To the Editor,
Following mastectomy for breast cancer approximately 5–10% of the patients will have a chest wall or regional nodal recurrence within 10 years. Between 50% and 70% of recurrences are located in the chest wall [Citation1].
In a multidisciplinary approach, oncological treatment may go hand in hand with surgery whenever possible. Thus, wide local excision may be an option for solitary or multiple nodules that are amenable to resection [Citation1].
Reconstruction of full-thickness chest wall defects is a significant therapeutic and technical challenge. The chest wall can be reconstructed using autologous tissue as local or distant tissue flaps. When semirigid reconstruction is indicated, synthetic meshes e.g. polypropylene (Marlex [Citation2], Prolene [Citation3]), expanded polytetrafluoroethylene (Gore-Tex [Citation2,Citation3]) or biological meshes (e.g. Alloderm™, Strattice™ [Citation4,Citation5] or Permacol™ [Citation6]) has been used in combination with soft tissue coverage of the mesh.
One of the biological meshes “Strattice” is derived from porcine dermis. The dermis is processed in order to eliminate all cellular elements from the tissue leaving only the intact extracellular matrix. The matrix provides a scaffold, promoting integration of the connective tissue and thus being revascularized and repopulated by the patient's own cells.
Patient
A 69-year-old female presented with local recurrent breast cancer for the fourth time on the left hemithorax. The patient was 34 years earlier diagnosed with a left-sided breast cancer. She underwent mastectomy and axillary dissection. In the following 28 years the patient experienced local recurrences three times. The first two recurrences were treated with surgical excision and direct suture followed by radiation therapy and endocrine therapy. The third recurrence required extensive surgery with full- thickness chest wall resection, including resection of the third, fourth and fifth rib. The thoracic wall was reconstructed using polypropylene mesh, an abdominal fasciocutaneous rotation flap and a partial ipsilateral latissimus dorsi flap. Postoperatively, she received endocrine therapy. The prolene mesh extruded during the postoperative period and caused intermittent ulceration, which resulted in discomfort to the patient.
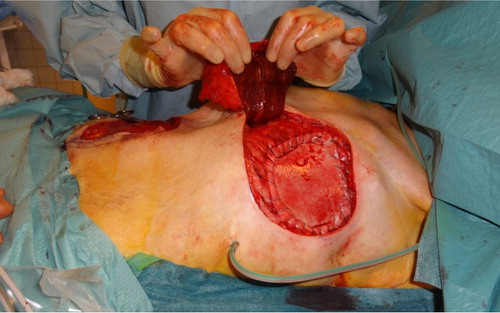
The present fourth time recurrence () required full-thickness chest wall resection and the chest wall defect measured 10 × 16 cm. The bony thorax reconstruction and stabilization was accomplished using Strattice Tissue Matrix Firm™ which was sutured to the chest wall. A contralateral TRAM (transverse rectus abdominus myocutaneous) flap was transposed to cover the biologic mesh with muscle, subcutaneous tissue and overlying skin (). The patient had an uneventful postoperative course and was discharged five days after surgery. Pathological examination showed positive tissue margins and 18 days later the patient underwent re-resection. At that time, adherence between the TRAM flap and the biologic mesh was observed. Additional soft tissue was resected, guided by frozen section pathological examination, and the TRAM flap could be used to fully cover the soft tissue defect again. Eighty days after the chest wall reconstruction the TRAM flap and biologic mesh was perfectly integrated (). Shortly after surgery the patient resumed all her daily activities.
Figure 1. (A) Fourth-time local recurrent breast cancer to the left hemithorax. (B) 80 days after full-thickness chest wall reconstruction.
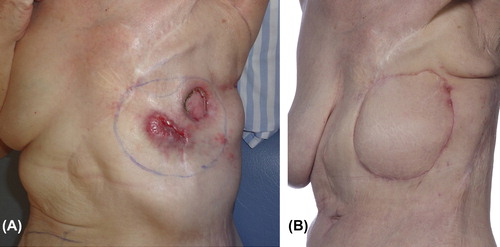
Figure 2. Strattice Tissue Matrix Firm™ sutured to the chest wall. Soft tissue defect covered with a contralateral TRAM flap.
The patient is followed up by the Departments of Oncology and Plastic Surgery and a computerized tomography scan is scheduled six months after surgery.
Discussion
The aim of chest wall resection is to achieve palliation for the patient, and most importantly, if possibly, to achieve a radical resection leading to local control of the cancer recurrence and a hopefully prolonged survival. Even though reconstruction of the chest wall is often very challenging, resection of locally recurrent breast cancer has shown a low morbidity and mortality rate [Citation3,Citation7].
In this present case of full-thickness chest wall reconstruction we chose Strattice™, a non-cross-linked, porcine acellular dermal matrix product, to act as a support for the pedicled TRAM flap. Once implanted, the biological mesh is infiltrated by host cells, revascularized, and remodeled into autologous tissue. This potentially improves the ability to resist infection and the mesh has successfully been used to repair infected or contaminated incisional hernias [Citation8]. This patient had skin ulceration and therefore the operation field may have been contaminated. Furthermore, the patient had previously received radiotherapy, which compromises the wound healing response, inhibits revascularization, and increases the rates of infection and dehiscence of surgical wounds [Citation9].
In this case, palliation was achieved and the chest wall integrity reestablished in a successful way using a biological mesh to provide structural support. Another way to possibly achieve palliation of cutaneous recurrent breast cancer is electrochemotherapy as shown Matthiessen et al. in a phase II study [Citation10]. Treatment was generally well tolerated and several patients reported having reduced oozing and bleeding. This is a promising method for treatment of relatively superficial recurrences, to be further investigated.
A multidisciplinary team including oncological surgeons, oncologists and plastic surgeons is needed when treating patients with recurrent cancer. Surgery – even using advanced techniques – can be offered to patients with recurrent breast cancer and the patient can in some cases be treated with extensive surgery more than once. It is worthwhile and feasible to offer these patients operation and hopefully achieve radical eradication of the malignancy, alternatively palliation, as surgery could be performed with a limited degree of morbidity.
Acknowledgements
The authors wish to acknowledge Lone Fischer, MD, Breast and Endocrine Unit, Aarhus University Hospital, Denmark and Hans K. Pilegaard, MD, Department of Cardiothoracic Surgery, Aarhus University Hospital, Denmark.
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Management of locoregional recurrence of breast cancer after mastectomy. [cited 2012 May 6]. Available from: http://www.uptodate.com/contents/management-of- locoregional-recurrence-of-breast-cancer-after-mastectomy.
- Pameijer CR, Smith D, McCahill LE, Bimston DN, Wagman LD, Ellenhorn JD. Full-thickness chest wall resection for recurrent breast carcinoma: An institutional review and meta-analysis. Am Surg 2005;71:711–5.
- Arnold PG, Pairolero PC. Chest-wall reconstruction: An account of 500 consecutive patients. Plast Reconstr Surg 1996;98:804–10.
- Kaplan KM, Chopra K, Feiner J, Gastman BR. Chest wall reconstruction with strattice in an immunosuppressed patient. Eplasty 2011;11:e46.
- Huston TL, Taback B, Rohde CH. Chest wall reconstruction with porcine acellular dermal matrix (strattice) and a latissimus myocutaneous flap. Am Surg 2011;77:e115–6.
- Mirzabeigi MN, Moore JH Jr, Tuma GA. The use of Permacol® for chest wall reconstruction in a case of desmoid tumour resection. J Plast Reconstr Aesthet Surg 2011;64:406–8.
- Bosc R, Lepage C, Hamou C, Matar N, Benjoar MD, Hivelin M, et al. Management of chest wall reconstruction after resection for cancer: A retrospective study of 22 consecutive patients. Ann Plast Surg 2011;67:263–8.
- Strattice RICH Study/KCI. [cited 2012 May 19]. Available from: http://www.kci-medical.ie/IE-ENG/richstudy.
- Butler CE, Langstein HN, Kronowitz SJ. Pelvic, abdominal, and chest wall reconstruction with AlloDerm in patients at increased risk for mesh-related complications. Plast Reconstr Surg 2005;116:1263–75; Discussion 1276–7.
- Matthiessen LW, Johannesen HH, Hendel HW, Moss T, Kamby C, Gehl J. Electrochemotherapy for large cutaneous recurrence of breast cancer: A phase II clinical trial. Acta Oncol 2012;51:713–21.