Abstract
The aim of this study was to assess the impact of conjugated linoleic acids (CLAs), vitamin E, and combination of these nutrients on serum lipid profiles and blood pressure (BP) in patients with active rheumatoid arthritis (RA). In a randomized, double-blind, placebo-controlled trial, 87 patients with active RA were divided into four groups receiving one of the following daily supplements for three months: Group C: CLAs 2.5 g equivalent to 2 g mixture of cis 9-trans 11 and trans 10-cis12 CLAs in a rate of 50/50; Group E: vitamin E: 400 mg; Group CE: CLAs and vitamin E at above doses: Group P: placebo. After supplementation, SBP levels decreased significantly in the group C in comparison with groups E and P and mean arterial pressure reduced significantly in groups C and CE. There weren’t significant differences in the levels of prostaglandin E2 (PGE2), triglycerides, cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), LDL/HDL, cholesterol/HDL, fasting blood sugar, C-reactive protein (CRP), arylestrase activity, platelet count and body mass index between groups. CRP dropped nonsignificantly in groups P, C, E and CE (19%, 24%, 55%, and 39%, respectively). Erythrocytes sedimentation rate levels decreased in groups C, E and CE (P ≤ 0.05, P ≤ 0.05, P ≤ 0.001, respectively). It is concluded that supplementation of CLAs decreased BP and vitamin E decreased CRP. Therefore cosupplementation of CLAs and vitamin E might be profitable for heart disease prevention in RA patients.
Introduction
Rheumatoid arthritis (RA), as the most common inflammatory disease, is associated with excess cardiovascular morbidity and mortality that is not entirely explained by traditional risk factors of cardiovascular disease (CVD) (CitationDe Pablo et al 2007). It might be due to an increased prevalence of cardiovascular risk factors such as dyslipidemia (CitationNurmohamed 2007). Risk factors for CVD, including inflammatory biomarkers, low intake of antioxidants, and vitamins, may contribute to excess CVD in persons with RA (CitationDessein et al 2006). There is a complex relationship between inflammation, antioxidant vitamin status, and the risk of CVD. Furthermore, low intakes of antioxidant vitamins and other micronutrients, including vitamin E, have been reported to be inversely associated with CVD incidence and mortality (CitationDe Pablo et al 2007). Conjugated linoleic acids (CLAs) are a group of naturally occurring isomers of linoleic acid that differ in the position or geometry of their double bonds (CitationKelly 2001; CitationBhattacharya et al 2006). The biological activities of CLAs have received considerable attention because of its anticancer (CitationParodi 1999), antiatherogenic (CitationRudel 1999; CitationArbonés-Mainar et al 2006) and antidiabetic effects (CitationPeck et al 1998). The atheroprotective properties of CLAs have been particularly well documented (CitationKritchevsky et al 2000; CitationToomey et al 2003). Inclusion of CLAs in a hypercholesterolemic diet in rabbits led to a significant reduction in serum triglyceride (TG), low-density lipoprotein cholesterol (LDL-C) and cholesterol (CHO) levels. In addition, there was a reduced incidence of atherosclerotic plaque formation in the rabbit aorta (CitationLee et al 1994). Similar results have also been reported for hamster hypercholesterolemia models (CitationNicolosi et al 1997; CitationWilson et al 2000). Despite these findings, some reports suggest that CLAs may possibly have proatherogenic effects (CitationMunday et al 1999; CitationGavino et al 2000). There are several molecular mechanisms for explaining the hypotriacylglycerolemic effect of CLAs. The cis-9, trans-11 and the trans-10, cis-12 isomers of CLAs are potent peroxisome proliferators-activated receptor (PPAR) agonists. PPARα is a key transcription factor that regulates hepatic lipid metabolism (CitationMoya-Camarena and Belury 1999a). Feeding CLAs-enriched diets increases the expression of PPARα-responsive genes in animals (CitationMoya-Camarena and Belury 1999b). Therefore, the TG-lowering effect of CLAs may be partly attributed to the effect of CLAs on PPARα. CLAs is also a PPARγ ligand. In adipose tissue, PPARγ regulates the expression of the genes that determine adipogenesis, lipid metabolism and insulin sensitivity (CitationLowell et al 1999; CitationMoya-Camarena et al 1999). Some other proposed mechanisms include their role on peroxisome proliferators-sterol regulatory element-binding proteins (SREBPs) (CitationPai et al 1998) and stearoyl-CoA desaturase (SCD) (CitationMiyazaki et al 2000, Citation2001; CitationNtambi 1999). SREBP1c isoforms regulate fatty acid and TG synthesis (CitationPai et al 1998). Studies suggest that liver SREBP-1c expression is dependent on the nuclear hormone liver X receptor (LXR). C9 t11 isomer was shown to down-regulate mRNA expression of LXR-α and SREBP-1c, whereas the t10 c12 isomer had no effect (CitationRoche et al 2002). The results suggested that c9t11 isomer positively influences lipid metabolism by reduced synthesis and cleavage of hepatic SREBP-1, which in turn is regulated by LXRα expression. SREBP-1c enhances the transcription of the genes required for fatty acid synthesis and fatty acid elongation including fatty acids synthetase (FAS) and SCD (CitationPai et al 1998; CitationKönig et al 2008). Hypertension is also a common pathological state associated with an increased risk of cardiovascular diseases. Some studies have shown that CLAs (50:50) or the t10c12 isomer decreases blood pressure (BP) and hypertension in various rat models prone to develop obesity, diabetes and hypertension (CitationNagao et al 2003a, Citation2003b; CitationAlice et al 2005). One possible mechanism is through inhibition of cyclooxygenases, both of them have been implicated in the development of atherosclerosis (CitationBelton et al 2003). c9, t11- and t10, c12-CLAs isomers are not substrates for cyclooxygenases (COX) but compete with arachidonic acid for access to the enzymes. In one study, it was seen that feeding rabbits with an atherogenic diet rich in CLAs reduced atherosclerotic lesion sizes (CitationValetille et al 2005). A number of studies in rabbits have considered the potential for the lipophilic antioxidant vitamin E to inhibit atherosclerosis in vitro by decreasing lipid peroxidation of LDL (CitationSchwenke et al 1998). Some epidemiological studies have shown an association between high dietary intake and high serum concentrations of vitamin E and lower rates of ischemic heart disease (Gey et al 1990; CitationRimm et al 1993; CitationStampfer et al 1993) and myocardial infarction (CitationStephens et al 1996). Here, we investigated the effect of 50:50 blend of c9, t11: t10, c12-CLAs on BP and lipid profiles in active RA patients.
Materials and methods
Subjects
A randomized double blind placebo controlled trial was conducted in a 12 week period in 87 patients with active RA. Patients were between 19–69 years old and had RA for at least 2 years fulfilling American College of Rheumatology (ACR) criteria of 1987 (CitationArnett et al 1988). The method for evaluation of disease activity was global physician assessment (GPA). Disease activity was determined by rheumatologist by medical history, pain in joints, morning stiffness, inflammation and tenderness and by acute phase reactants such as C-reactive protein (CRP), erythrocytes sedimentation rate (ESR), platelet (PLT), and hemoglobin (Hb). Data on dietary habits, dietary supplements, anthropometric indices including body mass index (BMI), smoking habits, and drug history were obtained by face to face interview. The exclusion criteria were: abnormal renal and/or hepatic function, smoking, and history of myocardial infarction, pregnancy, taking vitamins and/or mineral supplements, hyperlipidemia and taking drugs such as thyroid hormones, estrogens, progesterone, diuretics or β-blockers. A written informed consent was obtained from all participants. The research protocol was approved by the Ethics Committee of Tehran University of Medical Sciences. The patients were divided into four groups to receive randomly either CLAs (group C), vitamin E (group E), both CLAs and vitamin E (group CE) or placebo (group P). CLAs was prescribed 2 g daily as 2 capsules (each capsule was 1.25 g and contained 80% CLAs) containing both cis-9, trans-11 CLAs and trans-10, cis-12 triglyceride type CLAs in equal proportion and vitamin E (α-tocopherol, 400 mg). Corn oil was the placebo for vitamin E and high oleic sunflower (HOSF) was placebo for CLAs. These were assigned to one of the four treatment groups using random permuted blocks procedure. The amount of nutrients intakes were estimated using a 24-hour dietary recall questionnaire for two following days before and after the study and analyzing by Food Processor software (ESHA Research, Salem, OR).
The subjects were asked not to alter their usual diets and physical activity throughout the study and any changes in their medication were avoided whenever possible and if the patients did change their nonsteroidal anti-inflammatory drug (NSAIDs) intake they must write it in the special forms provided. Compliance with the supplementation was assessed by counting the number of capsules used and also by measuring changes in the serum vitamin E.
After 12–14 h overnight fasting, 10 ml blood samples were collected from each subject at the beginning and at the end of the three month trial.
Methods
Blood samples were collected in trace element-free tubes. Aliquots of serum were transferred to polystyrene tubes which were immediately stored at −70 °C until analysis. Prostaglandin E2 (PGE2) was measured by ELISA-Monoclonal Kit (No 514010.1; Cayman Chemical Co, Ann Arbor, MI). Plasma CHO and TG concentrations were determined by an enzymatic assay adapted to microtiter plates using commercially available kits (Randox Labs, Crumlin, North Ireland). High-density lipoprotein cholesterol (HDL-C) was determined by Immunoinhibition Colorimetric method (EliTech, Seppim SAS Industrial, Sees, France). LDL-C was measured by enzymatic colorimetric method (EliTech, Seppim SAS Industrial). Fasting blood sugar (FBS) was measured by enzymatic colorimetric method (colorimetric). CRP values were determined with immunoturbidometric assay. In this method CRP gives a complex with polyclonal antibody and creates turbidity that has positive relation with CRP in sample. Arylestrase activity (AEA) was measured by phenylacetate that was used as a substrate to measure the AEA. Enzymatic activity was calculated from the molar absorptive coefficient of the produced phenol, one unit of AEA was defined as 1 mmol phenol generated per min under the above conditions and expressed as units/l of serum (CitationKushi et al 1996). Serum α-tocopherol was determined by high performance liquid chromatography (CitationSanz and Santa-Cruz 1986). Hematological analyses were counted by an automated blood counter (Beckman Coulter, Miami, FL). ESR was measured using an ESR apparatus (Greiner Labor Technic GmbH, Germany). The systolic (SBP) and diastolic blood pressure (DBP) was measured after five minutes seated rest, the mean of two readings at baseline, and after three months of supplementation. Mean arterial pressure (MAP) was calculated using this formula (SBP + 2DBP)/3 (CitationCywinski 1980).
Statistical analysis
All values are expressed as mean ± standard error of mean. Log transformation was used to normalize the distribution of CRP. All other variables were normally distributed. Differences between four groups were compared by one-way analysis of variance (ANOVA) for continuous data and the χ2-test for categorical data. Post hoc comparisons were performed with Tukey test. Adjustment for differences in baselines covariates and changes in variables during study were performed by analysis of covariance. A value of P ≤ 0.05 was considered to be statistically significant. All data were analyzed using SPSS software (SPSS Inc., Chicago, IL).
Results
As shown in , groups were similar with respect to the sex, age, duration of RA, BMI, and daily intake of vitamin E at the beginning of the study. There were no significant changes in BMI, physical activity, dietary intake or medication during the study period (data not shown).
Table 1 Demographic, anthropometric and biological data for the four groups before study (Mean ± SEM)
shows the serum lipid, lipoprotein concentrations and BP before and after supplementation for subjects who completed the study. At baseline there were no significant differences between groups by ANOVA. Our study did not show any significant changes in plasma CHO or LDL-C concentrations in C and CE groups following three months of supplementation. Plasma levels of HDL-C increased nonsignificantly in the groups P, C, E, and CE by 0.9%, 1.23%, 2.35%, and 4.9%, respectively as compared with baseline. LDL-C increased in all groups, but this increase was significant in group E compared with baseline. We did not find any significant change in HDL-C and LDL-C concentrations among four groups. Total CHO, CHO/HDL-C and LDL-C/HDL-C ratios, and TG were not altered significantly after supplementation compared with baseline and between four groups. After three months of supplementation in the group C, SBP levels reduced significantly by 8% (117.36 ± 3.80 mmHg versus 108.04 ± 3.13 mmHg) (P < 0.05) and DBP by 8.5% (72.36 ± 2.37 mmHg versus 66.14 ± 1.66 mmHg) (P < 0.05). The MBP decreased as compared with baseline in groups C and CE, 8.3% and 5.66%, respectively (). The reduction of SBP in group C was significant compared with other groups (P < 0.05) ().
Figure 1 Levels of mean arterial pressure before and after three months’ vitamin E and conjugated linoleic acid (CLA) supplementation in patients with active rheumatoid arthritis.
Note: *p ≤ 0.05 for CLA group compared with baseline and group P.
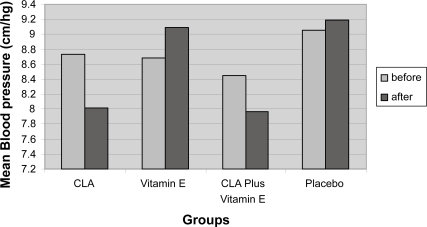
Table 2 Levels of lipid profile and blood pressure variables in patients with active rheumatoid arthritis before and after three months supplementation (Mean ± SEM)
shows the serum PGE2, FBS, CRP concentrations, AEA, ESR, WBC, PLT, and BMI before and after supplementation for subjects who completed the study. At baseline there were no significant differences between groups by ANOVA. PGE2 didn’t alter significantly among groups and in each group as compared with baseline (P > 0.05). ESR levels decreased significantly in groups C, E, and CE (P ≤ 0.05, P ≤ 0.05, P ≤ 0.001, respectively) in comparison with baseline and group CE had significantly lower ESR level than group P (P < 0.05). CRP decreased nonsignificantly in all groups as compared with baseline, after three months of supplementation (), but this reduction was 55% in group E compared with baseline (P= 0.054). The reduction of WBC in group CE was significant compared with other groups (P < 0.05) (). Platelet count decreased nonsignificantly in groups CE, C, and E. BMI and FBS changes weren’t significance in all groups. AEA decreased nonsignificantly in Groups E, C, and P (P ≥ 0.05) and there weren’t significant differences between 4 groups. Following the study Serum levels of α-tocopherol increased significantly in groups C, E and CE as compared with baseline (P ≤ 0.05, P ≤ 0.0001, P ≤ 0.0001, respectively) and in groups E and CE compared with group P (P ≤ 0.0001).
Figure 2 Levels of C-reactive protein (CRP) before and after three months vitamin E and conjugated linoleic acid (CLA) supplementation in patients with active rheumatoid arthritis.
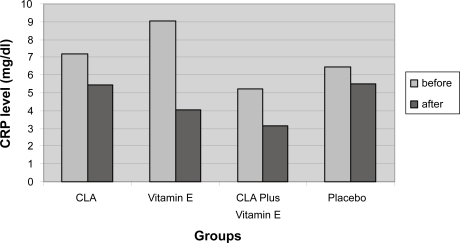
Table 3 Levels of blood pressure, oxidative stress variables in patients with active rheumatoid arthritis before and after three months supplementation (Mean ± SEM)
Table 4 Levels of FBS, inflammation variables and clinical tests in patients with active RA before and after three months supplementation (Mean ± SEM)
Discussion
In RA disease, chronic systemic inflammation may contribute to the higher incidence of CVD. (CitationHannawi et al 2007; Metosios et al 2007). To our knowledge; this is the first study which investigated the effects of CLAs and its combination with vitamin E in human with RA disease. In the present study, we didn’t find any significant changes in the plasma levels of TG and CHO in any of the studied groups. The magnitude of the change in plasma TG concentrations in our study is comparable with that achieved following 50:50 CLAs supplementation in CitationBenito and colleagues’ (2001) study. They did not find any effect on plasma CHO, lipoprotein, or TG. CitationNoone and colleagues’ (2002) study did not show any modification of plasma CHO and lipoprotein concentrations in healthy subjects receiving the 50:50 CLAs mix, however they showed a decrease in the TG concentration. Some other studies showed that trans-10, cis-12 CLAs isomer may be the effective hypotriglycerolemic isomer (CitationGavino et al 2000; CitationLin et al 2001). The TG-lowering effect of CLAs may be partly attributed to the effect of CLAs on PPARα. Furthermore, CLAs is a PPARγ ligand. In adipose tissue, PPARγ regulates the expression of the genes that determine adiposeness, lipid metabolism, and insulin sensitivity (CitationHouseknecht et al 1998; CitationLowell 1999). In our study, there was a nonsignificant decrease in CHO in CE group. We used CLAs capsules with equal proportions of the cis-9, trans-11 and trans-10, cis-12 CLAs in triglyceride form. Recent vitro and animal studies suggest that the individual CLAs isomers may have different effects on lipid metabolism. Human studies regarding CHO-related parameters were not in line with the animal ones. The inconsistency of results may arise from the effects of particular CLAs consists the usual CLAs mix. Moreover, the dose of CLAs mix administered to humans, on a weight basis, was much lower than that for animal models and may not reach the effective level (CitationSmedman and Vessby 2001). However, the effects of the CLAs-enriched dairy products on blood lipids were more complex. In our study similar to CitationDesroches and colleagues’ (2005) study after the consumption of the CLAs supplements, LDL-C, and HDL-C concentrations and the ratio of total to HDL-C and LDL-C to HDL-C were not significantly changed. CitationSinghal and colleagues (2001) study results in patients with coronary heart disease were similar to our study and plasma TG concentration didn’t change significantly in the vitamin E-supplemented group. A large base of epidemiological evidence suggests that a 1 mg/dl increment in HDL-C would be associated with a 2%–3% decrement in CVD risk (CitationGordon et al 1989). Our data show that supplementation of vitamin E and its combination with CLAs for at least three months can increase HDL-C nonsignificantly by 3.4% and 4.5%, respectively. Separate supplementation of CLAs did not improve serum HDL-C levels. The absence of such effects of CLAs supplementation should not be attributed to the interference of weight, physical activity, dietary intake or medication because these variables did not change during the study period and the patients had good compliance in taking their supplements. α-Tocopherol possibly has power to inhibit oxidation induced by α-tocopheroxyl radical (CitationSingh et al 2005) and known synergistic action between vitamin E and CLAs (CitationKim et al 2005; CitationSantoz-Zago et al 2007). Other studies have also shown that CLAs supplementation had no effect on HDL lipid composition (CitationNicolosi et al 1997; CitationGavino et al 2000; CitationStangl 2000). In our study, LDL-C increased in all groups and the difference between groups was not significant. CitationNicolosi and colleagues (2004) also showed that non-HDL-C concentrations were reduced by (9%–13%) in response to a CLAs-enriched diet. The LDL-C/HDL-C was reduced in the 80:20 CLAs group (27.2%) and the 50:50 CLAs group (22.4%), however these weren’t significant (CitationNicolosi et al 2004). In our investigation, CLAs plus vitamin E was more effective than CLAs in reducing LDL-C/HDL-C, CHO/HDL-C, and CRP. In the present study, FBS didn’t change significantly in four groups. Few studies have evaluated the effect of CLAs intake on glucose metabolism and insulin sensitivity in humans, but the available data suggest that glucose metabolism may be negatively affected by CLAs isomers. In CitationRiserus and colleagues’ (2001) study, a significant increase in glucose levels was observed in both the CLAs and the control group. The increase was more pronounced in the control group (CitationRiserus et al 2001). In another study, glucose levels increased significantly only in the group receiving the trans10 cis 12 isomer and not in the case of a mixture of different isomers. The increase was not significantly different from that which occurred in the control group (CitationRiserus et al 2002). CitationSmedman and Vessby (2001) also showed an increase (p = 0.054) in glycemia after the consumption of a mixture of CLAs isomers compared with the control group. They observed that insulin levels tended to increase after an intake of 2 g CLAs (CitationSmedman and Vessby 2001). Several other studies have shown no deleterious effects on glucose or insulin levels or sensitivity to insulin attributable to the consumption of different doses of CLAs enriched with cis 9, trans 11 or trans 10, cis 12 (CitationTricon et al 2004; CitationTaylor and Zahradka 2004). Results on the effect of CLAs on glucose metabolism from animal studies are controversial (CitationHouseknecht et al 1998; CitationStangl 2000; CitationTsuboyama-Kascoke et al 2000). In our study, CLAs supplementation had no effect on body weight. This finding is in agreement with CitationZambell and colleagues (2000) who found no changes in body weight or body composition attributed to CLAs supplementation in human subjects. However some studies have reported that CLAs supplementation (4 g/d) had no effect on body weight, but they significantly reduced body fat (CitationSmedman and Vessby 2001). It was shown that, 9c, 11t-CLAs, 10t, 12c-CLAs inhibited arachidonic acid and collagen-induced platelet aggregation (CitationTruitt et al 1999). In the present study, the number of platelet decreased nonsignificantly in groups P, C, and CE. CitationLeonard and colleagues (2007) studied the effects of vitamin E (400 IU) on cholesterol levels of hypercholesterolemic patients receiving statins. Similar to our study they didn’t find significant differences in any lipoprotein cholesterol fraction. However, unlike our study, a 6% decrease in HDL-C was detected within the vitamin E group (p < 0.05) (CitationLeonard et al 2007). Following our study, LDL-C increased in all groups, but this increase was only significant in group E. This increase may be due to increasing the size of LDL-C or changing its fractions or by increase of its half life by decreasing LDL-C oxidation. In our study AEA, decreased nonsignificantly in groups E, P, and C, but decreased as compared with baseline in group E (P = 0.068). We had nonsignificant differences between four groups. In one study on pedigreed baboons, dietary vitamin E improved LDL quality and showed two apparently paradoxical effects on HDL metabolism: lower HDL2 and higher apo A-I. These effects have contrasting associations with CVD risk and may help account for the mixed results from clinical trials of dietary vitamin E. In our study, nonsignificant reductions in serum AEA of lipoprotein-associated enzymes, Paraxonase1 (PON1) in all groups except group CE was shown. The presence of PON1 on HDL particles is considered to be a major source of protection from lipoprotein oxidation. Therefore, reduction of PON1 activity may be considered to be prooxidant and pro-atherogenic (CitationRainwater et al 2007). Our results contradict results in rabbits and humans (CitationJarvik et al 2002; CitationJeon et al 2005). This discrepancy may reflect differences in HDL size and composition properties among the species. Alternately, vitamin E-associated reduction in oxidative stress and oxidized LDL may have lowered the metabolic requirements for these enzymes to maintain the redox balance in the circulation. Then, through feedback mechanisms, the expression of PON1 may have been downregulated (CitationJarvik et al 2003; CitationRainwater et al 2005). Furthermore the differences between groups wasn’t significant. Following of our study ESR levels decreased significantly in the groups C, E, and CE compared with baseline (P ≤ 0.05 P ≤ 0.05, P ≤ 0.001, respectively). ESR levels in group CE was significantly lower than group P (P ≤ 0.05). Vitamin E was reported to regulate transcription or activity of a number of genes involved in oxidative stress, proliferation, inflammation, and apoptosis (CitationDutta and Dutta 2003; CitationTucker and Townsend 2005; CitationAldred et al 2006). CRP dropped in all groups in comparison with baseline, but this decreased more in group E (P = 0.054). The reduction of WBC in group CE was significant compare with other groups (P < 0.05) (). PLT count decreased nonsignificantly in CE, C, E groups. Consequently CLAs, vitamin E, and their combination are useful in reduction of inflammation. Many studies (CitationIgarashi and Miyazawa 2001; CitationKim et al 2002; CitationPalacios et al 2003; CitationBergamo et al 2006, Citation2007; CitationSantos-Zago et al 2007) show the antioxidant effect of CLAs, but several studies performed on healthy and obese subjects have observed that CLAs administration is associated with an increase in some lipid oxidation and peripheral inflammation parameters (CitationRiserus et al 2002, Citation2004; CitationBasu et al 2000a, Citation2000b; CitationSmedman et al 2004). One of our aims was to investigate the effect of α-tocopherol and CLAs supplementation on BP. BP levels decreased significantly in the group C by 8% and 8.5% for DBP and MAP respectively, after three months of supplementation as compared with baseline. The reduction of SBP in group C was significant compared with the groups E and P (). CitationHerrera and colleagues (2005) studied healthy primigravidas who had a family history of pre-eclampsia and diastolic notch. Calcium-CLAs supplementation reduced significantly the incidence of pregnancy-induced hypertension (PIH) in the study group (P = 0.01). Endothelial dysfunction was also significantly reduced after calcium-CLAs supplementation (CitationHerrera et al 2005). CitationHerrera and colleagues (2006) in another study on healthy primigravidas showed that the incidence of PIH was significantly reduced in women receiving the CLAs supplement compare with the placebo. There were no significant differences in the plasma concentrations of ionized calcium, PE2 (similar to our study), renin, angiotensin II, parathormone, and calcitonine (CitationHerrera et al 2006). In CitationRaff and colleagues’ (2006) investigation, a diet rich in CLAs didn’t alter SBP, DBP and pulse pressure, but this may be due to the short time for the study (five weeks). CitationNagao and colleagues (2003a) found that the relative mRNA expressions of angiotensinogen and leptin were suppressed by 10t, 12c-CLAs in adipose tissue. They speculate that the antihypertensive effect of 10t, 12c-CLAs can be attributed to the lowered secretion of hypertensive adipocytokines from abdominal adipose tissues (CitationNagao et al 2003a). In another study CitationNagao and colleagues (2003b) showed that CLAs suppresses the development of nonobese essential hypertension in spontaneously hypertensive rats (SHRs). They concluded that the antihypertensive effect of dietary CLAs may be due to the increase of plasma adiponectin level and is associated with the alleviation of membrane abnormality in SHRs (CitationNagao et al 2003b). In our study in group E, BP increased nonsignificantly, in CitationWard and colleagues’ (2007) study BP, pulse pressure, and heart rate increased versus placebo.
Conclusion
It is concluded that supplementation of 2 g CLAs 50:50 mix, and its combination with vitamin E for 12 weeks didn’t change lipid profiles and FBS significantly, but CLAs decreased BP and vitamin E decreased CRP so co-supplementation of CLAs and vitamin E could be helpful for heart disease prevention in RA patients.
Acknowledgments
This study was funded through a research grant from the Research Deputy of Tehran University of Medical Sciences (with Proposal No., 2804). We appreciate the kind cooperation of both Lipid Nutrition Company (Nederland) for providing CLAs capsules as Clarinol G-80, and Zahravi Company (Iran) for providing vitamin E pearls for this research.
References
- AldredSSozziTMudwayI2006Alpha tocopherol supplementation elevates plasma apolipoprotein A1 isoforms in normal healthy subjectsProteomics6169570316429457
- AliceVda CostaVViannaLM2005Effect of α-tocopherol supplementation on blood pressure and lipidic profile in streptozotocin-induced diabetes mellitus in spontaneously hypertensive ratsClinica Chimica Acta3511014
- Arbonés-MainarJMNavarroMAGuzmánMA2006Selective effect of conjugated linoleic acid isomers on atherosclerotic lesion development in apolipoprotein E knockout miceAtherosclerosis1893182716530768
- ArnettFEdworthySMBlochDA1988The American Rheumatoid Association 1987 revised criteria for the Classification of Rheumatoid ArthritisArthritis Rheum24315243358796
- BasuSSmedmanAVessbyB2000bConjugated linoleic acid induces lipid peroxidation in humansFEBS Lett46833610683436
- BasuSRiserusUTurpeinenA2000aConjugated linoleic acid induces lipid peroxidation in men with abdominal obesityClin Sci (Lond)99511611099394
- BeltonOADuffyAToomeyS2003Cyclooxygenase isoforms and platelet vessel wall interactions in the apolipoprotein E knockout mouse model of atherosclerosisCirculation10830172314638539
- BenitoPNelsonGJKelleyDS2001The effect of conjugated linoleic acid on plasma lipoproteins and tissue fatty acid composition in humansLipids362293611337977
- BergamoPLuongoDMauranoF2006Conjugated linoleic acid enhances glutathione synthesis and attenuates pathological signs in MRL/MpJ-Fas(lpr) miceJ Lipid Res4723829116877747
- BergamoPMauranoFRossiM2007Phase 2 enzyme induction by conjugated linoleic acid improves lupus-associated oxidative stressFree Radic Biol Med4371917561095
- BhattacharyaABanuJRahmanM2006Biological effects of conjugated linoleic acids in health and diseaseJ Nutr Biochem1778981016650752
- CywinskiJ1980The Essentials in Pressure MonitoringBoston, MAMartinus Nijhoff234
- De PabloPDietrichTKarlsonEW2007Antioxidants and other novel cardiovascular risk factors in subjects with rheumatoid arthritis in a large population sampleArthritis Rheum57Suppl 29536217665477
- DesrochesSChouinardPYGaliboisI2005Lack of effect of dietary conjugated linoleic acids naturally incorporated into butter on the blood lipid profile and body composition of overweight and obese menAm J Clin Nutr823091916087973
- DesseinPHStanwixAEJoffeBI2002Cardiovascular risk in rheumatoid arthritis versus osteoarthritis: acute phase response related decreased insulin sensitivity and high-density lipoprotein cholesterol as well as clustering of metabolic syndrome features in rheumatoid arthritisArthritis Res4Suppl 5R512223108
- DuttaADuttaSK2003Vitamin E and its role in the prevention of atherosclerosis and carcinogenesis: a reviewJ Am Coll Nutr22Suppl 42586812897039
- GavinoVCGavinoGLeblancMJ2000An isomeric mixture of conjugated linoleic acids but not pure cis-9, trans-11-octadecadienoic acid affects body weight gain and plasma lipids in hamstersJ Nutr13027910613761
- GeyKFMoserUKJordanP1993Increased risk of cardiovascular disease at suboptimal plasma concentrations of essential antioxidants: an epidemiological update with special attention to carotene and vitamin CAm J Clin Nutr57787S97S8475897
- GordonDJProbstfieldJLGarrisonRJ1989High-density lipoprotein cholesterol and cardiovascular disease: four prospective American studiesCirculation798152642759
- HannawiSHaluskaBMarwickTH2007Atherosclerotic disease is increased in recent-onset rheumatoid arthritis: a critical role for inflammationArthritis Res Ther9R11617986352
- HerreraJAArévalo-HerreraMShahabuddinAK2006Calcium and conjugated linoleic acid reduces pregnancy-induced hypertension and decreases intracellular calcium in lymphocytesAm J Hypertens9Suppl 43817J16580574
- HerreraJAShahabuddinGAKMErshengC2005Calcium plus linoleic acid therapy for pregnancy-induced hypertensionInt J Gynaecol Obstet91221716243339
- HouseknechtKLVandenJPMoya-CamarenaS1998Dietary conjugated linoleic acid normalises impared glucose tolerance in the Zucker diabetic fatty fa/fa ratBiochem Biophys Res Commun244678829535724
- IgarashiMMiyazawaT2001The growth inhibitory effect of conjugated linoleic acid on a human hepatoma cell line, HepG2, is induced by a change in fatty acid metabolism, but not the facilitation of lipid peroxidation in the cellsBiochim Biophys Acta15301627111239819
- JarvikGPHatsukamiTSCarlsonC2003Paraoxonase activity, but not haplotype utilizing the linkage disequilibrium structure, predicts vascular diseaseArterioscler Thromb Vasc Biol2314657112805074
- JarvikGPTsaiNTMcKinstryLA2002Vitamin C and E intake is associated with increased paraoxonase activityArterioscler Thromb Vasc Biol2213293312171796
- JeonS-MParkYBKwonO-S2005Vitamin E supplementation alters HDL-cholesterol concentration and paraoxonase activity in rabbits fed high-cholesterol diet: comparison with probucolJ Biochem Mol Toxicol193364616292755
- KellyGS2001Conjugated linoleic acid: A reviewAltern Med Rev63678211578253
- KimHKKimSRAhnJY2002Differential responses of hamsters and rats fed hig-fat or low-fat diets supplemented with conjugated linoleic acidNutr Res2271522
- KimHKKimSRAhnJY2005Dietary conjugated linoleic acid reduces lipid peroxidation by increasing oxidative stability in ratsJ Nutr Sci Vitaminol51Suppl 181515915662
- KönigBSpielmannJHaaseK2008Effects of fish oil and conjugated linoleic acids on expression of target genes of PPARa and sterol regulatory element-binding proteins in the liver of laying hensBr J Nutr2119
- KritchevskyDTepperSAWrightS2000Influence of conjugated linoleic acid (CLAs) on establishment and progression of atherosclerosis in rabbitsJ Am Coll Nutr19472S7S10963467
- KushiLHFolsomARPrineasRJ1996Dietary antioxidant vitamins and death from coronary heart disease in postmenopausal womenN Engl J Med3341156628602181
- LeeKNKritchevskyDParizaMW1994Conjugated linoleic acid and therosclerosis in rabbitsAtherosclerosis10819257980704
- LeonardSWJossJDMustacichDJ2007Effects of vitamin E on cholesterol levels of hypercholesterolemic patients receiving statinsAm J Health Syst Pharm64Suppl 2122576617959578
- LinYSchuurbiersEvan der VeenS2001Conjugated linoleic acid isomers have differential effects on triglyceride secretion in HepG2 cellsBiochim Biophys Acta1533384611514234
- LowellBB1999PPAR gamma: an essential regulator of adipogenesis and modulator of fat cell functionCell992394210555139
- MetsiosGSStavropoulos-KalinoglouJAGVeldhuijzen van ZantenJ2008Rheumatoid arthritis, cardiovascular disease and physical exercise: a systematic reviewRheumatology47Suppl 32394818045810
- MiyazakiMKimYCGray-KellerMP2000The biosynthesis of hepatic cholesterol esters and triglycerides is impaired in mice with a disruption of the gene for stearoyl-CoA desaturase 1J Biol Chem27530132810899171
- MiyazakiMKimYCNtambiJM2001A lipogenic diet in mice with a disruption of the stearoyl-CoA desaturase 1 gene reveals a stringent requirement of endogenous monounsaturated fatty acids for triglyceride synthesisJ Lipid Res4210182411441127
- Moya-CamarenaSYBeluryMA1999aSpecies differences in the metabolism and regulation of gene expression by conjugated linoleic acidNutr Rev573364010628184
- Moya-CamarenaSYBeluryMA1999bCLAs and PPARgamma activationJ Nutr1296026
- Moya-CarmarenaSYVanden-HeuvelJPBlanchardSG1999Conjugated linoleic acid is a potent naturally occuring ligand and activator of PPARaJ Lipid Res4014263310428978
- MundayJSThompsonKGJamesKAC1999Dietary conjugated linoleic acids promote fatty streak formation in the C57BL/6 mouse atherosclerosis modelBr J Nutr81251510434852
- NagaoKInoueNWangYM2003aThe 10 trans, 12 cis isomer of conjugated linoleic acid suppresses the development of hypertension in Otsuka Long–Evans Tokushima fatty ratsBiochem Biophys Res Commun306134812788078
- NagaoKInoueNWangYM2003bConjugated linoleic acid enhances plasma adiponectin level and alleviates hyperinsulinemia and hypertension in Zucker diabetic fatty (fa/fa) ratsBiochem Biophys Res Commun310562614521947
- NicolosiRJRogersEJKritchevskyD1997Dietary conjugated linoleic acid reduces plasma lipoproteins and early aortic atherosclerosis in hypercholesterolemic hamstersArtery22266779209699
- NicolosiRJWoolfreyBWilsonTA2004Decreased aortic early atherosclerosis and associated risk factors in hypercholesterolemic hamsters fed a high- or mid-oleic acid oil compared to a high-linoleic acid oilJ Nutr Biochem15540715350986
- NooneEJRocheHMNugentAP2002The effect of dietary supplementation using isomeric blends of conjugated linoleic acid on lipid metabolism in healthy human subjectsBr J Nutr882435112207834
- NtambiJM1999Regulation of stearoyl-CoA desaturase by polyunsaturated fatty acids and cholesterolJ Lipid Res4015495810484602
- NurmohamedMT2007Atherogenic lipid profiles and its management in patients with rheumatoid arthritisVasc Health Risk Manag3Suppl 68455218200805
- PaiJTGuryevOBrownMS1998Differential stimulation of cholesterol and unsaturated fatty acid biosynthesis in cells expressing individual nuclear sterol regulatory element-binding proteinsJ Biol Chem27326138489748295
- PalaciosAPiergiacomiVCataláA2003Antioxidant effect of conjugated linoleic acid and vitamin A during non enzymatic lipid peroxidation of rat liver microsomes and mitochondriaMol Cell Biochem250Suppl 1–21071312962148
- ParodiPW1999Conjugated linoleic acid and other anticarcinogenic agents of bovine milk fatJ Dairy Sci8213394910386321
- PeckLWNickelKPBeluryMA1998Dietary conjugated linoleic acid normalizes impaired glucose tolerance in Zucker diabetic fatty fa/fa ratBiochem Biophys Res Commun244678829535724
- RaffMTholstrupTSejrsenK2006Diets rich in conjugated linoleic acid and vaccenic acid have no effect on blood pressure and isobaric arterial elasticity in healthy young menJ Nutr136Suppl 4992716549463
- RainwaterDLMahaneyMCVandeBergJL2007Vitamin E dietary supplementation significantly affects multiple risk factors for cardiovascular disease in baboonsAm J Clin Nutr8659760317823422
- RimmEBStampferMJAscherioA1993Vitamin E consumption and the risk of coronary heart disease in menNew Engl J Med328145068479464
- RiserusUArnerPBrismarK2002Treatment with dietary trans10 cis12 conjugated linoleic acid causes isomer-specific insulin resistance in obese men with the metabolic syndromeDiabetes Care2515162112196420
- RiserusUBerglundLVessbyB2001Conjugated linoleic acid (CLAs) reduced abdominal adipose tissue in obese middle-aged men with signs of the metabolic syndrome: a randomised controlled trialInt J Obes Relat Metab Disord2511293511477497
- RocheHMNooneESewterC2002Isomer-dependent metabolic effects of conjugated linoleic acid: insights from molecular markers sterol regulatory element-binding protein-1c and LXRalphaDiabetes5120374412086931
- RudelLL1999Atherosclerosis and conjugated linoleic acidBr J Nutr81177910434843
- Santos-ZagoLFBotelhoAPde OliveiraAC2007Supplementation with commercial mixtures of conjugated linoleic acid in association with vitamin E and the process of lipid autoxidation in ratsLipids428455417665238
- SanzDCSanta-CruzMC1986Simultaneous measurement of retinol and α-tocopherol in human serum by high-performance liquid chromatography with ultraviolet detectionJ Chromatogr38014043745376
- SchwenkeDCBehrSR1998Vitamin E combined with selenium inhibits atherosclerosis in hypercholesterolemic rabbits independently of effects on plasma cholesterol concentrationsCirc Res83366779721693
- SinghAUDevarajSJialalI2005Vitamin E, oxidative stress, and inflammationAnnu Rev Nutr251517416011463
- SinghalSGuptaRGoyleA2001Comparison of antioxidant efficacy of vitamin E, vitamin C, vitamin A and fruits in coronary heart disease: a controlled trialJ Assoc Physicians India493273111291971
- SmedmanAVessbyBBasuS2004Isomer-specific effects of conjugated linoleic acid on lipid peroxidation in humans: regulation by alpha-tocopherol and cyclo-oxygenase-2 inhibitorClin Sci (Lond)106677312956622
- SmedmanAVessbyB2001Conjugated linoleic acid supplementation in humans metabolic effectsLipids36773811592727
- StampferMJHennekensCHMansonJE1993Vitamin E consumption and the risk of coronary disease in womenNew Engl J Med328144498479463
- StanglGI2000High dietary levels of conjugated linoleic acid mixture alter hepatic glycerophospholipid CLAss profile and cholesterol-carrying serum lipoproteins of ratsJ Nutr Biochem111849110827340
- StephensNGParsonsASchofieldPM1996Randomised controlled trial of vitamin E in patients with coronary disease: Cambridge Heart Antioxidant Study (CHAOS)Lancet34778168622332
- TaylorCGZahradkaP2004Dietary conjugated linoleic acid and insulin sensitivity and resistance in rodent modelsAm J Clin Nutr79Suppl 61164S1168S15159252
- ToomeySRocheHFitzgeraldD2003Regression of pre established atherosclerosis in the apoE−/− mouse by conjugated linoleic acidBiochem Soc Trans311075914505483
- TriconSBurdgeGCKewS2004Opposing effects of cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid on blood lipids in healthy humansAm J Clin Nutr806142015321800
- TruittAMcNeillGVanderhoekJY1999Antiplatelet effects of conjugated linoleic acid isomersBiochim Biophys Acta14382394610320806
- Tsuboyama-KasaokaNTakahashiMTanemuraK2000Conjugated linoleic acid supplementation reduces adipose tissue by apoptosis and develops lipodystrophy in miceDiabetes49Suppl 915344210969838
- TuckerJMTownsendDM2005Alpha-tocopherol: roles in prevention and therapy of human diseaseBiomed Pharmacother59380716081238
- ValeilleKFérézouJAmslerG2005A cis-9, trans-11-conjugated linoleic acid-rich oil reduces the outcome of atherogenic process in hyperlipidemic hamsterAm J Physiol Heart Circ Physiol289H652H65915778275
- WardNCWuJHClarkeMW2007The effect of vitamin E on blood pressure in individuals with type 2 diabetes: a randomized, double-blind, placebo-controlled trialJ Hypertens252273417143195
- WilsonTANicolosiRJChrysamM2000Conjugated linoleic acid reduces early aortic atherosclerosis greater than linoleic acid in hypercholesterolemic hamstersNutr Res201795805
- ZambellKLKeimNLVan LoanMD2000Conjugated linoleic acid supplementation in humans: effects on body composition and energy expenditureLipids357778210941879