- ArchboldKHBorghesaniPRMahurinRKKapurVKLandisCANeural activation patterns during working memory tasks and OSA disease severity: preliminary findingsJ Clin Sleep Med200951212719317377
- ZangYFHeYZhuCZAltered baseline brain activity in children with ADHD revealed by resting-state functional MRIBrain Dev2007292839116919409
- LuHZuoYGuHSynchronized delta oscillations correlate with the resting-state functional MRI signalProc Natl Acad Sci U S A2007104182651826917991778
- KiviniemiVJauhiainenJTervonenOSlow vasomotor fluctuation in fMRI of anesthetized child brainMagn Reson Med200044337337810975887
- ZuoXNDi MartinoAKellyCThe oscillating brain: complex and reliableNeuroimage20104921432144519782143
- DaiXJMinYJGongHHEvaluation of the post-effect of acupuncture at Sanyinjiao (SP 6) under sleep deprivation by resting-state amplitude of low-frequency fluctuation:a fMRI studyZhongguo Zhen Jiu2012321475222295826
- HoptmanMJZuoXNButlerPDAmplitude of low-frequency oscillations in schizophrenia: a resting state fMRI studySchizophr Res20101171132019854028
- HeYWangLZangYRegional coherence changes in the early stages of Alzheimer’s disease: a combined structural and resting-state functional MRI studyNeuroimage200735248850017254803
- PengDCDaiXJGongHHLiHJNieXZhangWAltered intrinsic regional brain activity in male patients with severe obstructive sleep apnea: a resting-state functional magnetic resonance imaging studyNeuropsychiatr Dis Treat2014101819182625278755
- DaiXJPengDCGongHHAltered intrinsic regional brain spontaneous activity and subjective sleep quality in patients with chronic primary insomnia: a resting-state fMRI studyNeuropsychiatr Dis Treat2014102163217525484585
- RedlineSBudhirajaRKapurVThe scoring of respiratory events in sleep: reliability and validityJ Clin Sleep Med20073216920017557426
- [No authors listed]EEG arousals: scoring rules and examples: a preliminary report from the Sleep Disorders Atlas Task Force of the American Sleep Disorders AssociationSleep199215217318411032543
- JohnsMWA new method for measuring daytime sleepiness: the Epworth sleepiness scaleSleep19911465405451798888
- WangWHHeGPXiaoXPGuCChenHYRelationship between brain-derived neurotrophic factor and cognitive function of obstructive sleep apnea/hypopnea syndrome patientsAsian Pac J Trop Med201251190691023146807
- ZhangXModern Sleep MedicineBeijingPeople’s Military Medical Press2007315
- SatterthwaiteTDElliottMAGerratyRTAn improved framework for confound regression and filtering for control of motion artifact in the preprocessing of resting-state functional connectivity dataNeuroimage20136424025622926292
- YanCGCheungBKellyCA comprehensive assessment of regional variation in the impact of head micromovements on functional connectomicsNeuroimage20137618320123499792
- FoxMDSnyderAZVincentJLThe human brain is intrinsically organized into dynamic, anticorrelated functional networksProc Natl Acad Sci U S A2005102279673967815976020
- GuoWJiangJXiaoCDecreased resting-state interhemispheric functional connectivity in unaffected siblings of schizophrenia patientsSchizophr Res2014152117017524325975
- SaadZSGottsSJMurphyKTrouble at rest: how correlation patterns and group differences become distorted after global signal regressionBrain Connect201221253222432927
- DaiXJGongHHWangYXGender differences in brain regional homogeneity of healthy subjects after normal sleep and after sleep deprivation: a resting-state fMRI studySleep Med201213672072722503940
- RaichleMEMacLeodAMSnyderAZA default mode of brain functionProc Natl Acad Sci U S A200198267668211209064
- PrilipkoOHuynhNSchwartzSTask positive and default mode networks during a parametric working memory task in obstructive sleep apnea patients and healthy controlsSleep20113429330121358846
- BeebeDWGroeszLWellsCNicholsAMcGeeKThe neuropsychological effects of obstructive sleep apnea: a meta-analysis of norm-referenced and case-controlled dataSleep200326329830712749549
- YaouhiKBertranFClochonPA combined neuropsychological and brain imaging study of obstructive sleep apneaJ Sleep Res2009181364819250174
- ChetelatGDesgrangesBde laSayetteVDissociating atrophy and hypometabolism impact on episodic memory in mild cognitive impairmentBrain20031261955196712821520
- PeifferCPolineJBThivardLAubierMSamsonYNeural substrates for the perception of acutely induced dyspneaAm J Respir Crit Care Med2001163495195711282772
- BanzettRBMulnierHEMurphyKRosenSDWiseRJAdamsLBreathlessness in humans activates insular cortexNeuroreport200011102117212010923655
- EvansKCBanzettRBAdamsLMcKayLFrackowiakRSCorfieldDRBOLD fMRI identifies limbic, paralimbic, and cerebellar activation during air hungerJ Neurophysiol20028831500151112205170
- BrannanSLiottiMEganGNeuroimaging of cerebral activations and deactivations associated with hypercapnia and hunger for airProc Natl Acad Sci U S A20019842029203411172070
- FrysingerRCHarperRMCardiac and respiratory relationships with neural discharge in the anterior cingulate cortex during sleep-walking statesExp Neurol19869422472633770117
- AyalonLAncoli-IsraelSAkaAAMcKennaBSDrummondSPRelationship between obstructive sleep apnea severity and brain activation during a sustained attention taskSleep200932337338119294957
- HendersonLAWooMAMaceyPMNeural responses during Valsalva maneuvers in obstructive sleep apnea syndromeJ Appl Physiol20039431063107412433858
- CavannaAETrimbleMRThe precuneus: a review of its functional anatomy and behavioural correlatesBrain2006129356458316399806
- AyalonLAncoli-IsraelSKlemfussZShalautaMDDrummondSPIncreased brain activation during verbal learning in obstructive sleep apneaNeuroimage20063141817182516626972
- DrummondSPMeloyMJYanagiMAOrffHJBrownGGCompensatory recruitment after sleep deprivation and the relationship with performancePsychiatry Res2005140321122316263248
- MorcomAMGoodCDFrackowiakRSRuggMDAge effects on the neural correlates of successful memory encodingBrain200312621321912477708
