Abstract
With the investigation of molecular targets, many agents, such as trastuzumab and ramucirumab, have attained a positive outcome in oncotherapy. Vascular endothelial growth factor (VEGF) is considered a potent factor in angiogenesis and plays an important role in the growth of tumors. Moreover, both VEGF and its receptor are usually excessively expressed in solid tumors and could be hopeful targets for the treatment of neoplasms. Apatinib (YN968D1) is an oral small-molecule tyrosine kinase inhibitor of VEGFR-2. By inhibiting several signaling transduction pathways, it restrains angiogenesis and subsequently controls tumorigenesis. According to current studies, apatinib shows promising application in various solid tumors as a post-second- and post-third-line treatment. It could significantly improve the median overall survival and progression-free survival of patients with tolerated adverse reactions. This paper aims to summarize the recent research on apatinib including the mechanism, pharmacokinetics, trials, adverse reactions, and prospect as a treatment.
Introduction
Solid tumors, as the primary cause of cancer death, have caused great societal and economic effects.Citation1 Generally, surgery is the most effective treatment for early-stage tumors, but most patients experience recurrence after radical surgery.Citation2 In recent years, the development and application of targeted drugs have enabled cancer treatment to enter the “precise” era.Citation3 In addition, the combined use of targeted therapy, immunotherapy, and traditional chemotherapy has innovated the standard treatment for many malignancies, bringing new light to patients with recurrent tumors.Citation4
A tumor requires large amounts of oxygen and nutrients for maintaining its activity, which causes large quantities of blood vessels to develop, creating a vicious cycle.Citation5 Therefore, pathological angiogenesis is considered a hallmark of tumorigenesis, and tumor development can be curbed by blocking this process.Citation6 Vascular endothelial growth factor (VEGF) signaling is the primary rate-limiting step in angiogenesis,Citation7 which is extremely active in most tumors.Citation8,Citation9 Angiogenesis is mainly mediated by two receptor tyrosine kinases VEGFR-1 and VEGFR-2.Citation10 VEGFR-1 has a high affinity for VEGF binding, but the kinases activated by VEGFR-2 appear more energetic and contribute more to angiogenesis.Citation10,Citation11 In addition, VEGFR-1 is expressed on vascular endothelial cells as well as many other cells, such as hematopoietic stem cells, monocytes, and macrophages, but the expression of VEGFR-2 is restricted to endothelial cells.Citation8,Citation12 Therefore, VEGFR-2 is a promising antitumor target.
Apatinib mesylate is a new small-molecule tyrosine kinase inhibitor (TKI) of VEGFR-2. It can significantly inhibit the angiogenesis of neoplasms and has demonstrated tolerance, safety, and efficacy in the clinic.Citation13 To date, apatinib has been investigated in Phase I/II/III trials and has shown positive results in multiple tumors, such as gastric cancer (GC), breast cancer, lung cancer, and esophageal cancer.Citation14–Citation21
The therapeutic mechanism of apatinib
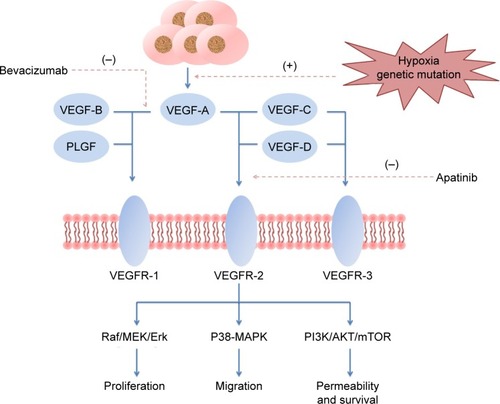
During tumor formation, environmental changes such as hypoxia and genetic mutations to molecules such as K-ras, p53, or HER2/ErbB2 induce the expression of VEGF in cancer cells.Citation22 Meanwhile, VEGFR-2 expression is also upregulated in tumor vasculature compared to the normal vascular system.Citation12,Citation23 After the binding of VEGF and VEGFR-2, the carboxyterminal TK domain of the receptor undergoes autophosphorylation, leading to downstream signaling transduction. Apatinib can block the phosphorylation of VEGFR-2 by binding its intracellular adenosine triphosphate sites, thereby restraining several signaling pathways: the Raf/MEK/Erk pathway, which results in the proliferation of endothelial cells; the p38-MAPK pathway, which stimulates the migration of endothelial cells; and the PI3K/AKT/mTOR pathway, which strengthens the survival of endothelial cells and enhances vascular permeability ().Citation12,Citation24 Additionally, the kinase activities of Ret, c-kit, and c-src have been proven to be suppressed by apatinib in vitro in enzyme experiments, but the specific mechanisms need further exploration.Citation25,Citation26 As a result, apatinib can effectively restrain proliferation, migration, and permeability of vascular endothelial cells.
Figure 1 The therapeutic mechanism of apatinib.Citation12,Citation22–Citation24
Recently, many studies have shown that apatinib could directly act on tumor cells and promote their apoptosis. Apatinib mediates this process by inhibiting the PI3K/Akt signaling pathway and subsequently upregulating the expression of proapoptotic genes Bax and caspase-9 and downregulating the expression of the anti-apoptotic gene Bcl-2. Further related mechanism research is in progress.Citation27–Citation31 These studies may broaden the therapeutic indications for drugs and provide guidance for drug regimens.
In addition, multidrug resistance (MDR) of tumor cells is an intractable obstacle for cancer treatment.Citation32 The mechanism of MDR is sophisticated, and a study has shown that it can be ascribed to the high expression of cell membrane–bound adenosine triphosphate-binding cassette (ABC) transporters.Citation33 After the signal activates the receptor, the transporter extrudes various types of chemotherapeutic agents out of tumor cells, hence reducing the cytotoxic effect.Citation33 Not by blocking the ERK1/2 or AKT pathway or decreasing the expression of ABCB1 or ABCG2, apatinib could act directly on ABCB1 or ABCG2 and inhibit its efflux function. Thus, apatinib reverses the MDR caused by ABC proteins.Citation34 This discovery proved that apatinib combined with traditional chemotherapeutic drugs could achieve effective results and resist the emergence of MDR.
Pharmacokinetic study of apatinib
Apatinib is mainly metabolized by cytochrome P450 (CYP)3A4/5 and CYP2D6, and the major routes of apatinib bioconversion contain E- and Z-cyclopentyl-3-hydroxylation, N-dealkylation, pyridyl-25-N-oxidation, 16-hydroxylation, dioxygenation, and O-glucuronidation after 3-hydroxylation. Nine primary metabolites have been verified by comparing them with the reference standards (M1-1, M1-2, M1-6, M4-7, M4-8, M6-9, M6-10, M8-2, and M9-2). Patients reach the Cmax of apatinib 2.9–4.7 hours after taking the medication. The cumulative recovery of the administered dose is 76.8%, which is mainly excreted in the feces (69.8% of the dose). Only a small proportion is eliminated via urine (7.02% of the dose). Moreover, ~59.0% of the intake dose is egested unaltered via feces and inappreciable amounts of unchanged apatinib are found in urine, which indicates that apatinib is widely metabolized in the biosystem.Citation35 These data could be used to predict potential drug interactions and pharmacokinetic variability and provide guidance for later experimental design.
A population pharmacokinetic model for apatinib was established and analyzed using data from 106 individuals, including healthy volunteers and patients with malignant tumors. The ultimate model was a two-compartment model that was composed of a complex absorption model including mixed first- and zero-order processes and first-order elimination. The absolute clearance (CL/F) of apatinib was 57.8 L/h, and its apparent volume at steady state was 112.5 L. Three factors influenced the pharmacokinetics of apatinib: dose, cancer status, and gastric surgery state. The exposure of apatinib was proportionately less than the dose increase, and the dose with a reduction in bioavailability was 766 mg. In addition, the administered dose of 425 mg twice daily appeared to have a higher exposure than the dose of 850 mg once daily.Citation36 This study provided a detailed description of the factors that influenced the metabolism of apatinib in the human body, providing guidance for subsequent medications.
A research study was implemented to assess the drug interactions of apatinib with a CYP3A4 inducer (rifampin) or a CYP3A inhibitor (itraconazole). In comparison to the single administration of apatinib, the combination of apatinib and rifampin led to a 5.6-fold absolute clearance (CL/F) and an 83% reduction in plasma area under the curve AUC0–t of apatinib. On the contrary, the combination of apatinib and itraconazole decreased the absolute clearance by 40% and enhanced its AUC0–t by 75%.Citation37 It proved that coadministration of apatinib with a CYP3A4 inducer had a strong influence on the pharmacokinetics of apatinib, while the CYP3A inhibitor had a weak influence. This suggests that we need to pay attention to the interaction between drugs in the formulation of future treatment plans.
The clinical research regarding apatinib
Gastric cancer
At present, the number of newly diagnosed GC patients has increased by ~679,100 each year, and GC has become the second leading primary cause of cancer death in China. Because of the low diagnosis rates, almost 80% of patients were found at advanced cancer stages.Citation38 However, even after receiving first- and second-line cytotoxic chemotherapy, many of them are not cured or relapse a few years later. At present, there are no treatment criteria for patients who have no response to second-line chemotherapy.Citation39
In 2010, a Phase I study was conducted on apatinib, which determined the maximum tolerated dose to be 850 mg once daily. The dose-limiting toxicities were hypertension and hand–foot syndrome that occurred with administration of 1,000 mg. Pharmacokinetic analysis revealed apatinib had an early absorption with a half-life of 9 hours. A total of 37 patients were eligible to evaluate tumor response. Seven patients (18.9%) had partial response (PR) and 24 patients (64.9%) achieved stable disease (SD) with a disease control rate of 83.8% at 8 weeks.Citation40 In 2013, a subsequent Phase II study was performed enrolling 144 patients. The result demonstrated that the progression-free survival (PFS) of patients who received apatinib 850 mg once daily was 3.67 months and the PFS of patients who took 425 mg twice daily was 3.20 months, while the PFS of patients who received placebo was 1.40 months; moreover, the median overall survival (OS) was 4.83, 4.27, and 2.50 months, respectively.Citation41 These two experiments initially evaluated the efficacy and safety of apatinib in GC patients and provided the basis for the design of clinical trials and the determination of dosing schedules.
In 2016, a randomized, double-blind, placebo-controlled Phase III study for GC was carried out to assess the effectiveness and safety of apatinib. A total of 267 patients from 32 centers in China were assigned to take apatinib 850 mg or placebo once daily. The median OS of the apatinib group was 6.5 months (95% CI: 4.8–7.6), in comparison with 4.7 months (95% CI: 3.6–5.4) for the placebo group (P=0.0149; hazard ratio: 0.709, 95% CI: 0.537–0.937; P=0.0156). In addition, apatinib meaningfully prolonged the median PFS compared with placebo (2.6 vs 1.8 months; hazard ratio: 0.444, 95% CI: 0.33–0.61; P<0.0001).Citation18 Furthermore, the efficacy of apatinib on OS was by prolonging PFS, rather than post-progression survival.Citation42 This experiment further proves that apatinib has a significant therapeutic effect on GC patients and has high safety, so it can be used as a drug for treating advanced GC patients.
The effectiveness of many antiangiogenic drugs for advanced GC is listed in . As we can see, all of them significantly improved the PFS and OS of patients. In terms of therapeutic effects (prolonged PFS and OS), multidrug combination therapy achieved better results. But compared to the control group, the effects of these programs (including ramucirumab and bevacizumab) were unsatisfactory. Among monotherapies, the average median PFS and OS were 2.4 and 6.1 months, respectively, and apatinib provided an above-average effect. As for disease control rate, apatinib, compared to its control group, was the most obvious group that could significantly control the disease. Recently, a network meta-analysis evaluated the efficacy and safety of targeted drugs (mainly including apatinib, regorafenib, rilotumumab, everolimus, bevacizumab, and sunitinib) with or without chemotherapy in advanced GC treatment and demonstrated that apatinib showed the best improvement in PFS, OS, and overall response rate.Citation43
Table 1 Efficacy of different antiangiogenic drugs in advanced gastric cancer
To find more detailed clinical effectiveness and safety of apatinib in GC, a real-world study is currently ongoing (ClinicalTrial.gov identifier NCT3333967). In addition, a series of combination drug therapies are being researched to determine the valid therapeutic method. Studies with apatinib in combination with S-1 (ClinicalTrial.gov identifier NCT3271073), XELOX (ClinicalTrial.gov identifier NCT3229096), Paclitaxel (ClinicalTrial.gov identifier NCT3144843), and SOX (ClinicalTrial.gov identifier NCT3007446) are presently underway.
Breast cancer
Breast cancer is heterogeneous in the clinic. There are three biomarkers, the expression of HER-2 and the receptors for progesterone and estrogen, which are usually used to assist in the choice of therapy. However, triple-negative breast cancer (TNBC) lacks the admitted targets for treatment. So far, several potential actionable pathways have been found in TNBC, such as the RAS/RAF/MEK pathway and the PI3K/mTOR pathway.Citation44 Based on the mechanism, apatinib could be applied in TNBC.
In 2014, a prospective, open-label, Phase II, multicenter trial was implemented to evaluate the effect and safety of apatinib monotherapy in patients with metastatic TNBC.Citation19 First, 25 patients were enrolled, who received apatinib 750 mg/day in a 4-week cycle. Then, according to toxicity response, he dose of apatinib was then adjusted to 500 mg/day. Finally, among the 56 appraisable patients, the overall response rate was 10.7% and the clinical benefit rate was 25.0%. The median OS and PFS were 10.6 (95% CI: 5.6–15.7) and 3.3 (95% CI: 1.7–5.0) months, respectively. As a result, the study recommended that a dose of 500 mg/day was suitable for metastatic TNBC.Citation19 In 2017, a non-clinical trial evaluated the potency of apatinib in metastatic breast cancer (MBC), and the result was consistent with previous reports. Moreover, it was found that preceding therapy with bevacizumab had no effect on the efficacy of apatinib.Citation45
So far, many antiangiogenic agents have shown a disappointing effect in MBC. Sunitinib and sorafenib, VEGF receptor tyrosine kinase inhibitors, were the first batch of drugs approved for clinical use. A study reported that high doses of these receptor tyrosine kinase inhibitors could lead to metastasis in a mouse model of breast cancer;Citation46 this finding may affect the clinical application of VEGF drugs. To elucidate their mechanism, many researchers conducted experiments and found that sunitinib and sorafenib were multi-target agents, and that the acceleration process may be ascribed to off-target effects. In addition to acting on VEGFR, these drugs also acted on platelet-derived growth factor receptor and KIT, which are important mediums for the recruitment of pericytes.Citation47,Citation48 As a result, pericytes decreased and led to hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by the Met signaling pathway.Citation49 However, apatinib has less activity toward receptors such as platelet-derived growth factor receptor and KIT.Citation25 Therefore, compared with other agents, this could be an advantage for the treatment of MBC. Currently, a prospective, multicenter, observational study of apatinib, single or combined with capecitabine, has been conducted in patients with metastatic Her-2–negative breast cancer (ClinicalTrial.gov identifier NCT3086785). This study is designed to provide further data about the effect and toxicity of apatinib. Furthermore, patients with TNBC are currently being recruited to demonstrate the antitumor activity of the combination of apatinib and paclitaxel (ClinicalTrial.gov identifier NCT3348098). A Phase II study of apatinib combined with oral vinorelbine in metastatic Her-2–negative breast cancer is also being conducted (ClinicalTrial.gov identifier NCT2768415).
Lung cancer
Lung cancer is the predominant cancer type in China,Citation1 and non-small-cell lung cancer (NSCLC) is the main histological subtype of the disease. With the increasing cognition of the molecular changes that regulate tumor progression, the management of NSCLC has totally changed. However, frequent resistance to therapy makes advanced NSCLC hard to cure.Citation50
In 2017, apatinib was tested as a salvage therapy for patients with advanced NSCLC. Among 42 patients administered a daily dose of 500 mg, 4 patients had PR and 22 patients experienced SD with a disease response rate of 9.5% and a control rate of 61.9%; the median PFS and OS were 4.2 and 6.0 months, respectively.Citation20 The study suggested that apatinib could be used as a valid regimen for advanced NSCLC.
In NSCLC, there were ~45% patients without epidermal growth factor receptor (EGFR) mutations, and these patients were often found to have progressive disease 4–6 months after first-line treatment.Citation51 Therefore, to find a suitable second-line treatment, the trials on the treatment of EGFR wild-type lung cancer were launched.Citation52,Citation53 A group of 16 patients were enrolled and administered apatinib 250–500 mg/day. Three patients exhibited a PR, and eight patients showed SD. The response rate and control rate were 18.8% and 68.8%, respectively. The median PFS was 4.4 months.Citation52 Thus, apatinib may be a choice for EGFR wild-type patients as a second-line treatment. Presently, the wide use of EGFR-TKIs in NSCLC caused excessive EGFR-TKI resistance. Therefore, methods to combat resistance have become a study hotspot. Recently, it was confirmed that combination therapy with apatinib and other EGFR-TKIs, such as erlotinib and gefitinib, could combat acquired EGFR-TKI resistance in NSCLC.Citation54,Citation55 Therefore, apatinib is a good choice as post-second-line treatment for advanced lung cancer.
At present, the Chinese Academy of Medical Sciences is recruiting patients with small cell lung cancer for a Phase II study of apatinib as a third-line and later therapy (Clinical-Trial.gov identifier NCT2995187). In addition, the studies of apatinib plus chemotherapy vs chemotherapy alone as the first-line treatment (ClinicalTrial.gov identifier NCT3164694) and the second-line treatment (ClinicalTrial.gov identifier NCT3256721) of NSCLC are underway.
Esophageal squamous cell carcinoma
Esophageal cancer is a complex disease that needs multidisciplinary therapy. The use of endoscopic procedures has improved the diagnosis and treatment of esophageal cancer. However, because the procedures are costly and often infeasible, many patients are diagnosed past the optimum stage for therapy.Citation56
In 2017, a study reported that apatinib was effective for advanced esophageal squamous cell carcinoma (ESCC). Among 62 patients administered a dose of 500 mg/day, 15 patients had a PR and 31 patients achieved SD, representing a response rate of 24.2% and a disease control rate of 74.2%. The median PFS and OS were 3.8 and 7.0 months, respectively. Compared with second-line chemotherapy, apatinib showed a better impact on advanced ESCC.Citation21
A study of apatinib and irinotecan combination treatment in unresectable or metastatic ESCC is currently in progress (ClinicalTrial.gov identifier NCT3251417) with the purpose of exploring the effectiveness as a second-line treatment for ESCC.
To sum up, the efficacy of apatinib in different trials is shown in .
Table 2 The efficacy of apatinib in different trials
The preclinical study of apatinib for other tumors
Due to antiangiogenesis effects, apatinib could be applied for the treatment of various solid tumors. Currently, there are numerous preclinical and clinical studies looking at the efficacy and safety of apatinib.
In vitro, colon cancer cells (HCT116 and SW480) were treated with different concentrations of apatinib (0, 10, 20, 30, 40, and 50 µM). The investigator found that apatinib could not only inhibit proliferation, migration, and invasion of colon cancer cells, but also regulate the autophagy and apoptosis of the cells.Citation28
In vivo, a murine model of nasopharyngeal carcinoma (NPC) was designed. In total, 132 nude mice, xenografted with CNE-2 NPC cells, were treated with six therapy regimens, including receiving apatinib alone and coadministration of apatinib and cisplatin (DDP), and so on. The results showed that concurrent administration of cisplatin and apatinib could treat NPC.Citation57 Therefore, patients with metastatic NPC are presently being recruited in order to assess apatinib as a maintenance therapy for NPC (ClinicalTrial.gov identifier NCT3180476).
A series of trials were designed to determine the potency of apatinib for osteosarcoma. The growth of osteosarcoma cells could be suppressed because apatinib not only leads to cycle arrest, but also causes apoptosis and autophagy of the cells. In addition, immunoprecipitation verified that VEGFR-2 is directly bound with the signal transducer and activator of transcription 3 (STAT-3). Apatinib inhibits VEGFR-2 and subsequently downregulates STAT-3 and BCL-2 in KHOS cells. The resulting low levels of VEGFR-2/STAT-3/BCL-2 led to apoptosis and autophagy of KHOS cells.Citation27 Therefore, a Phase II/III study of apatinib for advanced osteosarcoma after failure of standard multimodal therapy is underway (ClinicalTrial.gov identifier NCT2711007).
A study of apatinib for the treatment of intrahepatic cholangiocarcinoma indicated that VEGF signaling (through PI3K/Akt/mTOR) plays a role in the growth of the tumor. Apoptosis of tumor cells could be induced by blocking VEGFR-2.Citation58 Therefore, a Phase III study of second-line treatment with apatinib in patients with intrahepatic cholangiocarcinoma is presently ongoing (ClinicalTrial.gov identifier NCT3251443).
The details of the ongoing clinical trials of apatinib are shown in . Furthermore, there are many other trials not mentioned in this article because of space limitations. Based on the posted study results, apatinib demonstrates positive results that make it a good prospect for treatment of various cancer.
Table 3 Ongoing clinical trials with apatinib
Adverse reactions to apatinib
In contrast to placebo, the primary adverse reactions (ARs) of apatinib for GC are listed in . Hand–foot skin reaction, hypertension, and proteinuria were the most frequent ARs of antiangiogenic drugs. However, all of the ARs could be managed by reducing the dose of apatinib.Citation18 As for other solid tumors, the ARs during treatment were analogous.Citation21,Citation54,Citation59 In comparison to ramucirumab, apatinib showed a high rate of hypertension and proteinuria, but the occurrence rate of grade 3–4 events was low.Citation18,Citation60 In view of the present reports, apatinib shows acceptable safety.
Table 4 The safety of apatinib for GC
According to another study, the patients enrolled with GC always had gastric surgery, so they appeared to have lower absorption of and exposure to apatinib. However, no formal report explained the specific reason for the lower plasma concentration. However, it is known that the pharmacokinetics of apatinib in GC differs from that in other solid tumors. As a result, the dose of apatinib should be adjusted to maximize the benefits for patients with different solid tumors.Citation36
Additionally, there are no reliable biomarkers to assess the response of apatinib in GC. A recent study analyzed the relevance between the early appearance of primary ARs and the clinical results in metastatic GC patients. The results revealed that patients who experienced ARs in the first 4 weeks achieved prolonged median OS and PFS.Citation61 Therefore, the appearance of hypertension, proteinuria, and hand–foot syndrome in the first period of therapy could also be a feasible biomarker for the effectiveness of treatment in metastatic GC patients.
Resistance of antiangiogenic drugs including apatinib
Antiangiogenic drugs have achieved positive effects in clinical trials; however, drug resistance (DR) remains a problem that researchers cannot avoid. DR mainly includes the following mechanisms.Citation62
First, infiltration of tumors by different stromal cells (including immature myeloid cells, endothelial progenitor cells, or fibroblasts) can mediate resistance to VEGF-targeted drugs.Citation63–Citation65 Second, angiogenesis is regulated by multiple pathways. Although the VEGF pathway is the most important pathway, the compensatory effects of other pathways (such as c-kit and c-src) will gradually emerge as the treatment progresses.Citation66,Citation67 Third, tumors invade surrounding host tissues and form blood vessel-like structures and connect to host blood vessels, resulting in treatment failure.Citation68,Citation69 Fourth, the VEGF signaling system is complex and consists of five related ligands and three receptors. Because agents only work on one target, the efficacy will gradually decrease as treatment continues.Citation5,Citation6,Citation70,Citation71 Currently, there are no reports about DR to apatinib, but we can improve the treatment according to the common characteristics of antiangiogenic drugs. First, we can perform preconditioning to determine whether the tumor responds to VEGF-targeted therapy. Second, we can try to combine multiple antiangiogenic drugs to comprehensively suppress tumor progression. Of course, specific DR mechanisms still need to be explored. How to solve DR is one of the experimental directions in the future.
Discussion
Since precision medicine has become the hotspot of cancer therapy, multifarious molecular targets turn into crucial objects of study and the discovery of a specific target can be a breakthrough in antitumor research. Detection of VEGF signaling promoted the comprehension of the mechanism of angiogenesis.Citation5,Citation70,Citation72 Today, the agents that target VEGFR are being increasingly developed and put into use. Compared to drugs that target VEGFR-1, the antiangiogenesis ability of apatinib, which targets VEGFR-2, is affirmative.Citation17,Citation25,Citation73 Apatinib has been experimentally used in many cancer types and has proven to be a promising treatment in preclinical and clinical trials. For the moment, it has been shown that the curative effect of apatinib is slightly better than that of other antiangiogenic drugs such as ramucirumab, bevacizumab, and sunitinib.Citation39 In addition, apatinib can reverse the MDR caused by EGFR-TKIs and ABC proteins,Citation34 and it has a positive safety profile. Therefore, apatinib can be a promising treatment for different advanced cancers. Currently, extensive Phase II studies regarding different tumors are being conducted, and they will provide more detailed and accurate supporting data on the clinical application of apatinib.
However, there are a few issues that still require clarification.
First, the specific pathological make-up of the participants and the previous treatment plan are inexplicit.Citation18 In fact, the effect of treatment will be influenced by these two factors. It is necessary to make it clear which type of pathological patient is more sensitive to treatment and whether the previous treatment plan has influence on subsequent medications. These detailed data are helpful to guide future patient selection, so they should be collected in subsequent trials.
Second, there are no clear biomarkers used to predict patient response to treatment and to determine prognosis. Every mature treatment program has corresponding biomarkers to evaluate the current status of patients. This is very important for the adjustment of individualized treatment plans. Therefore, researchers need to carefully analyze the changes in each indicator during treatment and identify the characteristic indicators. As mentioned earlier, early ARs may be a potential biomarker.Citation61 In addition, biomarkers for other antiangiogenic drugs have been proposed, such as circulating markers and single-nucleotide polymorphisms,Citation62 but whether those biomarkers specifically relate to apatinib needs requires further exploration.
Third, patients’ quality of life should be taken seriously. In current times, patients and their families have an urgent need to improve quality of life. Doctors can no longer use therapeutic effects as an evaluation indicator for treatment; they also need to be responsible for patient mental health, social skills, and quality of life. How to achieve the best therapeutic effect while minimizing adverse drug reactions is the goal of further research. In addition to hematologic concerns, cardiac toxicity is also a common serious AR of antiangiogenic drugs.Citation74,Citation75 Although the incidence of cardiac toxicity is low in apatinib treatment,Citation18 the mechanism in this area needs further elucidation in order to properly solve the problem.
Fourth, the cost-effectiveness for the patient needs to be considered. A study developed a Markov model to assess the incremental cost-effectiveness ratio of apatinib to placebo. The study found that apatinib was currently not a cost-effective option for GC patients in China in whom second-line chemotherapy had failed.Citation76 However, the efficacy of apatinib was not inferior to that of ramucirumab (another VEGFR-TKI approved by the US Food and Drug Administration for advanced GC), and apatinib was an oral agent, which would also reduce costs. Therefore, apatinib is still a good option due to its positive clinical value. In addition, a charitable assistance program, the China Pharmaceutical Innovation and Research Development Association (PhIRDA) is underway, which greatly reduces the financial burden on patients.Citation76 The improvement of incremental cost-effectiveness ratio can be achieved through value-based pricing or increasing the effect of the therapeutic regimen. Therefore, new assistance programs or medical insurance are expected. Furthermore, multidisciplinary treatment is a promising means in oncotherapy. More research is required to explore the optimal therapy, such as the combination of apatinib and other antitumor drugs or therapy methods. We look forward to finding an effective way to maximize the benefit to patients.
Conclusion
Apatinib is now acknowledged as a novel oral small-molecule TKI in China. Due to antiangiogenesis activity, it could be applied to a variety of solid tumors. Currently, several studies are in progress, and many of them suggest that apatinib has good efficacy and safety in clinical trials. The ARs are tolerated, compared with other target agents, and the majority of them can be managed. Therefore, apatinib is a viable choice for the treatment of solid tumors.
Acknowledgments
This work was supported by the Taishan Scholar foundation (No tshw201502061), the Qingdao People’s Livelihood Science and Technology Program (16-6-2-3-nsh), and the third batch of the Qingdao Entrepreneurial Innovation Leading Talent Program.
Disclosure
The authors report no conflicts of interest in this work.
References
- ChenWZhengRBaadePDCancer statistics in China, 2015CA Cancer J Clin201666211513226808342
- van HagenPHulshofMCvan LanschotJJPreoperative chemoradiotherapy for esophageal or junctional cancerN Engl J Med2012366222074208422646630
- Le TourneauCDelordJPGonçalvesAMolecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trialLancet Oncol201516131324133426342236
- GotwalsPCameronSCipollettaDProspects for combining targeted and conventional cancer therapy with immunotherapyNat Rev Cancer201717528630128338065
- CarmelietPJainRKAngiogenesis in cancer and other diseasesNature2000407680124925711001068
- RoudsariLCWestJLStudying the influence of angiogenesis in in vitro cancer model systemsAdv Drug Deliv Rev20169725025926571106
- FerraraNGerberHPLecouterJThe biology of VEGF and its receptorsNat Med20039666967612778165
- EllisLMHicklinDJVEGF-targeted therapy: mechanisms of anti-tumour activityNat Rev Cancer20088857959118596824
- ShibuyaMVascular endothelial growth factor and its receptor system: physiological functions in angiogenesis and pathological roles in various diseasesJ Biochem20131531131923172303
- GoelHLMercurioAMVEGF targets the tumour cellNat Rev Cancer2013131287188224263190
- EbosJMBocciGManSA naturally occurring soluble form of vascular endothelial growth factor receptor 2 detected in mouse and human plasmaMol Cancer Res20042631532615235107
- HolmesKRobertsOLThomasAMCrossMJVascular endothelial growth factor receptor-2: structure, function, intracellular signalling and therapeutic inhibitionCell Signal200719102003201217658244
- GengRLiJApatinib for the treatment of gastric cancerExpert Opin Pharmacother201516111712225420417
- BrowerVApatinib in treatment of refractory gastric cancerLancet Oncol2016174e137
- DingLLiQJYouKYJiangZMYaoHRThe Use of Apatinib in Treating Nonsmall-Cell Lung Cancer: Case Report and Review of LiteratureMedicine20169520e359827196461
- LiFLiaoZZhaoJEfficacy and safety of Apatinib in stage IV sarcomas: experience of a major sarcoma center in ChinaOncotarget2017838644716448028969086
- ScottAJMessersmithWAJimenoAApatinib: a promising oral antiangiogenic agent in the treatment of multiple solid tumorsDrugs Today201551422322926020064
- LiJQinSXuJRandomized, Double-Blind, Placebo-Controlled Phase III Trial of Apatinib in Patients With Chemotherapy-Refractory Advanced or Metastatic Adenocarcinoma of the Stomach or Gastroesophageal JunctionJ Clin Oncol201634131448145426884585
- HuXZhangJXuBMulticenter phase II study of apatinib, a novel VEGFR inhibitor in heavily pretreated patients with metastatic triple-negative breast cancerInt J Cancer201413581961196924604288
- SongZYuXLouGShiXZhangYSalvage treatment with apatinib for advanced non-small-cell lung cancerOnco Targets Ther2017101821182528367065
- LiJWangLEfficacy and safety of apatinib treatment for advanced esophageal squamous cell carcinomaOnco Targets Ther2017103965396928860804
- BergersGBrekkenRMcmahonGMatrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesisNat Cell Biol200021073774411025665
- PlateKHBreierGMillauerBUllrichARisauWUp-regulation of vascular endothelial growth factor and its cognate receptors in a rat glioma model of tumor angiogenesisCancer Res19935323582258277694795
- RovielloGRavelliAPolomKApatinib: A novel receptor tyrosine kinase inhibitor for the treatment of gastric cancerCancer Lett2016372218719126797419
- TianSQuanHXieCYN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivoCancer Sci201110271374138021443688
- LiXXuALiHZhangBCaoBHuangJNovel role of apatinib as a multi-target RTK inhibitor in the direct suppression of hepatocellular carcinoma cellsBiochim Biophys Acta201818645 Pt A1693170129486282
- LiuKRenTHuangYApatinib promotes autophagy and apoptosis through VEGFR2/STAT3/BCL-2 signaling in osteosarcomaCell Death Dis201788e301528837148
- LuWKeHQianshanDZhenWGuoanXHonggangYApatinib has anti-tumor effects and induces autophagy in colon cancer cellsIran J Basic Med Sci201720999099529085592
- PengHZhangQLiJApatinib inhibits VEGF signaling and promotes apoptosis in intrahepatic cholangiocarcinomaOncotarget2016713172201722926967384
- YinLWangJHuangFCInhibitory effect of apatinib on HCT-116 cells and its mechanismNan Fang Yi Ke Da Xue Xue Bao201737336737228377354
- ZhangHCaoYChenYLiGYuHApatinib promotes apoptosis of the SMMC-7721 hepatocellular carcinoma cell line via the PI3K/Akt pathwayOncol Lett20181545739574329552208
- OzbenTMechanisms and strategies to overcome multiple drug resistance in cancerFEBS Lett2006580122903290916497299
- Pérez-TomásRMultidrug resistance: retrospect and prospects in anticancer drug treatmentCurr Med Chem200613161859187616842198
- MiYJLiangYJHuangHBApatinib (YN968D1) reverses multidrug resistance by inhibiting the efflux function of multiple ATP-binding cassette transportersCancer Res201070207981799120876799
- DingJChenXGaoZMetabolism and pharmacokinetics of novel selective vascular endothelial growth factor receptor-2 inhibitor apatinib in humansDrug Metab Dispos20134161195121023509226
- YuMGaoZDaiXPopulation Pharmacokinetic and Covariate Analysis of Apatinib, an Oral Tyrosine Kinase Inhibitor, in Healthy Volunteers and Patients with Solid TumorsClin Pharmacokinet2017561657627379402
- LiuXZhangYChenQPharmacokinetic Drug Interactions of Apatinib With Rifampin and ItraconazoleJ Clin Pharmacol2017
- ZongLAbeMSetoYJiJThe challenge of screening for early gastric cancer in ChinaLancet201638810060260627894662
- AoyamaTYoshikawaTTargeted therapy: Apatinib – new third-line option for refractory gastric or GEJ cancerNat Rev Clin Oncol201613526827027071350
- LiJZhaoXChenLSafety and pharmacokinetics of novel selective vascular endothelial growth factor receptor-2 inhibitor YN968D1 in patients with advanced malignanciesBMC Cancer20101052920923544
- LiJQinSXuJApatinib for chemotherapy-refractory advanced metastatic gastric cancer: results from a randomized, placebo-controlled, parallel-arm, phase II trialJ Clin Oncol201331263219322523918952
- HuangLWeiYShenSTherapeutic effect of apatinib on overall survival is mediated by prolonged progression-free survival in advanced gastric cancer patientsOncotarget2017817293462935427793017
- ZhaoTTXuHXuHMHmXThe efficacy and safety of targeted therapy with or without chemotherapy in advanced gastric cancer treatment: a network meta-analysis of well-designed randomized controlled trialsGastric Cancer201821336137129455269
- BianchiniGBalkoJMMayerIASandersMEGianniLTriple-negative breast cancer: challenges and opportunities of a heterogeneous diseaseNat Rev Clin Oncol2016131167469027184417
- LinYWuZZhangJApatinib for metastatic breast cancer in non-clinical trial setting: Satisfying efficacy regardless of previous anti-angiogenic treatmentTumour Biol2017396 1010428317711033
- EbosJMLeeCRCruz-MunozWBjarnasonGAChristensenJGKerbelRSAccelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesisCancer Cell200915323223919249681
- ChungASKowanetzMWuXDifferential drug class-specific metastatic effects following treatment with a panel of angiogenesis inhibitorsJ Pathol2012227440441622611017
- WeltiJCPowlesTFooSContrasting effects of sunitinib within in vivo models of metastasisAngiogenesis201215462364122843200
- CookeVGLebleuVSKeskinDPericyte depletion results in hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by met signaling pathwayCancer Cell2012211668122264789
- RotowJBivonaTGUnderstanding and targeting resistance mechanisms in NSCLCNat Rev Cancer2017171163765829068003
- JankuFGarrido-LagunaIPetruzelkaLBStewartDJKurzrockRNovel therapeutic targets in non-small cell lung cancerJ Thorac Oncol2011691601161221849857
- ZengDXWangCGLeiWHuangJAJiangJHEfficiency of low dosage apatinib in post-first-line treatment of advanced lung adenocarcinomaOncotarget2017839662486625329029508
- FangSCZhangHTZhangYMXieWPApatinib as post second-line therapy in EGFR wild-type and ALK-negative advanced lung adenocarcinomaOnco Targets Ther20171044745228176910
- LiFZhuTCaoBWangJLiangLApatinib enhances antitumour activity of EGFR-TKIs in non-small cell lung cancer with EGFR-TKI resistanceEur J Cancer20178418419228822888
- PengYCuiHLiuZApatinib to combat EGFR-TKI resistance in an advanced non-small cell lung cancer patient with unknown EGFR status: a case reportOnco Targets Ther2017102289229528490886
- LagergrenJSmythECunninghamDLagergrenPOesophageal cancerLancet2017390101102383239628648400
- PengQXHanYWZhangYLApatinib inhibits VEGFR-2 and angiogenesis in an in vivo murine model of nasopharyngeal carcinomaOncotarget2017832528135282228881773
- PengHZhangQLiJJlLApatinib inhibits VEGF signaling and promotes apoptosis in intrahepatic cholangiocarcinomaOncotarget2016713172201722926967384
- HuXCaoJHuWMulticenter phase II study of apatinib in non-triple-negative metastatic breast cancerBMC Cancer20141482025376790
- FuchsCSTomasekJYongCJRamucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trialLancet20143839911313924094768
- LiuXQinSWangZEarly presence of anti-angiogenesis-related adverse events as a potential biomarker of antitumor efficacy in metastatic gastric cancer patients treated with apatinib: a cohort studyJ Hematol Oncol201710115328870253
- VasudevNSReynoldsARAnti-angiogenic therapy for cancer: current progress, unresolved questions and future directionsAngiogenesis201417347149424482243
- ShojaeiFWuXMalikAKTumor refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+ myeloid cellsNat Biotechnol200725891192017664940
- ChungASWuXZhuangGAn interleukin-17-mediated paracrine network promotes tumor resistance to anti-angiogenic therapyNat Med20131991114112323913124
- ShakedYCiarrocchiAFrancoMTherapy-Induced Acute Recruitment of Circulating Endothelial Progenitor Cells to TumorsScience200631357941785178716990548
- LieuCHeymachJOvermanMTranHKopetzSBeyond VEGF: inhibition of the fibroblast growth factor pathway and antiangiogenesisClin Cancer Res201117196130613921953501
- LiJLSainsonRCOonCEDLL4-Notch signaling mediates tumor resistance to anti-VEGF therapy in vivoCancer Res201171186073608321803743
- KilarskiWWSamolovBPeterssonLKvantaAGerwinsPBiomechanical regulation of blood vessel growth during tissue vascularizationNat Med200915665766419483693
- DuRLuKVPetritschCHIF1alpha induces the recruitment of bone marrow-derived vascular modulatory cells to regulate tumor angiogenesis and invasionCancer Cell200813320622018328425
- GoelHLMercurioAMVEGF targets the tumour cellNat Rev Cancer2013131287188224263190
- TuguesSKochSGualandiLLiXClaesson-WelshLVascular endothelial growth factors and receptors: anti-angiogenic therapy in the treatment of cancerMol Aspects Med20113228811121565214
- HicklinDJEllisLMRole of the vascular endothelial growth factor pathway in tumor growth and angiogenesisJ Clin Oncol20052351011102715585754
- ZhangHApatinib for molecular targeted therapy in tumorDrug Des Devel Ther2015960756081
- TelliMLWittelesRMFisherGASrinivasSCardiotoxicity associated with the cancer therapeutic agent sunitinib malateAnn Oncol20081991613161818436521
- ChenXLLeiYHLiuCFAngiogenesis inhibitor bevacizumab increases the risk of ischemic heart disease associated with chemotherapy: a meta-analysisPLoS One201386e6672123818962
- ChenHDZhouJWenFCost-effectiveness analysis of apatinib treatment for chemotherapy-refractory advanced gastric cancerJ Cancer Res Clin Oncol2017143236136827798730
- BangYJKangYKKangWKPhase II study of sunitinib as second-line treatment for advanced gastric cancerInvest New Drugs20112961449145820461441
- PavlakisNSjoquistKMMartinAJRegorafenib for the Treatment of Advanced Gastric Cancer (INTEGRATE): A Multinational Placebo-Controlled Phase II TrialJ Clin Oncol201634232728273527325864
- Martin-RichardMGallegoRPericayCMulticenter phase II study of oxaliplatin and sorafenib in advanced gastric adenocarcinoma after failure of cisplatin and fluoropyrimidine treatment. A GEMCAD studyInvest New Drugs20133161573157924077981
- OhtsuAShahMAvan CutsemEBevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III studyJ Clin Oncol201129303968397621844504
- WilkeHMuroKvan CutsemERamucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trialLancet Oncol201415111224123525240821
- YoonHHBendellJCBraitehFSRamucirumab combined with FOLFOX as front-line therapy for advanced esophageal, gastroesophageal junction, or gastric adenocarcinoma: a randomized, double-blind, multicenter Phase II trialAnn Oncol201627122196220327765757
