 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
Background and purpose
Neuroimaging studies of relapsing–remitting multiple sclerosis (RRMS) have found structural disconnection and large-scale neural network dysfunction. However, few studies have explored the local brain activity of RRMS patients in the resting state.
Patients and methods
In this study, regional homogeneity (ReHo) and resting-state functional connectivity (FC) were used to investigate intra- and interregional synchronized activity in 22 patients with RRMS and 22 matched healthy controls (HCs).
Results
Compared with HCs, patients with RRMS showed significantly decreased ReHo in the left insula and right caudate. Through further seed-based FC analysis, we found decreased FC between the left insula and left precentral gyrus in patients with RRMS compared with HCs, as well as increased FC between the right caudate and right dorsolateral prefrontal cortex. Pearson’s correlation analysis showed that a decreased ReHo value in the left insula was associated with an increased total white matter lesion loads (TWMLL) score (r=−0.594, P=0.004) or a worsened paced auditory serial addition test score (r=0.536, P=0.010). No other significant correlations were observed between the FC value (left insula − left precentral gyrus) and clinical scores (P=0.246–0.982). The ReHo value of the right caudate was negatively correlated with disease duration (r=−0.526, P=0.012) and with the TWMLL score (r=−0.596, P=0.003). Moreover, a positive correlation was observed between the FC value (right caudate − right dorsolateral prefrontal cortex) and the TWMLL score (r=0.523, P=0.012) or the modified fatigue impact scale-5 score (r=0.608, P=0.003).
Conclusion
Together, these findings suggest that the insula with regional dysfunction involves disconnection with sensorimotor regions, and demyelinating lesion-related intra- and interregional dysfunction in the caudate is associated with the impact of fatigue on cognitive control functions. Abnormal synchronization of intra- and interregional activity in the insula and caudate may play important roles in the pathology of RRMS.
Introduction
Multiple sclerosis (MS) is a chronic, inflammatory, demyelinating, and neurodegenerative disease of the central nervous system which is a common cause of progressive neurological deficits in young adults.Citation1 Moreover, neurological symptoms including sensorimotor deficits, cognitive impairment, and fatigueCitation2–Citation4 in patients with MS affect a range of daily activities such as working, driving, and social integration.Citation5 Nevertheless, the mechanisms of MS-related neurocognitive deficits and psychological health issues remain unknown. Diffuse white matter and gray matter lesions are widely believed to account for these neurocognitive problems and disabilities. Conventional magnetic resonance imaging (MRI) currently serves as an important diagnostic tool for MS, but it possesses shortcomings, including low sensitivity to gray matter lesions and diffuse white matter involvement, and a low ability to predict clinical status.Citation6
Over the past two decades, advanced MRI techniques have enriched our understanding of the mechanisms underlying the structural and functional abnormalities that cause the neurological symptoms of MS. Together, cross-sectional and longitudinal studies have shown that gray matter atrophy occurs in the early stages of the disease,Citation7 and is associated with clinical disabilityCitation8 and cognitive deterioration.Citation9,Citation10 These studies have also shown that cortical atrophy preferentially affects the frontotemporal area in patients with relapsing–remitting MS (RRMS).Citation11 In addition to the white matter abnormalities identified by conventional MRI, structural studies utilizing diffusion-tensor MRI have revealed abnormalities in normal-appearing white matter, cortex, and deep gray matter nuclei, particularly in the frontal and temporal regions, the motor strip, and the basal ganglia at the earliest stages of MS.Citation12,Citation13 These studies reported that these abnormalities had a more pronounced association with increased disease duration and neurologic impairments.Citation12,Citation13 Studies of resting-state functional MRI (rs-fMRI) have demonstrated the integrity of intra-network functional connectivity (FC) and changes in inter-network coupling in MS patients in the default mode network,Citation14–Citation16 sensorimotor network,Citation17,Citation18 and visual network.Citation19 These findings suggest that functional disconnection parallels both structural damage and clinical impairment.
Regional homogeneity (ReHo) provides an approach to investigate the local synchronization of spontaneous activity within neighboring voxels in the resting state.Citation20 Previous studies have demonstrated that ReHo has high test–retest reliability and neurobiological relevance, and it could serve as a neuroimaging marker to investigate the human brain function.Citation21,Citation22 ReHo analysis has been successfully used to detect brain dysfunction in many diseases, such as neuromyelitis optica,Citation23 schizophrenia,Citation24 depression,Citation25 and autism.Citation26 Dogonowski et alCitation27 have shown a link between impaired regional integration in the cerebellum and general disability or ataxia using the ReHo method in MS. To the best of our knowledge, little is known about local functional homogeneity across the brain in MS. Furthermore, it is unclear whether such changes simply imply an abnormal local synchronization of low-frequency blood oxygenation level dependent (BOLD) signals reflective of abnormal metabolic activity or a successive relationship. FC is provided as a statistical correlation coefficient of BOLD signal coherence between remote brain regions and can help to further clarify this issue.
In this study, we hypothesized that regional function and subserving interregional connectivity networks would be affected by abnormal local functional homogeneity in RRMS. To test this hypothesis, we collected rs-fMRI data from patients with RRMS and compared the data from the RRMS group with data from healthy controls (HCs) to examine both intraregional synchronized activity (ie, ReHo) and interregional synchronized activity (ie, FC) within the whole brain in RRMS. We also predicted that RRMS patients would show abnormal brain activity associated with clinical marks. This study may enrich our understanding of the neural underpinnings of RRMS.
Materials and methods
Participants
Twenty-two MS patients (9 males, 13 females; mean age: 44.6 [range: 21–57] years) were recruited from the Department of Neurology of the First Affiliated Hospital of Nanchang University, according to McDonald’s criteria.Citation28 The inclusion criteria for the patients were as follows: 1) presenting with a relapsing–remitting courseCitation29 and 2) no clinical relapse symptoms occurring during scanning. The exclusion criteria were as follows: 1) an Expanded Disability Status Scale score >2.5 (corresponding to minimal disability),Citation30 2) treatment with immunomodulatory medication, and 3) any brain lesions attributed to tumor or stroke, as assessed on the basis of medical history or MRI.
In addition, we recruited 22 age- and sex-matched healthy subjects to form a control group. All of the subjects self-reported as right-handed. This study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board of the First Affiliated Hospital of Nanchang University. Written informed consent was obtained from each subject prior to the study.
Data acquisition
All MR images were obtained on a 3.0 T MR scanner (Trio Tim; Siemens Medical Systems, Erlangen, Germany). Foam pads and earplugs were used to reduce head motion and scanner noise, respectively. T2-weighted images, three-dimensional T1-weighted images, and rs-fMRI images were acquired using the following sequences: 1) T2-weighted turbo spin-echo imaging (repetition time [TR]/echo time [TE] =5,100/117 ms, number of excitations =3, echo train length =11, matrix =416×416, field of view [FOV] =240×240 mm, slice =22, slice thickness =6.5 mm, orientation = axial); 2) three-dimensional T1-weighted imaging (TR/TE =1,900/2.26 ms, number of excitations =1, matrix =240×256, FOV =215×230 mm, slices =176, slice thickness =1.0 mm, orientation = sagittal); and 3) rs-fMRI scan using a standard T2*-weighted gradient echo sequence with the following parameters: TR/TE =2,000/30 ms, flip angle =90°, FOV =200×200 mm, matrix =64×64, 30 interleaved axial slices with 4 mm thickness with an interslice gap of 1.2 mm, number of time points =240. During the fMRI scanning, subjects were instructed to keep their eyes closed, not to think about anything in particular, and not to fall asleep.
Data preprocessing
All rs-fMRI data were processed using Statistical Parametric Mapping (SPM8, Welcome Trust Centre for Neuroimaging, London, UK) and Data Processing Assistant for Resting-State fMRI Advanced Edition V2.2 (DPARSFA; Chinese Academy of Sciences, Beijing, People’s Republic of China). For each subject, we discarded the first ten volumes to avoid the possible effects of scanner instability and the adaptation of subjects to the scanner environment. The preprocessing steps included slice timing to correct within-scan acquisition time differences between slices, realignment to the first volume to correct head motion (a six-parameter spatial transformation), spatial normalization to the Montreal Neurological Institute template using a six-parameter spatial transformation, and resampling images into a spatial resolution of 3×3×3 mm3. We then performed signal linear detrending and voxelwise temporal bandpass filtering (0.01–0.08 Hz), and subsequently regressed out the nuisance covariates, including eight covariates (ie, signals from white matter and CSF, as well as six head motion parameters).
Recent studiesCitation31–Citation33 have indicated that head motion can significantly influence the results derived from rs-fMRI. So, we computed the voxel-specific framewise displacement (FDvox) for each subject by using the DPARSF toolbox as described in a previous study.Citation34 Using two-sample t-tests, we estimated the mean FDvox across all subjects and found no significant group difference (report in ). In this study, we excluded subjects for further analysis if the translation or rotation of head movement was greater than 2 mm or 2° in any direction.
Table 1 Demographics and clinical characteristics of healthy controls and RRMS patients
ReHo analysis
rs-fMRI data without spatial smoothing were used for ReHo analysis with DPARSFA.Citation35 Kendall’s coefficient of concordance was calculated to measure the local synchronization of 27 nearest neighboring voxels with EquationEquation 1(1) ,Citation20 and the ReHo value was assigned to the central voxel. Then, a ReHo map was obtained in a voxelwise manner. Thus, each individual ReHo map was generated. A standardized ReHo map was created by dividing every individual ReHo map by each participant’s global mean Kendall’s coefficient concordance value within the brain mask. Finally, the standardized ReHo maps were spatially smoothed using a Gaussian kernel (full width at half maximum) of 4 mm. Notably, spatial smoothing before ReHo calculation dramatically increased the ReHo value.Citation35
(1) Where W is the Kendall’s coefficient concordance for a given voxel, ranging from 0 to 1; Ri is the sum rank of the ith time point; K is the number of time series within a measured cluster (27, one given voxel plus the number of its neighbors); n is the number of ranks (n=230 time points).
FC analysis
A seed-based correlation approach was used for FC analysis. The seed was defined in the difference regions of the ReHo analysis. FC analysis was performed using the data resulting from preprocessing. We calculated the temporal correlation between the seed region and every other voxel within the brain. We used eight nuisance covariates: the six head motion parameters, a white matter time course, and a cerebrospinal fluid time course. Fisher’s z transformation was used to improve the normality of the correlation coefficients, and the resulting correlation coefficients were transformed into z values. All of the above procedures were executed using DPARSFA software.
Statistical analysis
Statistical analysis was performed using the software Statistical Package for the Social Sciences Version 17.0 (SPSS Inc., Chicago, IL, USA) for demographic and clinical data and SPM8 for fMRI data. Age and sex were included as covariates in the present and following statistical analysis of functional data. An independent two-sample t-test was performed on the standardized ReHo and FC maps using SPM8 software. ReHo and FC were reported at a two-tailed voxelwise significance level threshold of P<0.01 and cluster level of P<0.05 with Gaussian Random Field (GRF) correction. We used Pearson’s correlation analysis to assess the relationship between the rs-fMRI measurements (ie, ReHo values and the strength of FC from the connections indicating significant group differences) and the clinical presentation of the subjects. The statistical significance level was set at P<0.05 (uncorrection).
Results
Demographic and clinical data
provides the demographic information of the RRMS patients and HCs, and demonstrates the clinical features of RRMS patients. The two groups displayed no significant differences in age, sex, or handedness.
Decreased ReHo in RRMS patients
Compared with HCs, RRMS patients showed decreased ReHo in the left insula (Brodmann Area [BA] 13) and right caudate. No significant increase in ReHo was observed. and show the group differences in ReHo between RRMS patients and HCs (voxel-level P<0.01, cluster-level P<0.05, GRF correction).
Figure 1 Group differences in ReHo between RRMS patients and HCs (voxel-level P<0.01, cluster-level P<0.05, GRF correction).
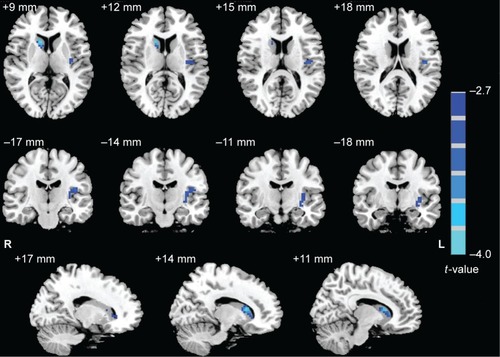
Table 2 Brain regions showing a significant difference in ReHo map between RRMS patients and HCs (voxel-level P<0.01 and cluster-level P<0.05, GRF correction)
Seed-based FC of altered ReHo regions
Based on group comparison in ReHo, the left insula and right caudate were selected as seed regions of interest for FC analysis. Consequently, we found decreased FC between the left insula and left precentral gyrus (lPreCG) (BA6) in patients with RRMS compared with HCs, while we observed increased FC between the right caudate and right dorsolateral prefrontal cortex (rDLPFC) (BA9) in patients (voxel-level P<0.01, cluster-level P<0.05, GRF correction) ( and ).
Figure 2 Group differences in seed-based functional connectivity between RRMS patients and HCs (voxel-level P<0.01, cluster-level P<0.05, GRF correction).
Abbreviations: FC, functional connectivity; GRF, Gaussian Random Field; HCs, healthy controls; RRMS, relapsing–remitting multiple sclerosis.
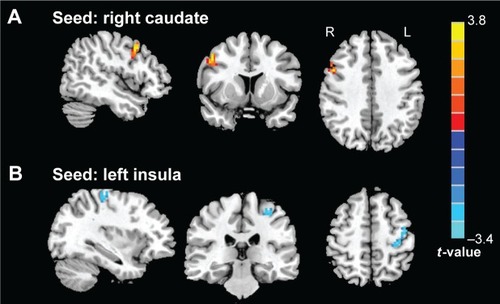
Table 3 Group differences in seed-based functional connectivity in altered ReHo regions (voxel-level P<0.01 and cluster-level P<0.05, GRF correction)
Correlations between ReHo/FC and clinical variables
We found that a decreased ReHo in the left insula was negatively correlated with total white matter lesion loads (TWMLL) score (), while it was positively correlated with paced auditory serial addition test (PASAT) score (). We found that a decreased ReHo in the right caudate was negatively correlated with TWMLL score () and disease duration score (). No other significant correlations were identified between altered ReHo and the other clinical scores assessed. A positive correlation was observed between the intensity of FC (right caudate − rDLPFC) and TWMLL score () or modified fatigue impact scale-5 (MFIS-5) score (). No other significant correlations were identified between the intensity of FC (left insula − lPreCG) and the clinical scores assessed ().
Figure 3 Correlations between ReHo/FC and clinical variables (uncorrection).
Abbreviations: FC, functional connectivity; MFIS-5, modified fatigue impact scale-5; PASAT, paced auditory serial addition test; rDLPFC, right dorsolateral prefrontal cortex; ReHo, regional homogeneity; TWMLL, total white matter lesion loads.
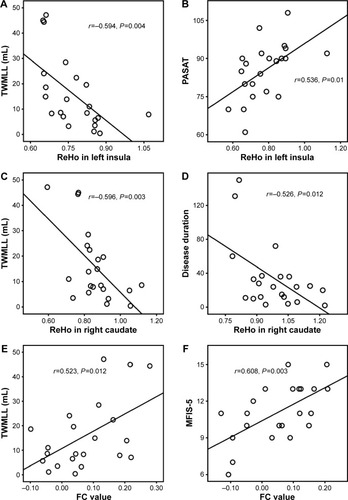
Table 4 Correlations between ReHo/FC and clinical variables
Discussion
In this study, the RRMS group presented a specifically decreased ReHo in the left insula and right caudate. Furthermore, the left insula showed decreased FC with the lPreCG, while the right caudate showed increased FC with the dorsolateral prefrontal cortex in patients with RRMS. We also identified an association between ReHo/FC and clinical variables such as lesion load (TWMLL) and PASAT in patients with RRMS. This evidence for local functional homogeneity and interregional FC contributes to our understanding of MS-related changes under neuropsychiatric conditions.
Decreased intraregional synchronized activity (ReHo) in RRMS
Our study found decreased ReHo in the left insula and right caudate in RRMS patients. ReHo is defined by the temporal coherence or synchronization of the BOLD time series between a given voxel and its nearest neighboring voxels,Citation20,Citation37 and can be interpreted as an index of network centrality to characterize the importance of the node in the human brain connectome by its local functional interactions.Citation37 Decreased ReHo may suggest a disease-related dysfunction caused by dysregulation over a short distance.
In this study, we observed a decreased ReHo in the left insula. The insula plays roles in sensorimotor function, emotion, and decision makingCitation38 as well as high-level cognitive processing,Citation39,Citation40 and is a hub of the salience network.Citation41 The insula comprises anterior and posterior sections: the anterior insula is specifically sensitive to salient environmental events,Citation41 while the posterior insula is involved in somatosensory–vestibular interactions.Citation42,Citation43 Moreover, decreased ReHo in left insula was positively correlated with PASAT score, which provides further evidence supporting a role for insula in high-level cognitive processing. Charil et alCitation44 have found that a relationship between TWMLL score and cortical loss in the insula was present in MS patients. Brain structure is considered to be the basis of function; our results show that decreased ReHo in the left insula is negatively correlated with TWMLL score, which corroborates this dogma.
The caudate is an important region involved in movement planning and executionCitation45 and is the main input node of the basal ganglia circuitry that receives glutamatergic fibers from the sensorimotor cortex.Citation46 Moreover, the caudate is linked to the neural networks underlying affective and cognitive function.Citation45,Citation47 In the current study, we detected decreased ReHo in the right caudate in patients with RRMS. These findings are consistent with previous reports that found abnormalities in caudate structureCitation48,Citation49 and functionCitation50,Citation51 in patients with MS. In this study, abnormal ReHo in the caudate might reflect changes in somatosensory, motor, and cognitive functions in RRMS patients. Moreover, the ReHo of the right caudate was negatively correlated with RRMS duration and TWMLL score, indicating that the state of the right caudate may be a marker for the progression of RRMS. The decreased regional coherence in the left insula and right caudate is consistent with the reduced functional activation and structural abnormality reported in previous literature.Citation38–Citation51
Abnormal interregional synchronized activity (FC) in RRMS
To clarify how regional coherence affects interregional connectivity, we investigated the FC between regions with altered ReHo (left insula and right caudate) and other brain regions.
We observed decreased FC between the left insula and the lPreCG in patients with RRMS, which suggested dysfunction in the insula-related motor network. Structural studies have demonstrated that the insula plays the role of the so-called “rich club” within the connectomes to form the basis of its central communication.Citation52–Citation54 Rs-fMRI studies also found that connections between the insula cortex and both the pre- and post-central cortex were mainly associated with the functional integration of somatosensory, motor, and auditory information.Citation55,Citation56 He et alCitation57 suggest that the decreased network efficiency in MS might reflect the disrupted functional neuronal organization of spatially distributed cortical regions as well as disruptions in the integrity of large-scale interconnected brain systems. In our study, the FC value between the left insula and lPreCG was found to be unassociated with clinical variables. Therefore, our interpretation of the current abnormal FC data in RRMS is cautious.
Connectivity between the rDLPFC and right caudate has been associated with increased restricted and repetitive behaviors that involve frontostriatal circuitry.Citation58 Also, few studies have found that increased FC might represent a neural mechanism to compensate for disease-induced dysfunction.Citation59,Citation60 In the current study, we found that increased FC between the right caudate and rDLPFC positively correlated with TWMLL or MFIS-5 score. MS lesions are predominantly located in the white matter and, therefore, mainly affect axonal conduction. We suggest that FC between the right caudate and rDLPFC may be affected by MS lesions. The fatigue-related MFIS-5 scale is an abbreviated version used to assess the impact of fatigue on physical, cognitive, and psychosocial functioning. Finke et alCitation61 found that fatigue severity was correlated with FC of basal ganglia nuclei (ie, reduction in caudate volume) with medial prefrontal cortex, precuneus, and motor cortex. Fatigue is associated with distinct alterations of basal ganglia FC independent of overall disability. In our view, the association of the caudate with interregional hyperconnectivity acts to compensate for fatigue on cognitive control functions.
Interestingly, we observed a laterality alteration in interregional FC with altered ReHo regions (left insula and right caudate). There are two possible explanations: 1) disruption of anatomic connectivity of the corpus callosum in MS may affect the interhemispheric functional connecting interactionsCitation62 and 2) compensatory functional reorganization ensues in both subcortical and cortical synchronization in the absence of corpus callosum.Citation63 In our study, we speculate that increased FC between the right caudate and rDLPFC may present compensatory function. In addition, compensatory functional reorganization as a complex and slow evolutionary fashion remains in need of further study to evaluate a specific functional pathway to explain decreased FC between the left insula and lPreCG.
Limitations
Certain MS lesions disrupted corresponding white matter pathways, which likely resulted in altered FC between gray matter regions and caused clinical impairment. However, cortical reorganization could improve the clinical symptoms caused by structural damage.Citation60 In future studies, the effects of lesion location should be considered when investigating the functional–structural–clinical relationships involved in MS. Second, decreased FC between the insula and PreCG subserving sensorimotor dysfunction in RRMS was found in our study. Future studies should be designed to include a scale to measure motor function in order to obtain more information about the mechanism of motor impairment. Our study involved Pearson’s correlation analysis for correlations between clinical data and neurocognitive metrics rather than multiple comparisons analysis, which should be applied in further studies.
Conclusion
This study suggests that RRMS patients present regional dysfunctions in the insula and caudate and decreased FC of the insula subserving sensorimotor dysfunction, and demy-elinating lesion-related intra- and interregional dysfunction in the caudate is associated with the impact of fatigue on cognitive control functions. The alteration of intra- and interregional activity in the insula and caudate may play an important role in the pathology of RRMS.
Acknowledgments
This study was supported by the National Science Foundation of China (Grant Nos 81101041, 81260217, 81460263, 81560284), the Science and Technology Support Program of Jiangxi, People’s Republic of China (No 20151BBG70224), the Science and Technology Project of Jiangxi Provincial Health and Family Planning Commission (Grant No 20143071), and the “Gan Po” engineering excellence 555 project of Jiangxi Province. The authors thank their patients and volunteers for participating in this study. They also thank the anonymous reviewers for their constructive suggestions to improve this work.
Disclosure
The authors report no conflicts of interest in this work.
References
- BrückWThe pathology of multiple sclerosis is the result of focal inflammatory demyelination with axonal damageJ Neurol2005252Suppl 5v3v916254699
- GuimarãesJSáMJCognitive dysfunction in multiple sclerosisFront Neurol201237422654782
- BakshiRFatigue associated with multiple sclerosis: diagnosis, impact and managementMult Scler20039321922712814166
- PantanoPMaineroCLenziDA longitudinal fMRI study on motor activity in patients with multiple sclerosisBrain2005128Pt 92146215315901646
- FilippiMRiccitelliGMattioliFMultiple sclerosis effects of cognitive rehabilitation on structural and functional MR imaging measures-an explorative studyRadiology2012262393294022357892
- LövbladKOAnzaloneNDörflerAMR imaging in multiple sclerosis: review and recommendations for current practiceAJNR Am J Neuroradiol201031698398920019103
- ChardDTGriffinCMParkerGJKapoorRThompsonAJMillerDHBrain atrophy in clinically early relapsing-remitting multiple sclerosisBrain2002125Pt 232733711844733
- PrinsterAQuarantelliMLanzilloRA voxel-based morphom-etry study of disease severity correlates in relapsing-remitting multiple sclerosisMult Scler2010161455420028706
- BenedictRHBruceJMDwyerMGNeocortical atrophy, third ventricular width, and cognitive dysfunction in multiple sclerosisArch Neurol20066391301130616966509
- AmatoMPPortaccioEGorettiBAssociation of neocortical volume changes with cognitive deterioration in relapsing-remitting multiple sclerosisArch Neurol20076481157116717698706
- BendfeldtKKapposLRadueEWBorgwardtSJProgression of gray matter atrophy and its association with white matter lesions in relapsing-remitting multiple sclerosisJ Neurol Sci20092851–226826919500803
- LiYJewellsVKimMDiffusion tensor imaging based network analysis detects alterations of neuroconnectivity in patients with clinically early relapsing-remitting multiple sclerosisHum Brain Mapp201334123376339122987661
- RovarisMAgostaFPaganiEFilippiMDiffusion tensor MR imagingNeuroimaging Clin N Am2009191374319064198
- HawellekDJHippJFLewisCMCorbettaMEngelAKIncreased functional connectivity indicates the severity of cognitive impairment in multiple sclerosisProc Natl Acad Sci U S A201110847190661907122065778
- ZhouFZhuangYGongHAltered inter-subregion connectivity of the default mode network in relapsing remitting multiple sclerosis: a functional and structural connectivity studyPLoS One201497e10119824999807
- RoccaMAAbsintaMValsasinaPAbnormal connectivity of the sensorimotor network in patients with MS: a multicenter fMRI studyHum Brain Mapp20093082412242519034902
- CeccarelliARoccaMAValsasinaPStructural and functional magnetic resonance imaging correlates of motor network dysfunction in primary progressive multiple sclerosisEur J Neurosci20103171273128020345920
- RoosendaalSDSchoonheimMMHulstHEResting state networks change in clinically isolated syndromeBrain2010133Pt 61612162120356855
- TewariePSchoonheimMMStamCJCognitive and clinical dysfunction, altered MEG resting-state networks and thalamic atrophy in multiple sclerosisPLoS One201387e6931823935983
- ZangYJiangTLuYHeYTianLRegional homogeneity approach to fMRI data analysisNeuroimage200422139440015110032
- JiangLXuTHeYToward neurobiological characterization of functional homogeneity in the human cortex: regional variation, morphological association and functional covariance network organizationBrain Struct Funct201522052485250724903825
- ZuoXNXuTJiangLToward reliable characterization of functional homogeneity in the human brain: preprocessing, scan duration, imaging resolution and computational spaceNeuroimage20136537438623085497
- LiangPLiuYJiaXRegional homogeneity changes in patients with neuromyelitis optica revealed by resting-state functional MRIClin Neurophysiol2011122112112720621555
- LiuHLiuZLiangMDecreased regional homogeneity in schizophrenia: a resting state functional magnetic resonance imaging studyNeuroreport2006117192216361943
- YaoZWangLLuQLiuHTengGRegional homogeneity in depression and its relationship with separate depressive symptom clusters: a resting-state fMRI studyJ Affect Disord2009115343043819007997
- PaakkiJJRahkoJLongXAlterations in regional homogeneity of resting-state brain activity in autism spectrum disordersBrain Res2010132116917920053346
- DogonowskiAMAndersenKWMadsenKHMultiple sclerosis impairs regional functional connectivity in the cerebellumNeuroimage Clin2014413013824371795
- PolmanCHReingoldSCBanwellBDiagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteriaAnn Neurol201169229230221387374
- HurwitzBJThe diagnosis of multiple sclerosis and the clinical subtypesAnn Indian Acad Neurol200912422623020182569
- KurtzkeJFHistorical and clinical perspectives of the expanded disability status scaleNeuroepidemiology20083111918535394
- Van DijkKRSabuncuMRBucknerRLThe influence of head motion on intrinsic functional connectivity MRINeuroimage201259143143821810475
- YanCGCraddockRCHeYMilhamMPAddressing head motion dependencies for small-world topologies in functional connectomicsFront Hum Neurosci2013791024421764
- PowerJDBarnesKASnyderAZSchlaggarBLPetersenSESpurious but systematic correlations in functional connectivity MRI networks arise from subject motionNeuroimage20125932142215422019881
- YanCGCheungBKellyCA comprehensive assessment of regional variation in the impact of head micromovements on functional connectomicsNeuroimage20137618320123499792
- Chao-GanYYu-FengZDPARSF: A MATLAB toolbox for “pipeline” data analysis of resting-state fMRIFront Syst Neurosci201041320577591
- ShuNLiuYLiKDiffusion tensor tractography reveals disrupted topological efficiency in white matter structural networks in multiple sclerosisCereb Cortex201121112565257721467209
- JiangLZuoXNRegional homogeneity: A multimodal, multiscale neuroimaging marker of the human connectomeNeuroscientist Epub2015713
- ScottTRInsular cortex as a mediator of emotion: commentary on emotion and decision-making explained, by Edmund T. RollsCortex20145920620724856289
- WangXFanYZhaoFAltered regional and circuit resting-state activity associated with unilateral hearing lossPLoS One201495e9612624788317
- FangWLvFLuoTAbnormal regional homogeneity in patients with essential tremor revealed by resting-state functional MRIPLoS One201387e6919923869236
- MenonVUddinLQSaliency, switching, attention and control: a network model of insula functionBrain Struct Funct20102145–665566720512370
- HashimotoTTaokaMObayashiSHaraYTanakaMIrikiAModulation of cortical vestibular processing by somatosensory inputs in the posterior insulaBrain Inj20132713–141685169124266797
- CeraNTartaroASensiSLModafinil alters intrinsic functional connectivity of the right posterior insula: a pharmacological resting state fMRI studyPLoS One201499e10714525237810
- CharilADagherALerchJPZijdenbosAPWorsleyKJEvansACFocal cortical atrophy in multiple sclerosis: relation to lesion load and disabilityNeuroimage200734250951717112743
- RobinsonJLLairdARGlahnDCThe functional connectivity of the human caudate: an application of meta-analytic connectivity modeling with behavioral filteringNeuroimage201260111712922197743
- LoitfelderMFazekasFKoschutnigKBrain activity changes in cognitive networks in relapsing-remitting multiple sclerosis – insights from a longitudinal FMRI studyPLoS One201494e9371524718105
- Di MartinoAScheresAMarguliesDSFunctional connectivity of human striatum: a resting state FMRI studyCereb Cortex200818122735274718400794
- CavallariMCeccarelliAWangGYMicrostructural changes in the striatum and their impact on motor and neuropsychological performance in patients with multiple sclerosisPLoS One201497e10119925047083
- MotlRWHubbardEASreekumarNPallidal and caudate volumes correlate with walking function in multiple sclerosisJ Neurol Sci20153541–2333625959979
- DeLucaJGenovaHMHillaryFGWylieGNeural correlates of cognitive fatigue in multiple sclerosis using functional MRIJ Neurol Sci20082701–2283918336838
- NebelKWieseHSeyfarthJActivity of attention related structures in multiple sclerosis patientsBrain Res2007115115016017397807
- van den HeuvelMPKahnRSGoñiJSpornsOHigh-cost, high-capacity backbone for global brain communicationProc Natl Acad Sci U S A201210928113721137722711833
- van den HeuvelMPSpornsORich-club organization of the human connectomeJ Neurosci20113144157751578622049421
- CollinGSpornsOMandlRCvan den HeuvelMPStructural and functional aspects relating to cost and benefit of rich club organization in the human cerebral cortexCereb Cortex20142492258226723551922
- HeYWangJWangLUncovering intrinsic modular organization of spontaneous brain activity in humansPLoS One200944e522619381298
- MeunierDAchardSMorcomABullmoreEAge-related changes in modular organization of human brain functional networksNeuroimage200944371572319027073
- HeYDagherAChenZImpaired small-world efficiency in structural cortical networks in multiple sclerosis associated with white matter lesion loadBrain2009132Pt 123366337919439423
- DelmonteSGallagherLO’HanlonEMcGrathJBalstersJHFunctional and structural connectivity of frontostriatal circuitry in autism spectrum disorderFront Hum Neurosci2013743023964221
- ZhouFGongHLiFAltered motor network functional connectivity in amyotrophic lateral sclerosis: a resting-state functional magnetic resonance imaging studyNeuroreport2013241265766223839257
- ZhouFQTanYMWuLZhuangYHeLCGongHHIntrinsic functional plasticity of the sensory-motor network in patients with cervical spondylotic myelopathySci Rep20155997525897648
- FinkeCSchlichtingJPapazoglouSAltered basal ganglia functional connectivity in multiple sclerosis patients with fatigueMult Scler201521792593425392321
- ZhouYMilhamMZuoXNFunctional homotopic changes in multiple sclerosis with resting-state functional MR imagingAJNR Am J Neuroradiol20133461180118723348760
- CifelliAMatthewsPMCerebral plasticity in multiple sclerosis; insights from fMRIMult Scler20028319319912120689
