Abstract
Dipeptidyl peptidase-4 (DPP-4) inhibitors, a new class of oral hypoglycemic agents, augment glucose-dependent insulin secretion and suppress glucagon levels through enhancement of the action of endogenous incretin by inhibiting DPP-4, an incretin-degrading enzyme. DPP-4 inhibitors are generally well tolerated because of their low risk of hypoglycemia and other adverse events. Moreover, with their potential to improve beta cell function, a core defect of type 2 diabetes, DPP-4 inhibitors are becoming a major component of treatment of type 2 diabetes. Alogliptin benzoate is a newly developed, highly selective DPP-4 inhibitor which has been approved in many countries throughout the world. Once-daily administration of alogliptin as either monotherapy or combination therapy with other oral antidiabetic drugs or insulin has a potent glucose-lowering effect which is similar to that of other DPP-4 inhibitors, with a low risk of hypoglycemia and weight gain. The cardiovascular safety of this drug has been confirmed in a recent randomized controlled trial. This review summarizes the efficacy and safety of alogliptin, and discusses the role of DPP-4 inhibitors in the treatment of type 2 diabetes.
Introduction
Dipeptidyl peptidase-4 (DPP-4) inhibitors, a new class of oral hypoglycemic agents, augment glucose-dependent insulin secretion through enhancement of the action of endogenous incretin (ie, glucagon-like peptide-1 and glucose-dependent insulinotropic polypeptide) by inhibiting DPP-4, a degrading enzyme of incretin. DPP-4 inhibitors also suppress glucagon secretion via elevation of glucagon-like peptide-1. They are generally well tolerated because of their low risk of hypoglycemia and other adverse events.Citation1
Alogliptin benzoate is a newly developed DPP-4 inhibitor developed by Takeda Inc (Osaka, Japan) that was approved for clinical use in the treatment of type 2 diabetes (T2DM) in Japan in April 2010 with the trade name Nesina®. Alogliptin was also approved by the US Food and Drug Administration and the European Medicines Agency in January 2013 and September 2013, respectively (with the trade name Vipidia® in Europe) and, as of September 2014, is available in 39 countries. In Japan, sitagliptin was the first approved DPP-4 inhibitor in December 2009, and first approved by the US Food and Drug Administration in 2006. Currently, seven DPP-4 inhibitors, ie, sitagliptin, vildagliptin, alogliptin, linagliptin, teneligliptin, anagliptin, and saxagliptin, have been approved in Japan.
Alogliptin was developed by structure-based design and proved to have high selectivity for DPP-4.Citation2 The efficacy and safety of alogliptin has been confirmed to be similar to that of other DPP-4 inhibitors in recent clinical trials.Citation3 This updated review of a previous publicationCitation4 focuses on the clinical efficacy and safety of alogliptin for the treatment of T2DM. The pharmacokinetics and pharmacodynamics of alogliptin have also been reviewed elsewhere.Citation5,Citation6
Methods
This review is based upon a MEDLINE search and personal experience with alogliptin. A MEDLINE search (1966 to December 2014) for published clinical trials and pertinent review articles published in English was conducted with the keywords “alogliptin”, “incretin”, and “DPP-4 inhibitor”. References of identified articles were searched for additional relevant sources. Relevant articles were also obtained from the author’s personal list of references.
Chemistry
The chemical structure of alogliptin is shown in . Alogliptin shows high selectivity for DPP-4 (>10,000-fold over other DPP isozymes such as DPP-2, DPP-8, and DPP-9.Citation2,Citation7 High selectivity of alogliptin for DPP-4 has also been confirmed in a DPP-8/9-expressing cell model.Citation8 A recent study using X-ray crystallography revealed that alogliptin interacts with the S1 and/or S2 subsites in addition to the S1 and S2 subsites of DPP-4, where vildagliptin interacts, suggesting that alogliptin offers more potent DPP-4 inhibition compared with vildagliptin.Citation9
Pharmacokinetics
Alogliptin is rapidly absorbed after oral administration, with a median time taken to reach peak plasma concentration of 2 hours and a mean half-life of 12–21 hours across various doses (6.25–800 mg). Food does not affect the absorption of alogliptin.Citation10–Citation12
To date, no drug interaction with alogliptin has been reported, although among the DPP-4 inhibitors, the level of saxagliptin may be affected by inhibitors or inducers of the cytochrome P450 3A4/5 isozyme (eg, ketoconazole, diltiazem, and rifampicin).
The pharmacokinetics of alogliptin have also been evaluated in subjects with renal and hepatic impairment. Following administration of a single 50 mg dose of alogliptin, 1.7-fold, 2.1-fold, 3.2-fold, and 3.8-fold increases in alogliptin exposure in subjects with mild, moderate, severe renal insufficiency, and end-stage renal disease, respectively, were observed compared with levels in healthy volunteers.Citation6,Citation13 Thus, dose adjustment of alogliptin, as well as sitagliptin and saxagliptin, is recommended for patients with renal dysfunction ().
Table 1 Dosage modification of alogliptin for patients with renal impairment in Japan
A single 25 mg dose of alogliptin was administered to eight patients with moderate hepatic impairment based on the Child-Pugh Scale, and there was no significant increase in serum concentration of alogliptin when compared with that in healthy volunteers. Thus, dose adjustment of alogliptin is not needed in patients with hepatic impairment.Citation6,Citation13
Pharmacodynamics
Mean plasma DPP-4 inhibition after a single dose of alogliptin (25–800 mg) ranged from 74% to 94% at 24 hours and 48% to 83% at 72 hours. After 14 days of dosing (25–400 mg/day), mean peak DPP-4 inhibition ranged from 94% to 99%, with mean inhibition at 24 hours after dosing of 82%–97%, supporting once-daily administration.Citation10,Citation11 A recent qualitative model of DPP-4 inhibition showed that with once-daily administration of alogliptin 25 mg, the usual recommended dosage of alogliptin, the percentage of time when DPP-4 inhibition was.80% was 88%, the percentage of DPP-4 inhibition at trough was 80%, and the percentage of weighted average DPP-4 inhibition over 24 hours was 88%, which is comparable with the results with once-daily administration of sitagliptin 100 mg.Citation14
Clinical efficacy
Glucose-lowering effect
The glucose-lowering effect of alogliptin has been evaluated in double-blind, placebo-controlled Phase II and III trials and also in recent Phase IV trials. The results of major clinical trials assessing the clinical efficacy of alogliptin compared with placebo or active comparators are summarized in .
Table 2 Selected efficacy outcomes from clinical trials of alogliptin
Monotherapy
To evaluate the efficacy of alogliptin as monotherapy, 329 drug-naïve patients with poorly controlled T2DM (mean HbA1c 7.9%) were randomized to once-daily treatment with alogliptin 12.5 mg, alogliptin 25 mg, or placebo for 26 weeks.Citation15 At week 26, the reduction in HbA1c from baseline was −0.56% and −0.59% in the alogliptin 12.5 mg and 25 mg groups, respectively, compared with 0.02% in the placebo group (P<0.001). Another dose-ranging study examined the efficacy of alogliptin (6.25–50 mg) in 480 drug-naïve Japanese patients with T2DM.Citation16 HbA1c at week 12 was dose-dependently reduced by alogliptin, and the reduction in HbA1c from baseline was −0.68% and −0.77% in the alogliptin 12.5 mg and 25 mg groups, respectively, compared with 0.05% in the placebo group (P<0.0001).
Combination with other antidiabetic medications
Sulfonylureas
The efficacy of alogliptin in combination with sulfonylureas has also been evaluated.Citation17 In one study, 500 patients with T2DM inadequately controlled by sulfonylurea monotherapy (mean HbA1c 8.1%) were randomized to treatment with glyburide plus alogliptin 12.5 mg or 25 mg or placebo for 26 weeks. At week 26, the reduction in HbA1c was −0.38% and −0.52% with alogliptin 12.5 mg and 25 mg, respectively, compared with 0.01% for placebo (P<0.001). In another study, 312 Japanese patients with T2DM inadequately controlled by sulfonylurea monotherapy (mean HbA1c 8.5%) were randomized to alogliptin 12.5 mg or 25 mg or placebo in addition to glimepiride 1–4 mg for 12 weeks.Citation18 The HbA1c reduction at week 12 was −0.59% and −0.65% with alogliptin 12.5 mg and 25 mg, respectively, compared with 0.35% for placebo (P<0.0001).
Metformin
The efficacy of alogliptin in combination with metformin was evaluated in a study that included 527 patients with T2DM inadequately controlled on metformin monotherapy (mean HbA1c 8%).Citation19 The patients were randomized to add-on treatment with alogliptin 12.5 mg or 25 mg or placebo for 26 weeks. At week 26, the reduction in HbA1c with either dose of alogliptin was −0.6% (P<0.001 versus placebo). In another Japanese study, a total of 288 patients with T2DM inadequately controlled with metformin 500 or 750 mg/day (mean HbA1c 8.0%) were randomized to receive alogliptin 12.5 mg or 25 mg or placebo in addition to metformin for 12 weeks.Citation20 At week 12, the reduction in HbA1c was −0.55% and −0.64% with alogliptin 12.5 mg and 25 mg, respectively, compared with 0.22% for placebo (P<0.0001).
Two more studies have been published recently. One trial was designed to evaluate the efficacy of initial therapy with alogliptin plus metformin.Citation21 A total of 784 drug-naïve patients (mean HbA1c 8.45%) were randomized to receive placebo, alogliptin 12.5 mg twice daily or 25 mg once daily, metformin 500 mg or 1,000 mg twice daily, or a combination of alogliptin 12.5 mg plus metformin 500 mg or 1,000 mg twice daily. The reduction in HbA1c at week 26 was −1.22% and −1.55% with the combination of alogliptin 12.5 mg plus metformin 500 mg or 1,000 mg twice daily, respectively, compared with −0.52% and −1.11% with alogliptin 25 mg once daily and metformin 1,000 mg twice daily, respectively (P<0.001). The number of patients who achieved HbA1c <7% was greater for combination therapy compared with monotherapy (47.1%–59.5% versus 20.2%–34.3%). The second study was designed to compare the long-term efficacy and safety of alogliptin plus metformin with that of glipizide plus metformin.Citation22 A total of 2,639 patients with a mean age 55.4 years, a mean duration of diabetes of 5.5 years, and a mean HbA1c of 7.6% were randomized to receive alogliptin 12.5 mg or 25 mg once daily or glipizide 5–20 mg once daily in addition to metformin for 104 weeks. The HbA1c reduction at week 104 was significantly greater with alogliptin 25 mg than with glipizide (−0.72% versus −0.59%, P=0.010), with less hypoglycemia (1.4% versus 23.2%) and weight gain (−0.89 versus 0.95 kg, P<0.001).
Thiazolidinediones
The efficacy of alogliptin in combination with a thiazolidinedione (TZD) was also evaluated. A total of 493 patients with T2DM inadequately controlled on a TZD (mean HbA1c 8.0%) were randomized to treatment with pioglitazone plus alogliptin 12.5 mg, alogliptin 25 mg, or placebo for 26 weeks.Citation23 At week 26, the reduction in HbA1c was −0.66% with alogliptin 12.5 mg and −0.80% with alogliptin 25 mg, compared with −0.19% for placebo (P<0.001). In another Japanese study,Citation24 339 patients with T2DM inadequately controlled on pioglitazone (mean HbA1c 7.9%) were randomized to add-on treatment with alogliptin 12.5 mg, alogliptin 25 mg, or placebo for 12 weeks. At week 12, the reduction in HbA1c was −0.91% with alogliptin 12.5 mg and −0.97% with alogliptin 25 mg compared with −0.19% for placebo (P<0.001).
Initial combination therapy of alogliptin and pioglitazone in drug-naïve patients with T2DM was also evaluated. A total of 655 drug-naïve patients with inadequately controlled diabetes (mean HbA1c 8.8%) were randomized into four treatment arms: alogliptin 25 mg monotherapy, pioglitazone 30 mg monotherapy, or alogliptin 12.5 mg or 25 mg plus pioglitazone 30 mg combination therapy for 26 weeks.Citation25 At week 26, combination therapy with alogliptin 25 mg plus pioglitazone 30 mg resulted in a greater reduction of HbA1c (–1.7%) versus alogliptin 25 mg monotherapy (–1.0%) or pioglitazone 30 mg monotherapy (–1.2%; P<0.001).
Combination therapy with alogliptin and pioglitazone in addition to metformin monotherapy was evaluated in another study.Citation26 A total of 1,554 patients with T2DM inadequately controlled on metformin monotherapy (mean HbA1c 8.5%) were randomized to add-on treatment with alogliptin (12.5 mg or 25 mg) alone or combined with pioglitazone (15, 30, or 45 mg) for 26 weeks. At week 26, the reduction in HbA1c was −0.9% in the pioglitazone alone group and −1.4% in the alogliptin 12.5 mg and alogliptin 25 mg plus pioglitazone groups (P<0.001).
The efficacy of a combination of alogliptin and pioglitazone was also evaluated in another study.Citation27 Seventy-one patients with well-controlled T2DM (mean HbA1c 6.7%) treated with metformin, sulfonylurea, or glinide monotherapy were randomized to receive alogliptin 25 mg plus pioglitazone 30 mg, alogliptin 25 mg monotherapy, or placebo for 16 weeks. The reduction in HbA1c at week 16 was −0.9%, −0.4%, and 0.4%, respectively (P<0.001 versus placebo).
Insulin
The efficacy of alogliptin when added to insulin therapy in patients with T2DM was assessed in two trials. In one study, 390 patients with T2DM inadequately controlled with insulin alone or combined with metformin (mean HbA1c 9.3%) were randomized to receive add-on therapy with alogliptin 12.5 mg, alogliptin 25 mg, or placebo for 26 weeks.Citation28 At week 26, the reduction in HbA1c was −0.63% with alogliptin 12.5 mg and −0.71% with alogliptin 25 mg, compared with −0.13% for placebo (P<0.001). In another Japanese study, 179 patients with T2DM inadequately controlled with insulin monotherapy were randomized to receive alogliptin 25 mg or placebo in addition to insulin therapy for 12 weeks.Citation29 The HbA1c reduction at week 12 was−0.96% for alogliptin compared with −0.29% for placebo (P<0.0001). The proportion of patients who achieved HbA1c <7% was greater with alogliptin than with placebo (23.3% versus 5.7%).
Lastly, the large-scale, randomized, controlled EXAMINE (Examination of Cardiovascular Outcomes with Alogliptin versus Standard of Care) study was conducted to assess the cardiovascular safety of alogliptin.Citation30 In this study, treatment with alogliptin on top of other antidiabetic medications showed a significant reduction in HbA1c (−0.36%, P<0.001) compared with placebo during a median follow-up of 18 months.
Recently, Berhan et al conducted a meta-analysis of ten randomized controlled trials to assess the efficacy of alogliptin in T2DM.Citation3 The pooled standardized mean difference for the reduction in HbA1c in patients treated with alogliptin 12.5 mg was −0.81% (95% confidence interval [CI] −1.11, −0.51) and that in patients treated with alogliptin 25 mg was −0.98% (95% CI −1.30, −0.66). The standardized mean difference of the reduction in fasting plasma glucose in patients treated with alogliptin 25 mg was −0.51 mmol/L (95% CI −0.68, −0.34).
Although there has been no head-to-head trial comparing the efficacy of alogliptin with that of other DPP-4 inhibitors, the improvement of HbA1c seen with alogliptin seems to be in the same range as that seen with other DPP-4 inhibitors,Citation31,Citation32 with the exception of vildagliptin 50 mg twice daily, which was reported to have a small advantage in glucose-lowering effect compared with other DPP-4 inhibitors.Citation33–Citation35
Predictors of HbA1c reduction by DPP-4 inhibitors
Recent meta-analyses of randomized controlled trials have shown that the efficacy of DPP-4 inhibitors is mainly related to baseline HbA1c level, as is seen with other antidiabetic medications.Citation36,Citation37 Esposito et al examined the clinical characteristics that predict the HbA1c response to DPP-4 inhibitors in their meta-regression analysis of 78 randomized controlled trials with 20,053 patients, and found a greater absolute reduction of baseline HbA1c in patients with higher baseline HbA1c and lower fasting glucose levels.Citation37
Recent meta-analyses have also suggested the efficacy of incretin therapy differs between ethnic groups. It has been shown that the efficacy of HbA1c reduction by DPP-4 inhibitors or glucagon-like peptide-1 receptor agonists is greater in Asian populations than in non-Asian populations,Citation38,Citation39 suggesting that lower body mass index is associated with a better HbA1c response to incretin therapy.
The long-term glycemic durability of DPP-4 inhibitors in patients with T2DM has also been reported. A meta-analysis of 12 trials with 14,829 patients showed that the effect of DPP-4 inhibitors on HbA1c declined significantly by ~0.2% during the second year of treatment,Citation40 while a greater HbA1c reduction at 2 years was reported for alogliptin compared with glipizide in the above-mentioned trial.Citation22
Efficacy in the elderly
The efficacy of alogliptin in the elderly was evaluated in a randomized, double-blind, active-controlled study.Citation41 In this study, 441 elderly type 2 diabetic patients of mean age 70 (range 65–90) years and with mild hyperglycemia on diet/exercise therapy alone or combined with oral antidiabetic monotherapy (mean HbA1c 7.5%) were randomized to alogliptin 25 mg or glipizide 5–10 mg for 52 weeks. The reduction in HbA1c at the end of study was −0.42% with alogliptin and −0.33% with glipizide, demonstrating no inferiority of alogliptin to glipizide. A post hoc analysis using this cohort showed that the proportion of patients achieving HbA1c ≤7.0% without hypoglycemia or weight gain was significantly higher with alogliptin than with glipizide (24% versus 13%, P<0.03).Citation42 It has also been shown that patients with baseline HbA1c <8.0% receiving alogliptin are also more likely to achieve HbA1c ≤7.0% without hypoglycemia or weight gain than those receiving glipizide (29% versus 13%, P<0.03).Citation42
A pooled analysis of Phase II and III studies to assess the efficacy of alogliptin in elderly patients with T2DM has also been reported.Citation43 Analysis of six randomized controlled trials of alogliptin showed a similar improvement in HbA1c in elderly patients (mean age 70 years) compared with younger patients (mean age 52 years).
Efficacy in hemodialysis patients
Since alogliptin is excreted by the kidney, the dose of alogliptin should be reduced in patients with moderate to severe renal impairment, as shown in . In a single-arm study, treatment with alogliptin 6.25 mg reduced HbA1c and glycated albumin levels from 7.1%±0.2% to 5.8%±1.6% and from 22.5%±0.7% to 19.6%±0.6%, respectively, after 24 months in 16 hemodialysis patients with T2DM.Citation44
Effect on beta cell function
Alogliptin has been shown to improve beta cell function and increase beta cell mass in rodents.Citation45–Citation48 In humans, it was reported that there was no or only modest improvement of beta cell function assessed by proinsulin to insulin ratio or homeostasis model assessment (HOMA)-β by alogliptin monotherapy or combination therapy with metformin or a sulfonylurea.Citation15,Citation17,Citation19 On the other hand, significant improve ment of beta cell function was reported when patients were treated with alogliptin combined with pioglitazone.Citation24,Citation26,Citation49 It has been reported that treatment with a combination of alogliptin 25 mg and pioglitazone 30 mg for 16 weeks significantly improved beta cell glucose sensitivity and fasting secretory tone compared with placebo, while alogliptin 25 mg monotherapy did not change beta cell function parameters.Citation27 A recent study has shown that treatment with alogliptin improved the function and proliferation of adult human beta cells engrafted in diabetic immunodeficient mice;Citation50 however, the effect of DPP-4 inhibitors on beta cell mass in humans remains unclear.Citation51
Effect on lipids
Eliasson et al reported a change in postprandial lipids during treatment with alogliptin.Citation52 Seventy-one patients with inadequately controlled T2DM were randomized to treatment with alogliptin 25 mg, alogliptin 25 mg plus pioglitazone 30 mg, or placebo for 16 weeks. At 16 weeks, a significant reduction in postprandial triacylglycerol and triacylglycerol-rich lipoproteins was observed with alogliptin monotherapy and alogliptin plus pioglitazone combination therapy compared with placebo. A meta-analysis of 17 studiesCitation53 also showed a significant reduction in total cholesterol (−0.18 mmol/L [−7 mg/dL]) and triacylglycerol (−0.15 mmol/L [−13 mg/dL]) on treatment with a DPP-4 inhibitor, although the reduction was small. A separate analysis of the effect of individual DPP-4 inhibitors showed a significant reduction in total cholesterol with alogliptin and vildagliptin, but not with sitagliptin.Citation53 However, in the EXAMINE study, no significant difference in lipoprotein levels was found between the alogliptin and placebo groups.Citation30
Effect on atherosclerosis and cardiovascular events
In animal studies, it was reported that alogliptin suppressed the progression of atherosclerosis in diabetic mice through downregulation of proinflammatory cytokines.Citation54,Citation55 It was also reported that alogliptin reduced the volume of the infarct lesion and neurological deficit after the induction of temporary focal ischemia in nondiabetic male C57BL/6J mice,Citation56 suggesting that the neuroprotective effect of alogliptin is independent of its glucose-lowering effect.
The effect of DPP-4 inhibitors on atherosclerosis in humans is unclear. In a preliminary study of ten healthy volunteers, treatment with alogliptin 25 mg for 1 week significantly suppressed the postprandial elevation in serum triacylglycerol and improved postprandial endothelial dysfunction, as assessed by brachial artery flow-mediated dilation.Citation57
Effect on kidney function
Although a protective effect of DPP-4 inhibitors against renal injury has been reported in rodent studies,Citation58–Citation61 the presence of such an effect remains to be established in humans. It has been reported that switching from sitagliptin to alogliptin, combined with an angiotensin II type 1 receptor blocker, resulted in a significant reduction in the urinary albumin to creatinine ratio in 12 patients with T2DM and albuminuria.Citation62 This improvement in albuminuria was accompanied by an elevated plasma level of stromal cell-derived factor-1 alpha and a decrease in urinary 8-hydroxy-2′-deoxyguanosine, an oxidative stress marker, suggesting that alogliptin ameliorated albuminuria by reducing oxidative stress. Sakata et al reported that treatment with alogliptin for 12 weeks significantly reduced the urinary albumin to creatinine ratio, as well as HbA1c and the circulating soluble form of the receptor for advanced glycation end products, in 61 patients with T2DM.Citation63
In the EXAMINE trial, no difference in change in estimated glomerular filtration rate was reported between alogliptin and placebo.Citation30 On the other hand, fewer patients showed worsened urinary albumin to creatinine ratio with saxagliptin versus placebo in the SAVOR (Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus)-TIMI (Thrombolysis in Myocardial Infarction) 53 trial,Citation64 although it remains unclear whether this effect of a DPP-4 inhibitor is independent of its glucose-lowering effect.
The effect of DPP-4 inhibitors on other types of microangiopathy such as retinopathy and neuropathy in humans remains largely unknown.Citation65
Safety
In general, the incidence of adverse events with DPP-4 inhibitors is reported to be low, and these agents are well tolerated.
Hypoglycemia
The risk of hypoglycemia is low on treatment with alogliptin. The incidence of hypoglycemia with alogliptin was reported to be as low as that with placebo or metformin monotherapy and lower than that with sulfonylureas.Citation15,Citation17,Citation19,Citation41 Adding alogliptin to insulin therapy resulted in improvement of HbA1c, without increasing hypoglycemia compared with insulin therapy alone.Citation28,Citation29 However, use of a DPP-4 inhibitor as combination therapy with a sulfonylurea can increase the risk of hypoglycemia.Citation1 Therefore, caution is needed when a DPP-4 inhibitor is added to a sulfonylurea, especially in patients at risk of hypoglycemia, such as the elderly, patients with renal impairment, patients treated with a high dose of sulfonylureas, and patients with autonomic neuropathy.
Effect on body weight
The effect of DPP-4 inhibitors on body weight is generally reported to be neutral.Citation1 Treatment with alogliptin has shown less weight gain compared with sulfonylureas.Citation22,Citation41 It was reported that alogliptin neither prevented nor exacerbated weight gain when it was combined with pioglitazone.Citation23–Citation26,Citation49
Cardiovascular safety
Recently, regulatory agencies have created cardiovascular safety guidelines for the development of therapies for diabetes. A pooled analysis of alogliptin trials including 4,168 patients showed no sign of increased cardiovascular risk with alogliptin in patients with T2DM (hazard ratio, 0.635, 95% CI 0.0–1.41).Citation66
The results of the EXAMINE study assessing the long-term cardiovascular safety of alogliptin were published in September 2013.Citation30 In this study, 5,380 patients with T2DM and recent acute coronary syndrome were randomized to receive alogliptin or placebo in addition to existing antihyperglycemic therapy. During a median follow-up period of 1.8 years, the rate of major adverse cardiovascular events, a composite of death from cardiovascular causes, nonfatal myocardial infarction, and nonfatal stroke, was not increased with alogliptin compared with placebo (11.3% versus 11.8%, hazard ratio 0.96; upper boundary of one-sided repeated CI, 1.16; P<0.001 for noninferiority).
Another cardiovascular outcome trial of a DPP-4 inhibitor was published at the same time. The SAVOR-TIMI 53 trialCitation64 was designed to evaluate the safety and efficacy of saxagliptin with respect to cardiovascular outcomes in patients with T2DM and at risk of a cardiovascular event. A total of 16,492 patients were randomized to receive saxagliptin or placebo and followed for a median of 2.1 years. There was no significant difference in incidence of a primary endpoint event, a composite of cardiovascular death, myocardial infarction, and ischemic stroke, between the groups (hazard ratio 1.00, 95% CI 0.89–1.12; P=0.99 for superiority; P<0.001 for noninferiority).
The results of these two randomized controlled trials confirmed the cardiovascular safety of DPP-4 inhibitors. However, in the SAVOR-TIMI 53 trial, more patients in the saxagliptin group than in the placebo group were hospitalized for heart failure (3.5% versus 2.8%; hazard ratio 1.27, 95% CI 1.07–1.51, P=0.007).Citation64 Meta-analyses of trials using different DPP-4 inhibitors including alogliptin also suggested an increased risk of heart failure with DPP-4 inhibitor treatment,Citation67,Citation68 although in the EXAMINE study the incidence of heart failure was not significantly increased in the alogliptin group (hazard ratio 1.19, 95% CI 0.89–1.58, P=0.24).Citation30 Further investigation is needed to clarify whether DPP-4 inhibitors are associated with a risk of heart failure.
Other adverse events
Angioedema has been reported as an adverse event of DPP-4 inhibitors, especially when combined with an angiotensin-converting enzyme inhibitor.Citation69 We have had a patient who developed facial angioedema after the introduction of vildagliptin, which improved after switching from vildagliptin to alogliptin.Citation70 The incidence of angioedema due to DPP-4 inhibitors is generally low, and no difference in its incidence was reported between the alogliptin and placebo groups in the EXAMINE trial.Citation30
To date, no meta-analyses of clinical trials or large-scale, randomized controlled trials of DPP-4 inhibitors suggest any increase in specific adverse events, such as nasopharyngitis, upper respiratory tract infection, skin reaction, hepatotoxicity, or pancreatitis, with the exception of heart failure as described above.Citation1,Citation30,Citation64,Citation71,Citation72 Concern has been raised regarding the risk of pancreatitis or pancreatic cancer associated with incretin therapy;Citation73,Citation74 however, no increase in the incidence of acute pancreatitis has been reported in patients treated with incretin therapy in recent health insurance claims database and case-control studies,Citation75,Citation76 or in randomized controlled trials.Citation30,Citation64 Although case reports of liver injury in patients treated with alogliptin have raised the question of possible hepatotoxicity with this agent, no evidence of liver injury was reported in the EXAMINE trial.Citation30,Citation77 Completion of pooled analysis of ongoing long-term, large-scale clinical trials is needed to determine the longer-term safety profile, such as the incidence of cancer ().
Table 3 Issues concerning dipeptidyl peptidase-4 inhibitors in need of further clarification
Role of DPP-4 inhibitors in treatment of T2DM
It is now well appreciated that not only type 1 diabetes but also T2DM is characterized by a deficit of beta cells.Citation78–Citation81 Beta cell dysfunction is a strong predictor of future development of T2DM,Citation82–Citation87 suggesting that beta cell dysfunction is a core pathogenetic feature of the disease. Further, beta cell function in patients with T2DM progressively declines with duration of disease and is negatively associated with glycemic control.Citation88–Citation92 Thus, preservation or recovery of beta cell function is an important treatment strategy in T2DM.Citation93
Despite recent advances in the treatment of T2DM, several issues remain to be resolved (), all of which are related to nonphysiological, inappropriate insulin supplementation by treatment, such as with sulfonylureas or insulin. In contrast, DPP-4 inhibitors have benefits with respect to all these issues (). Further, DPP-4 inhibitors improve the dynamics of glucagon,Citation94,Citation95 whereas raised plasma glucagon levels and a paradoxical increase in postprandial glucagon are another major defect in diabetes.Citation96 Therefore, one of the most important characteristics of DPP-4 inhibitors is recovery of endogenous insulin (and glucagon) secretion in a more physiological fashion compared with previous antidiabetic medications.
Table 4 Remaining issues in the treatment of type 2 diabetes and potential of DPP-4 inhibitors
A proposed concept with regard to a treatment strategy for T2DM in relation to beta cell function is shown in . T2DM is a progressive disease, and usually medication needs to be uptitrated with time.Citation97 Currently, the most effective strategy to preserve or restore beta cell function is to reduce the beta cell workload.Citation98 Since metformin reduces the insulin demand and beta cell workload by lowering hepatic glucose production, use of metformin in addition to lifestyle modification should be considered at as early a stage of diabetes as possible, unless contraindicated. In addition to the glucose-lowering efficacy of metformin, the less weight gain or modest weight loss, low risk of hypoglycemia, and low cost of this drug support metformin as a first-line treatment for T2DM, as recommended in most guidelines.Citation99–Citation101 Other non-insulin secretagogues, such as TZDs, alpha-glucosidase inhibitors, and sodium-glucose cotransporter 2 inhibitors should also be considered as treatment options to reduce beta cell workload, in addition to their clinical utility, ie, low risk of hypoglycemia.
Figure 2 Proposed concept of treatment strategy for T2DM in relation to beta cell function.
Abbreviations: IGT, impaired glucose tolerance; T2DM, type 2 diabetes; DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1; SGLT2, sodium-glucose cotransporter 2.
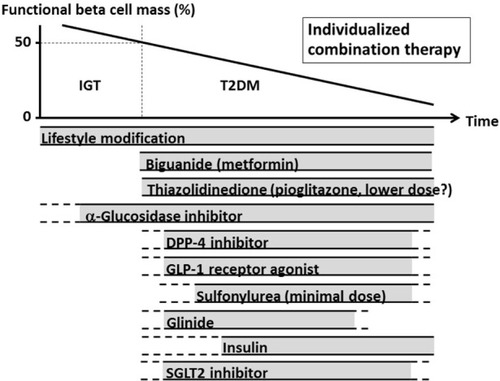
On the other hand, although DPP-4 inhibitors are “insulin secretagogues”, they are expected to improve beta cell function in addition to having a glucose-lowering effect and other advantages, as described in . Therefore, use of DPP-4 inhibitors can be considered for a broad range of disease stages. Combination therapy with metformin and a DPP-4 inhibitor is expected to have high glucose-lowering efficacy, a low risk of hypoglycemia, less weight gain or modest weight loss, and a low risk of adverse events, except for the gastrointestinal side effects of metformin, as described in a meta-analysis.Citation102
The glucose-lowering effect of TZDs has been shown to have greater durability compared with that of sulfonylureas or even metformin.Citation103 Thus, combination therapy with a DPP-4 inhibitor and a TZD is also expected to have a glucose- lowering effect with high efficacy and durability and a low risk of hypoglycemia, and to effectively improve beta cell function.Citation27 However, since TZDs are associated with weight gain, weight reduction should not be expected. In addition, a risk of bladder cancer and bone fractures are a concern with TZDs, although a recent large cohort study showed no increased risk of bladder cancer in patients treated with pioglitazone.Citation104 Thus, it might be advisable to use TZDs such as pioglitazone at a lower dosage to reduce the risk of adverse effects.
Fixed-dose combination tablets of alogliptin and metformin (Kazano®) or pioglitazone (Liovel® or Oseni®) are now available in many countries. It has been reported that use of fixed-dose combination tablets improves patient adherence to treatment and treatment satisfaction,Citation105,Citation106 possibly resulting in improved glycemic control. Thus, fixed-dose combinations containing alogliptin will be an important option for the treatment of T2DM. A combination of a DPP-4 inhibitor and another non-insulin secretagogue such as an alpha-glucosidase inhibitor or sodium-glucose cotransporter 2 inhibitor has also been shown to be effective for improving HbA1c and glycemic variability,Citation107,Citation108 with a low risk of hypoglycemia and weight neutrality or modest weight loss, although gastrointestinal disturbance with alpha-glucosidase inhibitors often limits their use. The long-term efficacy and safety of the combination of a DPP-4 inhibitor and a sodium-glucose cotransporter 2 inhibitor remains to be established.
In contrast, sulfonylureas (insulin secretagogues) may not be considered as initial therapy, but rather for use at a lower dose to support the insulinotropic effect of incretin therapy, because of their risk of hypoglycemia and weight gain. Glinides, short-acting insulin secretagogues, correct postprandial hyperglycemia. Although the glucose-lowering effect of glinides is modest, the risk of hypoglycemia with glinides is lower than that with sulfonylureas. Therefore, combined use of a DPP-4 inhibitor and a glinide may be useful to manage postprandial hyperglycemia while minimizing the risk of hypoglycemia and weight gain.
Insulin shows the greatest efficacy in reducing HbA1c, and has been shown to improve beta cell function.Citation109,Citation110 Thus, combination of a DPP-4 inhibitor and insulin should be considered for patients with various stages of T2DM, although the risk of hypoglycemia, weight gain, and fear or unwillingness to carry out injections may limit this combination. The efficacy of combination of a DPP-4 inhibitor and a glucagon-like peptide-1 agonist remains to be determined.
Since there is no drug to date that can cure diabetes, combination therapy should be considered in most cases. To date, on top of metformin, each combination of antidiabetic medication has its advantages and disadvantages (). The results of the ACCORD (Action to Control Cardiovascular Risk in Diabetes) study have warned us not to simply intensify medical treatment to reduce HbA1c to near-normal levels.Citation111 Hospital admission rates for hypoglycemia now exceed those for hyperglycemia among older adults.Citation112 The burden of medical treatment may rather result in reducing the patient’s quality-adjusted life years.Citation113 Therefore, each combined drug should be selected for each individual patient with consideration of the patient’s preference.Citation99,Citation114 Although studies suggest that initial combination therapy reduces HbA1c more effectively than monotherapy in the short term,Citation21,Citation25 the longer-term efficacy remains unknown.
Table 5 Summary of characteristics of antidiabetic medications when combined with metformin
It has been reported that the glucose-lowering effect of DPP-4 inhibitors is more potent in Asians than in Caucasians.Citation38 Moreover, recent studies suggest that there is a difference in the pathophysiological features of T2DM according to ethnicity.Citation115–Citation117 Thus, the above-mentioned treatment strategy may not be applicable to other ethnicities or countries, and may need to be modified in each situation.
Conclusion
Alogliptin, a highly selective DPP-4 inhibitor, has a glucose-lowering effect, the degree of which is similar to that of other DPP-4 inhibitors, with a low risk of hypoglycemia and weight gain. The safety of alogliptin, including cardiovascular adverse events, has been confirmed in a large-scale trial. Having a low risk of hypoglycemia, weight neutrality, and the potential to improve beta cell function, DPP-4 inhibitors are expected to become an important component of the treatment of T2DM. In Japan, currently more than half of patients with T2DM are treated with DPP-4 inhibitors. The cost-effectiveness of DPP-4 inhibitors needs to be assessed in future trials.
Acknowledgments
The author is grateful to Dr Wendy Gray for editing the manuscript.
Disclosure
The author reports no conflicts of interest in this work.
References
- KaragiannisTPaschosPPaletasKMatthewsDRTsapasADipeptidyl peptidase-4 inhibitors for treatment of type 2 diabetes mellitus in the clinical setting: systematic review and meta-analysisBMJ2012344e136922411919
- FengJZhangZWallaceMBDiscovery of alogliptin: a potent, selective, bioavailable, and efficacious inhibitor of dipeptidyl peptidase IVJ Med Chem200750102297230017441705
- BerhanABerhanYEfficacy of alogliptin in type 2 diabetes treatment: a meta-analysis of randomized double-blind controlled studiesBMC Endocr Disord2013131923452780
- SaishoYAlogliptin for the treatment of type 2 diabetesJ Symptoms Signs2014312634
- RendellMDrincicAAndukuriRAlogliptin benzoate for the treatment of type 2 diabetesExpert Opin Pharmacother201213455356322296609
- WhiteJRAlogliptin for the treatment of type 2 diabetesDrugs Today (Barc)20114729910721431099
- LeeBShiLKasselDBAsakawaTTakeuchiKChristopherRJPharmacokinetic, pharmacodynamic, and efficacy profiles of alogliptin, a novel inhibitor of dipeptidyl peptidase-4, in rats, dogs, and monkeysEur J Pharmacol20085891–330631418538760
- HuanYJiangQLiuJ-LShenZ-FEstablishment of a dipeptidyl peptidases (DPP) 8/9 expressing cell model for evaluating the selectivity of DPP4 inhibitorsJ Pharmacol Toxicol Methods20157181225464020
- NabenoMAkahoshiFKishidaHA comparative study of the binding modes of recently launched dipeptidyl peptidase IV inhibitors in the active siteBiochem Biophys Res Commun2013434219119623501107
- ChristopherRCovingtonPDavenportMPharmacokinetics, pharmacodynamics, and tolerability of single increasing doses of the dipeptidyl peptidase-4 inhibitor alogliptin in healthy male subjectsClin Ther200830351352718405789
- CovingtonPChristopherRDavenportMPharmacokinetic, pharmacodynamic, and tolerability profiles of the dipeptidyl peptidase-4 inhibitor alogliptin: a randomized, double-blind, placebo-controlled, multiple-dose study in adult patients with type 2 diabetesClin Ther200830349951218405788
- KarimACovingtonPChristopherRPharmacokinetics of alogliptin when administered with food, metformin, or cimetidine: a two-phase, crossover study in healthy subjectsInt J Clin Pharmacol Ther2010481465820040339
- BaettaRCorsiniAPharmacology of dipeptidyl peptidase-4 inhibitors: similarities and differencesDrugs201171111441146721812507
- GibbsJPFredricksonJBarbeeTQuantitative model of the relationship between dipeptidyl peptidase-4 (DPP-4) inhibition and response: meta-analysis of alogliptin, saxagliptin, sitagliptin, and vildagliptin efficacy resultsJ Clin Pharmacol201252101494150522162539
- DeFronzoRAFleckPRWilsonCAMekkiQEfficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin in patients with type 2 diabetes and inadequate glycemic control: a randomized, double-blind, placebo-controlled studyDiabetes Care200831122315231718809631
- SeinoYFujitaTHiroiSHirayamaMKakuKEfficacy and safety of alogliptin in Japanese patients with type 2 diabetes mellitus: a randomized, double-blind, dose-ranging comparison with placebo, followed by a long-term extension studyCurr Med Res Opin20112791781179221806314
- PratleyREKipnesMSFleckPRWilsonCMekkiQEfficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin in patients with type 2 diabetes inadequately controlled by glyburide monotherapyDiabetes Obes Metab200911216717619125778
- SeinoYHiroiSHirayamaMKakuKEfficacy and safety of alogliptin added to sulfonylurea in Japanese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial with an open-label, long-term extension studyJ Diabetes Invest201236517525
- NauckMAEllisGCFleckPRWilsonCAMekkiQEfficacy and safety of adding the dipeptidyl peptidase-4 inhibitor alogliptin to metformin therapy in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a multicentre, randomised, double-blind, placebo-controlled studyInt J Clin Pract2009631465519125992
- SeinoYMiyataYHiroiSHirayamaMKakuKEfficacy and safety of alogliptin added to metformin in Japanese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial with an open-label, long-term extension studyDiabetes Obes Metab2012141092793622583697
- PratleyREFleckPWilsonCEfficacy and safety of initial combination therapy with alogliptin plus metformin versus either as monotherapy in drug-naïve patients with type 2 diabetes: a randomized, double-blind, 6-month studyDiabetes Obes Metab201416761362124400655
- Del PratoSCamisascaRWilsonCFleckPDurability of the efficacy and safety of alogliptin compared with glipizide in type 2 diabetes mellitus: a 2-year studyDiabetes Obes Metab201416121239124625132212
- PratleyREReuschJEFleckPRWilsonCAMekkiQEfficacy and safety of the dipeptidyl peptidase-4 inhibitor alogliptin added to pioglitazone in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled studyCurr Med Res Opin200925102361237119650752
- KakuKItayasuTHiroiSHirayamaMSeinoYEfficacy and safety of alogliptin added to pioglitazone in Japanese patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial with an open-label long-term extension studyDiabetes Obes Metab201113111028103521682833
- RosenstockJInzucchiSESeufertJFleckPRWilsonCAMekkiQInitial combination therapy with alogliptin and pioglitazone in drug-naive patients with type 2 diabetesDiabetes Care201033112406240820724648
- DeFronzoRABurantCFFleckPWilsonCMekkiQPratleyREEfficacy and tolerability of the DPP-4 inhibitor alogliptin combined with pioglitazone, in metformin-treated patients with type 2 diabetesJ Clin Endocrinol Metab20129751615162222419732
- Van RaalteDHvan GenugtenREEliassonBThe effect of alogliptin and pioglitazone combination therapy on various aspects of β-cell function in patients with recent-onset type 2 diabetesEur J Endocrinol2014170456557424421302
- RosenstockJRendellMSGrossJLFleckPRWilsonCAMekkiQAlogliptin added to insulin therapy in patients with type 2 diabetes reduces HbA(1C) without causing weight gain or increased hypoglycaemiaDiabetes Obes Metab200911121145115219758359
- KakuKMoriMKanooTKatouMSeinoYEfficacy and safety of alogliptin added to insulin in Japanese patients with type 2 diabetes: a randomized, double-blind, 12-week, placebo-controlled trial followed by an open-label, long-term extension phaseExpert Opin Pharmacother201415152121213025190226
- WhiteWBCannonCPHellerSRAlogliptin after acute coronary syndrome in patients with type 2 diabetesN Engl J Med2013369141327133523992602
- ArodaVRHenryRRHanJEfficacy of GLP-1 receptor agonists and DPP-4 inhibitors: meta-analysis and systematic reviewClin Ther201234612471258e122222608780
- CraddyPPalinH-JJohnsonKIComparative effectiveness of ddipeptidylpeptidase-4 iinhibitors in type 2 diabetes: a systematic review and mixed treatment comparisonDiabetes Ther20145114124664619
- MarfellaRBarbieriMGrellaRRizzoMRNicolettiGFPaolissoGEffects of vildagliptin twice daily vs. sitagliptin once daily on 24-hour acute glucose fluctuationsJ Diabetes Complications2010242798319261490
- SignorovitchJEWuEQSwallowEKantorEFanLGruenbergerJBComparative efficacy of vildagliptin and sitagliptin in Japanese patients with type 2 diabetes mellitus: a matching-adjusted indirect comparison of randomized trialsClin Drug Invest2011319665674
- SakamotoMNishimuraRIrakoTTsujinoDAndoKUtsunomiyaKComparison of vildagliptin twice daily vs sitagliptin once daily using continuous glucose monitoring (CGM): crossover pilot study (J-VICTORIA study)Cardiovasc Diabetol2012119222867630
- DeFronzoRAStonehouseAHHanJWintleMERelationship of baseline HbA1c and efficacy of current glucose-lowering therapies: a meta-analysis of randomized clinical trialsDiabet Med201027330931720536494
- EspositoKChiodiniPCapuanoAMaiorinoMBellastellaGGiuglianoDBaseline glycemic parameters predict the hemoglobin A1c response to DPP-4 inhibitorsEndocrine2014461435124248503
- KimYGHahnSOhTJKwakSHParkKSChoYMDifferences in the glucose-lowering efficacy of dipeptidyl peptidase-4 inhibitors between Asians and non-Asians: a systematic review and meta-analysisDiabetologia201356469670823344728
- KimYGHahnSOhTJParkKSChoYMDifferences in the HbA1c-lowering efficacy of glucagon-like peptide-1 analogues between Asians and non-Asians: a systematic review and meta-analysisDiabetes Obes Metab2014161090090924655583
- EspositoKChiodiniPMaiorinoMIBellastellaGCapuanoAGiuglianoDGlycaemic durability with dipeptidyl peptidase-4 inhibitors in type 2 diabetes: a systematic review and meta-analysis of long-term randomised controlled trialsBMJ Open201446e005442
- RosenstockJWilsonCFleckPAlogliptin versus glipizide monotherapy in elderly type 2 diabetes mellitus patients with mild hyperglycaemia: a prospective, double-blind, randomized, 1-year studyDiabetes Obes Metab2013151090691423531118
- BronMWilsonCFleckPA post hoc analysis of HbA1c, hypoglycemia, and weight change outcomes with alogliptin vs glipizide in older patients with type 2 diabetesDiabetes Ther2014552153425424968
- PratleyREMcCallTFleckPRWilsonCAMekkiQAlogliptin use in elderly people: a pooled analysis from phase 2 and 3 studiesJ Am Geriatr Soc200957112011201919793357
- NakamuraYInagakiMShimizuTLong-term effects of alogliptin benzoate in hemodialysis patients with diabetes: a 2-year studyNephron Clin Pract20131231–2465123774306
- MoritohYTakeuchiKAsakawaTKataokaOOdakaHChronic administration of alogliptin, a novel, potent, and highly selective dipeptidyl peptidase-4 inhibitor, improves glycemic control and beta-cell function in obese diabetic ob/ob miceEur J Pharmacol20085882-332533218499100
- KawashimaSMatsuokaTAKanetoHEffect of alogliptin, pioglitazone and glargine on pancreatic beta-cells in diabetic db/db miceBiochem Biophys Res Commun2011404153454021144823
- ZhangXWangZHuangYWangJEffects of chronic administration of alogliptin on the development of diabetes and beta-cell function in high fat diet/streptozotocin diabetic miceDiabetes Obes Metab201113433734721205126
- YinHParkSYWangXJEnhancing pancreatic beta-cell regeneration in vivo with pioglitazone and alogliptinPLoS One201386e6577723762423
- BosiEEllisGCWilsonCAFleckPRAlogliptin as a third oral antidiabetic drug in patients with type 2 diabetes and inadequate glycaemic control on metformin and pioglitazone: a 52-week, randomized, double-blind, active-controlled, parallel-group studyDiabetes Obes Metab201113121088109621733058
- JurczykADiiorioPBrostowinDImproved function and proliferation of adult human beta cells engrafted in diabetic immunodeficient NOD-scid IL2rgamma(null) mice treated with alogliptinDiabetes Metab Syndr Obes Target Ther20136493499
- FoleyJEBunckMCMoller-GoedeDLBeta cell function following 1 year vildagliptin or placebo treatment and after 12 week washout in drug-naive patients with type 2 diabetes and mild hyperglycaemia: a randomised controlled trialDiabetologia20115481985199121547496
- EliassonBMoller-GoedeDEeg-OlofssonKLowering of postprandial lipids in individuals with type 2 diabetes treated with alogliptin and/or pioglitazone: a randomised double-blind placebo-controlled studyDiabetologia201255491592522237690
- MonamiMLamannaCDesideriCMMannucciEDPP-4 inhibitors and lipids: systematic review and meta-analysisAdv Ther2012291142522215383
- ShahZKampfrathTDeiuliisJALong-term dipeptidyl-peptidase 4 inhibition reduces atherosclerosis and inflammation via effects on monocyte recruitment and chemotaxisCirculation2011124212338234922007077
- TaNNSchuylerCALiYLopes-VirellaMFHuangYDPP-4 (CD26) inhibitor alogliptin inhibits atherosclerosis in diabetic apolipoprotein E-deficient miceJ Cardiovasc Pharmacol201158215716621558879
- YangDNakajoYIiharaKKataokaHYanamotoHAlogliptin, a dipeptidylpeptidase-4 inhibitor, for patients with diabetes mellitus type 2, induces tolerance to focal cerebral ischemia in non-diabetic, normal miceBrain Res2013151710411323602966
- NodaYMiyoshiTOeHAlogliptin ameliorates postprandial lipemia and postprandial endothelial dysfunction in non-diabetic subjects: a preliminary reportCardiovasc Diabetol201312823298374
- MegaCde LemosETValaHDiabetic nephropathy amelioration by a low-dose sitagliptin in an animal model of type 2 diabetes (Zucker diabetic fatty rat)Exp Diabetes Res2011201116209222203828
- NakashimaSMatsuiTTakeuchiMYamagishiSILinagliptin blocks renal damage in type 1 diabetic rats by suppressing advanced glycation end products-receptor axisHorm Metab Res2014461071772124710699
- LiuWJXieSHLiuYNDipeptidyl peptidase IV inhibitor attenuates kidney injury in streptozotocin-induced diabetic ratsJ Pharmacol Exp Ther2012340224825522025647
- VavrinecPHenningRHLandheerSWVildagliptin restores renal myogenic function and attenuates renal sclerosis independently of effects on blood glucose or proteinuria in Zucker diabetic fatty ratCurr Vasc Pharmacol201412683684424066937
- FujitaHTaniaiHMurayamaHDPP-4 inhibition with alogliptin on top of angiotensin II type 1 receptor blockade ameliorates albuminuria via up-regulation of SDF-1alpha in type 2 diabetic patients with incipient nephropathyEndocr J201461215916624225429
- SakataKHayakawaMYanoYEfficacy of alogliptin, a dipeptidyl peptidase-4 inhibitor, on glucose parameters, the activity of the advanced glycation end product (AGE) – receptor for AGE (RAGE) axis and albuminuria in Japanese type 2 diabetesDiabetes Metab Res Rev201329862463023861159
- SciricaBMBhattDLBraunwaldESaxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitusN Engl J Med2013369141317132623992601
- AvogaroAFadiniGPThe effects of dipeptidyl peptidase-4 inhibition on microvascular diabetes complicationsDiabetes Care201437102884289425249673
- WhiteWBPratleyRFleckPCardiovascular safety of the dipetidyl peptidase-4 inhibitor alogliptin in type 2 diabetes mellitusDiabetes Obes Metab201315766867323489301
- MonamiMDicembriniIMannucciEDipeptidyl peptidase-4 inhibitors and heart failure: a meta-analysis of randomized clinical trialsNutr Metab Cardiovasc Dis201424768969724793580
- CliftonPDo dipeptidyl peptidase IV (DPP-IV) inhibitors cause heart failure?Clin Ther201436122072207925453730
- BrownNJByiersSCarrDMaldonadoMWarnerBADipeptidyl peptidase-IV inhibitor use associated with increased risk of ACE inhibitor-associated angioedemaHypertension200954351652319581505
- SaishoYItohHDipeptidyl peptidase-4 inhibitors and angioedema: a class effect?Diabet Med2013304e149e15023323612
- MonamiMDicembriniIMartelliDMannucciESafety of dipeptidyl peptidase-4 inhibitors: a meta-analysis of randomized clinical trialsCurr Med Res Opin201127Suppl 3576422106978
- NauckMAA critical analysis of the clinical use of incretin-based therapies: the benefits by far outweigh the potential risksDiabetes Care20133672126213223645884
- ElashoffMMatveyenkoAVGierBElashoffRButlerPCPancreatitis, pancreatic, and thyroid cancer with glucagon-like peptide-1- based therapiesGastroenterology2011141115015621334333
- SinghSChangHYRichardsTMWeinerJPClarkJMSegalJBGlucagonlike peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes mellitus: a population-based matched case-control studyJAMA Intern Med2013173753453923440284
- YabeDKuwataHKanekoMUse of the Japanese health insurance claims database to assess the risk of acute pancreatitis in patients with diabetes: comparison of DPP-4 inhibitors with other oral antidiabetic drugsDiabetes Obes Metab8222014 Epub ahead of print
- GiordaCSacerdoteCNadaEMarafettiLBaldiIGnaviRIncretin-based therapies and acute pancreatitis risk: a systematic review and meta-analysis of observational studiesEndocrine8222014 Epub ahead of print
- ScheenAAlogliptin: concern about hepatotoxicity?Clin Pharmacokinet201453111057105925336393
- ButlerAEJansonJBonner-WeirSRitzelRRizzaRAButlerPCBeta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetesDiabetes200352110211012502499
- YoonKHKoSHChoJHSelective beta-cell loss and alpha-cell expansion in patients with type 2 diabetes mellitus in KoreaJ Clin Endocrinol Metab20038852300230812727989
- SakurabaHMizukamiHYagihashiNWadaRHanyuCYagihashiSReduced beta-cell mass and expression of oxidative stress-related DNA damage in the islet of Japanese type II diabetic patientsDiabetologia2002451859611845227
- RahierJGuiotYGoebbelsRMSempouxCHenquinJCPancreatic beta-cell mass in European subjects with type 2 diabetesDiabetes Obes Metab200810Suppl 4324218834431
- SkarforsETSelinusKILithellHORisk factors for developing non-insulin dependent diabetes: a 10 year follow up of men in UppsalaBMJ199130368057557601932936
- ErikssonKFLindgardeFPoor physical fitness, and impaired early insulin response but late hyperinsulinaemia, as predictors of NIDDM in middle-aged Swedish menDiabetologia19963955735798739917
- WeyerCBogardusCMottDMPratleyREThe natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitusJ Clin Invest1999104678779410491414
- HaffnerSMMiettinenHGaskillSPSternMPDecreased insulin secretion and increased insulin resistance are independently related to the 7-year risk of NIDDM in Mexican-AmericansDiabetes19954412138613917589843
- LyssenkoVAlmgrenPAnevskiDPredictors of and longitudinal changes in insulin sensitivity and secretion preceding onset of type 2 diabetesDiabetes200554116617415616025
- TabakAGJokelaMAkbaralyTNBrunnerEJKivimakiMWitteDRTrajectories of glycaemia, insulin sensitivity, and insulin secretion before diagnosis of type 2 diabetes: an analysis from the Whitehall II studyLancet200937396822215222119515410
- UK Prospective Diabetes Study GroupUK Prospective Diabetes Study 16. Overview of 6 years’ therapy of type II diabetes: a progressive diseaseDiabetes19954411124912587589820
- LevyJAtkinsonABBellPMMcCanceDRHaddenDRBeta-cell deterioration determines the onset and rate of progression of secondary dietary failure in type 2 diabetes mellitus: the 10-year follow-up of the Belfast Diet StudyDiabet Med19981542902969585393
- SaishoYTanakaKAbeTShimadaAKawaiTItohHEffect of obesity on declining beta cell function after diagnosis of type 2 diabetes: a possible link suggested by cross-sectional analysisEndocr J201259318719522156325
- SaishoYKouKTanakaKAssociation between beta cell function and future glycemic control in patients with type 2 diabetesEndocr J201360451752323268927
- SaishoYTanakaKAbeTShimadaAKawaiTItohHGlycated albumin to glycated hemoglobin ratio reflects postprandial glucose excursion and relates to beta cell function in both type 1 and type 2 diabetesDiabetol Int201123146153
- SaishoYBeta cell dysfunction: its critical role in prevention and management of type 2 diabetesWorld J Diabetes20156110912425685282
- MuscelliECasolaroAGastaldelliAMechanisms for the antihyperglycemic effect of sitagliptin in patients with type 2 diabetesJ Clin Endocrinol Metab20129782818282622685234
- Solis-HerreraCTriplittCGarduno-GarciaJDAdamsJDefronzoRACersosimoEMechanisms of glucose lowering of dipeptidyl peptidase-4 inhibitor sitagliptin when used alone or with metformin in type 2 diabetes: a double-tracer studyDiabetes Care20133692756276223579178
- MullerWAFaloonaGRAguilar-ParadaEUngerRHAbnormal alpha-cell function in diabetes. Response to carbohydrate and protein ingestionN Engl J Med197028331091154912452
- Del PratoSFeltonAMMunroNNestoRZimmetPZinmanBImproving glucose management: ten steps to get more patients with type 2 diabetes to glycaemic goalInt J Clin Pract200559111345135516236091
- DeFronzoRAAbdul-GhaniMAPreservation of beta-cell function: the key to diabetes preventionJ Clin Endocrinol Metab20119682354236621697254
- InzucchiSEBergenstalRMBuseJBManagement of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: Update to a position statement of the American Diabetes Association and the European Association for the Study of DiabetesDiabetes Care201538114014925538310
- GarberAJAbrahamsonMJBarzilayJIAACE comprehensive diabetes management algorithm 2013Endocr Pract201319232733623598536
- International Diabetes Federation Guideline Development GroupGlobal guideline for type 2 diabetesDiabetes Res Clin Pract2014104115224508150
- PhungOJScholleJMTalwarMColemanCIEffect of noninsulin antidiabetic drugs added to metformin therapy on glycemic control, weight gain, and hypoglycemia in type 2 diabetesJAMA2010303141410141820388897
- KahnSEHaffnerSMHeiseMAGlycemic durability of rosiglitazone, metformin, or glyburide monotherapyN Engl J Med2006355232427244317145742
- LevinDBellSSundRPioglitazone and bladder cancer risk: a multipopulation pooled, cumulative exposure analysisDiabetologia201458349350425481707
- BangaloreSKamalakkannanGParkarSMesserliFHFixed-dose combinations improve medication compliance: a meta-analysisAm J Med2007120871371917679131
- HutchinsVZhangBFleurenceRLKrishnarajahGGrahamJA systematic review of adherence, treatment satisfaction and costs, in fixed-dose combination regimens in type 2 diabetesCurr Med Res Opin20112761157116821466277
- KurozumiAOkadaYMoriHAraoTTanakaYEfficacy of α-glucosidase inhibitors combined with dipeptidyl-peptidase-4 inhibitor (alogliptin) for glucose fluctuation in patients with type 2 diabetes mellitus by continuous glucose monitoringJ Diabetes Invest201344393398
- JabbourSAHardyESuggJParikhSStudy 10 GroupDapagliflozin is effective as add-on therapy to sitagliptin with or without metformin: a 24-week, multicenter, randomized, double-blind, placebo-controlled StudyDiabetes Care201437374075024144654
- WengJLiYXuWEffect of intensive insulin therapy on beta-cell function and glycaemic control in patients with newly diagnosed type 2 diabetes: a multicentre randomised parallel-group trialLancet200837196261753176018502299
- PennartzCSchenkerNMengeBASchmidtWENauckMAMeierJJChronic reduction of fasting glycemia with insulin glargine improves first- and second-phase insulin secretion in patients with type 2 diabetesDiabetes Care20113492048205321775756
- GersteinHCMillerMEByingtonRPEffects of intensive glucose lowering in type 2 diabetesN Engl J Med2008358242545255918539917
- LipskaKJRossJSWangYNational trends in US hospital admissions for hyperglycemia and hypoglycemia among Medicare beneficiaries, 1999 to 2011JAMA Intern Med201417471116112424838229
- VijanSSussmanJBYudkinJSHaywardRAEffect of patients’ risks and preferences on Health gains with plasma glucose level lowering in type 2 diabetes mellitusJAMA Intern Med201417481227123424979148
- RazIRiddleMCRosenstockJPersonalized management of hyperglycemia in type 2 diabetes: reflections from a Diabetes Care Editors’ Expert ForumDiabetes Care20133661779178823704680
- HsuWCBoykoEJFujimotoWYPathophysiologic differences among Asians, native Hawaiians, and other Pacific Islanders and treatment implicationsDiabetes Care20123551189119822517940
- KodamaKTojjarDYamadaSTodaKPatelCJButteAJEthnic differences in the relationship between insulin sensitivity and insulin response: a systematic review and meta-analysisDiabetes Care20133661789179623704681
- KouKSaishoYSatohSYamadaTItohHChange in beta cell mass in Japanese non-diabetic obese individualsJ Clin Endocrinol Metab20139893724373023766518

![Figure 1 Chemical structure of alogliptin benzoate: 2-({6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl}methyl) benzonitrile monobenzoate.](/cms/asset/fe446692-666d-4f9e-b276-597f24560a71/dvhr_a_68564_f0001_b.jpg)