Abstract
Background. Almost the same pathophysiological mechanism has been suggested for both atherosclerosis and calcific aortic stenosis (AS). In this study, we examined any association between ascending aortic pressure-derived indices and hemodynamic characteristics of calcific AS. Methods. A total of 90 patients were studied (26 males, 64 females; mean age: 64.4 ± 11.3 years). The study population consisted of two groups: AS and a control group. Both groups were well matched. Ascending aortic pressure-derived indices were obtained from all patients via catheterization. Results. Two groups were well matched according to demographic characteristics. Aortic pulse/FPPs (fractional pulse pressure = aortic pulse pressure/ aortic mean pressure), and PI (pulsatility index = aortic pulse pressure/aortic diastolic pressure) were significantly higher in patients with AS than in those without. Mean aortic mean gradient had significant positive correlation with aortic diastolic pressure, FPP and PI. The multiple-adjusted odds ratios of the risk of AS was 4.51 (95% CI 1.63–12.48) and 4.34 (95% CI 1.59–11.88) for the higher aortic FPP and PI levels compared with lower levels, respectively. Conclusion. Ascending aortic pressure-derived indices were significantly and independently associated with the presence and severity of calcific AS. This confirms the participation of blood pressure in the pathogenesis of AS.
Introduction
Aortic valve disease, especially aortic stenosis (AS), is the most frequently seen valvular heart disease in the western world (Citation1). Its prevalence increases with age. After 85 years old, the prevalence has been reported as high as 4% by echocardiography (Citation2). Although some recent studies have been tried to demonstrate any relation between AS and atherosclerosis, the presence of such association is still unknown.
Invasively measured blood pressure-derived indices through catheterization give more reliable and accurate values than indirect measurements through a sphygmomanometer. Ascending aortic fractional pulse pressure (FPP) and pulsatility index (PI) are the parameters that most extensively studied. These indices have been studied and significant associations have been demonstrated in various cardiovascular pathologies (Citation3–9). No study, however, has examined the association between ascending aortic FPP/PI and the presence and severity of calcific AS.
The aim of the present study was to assess aortic FPP/PI in subjects with AS, compared with well-matched controls.
Methods
Study population
A total of 45 patients with AS diagnosed by echocardiography were included in the study. As the control group, 45 subjects without AS were recruited for the study. All patients (26 males, 64 females; mean age 64.4±11.3 years; range 43–81 years) underwent selective coronary angiography. The measurement of ascending aortic blood pressures was performed during cardiac catheterization. Two consecutive weekly clinic visits during each of which blood pressure measurements were obtained for all patients. Hypertension was diagnosed if the average of the three blood pressure measurements at the two clinic visits was consistently elevated over 140 systolic and/ or 90 diastolic. In addition, previously diagnosed and treated hypertensive states were accepted as hypertension, although measured blood pressures were lower than 140 systolic and/or 90 diastolic, and diabetes mellitus was defined as having fasting blood glucose >126 mg/dl on two occasions or being on treatment. All patients were questioned for any cardiovascular drug use, and underwent transthoracic echocardiography for the measurement of maximum and mean aortic valve gradients. Exclusion criteria included the following: known coronary artery disease; left ventricular dysfunction (left ventricular ejection fraction <50%); unstable ischemic conditions (unstable angina pectoris and myocardial infarction); valvular heart disease other than AS; and detection of coronary atherosclerotic lesion and/or coronary slow flow (CSF) after selective coronary artery angiography.
Echocardiography
Standard continuous-wave Doppler-derived gradient values of maximum and mean at the aortic valve level in apical five-chamber view were used (GE Medical Systems, Vivid 7 Dimension, Horten, Norway). The continuous-wave Doppler cursor was placed parallel to flow direction with no more than 20° angle. All echocardiograms were performed by an expert cardiologist blinded to the study.
Cardiac catheterization
All patients in the study underwent selective coronary artery angiography after appropriate patient preparation. Femoral artery cannulation was used for the arterial access site and a Judkins system was applied for cannulating the left and right coronary arteries. All angiograms were evaluated by two experienced physicians blinded to the study. Angiograms without stenotic lesion in all major epicardial coronary arteries including left main (LM), left anterior descending (LAD), left circumflex (LCX) and right coronary arteries (RCAs) were considered normal angiograms. In addition, CSF was investigated by using thrombosis in myocardial infarction (TIMI) frame count method described by Gibson et al. (Citation10). Previously published normal TIMI frame counts were 21.1 ±1.5 frames for LAD (after correction), 22.2±4.1 frames for LCX and 20.4±3.0 frames for RCA (Citation10). For a given artery, any value above this published range was considered CSF and accepted as an exclusion criterion.
Measurement of hemodynamic parameters
Hemodynamic measurements including systolic and diastolic blood pressures were assessed using the pigtail system during cardiac catheterization for each patient. The average of at least five pressure waveforms on the paper with a speed of 25 mm/s was used for analysis by a physician who was blinded to the study.
Calculation of hemodynamic parameters
Aortic pulse pressure = aortic systolic pressure – aortic diastolic pressure;
Aortic mean pressure =1/3 aortic systolic pressure + 2/3 aortic diastolic pressure;
Aortic fractional pulse pressure = aortic pulse pressure/aortic mean pressure; and
Aortic pulsatility index = aortic pulse pressure/ aortic diastolic pressure.
Anthropometric measurement
Height and weight of patients were measured and body mass index (BMI) was calculated by dividing weight in kilograms by height in meters squared, and described as kg/m2.
Statistical analysis
Data were analyzed with the SPSS software version 15.0 for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables were presented as mean±SD and categorical variables as frequency and percentage. Student's t -test was used to compare normally distributed continuous variables and the Mann–Whitney U test for variables without normal distribution. The χ2 test was used to compare categorical variables. Any correlation between aortic pressure-derived indices and aortic gradients was analyzed by the linear regression model. A two-tailed p -value of <0.05 was considered statistically significant. Multiple logistic regression analysis was used to evaluate the independent associates of the risk of AS. Parameters with a p -value of less than 0.1 in univariate analysis were included in the model. The odds ratios and 95% confidence intervals (CI) were calculated.
Results
Baseline characteristics
Baseline demographic characteristics of patients in both groups were outlined in .
Table I. Baseline demographic characteristics of study participants.
Twenty-two percent of patients were male with a mean age of 71.0 ±6.0 years and 78% of the cases were female with a mean age of 64.0±15.7 years in the AS group; 36% of patients were male and 64% were female with a mean age of 65.4±7.9 and 62.0 ±6.5 years in the control group, respectively.
No significant differences between the two groups concerning age, sex and other cardiovascular risk factors, including hypertension, hypercholesterolemia, diabetes mellitus and smoking were observed. In addition, both groups did not significantly differ in BMI, heart rate and use of any cardiovascular drug except calcium-channel blockers. Therefore, the two groups were well matched.
Hemodynamic parameters
The hemodynamic characteristics of patients in both groups were outlined in .
Table II. Ascending aortic pressure-derived indices of study participants.
Hemodynamic parameters including aortic diastolic pressure measured during catheterization and calculated aortic mean pressure were not different between the two groups. Measured aortic systolic, calculated aortic pulse/FPPs and PI were, however, significantly higher in patients with AS than in those without.
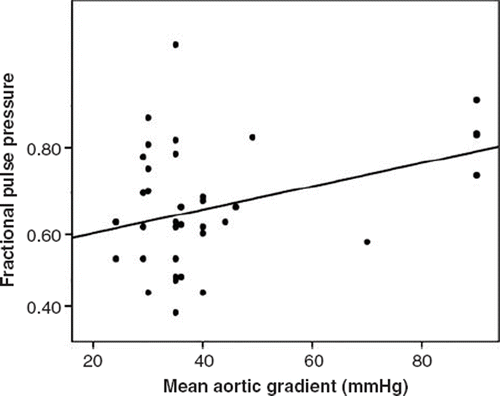
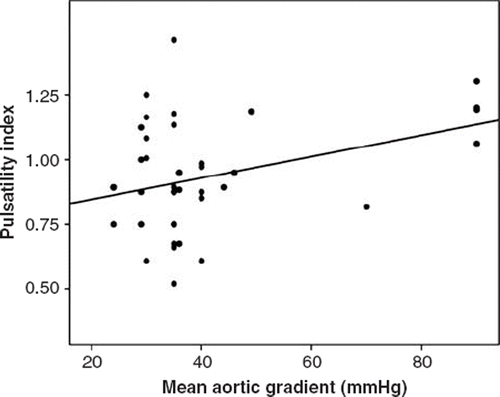
There was no significant correlation between mean aortic maximum gradient and ascending aortic pressure-derived indices including systolic, diastolic, pulse, mean, FPP or PI. Mean aortic mean gradient had, however, significant positive correlation with aortic diastolic pressure, FPP and PI ( and ). No significant correlation was detected between mean aortic mean gradient and systolic, pulse and mean pressures ().
Table III. Correlation parameters between ascending aortic pressure-derived indices and aortic gradients.
In addition, there was no significant gender influence on the aortic FPP and PI–AS relationship in our study: women 0.64±0.13; men 0.62±0.12, p = 0.716 for aortic FPP; women 0.83 ±0.20; men 0.80±0.20, p = 0.693 for aortic PI.
Multivariate analysis of the presence of AS
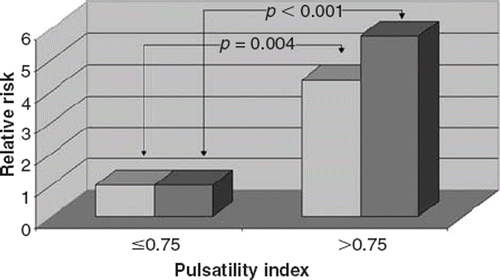
Two cut-off points of 0.60 and 0.75 were determined by rank cases analysis for aortic FPP and PI, respectively. The higher aortic FPP and PI were significantly associated with the risk of AS such that 68.9% and 71.1% of patients in the AS group had higher aortic FPP and PI, respectively, whereas 28.9% and 31.1% of patients in the control group had a higher aortic FPP and PI, respectively (p<0.001 and p<0.001, respectively). Having higher aortic FPP (>0.60) increased the risk of AS 5.45-fold (95% CI 2.21–13.44). In addition, having higher aortic PI (>0.75) increased the risk of AS 5.75-fold (95% CI 2.29–14.39). After adjustment for age, sex, BMI, the presence of hypertension, hypercholesterolemia, diabetes mellitus, smoking, heart rate and the current use of medications, the multiple-adjusted odds ratios of the risk of AS was 4.51 (95% CI 1.63–12.48) and 4.34 (95% CI 1.59–11.88) for the higher aortic FPP and PI levels compared with lower levels, respectively ( and ).
Figure 3. Relative risks of calcific aortic stenosis (AS) according to cut-off value of 0.60 of aortic fractional pulse pressure. The reference group was subjects with an aortic fractional pulse pressure of ≤0.60. Adjustments were made for age, sex, hypertension, hypercholesterolemia, diabetes, smoking, body mass index, heart rate and medication use. Darker bars indicate the results of univariate analysis and the others indicate the results of multivariate analysis.
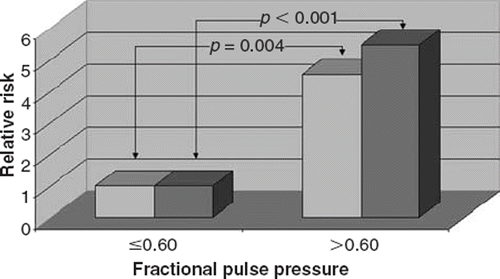
Figure 4. Relative risks of calcific aortic stenosis (AS) according to cut-off value of 0.75 of aortic pulsatility index. The reference group was subjects with an aortic pulsatility index of ≤0.75. Adjustments were made for age, sex, hypertension, hypercholesterolemia, diabetes, smoking, body mass index, heart rate and medication use. Darker bars indicate the results of univariate analysis and the others indicate the results of multivariate analysis.
In addition, each increase in aortic FPP/PI was significantly associated with calcific AS ().
Table IV. Regression analysis of fractional pulse pressure and pulsatility index.
Discussion
In our study, we found for the first time that hemo-dynamic indices obtained from ascending aorta invasively were significantly associated with the presence and severity of AS in patients with calcific AS. In addition, these significant and positive correlations were independent of risk factors for coronary artery disease.
To date, many investigations have been performed to answer the questions ‘Is there any relationship between atherosclerosis and calcific AS? If there is, which mechanisms play the role?’ In the majority of these studies, the same pathophysiological mechanism has been suggested in both calcific AS and atherosclerosis. In addition, the same treatment modalities such as statin drugs have been used to prevent progression of stenosis (Citation11). Mechanistically, after disruption of the basement membrane, lipids and lipoproteins accumulate in subendothelial region. Then inflammatory cells migrate to the region to start both local and systemic inflammation. This is an active atheroinflammatory process. The main difference from atherosclerosis in the vascular tissue is much more calcification of the aortic valves and adjacent structures (Citation12).
Increased PP has been shown to be independently associated with adverse cardiovascular events in large cohorts of population (Citation13,Citation14). Although these studies have included male subjects largely, it has been shown that increased aortic pulse pressure (PP) and FPPs are significantly associated with the presence of angiographic coronary artery disease in women (Citation3). Although measured brachial PP has been shown to be significantly associated with the risk of coronary heart disease (Citation14), invasively measured aortic PP and evaluated FPP via catheterization gives more accurate and reliable values compared with sphygmomanometer-measured brachial blood pressure. PP also increases from central to peripheral arteries. The reliability of derived aortic BP is crucially dependent on the validity of the transfer function used to generate the central aortic waveforms. Increased aortic PP is associated with increased aortic stiffness. In addition, variations in PP are commonly a consequence of variations in the mean pressure. Increased PP may accordingly be related to distension of the aortic wall, without changes in the wall structure. It may be possible to correct for this by calculating the FPP and PI. Therefore, accurate FPP/PI measures aortic stiffness. Atherosclerosis produces many pathological changes in the arterial wall, one of the most important being a progressive increase in arterial stiffening although hypertension may also increase aortic stiffness. When the aortic wall has stiffened because of atherosclerotic process, systolic blood pressure increases and the diastolic blood pressure decreases relative to mean pressure because of shortened time constant of diastolic blood pressure decay, resulting in increased aortic PP. Therefore, central aortic PP is a more reliable and accurate indicator of aortic stiffness. Aortic FPP has also been shown to be strongly associated with angiographic coronary heart disease in men and women (Citation3,Citation15). PI has been found to be associated with the extent of coronary artery disease (Citation8). Because aortic stiffness has been shown to be related to coronary atherosclerosis (Citation16), aortic PP is more strongly associated with coronary atherosclerosis than the peripheral PP. In addition, other than aortic PP, it has been also shown that FPP is independently associated with coronary atherosclerosis (Citation3,Citation15). In the present study, we studied patients with normal coronary angiograms. Therefore, which mechanism is responsible for the significant increase in aortic PP and FPP/PI in patients with calcific AS compared with control subjects? The endothelium of the vascular tissue has many functions for the continuation of the normal vascular physiology. It plays a key role in maintaining vascular homeostasis through balancing endothelial-derived relaxing and contracting factors. The role of the endothelium in controlling the vascular tone, especially vasodilatation, has been shown via the endothelial-derived relaxing factor, which was later named as nitric oxide (NO) (Citation17,Citation18). Endothelial dysfunction occurs when NO activity in the vascular tissue is decreased, resulting in decreased endothelium-dependent vasodilatation. McEniery et al. (Citation19) have showed that endothelial function was inversely associated with aortic pulse wave velocity and augmentation index in a large cohort of healthy volunteers. In their study, both endothelium-dependent and endothelium-independent vasodilatations were evaluated through the change in augmentation index. This association was also more prominent with central PP compared with brachial PP (Citation19).
Study limitations
The major limitation of this study is the limited sample size, which means that the results have to be confirmed in a larger study. In addition, this is a clinical study. Therefore, at the molecular level, endothelial function/dysfunction might be evaluated and results might be combined with clinical factors.
Conclusion
We have demonstrated that aortic PP and FPP/PI, indicators of the risk of coronary artery disease, in patients with calcific AS was significantly elevated compared with control subjects. Aortic FPP and PI were significantly correlated with mean aortic gradient. In addition, these significant and positive correlations were independent of risk factors for coronary artery disease. Prospective studies with large sample sizes are needed for prognostic evaluation of increased aortic PP and FPPs in patients with calcific AS in real-life practice on long-term follow-up.
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Iung B, Baron G, Butchart EG, Delahaye F, GohlkeBärwolf C, Levang OW, . A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. Eur Heart J. 2003;24: 1231–1243.
- Stewart BF, Siscovick D, Lind BK, Gardin JM, Gottdiener JS, Smith VE, . Clinical factors associated with calcific aortic valve disease. Cardiovascular Health Study. J Am Coll Cardiol. 1997;29:630–634.
- Guray Y, Guray U, Altay H, Cay S, Yilmaz MB, Kisacik HL, . Aortic pulse pressure and aortic pulsatility are associated with angiographic coronary artery disease in women. Blood Press. 2005;14:293–297.
- Guray U, Guray Y, Yilmaz MB, Caldir V, Cay S, Sasmaz H, . Aortic pulse pressure and aortic pulsatility in patients with coronary slow flow. Cardiology. 2007;107:233–238.
- Cay S, Guray U, Korkmaz S. Increased aortic pulse and fractional pulse pressures in patients with cardiac syndrome X. Blood Press. 2006;15:179–184.
- Cay S, Ozturk S, Funda Biyikoglu S, Atak R, Balbay Y, Aydogdu S. Association of aortic pressures with fasting plasma glucose in patients with and without impaired fasting glucose. Blood Press. 2008;17:164–169.
- Cay S, Cagirci G, Balbay Y, Atak R, Maden O, Aydogdu S. Effect of aortic pulse and fractional pulse pressures on early patency of sapheneous vein grafts. Coron Artery Dis. 2008;19:435–439.
- Jankowski P, Kawecka-Jaszcz K, Czarnecka D, BrzozowskaKiszka M, Styczkiewicz K, Styczkiewicz M, . Ascending aortic but not brachial blood pressure-derived indices are related to coronary atherosclerosis. Atherosclerosis. 2004;176:151–155.
- Nakayama Y, Tsumura K, Yamashita N, Yoshimaru K, Hayashi T. Pulsatility of ascending aortic pressure waveform is a powerful predictor of restenosis after percutaneous transluminal coronary angioplasty. Circulation. 2000;101:470–472.
- Gibson CM, Cannon CP, Daley WL, Dodge JT Jr, Alexander B Jr, Marble SJ, . TIMI frame count: A quantitative method of assessing coronary artery flow. Circulation. 1996;93:879–888.
- Bellamy MF, Pellikka PA, Klarich KW, Tajik AJ, EnriquezSarano M. Association of cholesterol levels hydroxymethylglutaryl coenzyme-A reductase inhibitor treatment, and progression of aortic stenosis in the community. J Am Coll Cardiol. 2002;40:1723–1730.
- Rajamannan NM, Otto CM. Targeted therapy to prevent progression of calcific aortic stenosis. Circulation. 2004;110:1180–1182.
- Benetos A, Safar M, Rudnichi A, Smulyan H, Richard JL, Ducimetieère P, . Pulse pressure: A predictor of long-term cardiovascular mortality in a French male population. Hypertension. 1997;30:1410–1415.
- Franklin SS, Khan SA, Wong ND, Larson MG, Levy D. Is pulse pressure useful in predicting risk for coronary heart disease? The Framingham Heart Study. Circulation. 1999;100:354–360.
- Nishijima T, Nakayama Y, Tsumura K, Yamashita N, Yoshimaru K, Ueda H, . Pulsatility of ascending aortic blood pressure waveform is associated with an increased risk of coronary heart disease. Am J Hypertens. 2001;14:469–473.
- Weber T, Auer J, O’Rourke MF, Kvas E, Lassnig E, Berent R, . Arterial stiffness, wave reflections, and the risk of coronary artery disease. Circulation. 2004;109:184–189.
- Furchgott RF, Zawadzki JV. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980;288:373–376.
- Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002–2012.
- McEniery CM, Wallace S, Mackenzie IS, McDonnell B, Yasmin, Newby DE, . Endothelial function is associated with pulse pressure, pulse wave velocity, and augmentation index in healthy humans. Hypertension. 2006;48:602–608.