Abstract
Clinical observations have revealed a strong correlation between loss of bone density in HIV-infected individuals, particularly in conjunction with the antiretroviral drug tenofovir, a nucleotide analog that inhibits HIV reverse transcriptase. The most compelling correlations have been observed in clinical studies involving young children and adolescents. These observations strongly suggest that bone density is being affected during active bone growth and development, implicating a role for tenofovir in bone loss. Here we discuss the literature and potential mechanisms for how tenofovir-associated bone loss may arise, which likely involves perturbation of cellular DNA synthesis and gene expression. Elucidation of the mechanism(s) involved in tenofovir-mediated bone loss will help in developing adjuvant therapies to reduce tenofovir-associated bone density loss.
Introduction
Nucleoside reverse transcriptase inhibitors (NRTIs) represent the backbone of highly active antiretroviral therapy (HAART) for the clinical management of human immunodeficiency virus (HIV) infection. NRTIs target HIV reverse transcriptase and prevent the synthesis of viral DNA. NRTIs lack a 3′-hydroxyl group on the deoxyribose and following incorporation of the next incoming deoxynucleotide cannot form the new 5′-3′ phosphodiester bond required to extend the synthesis of the DNA chain. Thus, NRTIs act as chain terminators of HIV DNA synthesis. Due to the development of HIV drug resistance, there has been a continual need for the discovery and development of new drug targets and new anti-HIV drugs.
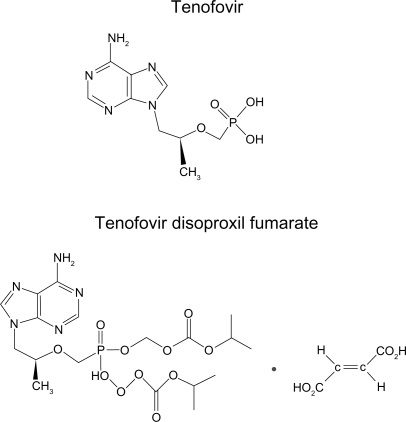
Tenofovir, also called 9-(R)-{2-(phosphonomethoxy)propyl}adenine or PMPA, is a nucleotide analog () and was approved in 2001 for use in treating HIV infection and more recently for chronic hepatitis B infection.Citation1 The prodrug form is tenofovir disoproxil fumarate (TDF) (). Tenofovir has improved potency as it is a nucleotide and has an abbreviated intracellular activation pathway to allow a more rapid and complete conversion to the active drug. The prodrug TDF has a labile lipophilic group to facilitate penetration through target cell membranes. Tenofovir is not known to be a substrate, inducer or inhibitor of human cytochrome P450 enzymes in vitro or in vivo. Tenofovir has high potency and an unusually durable response when used in trials of single-agent therapy intensification in highly treatment-experienced individuals.Citation2 The active metabolite, tenofovir diphosphate, exhibits a long intracellular half-life in both resting and activated peripheral blood mononuclear cells, which allows for single daily dosing. The clinical efficacy of TDF has been shown in Phase II (ie, GS-98-902) and III (ie, GS-99-907) clinical trials, with highly treatment-experienced patients.Citation3,Citation4 The Phase II and III studies were both randomized, double-blind, placebo-controlled, multicenter intensification studies of TDF that confirmed the utility of TDF in which patients added TDF therapy (300 mg once daily) in addition to their existing regimen.Citation5
While there has been great success in the use of tenofovir in treating HIV infection, there have been clinical reports of tenofovir-associated side effects. Among those side effects, multiple studies have implicated tenofovir-associated bone loss. This review discusses the literature on tenofovir-associated bone loss and presents several plausible mechanisms that might explain the basis for bone loss.
Tenofovir-associated bone loss
Bone is a dynamic tissue that is formed and maintained by osteoblasts (which form bone) and osteoclasts (which resorb bone). An extensive cell signaling network between osteoblasts and osteoclasts is required for maintaining a balance in the activities of these two cell types in bone remodeling and bone health. Recent studies have indicated that direct signals between bone cells are important for regulating bone remodeling.Citation6–Citation8 For example, it has been shown that osteoclasts can initiate bone formation via signaling to osteoblasts independent of their ability to resorb bone.Citation9,Citation10 In patients with autosomal dominant osteopetrosis type II it was demonstrated that the number of osteoclasts but not their activity controls bone formation.Citation11 Conversely, osteoblasts regulate osteoclast differentiation by expressing two factors that are necessary and sufficient for osteoclast formation: M-CSFCitation12 and RANKLCitation13; M-CSF is required for survival and proliferation of early osteoclast precursors.Citation14–Citation16 Binding of RANKL, produced by osteoblasts, and the RANK receptor on osteoclasts stimulates expression of genes necessary for osteoclast differentiation, cellular fusion and bone resorption. Additionally, osteoblasts express osteoprotegerin, a soluble decoy receptor for RANKL, which inhibits activation of RANK by RANKL. The ratio of RANKL to osteoprotegerin produced by osteoblasts is a major determinant of osteoclast forming activity within the bone microenvironment and allows for close coordination between bone formation and bone resorption under normal physiological conditions.
Perturbations in the function of osteoblasts or osteoclasts can lead to bone loss, clinically presented as osteopenia or osteoporosis. Osteoporosis commonly occurs in women following menopause, where it is called postmenopausal osteoporosis, but can also occur in men or anyone with certain hormonal disorders, other chronic diseases, or due to medications such as glucocorticoids.Citation17–Citation23 Vitamin D deficiencies can result in bone softening, resulting in osteomalacia – in children called rickets.Citation24–Citation26 Loss of osteoclast function increases bone density, resulting in osteopetrosis. One example of a human condition involving osteopetrosis is a rare inherited disorder, called Albers–Schonberg disease. In osteopetrosis, osteoclast numbers may be lower, normal, or elevated, however the critical factor in the development of osteopetrosis is the dysfunction of osteoclasts. For example, a reduction of carbonic anhydrase expression in osteoclasts can result in defects in hydrogen ion pumping, which can cause a dysfunction in osteoclast resorption of bone.Citation27,Citation28 However, osteoblast dysfunction, particularly hyperactivity, can also result in osteopetrosisCitation29,Citation30 while a decrease in function can contribute to osteoporosis.
HIV infection has been implicated as a risk factor for alteration of bone mineral density in children as well as in adults.Citation31–Citation34 Imbalances in growth factors and cytokines, as well as the use of HAART could contribute to bone loss by increasing bone resorption. Previous studies have demonstrated adverse effects on bone metabolism by the administration of tenofovir in macaques, resulting primarily in decreased bone density.Citation35–Citation37 Infection of macaques by simian immunodeficiency virus was also implicated in affecting bone density. Histomorphometric analysis revealed in one of these studies an increase in tibial osteoid seam width, which can result in bone softening (and can develop into osteomalacia).Citation35 The increase in osteoid seam width is likely associated with reduced activity in osteoblasts. Bone density reductions due to tenofovir have also been reported in human adults.Citation38 Since tenofovir has a distinct and unique drug resistance profile, it satisfies the significant need for new non-cross-resistant HAART regimens in salvage therapy, particularly with children and adolescents. In light of this, off-label clinical studies of tenofovir with HIV-infected children and adolescents have been conducted to investigate the efficacy of tenofovir-containing HAART regimens and to investigate whether tenofovir-associated bone loss is greater in children and adolescents than in adults.Citation39–Citation41 Increased bone resorption could cause a compensatory increase in osteoblast activity, which would be revealed by increased serum alkaline phosphatase levels.Citation42 General conclusions drawn from these studies include that tenofovir treatment decreased bone mineral density as well as increased urinary calcium excretion. One limitation of these studies was the relatively small study size (ranging from 18 to 6 children). Another limitation of these studies is that the measurement of bone density is difficult in children and adolescents because of the dynamic yet variable rate of bone growth. Presently, there are different methods that are commonly used to measure bone density: dual-energy X-ray absorbtiometry (DXA), quantitative computed tomography (QCT), and quantitative ultrasound (QUS). Each approach has limitations and comparisons between these methods are difficult as comparative data analysis can be somewhat subjective. A study that analyzed the effect of TDF treatment of a group of 16 HIV-infected children and adolescents over a 1-year period concluded that TDF did not affect bone density when stavudine and protease inhibitor-containing regimens were compared with TDF/lami-vudine/efavirenz.Citation43 Differences in this study compared to that of the previously described studies include that the patients were older, had greater height and weight, and the majority were either in middle or late puberty or postpubertal. The children under study were also required to have long-lasting viral suppression prior to the switch to TDF and were exposed to TDF levels that may have been lower, as fractions of pills were administered. This study design is different to the other studies, which involved relatively younger children that did not have long lasting viral suppression prior to the switch to TDF, which were entire pills (ie, 300 mg).Citation39–Citation41
Mechanisms of tenofovir-associated bone loss
A significant literature exists on NRTIs that have implicated mitochondrial dysfunction, reviewed in.Citation44–Citation49 The inhibition of mitochondrial polymerase gamma could occur by competitive inhibition of polymerase gamma and/or the incorporation and subsequent DNA chain termination. The result would be a reduction in mitochondrial DNA synthesis. NRTIs could also indirectly inhibit DNA synthesis by inducing nucleotide pool imbalances, oxidative stress, and DNA damage. Both direct and indirect mechanisms would result in the alteration of mitochondrial gene expression, which would cause mitochondrial dysfunction. NRTIs have greater affinity for affecting different tissue types as well as the nature of the mitochondrial-induced dysfunction in these tissue types, depending upon the NRTI. The analysis of NRTI-associated cellular stress has indicated that the mechanisms involved can be quite complex. NRTIs can result in altered gene expression profiles.Citation50,Citation51 Altered gene expression profiles have been observed in the absence of mitochondrial DNA depletion, suggesting that NRTIs can cause mitochondrial dysfunction and not inhibit mitochondrial DNA polymerase gamma.Citation52
The association of tenofovir with mitochondrial dysfunction has been investigated. In general, no mitochondrial dysfunction was observed with tenofovir.Citation53–Citation56 Other studies have reported a lowering of mitochondrial dysfunction when drug regimens were changed and NRTIs were replaced with tenofovir.Citation57–Citation59 A recent study in rats found that very high doses (ie, 100 mg/kg/day) of tenofovir reduced mitochondrial DNA and gene expression in kidney cells.Citation60 The relevance of this observation given the very high dosing used is unclear. In stark contrast, in an HIV transgenic mouse model tenofovir treatment was found to increase mitochondrial DNA content in kidney cells.Citation61 Further analysis of mitochondrial DNA obtained from renal proximal tubules microdissected from kidney sections suggested that tenofovir lowered mitochondrial DNA levels in the renal proximal tubules.Citation61 To date, no studies of mitochondrial dysfunction being associated with tenofovir-mediated bone loss have been reported. Overall, the current literature does not provide strong evidence for mitochondrial dysfunction being a major mechanism for tenfovir-mediated cellular stress, particularly in regards to bone loss.
In vitro studies have previously reported that tenofovir diphosphate is a poor substrate and weak inhibitor of rat DNA polymerase alpha, delta and epsilon.Citation55 Combined with the report of tenofovir diphosphate being poorly incorporated into DNA by the human mitochondrial DNA polymerase gamma,Citation49 data are limited that would support a direct role of tenofovir in bone loss by the inhibition of either nuclear or mitochondrial DNA replication. While data are lacking for tenofovir diphosphate being a good substrate for cellular DNA polymerases, the in vivo data correlating tenofovir therapy with bone loss, particularly in children and adolescents who have very active and ongoing bone growth, implicate a role for tenofovir’s ability to impact cellular DNA synthesis. Such an impact could be either direct or indirect, such as 1) incorporation and DNA chain termination, 2) DNA damage, 3) alteration of deoxynucleotide transport, and/or 4) nucleotide pool imbalances, which would perturb cellular DNA synthesis. The perturbation of cellular (ie, nuclear and/or mitochondrial) DNA synthesis would result in altered gene expression. The alteration of gene expression for genes involved in bone maintenance could explain the clinical observation of bone density loss during tenofovir treatment.
We propose three potential mechanisms for tenofovir-associated bone loss. These include 1) preferential uptake by osteoclasts (altering gene expression and resulting in increased bone resorption), 2) update by osteoblasts (altering gene expression and decreasing bone formation), and 3) uptake by both osteoclasts and osteoblasts (altering gene expression of both cells types and ultimately the balance between bone resorption and bone formation – resulting in bone loss).
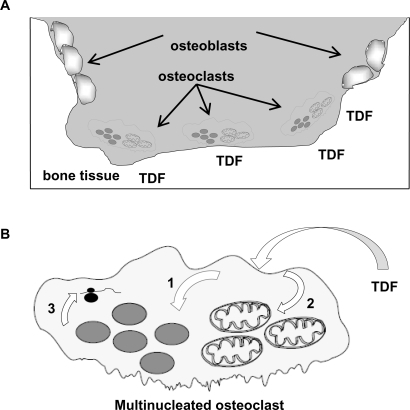
Since TDF is a phosphonate, it is possible that this could enhance its uptake into cells (), which would increase the probability of cellular stress. Bisphosphonates (ie, diphosphonates) are drugs used clinically to prevent loss of bone density, particularly in diseases such as osteoporosis, bone metastasis, multiple myeloma, Paget’s disease of bone, and primary hyperparathyroidism. Bisphosphonates target bone and inhibit osteoclast function after their cellular uptake by inducing apoptosis.Citation62,Citation63 Since tenofovir and TDF are both phosphonates, it is conceivable that they could also have an association with bone and be selectively taken up by osteoclasts by a mechanism similar to that of bisphosphonates, ultimately causing cellular stress. The resulting cellular stress would likely perturb cellular DNA synthesis (ie, nuclear and/or mitochondrial) and gene expression (). For example, the reduction of gene expression for an osteoclast gene that is involved in signaling osteoblast activity could ultimately result in a loss of bone density. It is formally possible that adjuvant treatment of bone density loss could improve the durability of tenofovir-containing HAART. In the case of osteoclast hyperactivity, it is possible that bisphosphonates could reduce bone density loss associated with tenofovir treatment.
Figure 2 The osteoclast as a target for TDF. A) Bone tissue, osteoblasts and osteoclasts. TDF, as a phosphonate, associates with bone tissue. Bone resorption by osteoclasts would result in the preferential uptake of TDF. B) Impact of TDF on osteoclast DNA synthesis and gene expression. Following TDF uptake by osteoclasts, TDF can target the nucleus (1) and/or mitochondria (2), where it may directly or indirectly perturb DNA synthesis by 1) incorporation and DNA chain termination, 2) DNA damage, 3) alteration of deoxynucleotide transport, and/or 4) nucleotide pool imbalances. The impact of TDF on cellular DNA synthesis would result in altered gene expression (3).
The loss of bone density due to TDF exposure could also be associated with tenofovir-induced renal dysfunction – particularly renal proximal tubule dysfunction.Citation42,Citation61,Citation64–Citation72 The failure of renal proximal tubular cells to reabsorb filtered bicarbonate from the urine would result in urinary bicarbonate wasting and subsequent acidemia and a more general dysfunction of the proximal tubular cells – called Fanconi’s syndrome. Commonly observed conditions in Fanconi syndrome include aminoaciduria, glycosuria, tubular proteinuria, and uricosuria. Importantly, the main clinical feature of Fanconi’s syndrome is bone demineralization (osteomalacia or rickets) due to phosphate wasting. Therefore, TDF-associated bone density loss may an outcome of renal dysfunction.
Summary
The distinctive resistance profile, high potency and limited side effects of tenofovir have made it an important component of HAART. The off-label use of tenofovir for treatment of children and adolescents is important for providing alternatives to treatment-experienced patients. Also, tenofovir will likely play an important role in preventing mother-to-child transmission, particularly in the context of the appeal of TDF-containing fixed dose combinations in a single pill (ie, Atripla® and Truvada®). A concern in off-label tenofovir use is bone density loss. Current data indicate that tenofovir-mediated bone loss is likely mediated by novel mechanisms not solely related to the known mitochondrial dysfunction associated with NRTIs. Future research will further aid in the elucidation the mechanism(s) involved in tenofovir-mediated bone loss, which would help in developing adjuvant therapies to reduce tenofovir-associated bone density loss.
Acknowledgements
We thank Steve Patterson for stimulating discussions. Supported by NIH grants AR53946 (K.C.M.), DE16093 (R.G.), and GM56615 (L.M.M.). I.F.G. and L.P. were supported by the MinnCResT Program, T32DE07288.
Disclosures
The authors declare no conflicts of interest.
References
- PozniakATenofovir: what have over 1 million years of patient experience taught us?Int J Clin Pract20086281285129318705824
- StephanCExperience with tenofovir disoproxil fumarate for antiretroviral therapyExpert Opin Pharmacother2008971197120918422476
- MargotNAIsaacsonEMcGowanIChengAKSchooleyRTMillerMDGenotypic and phenotypic analyses of HIV-1 in antiretroviral-experienced patients treated with tenofovir DFAIDS20021691227123512045487
- SchooleyRTRuanePMyersRATenofovir DF in antiretroviral-experienced patients: results from a 48-week, randomized, double-blind studyAIDS20021691257126312045491
- SquiresKPozniakALPieroneGJrTenofovir disoproxil fumarate in nucleoside-resistant HIV-1 infection: a randomized trialAnn Intern Med20031395 Pt 131332012965939
- KarsdalMAMartinTJBollerslevJChristiansenCHenriksenKAre nonresorbing osteoclasts sources of bone anabolic activity?J Bone Miner Res200722448749417227224
- KarsdalMAHenriksenKSorensenMGAcidification of the osteoclastic resorption compartment provides insight into the coupling of bone formation to bone resorptionAm J Pathol2005166246747615681830
- MartinTJSimsNAOsteoclast-derived activity in the coupling of bone formation to resorptionTrends Mol Med2005112768115694870
- BollerslevJMarksSCJrPockwinseSUltrastructural investigations of bone resorptive cells in two types of autosomal dominant osteopetrosisBone19931468658698155410
- BollerslevJSteinicheTMelsenFMosekildeLStructural and histomorphometric studies of iliac crest trabecular and cortical bone in autosomal dominant osteopetrosis: a study of two radiological typesBone198910119242660883
- AlataloSLIvaskaKKWaguespackSGEconsMJVaananenHKHalleenJMOsteoclast-derived serum tartrate-resistant acid phosphatase 5b in Albers-Schonberg disease (type II autosomal dominant osteopetrosis)Clin Chem200450588389015016726
- CecchiniMGHofstetterWHalasyJWetterwaldAFelixRRole of CSF-1 in bone and bone marrow developmentMol Reprod Dev19974617583 discussion 83–74.8981367
- WadaTNakashimaTHiroshiNPenningerJMRANKL-RANK signaling in osteoclastogenesis and bone diseaseTrends Mol Med2006121172516356770
- FelixRCecchiniMGHofstetterWElfordPRStutzerAFleischHImpairment of macrophage colony-stimulating factor production and lack of resident bone marrow macrophages in the osteopetrotic op/op mouseJ Bone Miner Res1990577817892204254
- Wiktor-JedrzejczakWBartocciAFerranteAWJrTotal absence of colony-stimulating factor 1 in the macrophage-deficient osteopetrotic (op/op) mouseProc Natl Acad Sci U S A19908712482848322191302
- YoshidaHHayashiSKunisadaTThe murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor geneNature199034562744424442188141
- BonnelyeEMerdadLKungVAubinJEThe orphan nuclear estrogen receptor-related receptor alpha (ERRalpha) is expressed throughout osteoblast differentiation and regulates bone formation in vitroJ Cell Biol2001153597198411381083
- LiuRHWerthVPWhat is new in the treatment of steroid-induced osteoporosis?Semin Cutan Med Surg200726420320918395668
- ShakerJLLukertBPOsteoporosis associated with excess glucocorticoidsEndocrinol Metab Clin North Am2005342341356viiiix15850846
- CaplanLSaagKGGlucocorticoids and the risk of osteoporosisExpert Opin Drug Saf200981334719236216
- EbelingPROsteoporosis in men. New insights into aetiology, pathogenesis, prevention and managementDrugs Aging19981364214349883398
- McIlwainHHGlucocorticoid-induced osteoporosis: pathogenesis, diagnosis, and managementPrev Med200336224324912591000
- PietschmannPRaunerMSiposWKerschan-SchindlKOsteoporosis: an age-related and gender-specific disease – a mini-reviewGerontology200855131218948685
- FrameBParfittAMOsteomalacia: current conceptsAnn Intern Med1978896966982363010
- MisraMPacaudDPetrykACollett-SolbergPFKappyMVitamin D deficiency in children and its management: review of current knowledge and recommendationsPediatrics2008122239841718676559
- UkincKSevere osteomalacia presenting with multiple vertebral fractures: a case report and review of the literatureEndocrine2009361303619543841
- Segovia-SilvestreTNeutzsky-WulffAVSorensenMGAdvances in osteoclast biology resulting from the study of osteopetrotic mutationsHum Genet2009124656157718987890
- SlyWSHewett-EmmettDWhyteMPYuYSTashianRECarbonic anhydrase II deficiency identified as the primary defect in the autosomal recessive syndrome of osteopetrosis with renal tubular acidosis and cerebral calcificationProc Natl Acad Sci U S A1983809275227566405388
- BoydenLMMaoJBelskyJHigh bone density due to a mutation in LDL-receptor-related protein 5N Engl J Med2002346201513152112015390
- Van WesenbeeckLCleirenEGramJSix novel missense mutations in the LDL receptor-related protein 5 (LRP5) gene in different conditions with an increased bone densityAm J Hum Genet200372376377112579474
- ArpadiSMHorlickMThorntonJCuffPAWangJKotlerDPBone mineral content is lower in prepubertal HIV-infected childrenJ Acquir Immune Defic Syndr200229545045411981360
- TebasPPowderlyWGClaxtonSAccelerated bone mineral loss in HIV-infected patients receiving potent antiretroviral therapyAIDS2000144F636710770534
- ViganoAMoraSAdverse effects of antiretroviral therapy: focus on bone densityExpert Opin Drug Saf20043319920815155148
- MadedduGSpanuASolinasPBone mass loss and vitamin D metabolism impairment in HIV patients receiving highly active antiretroviral therapyQ J Nucl Med Mol Imaging2004481394815195003
- CastilloABTarantalAFWatnikMRMartinRBTenofovir treatment at 30 mg/kg/day can inhibit cortical bone mineralization in growing rhesus monkeys (Macaca mulatta)J Orthop Res20022061185118912472227
- TarantalAFMarthasMLShawJPCundyKBischofbergerNAdministration of 9-[2-(R)-(phosphonomethoxy)propyl]adenine (PMPA) to gravid and infant rhesus macaques (Macaca mulatta): safety and efficacy studiesJ Acquir Immune Defic Syndr Hum Retrovirol199920432333310096575
- Van RompayKKBrignoloLLMeyerDJBiological effects of short-term or prolonged administration of 9-[2-(phosphonomethoxy) propyl]adenine (tenofovir) to newborn and infant rhesus macaquesAntimicrob Agents Chemother20044851469148715105094
- GallantJEStaszewskiSPozniakALEfficacy and safety of tenofovir DF vs stavudine in combination therapy in antiretroviral-naive patients: a 3-year randomized trialJAMA2004292219120115249568
- HazraRGafniRIMaldarelliFTenofovir disoproxil fumarate and an optimized background regimen of antiretroviral agents as salvage therapy for pediatric HIV infectionPediatrics20051166e846e85416291735
- GafniRIHazraRReynoldsJCTenofovir disoproxil fumarate and an optimized background regimen of antiretroviral agents as salvage therapy: impact on bone mineral density in HIV-infected childrenPediatrics20061183e711e71816923923
- PurdyJBGafniRIReynoldsJCZeichnerSHazraRDecreased bone mineral density with off-label use of tenofovir in children and adolescents infected with human immunodeficiency virusJ Pediatr2008152458258418346519
- FuxCARauchASimcockMTenofovir use is associated with an increase in serum alkaline phosphatase in the Swiss HIV Cohort StudyAntivir Ther20081381077108219195333
- GiacometVMoraSMartelliLMerloMSciannambloMViganoAA 12-month treatment with tenofovir does not impair bone mineral accrual in HIV-infected childrenJ Acquir Immune Defic Syndr200540444845016280700
- CoteHCMechanisms of antiretroviral therapy-induced mitochondrial dysfunctionCurr Opin HIV AIDS20072425326019372896
- LewisWCopelandWCDayBJMitochondrial dna depletion, oxidative stress, and mutation: mechanisms of dysfunction from nucleoside reverse transcriptase inhibitorsLab Invest200181677779011406640
- LewisWDayBJCopelandWCMitochondrial toxicity of NRTI antiviral drugs: an integrated cellular perspectiveNat Rev Drug Discov200321081282214526384
- LewisWKohlerJJHosseiniSHAntiretroviral nucleosides, deoxynucleotide carrier and mitochondrial DNA: evidence supporting the DNA pol gamma hypothesisAIDS200620567568416514297
- KohlerJJLewisWA brief overview of mechanisms of mitochondrial toxicity from NRTIsEnviron Mol Mutagen20074834166172
- LeeHHanesJJohnsonKAToxicity of nucleoside analogues used to treat AIDS and the selectivity of the mitochondrial DNA polymeraseBiochemistry20034250147111471914674745
- DesaiVGLeeTDelongchampRRNucleoside reverse transcriptase inhibitors (NRTIs)-induced expression profile of mitochondria-related genes in the mouse liverMitochondrion20088218119518313992
- DesaiVGLeeTMolandCLEffect of short-term exposure to zidovudine (AZT) on the expression of mitochondria-related genes in skeletal muscle of neonatal miceMitochondrion20099191618824140
- MallonPWUnemoriPSedwellRIn vivo, nucleoside reverse-transcriptase inhibitors alter expression of both mitochondrial and lipid metabolism genes in the absence of depletion of mitochondrial DNAJ Infect Dis2005191101686169615838796
- VidalFDomingoJCGuallarJIn vitro cytotoxicity and mitochondrial toxicity of tenofovir alone and in combination with other antiretrovirals in human renal proximal tubule cellsAntimicrob Agents Chemother200650113824383216940060
- CihlarTBirkusGGreenwaltDEHitchcockMJTenofovir exhibits low cytotoxicity in various human cell types: comparison with other nucleoside reverse transcriptase inhibitorsAntiviral Res2002541374511888656
- BirkusGHitchcockMJCihlarTAssessment of mitochondrial toxicity in human cells treated with tenofovir: comparison with other nucleoside reverse transcriptase inhibitorsAntimicrob Agents Chemother200246371672311850253
- BieseckerGKarimiSDesjardinsJEvaluation of mitochondrial DNA content and enzyme levels in tenofovir DF-treated rats, rhesus monkeys and woodchucksAntiviral Res200358321722512767469
- GerschensonMKimCBerzinsBMitochondrial function, morphology and metabolic parameters improve after switching from stavudine to a tenofovir-containing regimenJ Antimicrob Chemother20096361244125019321503
- RiberaEParadineiroJCCurranAImprovements in subcutaneous fat, lipid profile, and parameters of mitochondrial toxicity in patients with peripheral lipoatrophy when stavudine is switched to tenofovir (LIPOTEST study)HIV Clin Trials20089640741719203906
- MiroOGarrabouGLopezSShort communication metabolic and mitochondrial effects of switching antiretroviral-experienced patients to enfuvirtide, tenofovir and saquinavir/ritonavirAntivir Ther200611562563016964831
- LebrechtDVenhoffACKirschnerJWiechTVenhoffNWalkerUAMitochondrial tubulopathy in tenofovir disoproxil fumarate-treated ratsJ Acquir Immune Defic Syndr200951325826319582894
- KohlerJJHosseiniSHHoying-BrandtATenofovir renal toxicity targets mitochondria of renal proximal tubulesLab Invest200989551351919274046
- KwakHBKimJYKimKJRisedronate directly inhibits osteoclast differentiation and inflammatory bone lossBiol Pharm Bull20093271193119819571384
- MoreauMFGuilletCMassinPComparative effects of five bisphosphonates on apoptosis of macrophage cells in vitroBiochem Pharmacol200773571872317157266
- EarleKESeneviratneTShakerJShobackDFanconi’s syndrome in HIV+ adults: report of three cases and literature reviewJ Bone Miner Res200419571472115068493
- ParsonageMJWilkinsEGSnowdenNIssaBGSavageMWThe development of hypophosphataemic osteomalacia with myopathy in two patients with HIV infection receiving tenofovir therapyHIV Med20056534134616156882
- WilliamsJChadwickDRTenofovir-induced renal tubular dysfunction presenting with hypocalcaemiaJ Infect2006524e107e10816125779
- NelsonMRKatlamaCMontanerJSThe safety of tenofovir disoproxil fumarate for the treatment of HIV infection in adults: the first 4 yearsAIDS200721101273128117545703
- LabargaPBarreiroPMartin-CarboneroLKidney tubular abnormalities in the absence of impaired glomerular function in HIV patients treated with tenofovirAIDS200923668969619262355
- WoodwardCLHallAMWilliamsIGTenofovir-associated renal and bone toxicityHIV Med200910848248719459988
- PerrotSAslangulESzwebelTCaillat-VigneronNLe JeunneCBone pain due to fractures revealing osteomalacia related to tenofovir-induced proximal renal tubular dysfunction in a human immunodeficiency virus-infected patientJ Clin Rheumatol2009152727419265350
- CalmyAFuxCANorrisRLow bone mineral density, renal dysfunction, and fracture risk in HIV infection: a cross-sectional studyJ Infect Dis2009200111746175419874178
- Di BiagioARossoRMontefortePRussoRRovettaGViscoliCWhole body bone scintigraphy in tenofovir-related osteomalacia: a case reportJ Med Case Reports20093813619830218