Abstract
Human plasma fraction IV is an intermediate precipitate during the production of human serum albumin using cold ethanol method. Haptoglobin locates in this fraction can be purified for various applications. A new process integration of polyethylene glycol (PEG) precipitation and ion-exchange chromatography (IEC) was developed for purification of haptoglobin, which could effectively purify the haptoglobin from 16.6% to 95%. The recovery of the new process was 58.2% in comparison to 30.3% of the conventional affinity chromatography. Furthermore, 175 mg haptoglobin production in a scaled-up process showed the method to be simple, fast, and low-cost.
| Abbreviations | ||
| AFC | = | affinity chromatography |
| Hb | = | hemoglobin |
| Hp | = | haptoglobin |
| IEC | = | ion-exchange chromatography |
| PEG | = | polyethylene glycol |
| PT | = | pertussis toxin |
| CV | = | column volume |
INTRODUCTION
Haptoglobin (Hp), an acidic glycoprotein component of human plasma α2 globulin, can bind with free hemoglobin (Hb) [Citation1] and is involved in the body's endogenous defense system against inflammation and viral infection [Citation2]. Hp is a biomarker for infection and can also be used as a potent antioxidant to prevent the damage of renal tubules caused by free Hb, thereby avoiding the occurrence of hemolytic kidney failure. A potential therapeutic value of Hp is in the treatment of hemolytic renal failure through removal of free hemoglobin. In addition, Hp has the ability of binding pertussis toxin (PT) and hemagglutinin, toxin proteins secreted by Bordetella pertussis [Citation3,Citation4]. This feature makes it an affinity ligand for purification of pertussis toxin that is used for pertussis vaccines.
Human plasma fraction IV is a by-product in fractionation of human serum albumin through cold ethanol precipitation [Citation5]. This fraction is often discarded in the manufacturing process because it is a mixture of proteins (e.g. haptoglobin transferrin, anti-thrombin III) that could not be directly used. Separation of these proteins needs extra chromatographic procedures. The economic benefit has to be verified. In view of the shortage of blood resources all over the world, it is worthwhile to conduct research for making full use of all plasma compositions.
Structurally similar to immunoglobulin, Hp is a tetramer composed of two heavy chains (b chains) and two light chains (α chains), with the molecular weight of 40 kDa (b chains), 9.1 kDa and 16.0 kDa (α1 and α2), respectively [Citation6,Citation7].
Purification processes with possible low cost and high concentration of target products are very attractive. In the literature, Hp was mainly purified by affinity chromatography using hemoglobin as the ligand [Citation6,Citation8]. This technique offers high selectivity and gives a highly pure product. However, the recovery of target protein as our experiments shown was very low, only 30.3%. On the other hand, the application is limited because hemoglobin is not a stable ligand. It tends to dissociate into α–β dimers and oxidize into methemoglobin, especially during the elution stage with 8M urea. It is difficult for repeat uses of the column. Affinity chromatography with monoclonal antibodies as ligands is a good alternative [Citation9–11], but the cost and stability of ligands in harsh elution conditions are questionable. Ion-exchange chromatography (IEC) and gel filtration chromatography were attempted [Citation12], but these had not achieved high purification. For instance, Akaiwa et al. reported that an IEC purification only improved the purity of Hp from 4.8% to 34.2% [Citation12].
In this study, a two-step protocol for purification of Hp from human plasma fraction IV was designed and compared with conventional affinity chromatography. The fraction was first treated with polyethylene glycol precipitation, followed by DEAE ion exchange chromatography. Under optimal experimental conditions, 175 mg Hp was purified from 120 ml human plasma fraction IV redissolved in 0.9% NaCl, with purity more than 95% and recovery of 57%.
MATERIALS
Human plasma fraction IV was kindly provided by Hualan Biological Engineering Inc. (Xinxiang, China). PEG 10 000 was obtained from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Chromatography media (SP-Sepharose FF, DEAE-Sepharose FF and Sepharose 4 FF) and Chromatography systems (ÄKTA Explorer 100 and Biopilot) were products from GE Healthcare Bio-Sciences (Sweden). Chromatography columns (20 cm × 1.0 cm I.D. and 20 cm × 5.0 cm I.D.) were from Jinhua (Shanghai, China). Mini-PROTEIN®П electrophoresis apparatus was the product of Bio-Rad (USA) and 96-well plates were from Costar (USA). All chemicals were analytical grade reagents. All solutions were prepared with Milli-Q grade water (Millipore, USA).
METHODS
PEG Precipitation
The human plasma fraction, obtained from Hualan Biological Engineering, was re-dissolved in 0.9% NaCl and centrifuged to remove indiscerptible materials, and the pH of the supernatant (protein concentration was around 15 mg/ml) was adjusted to 6.5 with 1 M HCl. PEG 10 000 was introduced slowly into the fraction with stirring to a final concentration of 10% (w/v) and kept for 30min at room temperature. Afterwards, the suspension was centrifuged at 10 000 × g for 20min at 4°C and the supernatant was discarded. The pellet was resuspended with equal volume of Buffer A (20mM PB-citric acid, pH 5.0) compared with buffer B (20mM PB, pH 7.0), and then centrifuged again as above. The supernatants containing Hp were collected and used in the following steps. Unless noted, the samples loaded onto chromatography columns were pre-treated with PEG as described above.
Hb-affinity Chromatography
For comparison purposes, affinity chromatography with hemoglobin as the ligand was performed. Bovine hemoglobin, purified by DEAE Sepharose FF chromatography [Citation13], was coupled to Sepharose 4FF, which was pre-activated with CNBr, to prepare Hb-affinity medium as described in the literature [Citation6]. 30 ml of pre-treated sample was loaded onto the as-synthesized Hb affinity column (5.5 cm × 1.0 cm I.D., CV = 4.3 ml, CV, column volume) pre-equilibrated with Buffer B (20 mM PB, pH 7.0). The unbound materials and the non-specific bound proteins were washed out with 2 CV of buffer B and 3 CV of buffer C (0.15 M NaCl, pH 11.0) in sequence. The target protein Hp was eluted out with 10 CV of Buffer D (5M Urea in 0.15M NaCl, pH 11.0). All the processes were operated at room temperature. The flow rate was kept at constant 1 ml/min controlled by ÄKTA Explorer 100 chromatography workstation with the detection wavelength set at 280 nm.
Ion-exchange Chromatography (IEC)
PEG pre-treated samples were loaded onto the DEAE-Sepharose FF column (8.5 cm × 1.0 cm I.D., CV = 6.7 ml) pre-equilibrated with Buffer A or Buffer B. The column was washed with 3–5 CV of the same buffer (A/B) to wash out unbound fractions. At last, a linear gradient was produced with 10 CV buffer (A/B) from 0 to 0.4 M NaCl in the mobile phase to elute the bound proteins. The process was operated at room temperature. Finally, the IEC was scaled up to 130 ml of ion-exchange column performed at a flow rate of 10 ml/min.
Determination of Protein Concentration
Protein concentration was determined based on the Bradford method with slight modification as described previously [Citation14]. In brief, 96-well plates were employed for analysis and bovine serum albumin (BSA) was taken as standard reference. The detection wavelength is 595 nm.
SDS-PAGE and High Performance Size Exclusion Chromatography (HPSEC)
The purity of the eluted fraction was measured with electrophoresis (SDS-PAGE) and high performance size exclusion chromatography (HPSEC). Electrophoresis was performed on 15% gel with silver staining. HPSEC with pre-packed Superdex 200 10/300 GL column (GE Healthcare) was carried out using an Agilent 1100 HPLC system with a degasser and a variable wavelength detector monitoring set up at 280 nm. Elution buffer was 20 mM sodium phosphate, pH 6.8, containing 100 mM sodium sulphate running at a flow rate of 0.5 ml/min. Samples were filtered with 0.45 mm membrane before loading. The injection volume was 100 μl at a protein concentration of 0.1– 0.2 mg/ml.
Haptoglobin Assay
The purified Hp was identified indirectly by getting PT from Hp-affinity chromatography column with high purity, as Poolman et al. pointed out that Bordetella pertussis toxin (PT) binds with haptoglobin in an unspecific manner [Citation4]. The identification and quantification of PT were done as described by Sekura et al. [Citation15].
RESULTS
Identification of Hp and its Phenotypes
Hp purified in this study was employed for preparation of Hp-affinity gel with different ligand densities, and PT was purified from Bordetella pertussis culture supernatant by Hp-affinity chromatography with a purity as high as 99% quantified by ELISA and SDS-PAGE (unpublished data), which indirectly confirmed the identity of Hp. As a tetramer composed of 2α and 2β chains, human Hp has three major phenotypes known: Hp1-1, 2-1, and 2-2. All three phenotypes share the same 2β chains (Mr∼40 kDa), but contain different contents of α chains: two α1 (∼9 kDa) in Hp1-1, two α2 chains (Mr∼16.5 kDa) existing in Hp2-2, and one α1 and one α2 in Hp2-1 [Citation11]. In the present study, the bands of Hp near 40 kDa (β chain), and around 20.1 kDa (α2 chain) and 14.4 kDa (α1 chain) appearing in the SDS-PAGE gel, are in accordance with the observation of Mao's group, whose immunoblot analysis confirmed the molecular weights of the different chains of Hp [Citation6,Citation11]. Based on these results, the phenotype of Hp2-2 was further identified by the molecular weight of β and α2 chains which predominantly appeared in the SDS-PAGE gel.
Pretreatment of PEG Precipitation
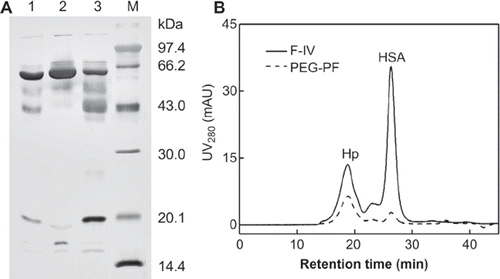
Based on our pre-experiments (data not shown), 10% (w/v) of PEG 10 000 was selected for precipitation of human plasma fraction IV under room temperature. In this fraction (, lane 1), human serum albumin (HSA, 66 kDa) was present in large quantities (42.9%), and was the major impurity interfering with the haptoglobin purification. After the precipitation, most of the HSA was left in the supernatant (, lane 2). The buffer of 20 mM PB-citric acid with pH 5.0 was employed for dissolving the precipitate. After re-dissolving, the purity of Hp, including α1, α2, and β subunits, was increased from about 16.6 % to 46.0 – 52.9% (, lane 3) analyzed by Quantity One software (Bio-Rad). The effect of PEG precipitation was verified by high performance size exclusion chromatography, as shown in . It's clear to see that after PEG precipitation and redissolving, there was very few HSA left with Hp. The advantage of this precipitation is that it can be operated at room temperature without denaturation of the target products.
Figure 1. Analysis of the effect of PEG precipitation on Hp purity. The original protein components of human plasma fraction IV were show in lane 1. After PEG precipitation, the supernatant (A, lane 2) was discarded. The precipitate redissolved in 20 mM PB-citric acid, pH 5.0 (A, lane 3) was for further purification. M stands for standard molecular markers. The effect of PEG precipitation was verified by high performance size exclusion chromatography as shown in . The real line (−) stands for the human plasma fraction IV, and the dashed line (--) represents the PEG precipitate redissolved in 20 mM PB-citric acid buffer, pH 5.0. 99 × 57 mm (300 × 300 DPI).
Hb-affinity Purification of Hp
The ligand density plays an important role in the affinity chromatography separation process. In our experiment, two Hb-affinity media with ligand densities of 7.0 and 1.42 mg Hb/g gel were prepared, respectively, by controlling the amount of Hb added during the process of coupling.
After having been pretreated with PEG precipitation, the sample containing Hp was loaded onto the Hb-Sepharose 4FF chromatography column. The chromatographic efficiency of Hb-Sepharose 4FF with a ligand density of 7.0 mg Hb/g gel was unsatisfied due to non-specific and irreversible adsorption (data not shown). When the ligand density was reduced to 1.42 mg Hb/g, however, the non-specific and irreversible adsorption could be restrained to the minimum. The elution profile is as shown in . Under this condition, the target protein Hp has been purified to a high purity greater than 99 % (, lane 2 and 3). However, the whole recovery of proteins in all of the peaks was only 35% compared to the loaded proteins to the column, which means that most proteins were not washed out from the affinity chromatography column. In addition, there was still some Hp2-2 in the flow-through fractions (, lane 4). Therefore, the whole recovery of Hp in P1 and P2 was only 30.3% (), calculated from protein concentration and Hp purify. The low recovery of Hp in Hb-affinity chromatography may be due to the intense binding of Hp with Hb, resulting in the difficult elution of Hp2-2 from Hb even using 5M urea. Additionally, the harsh-elution condition may cause the dissociation of an Hb subunit from the Sepharose and the unfolding of Hp [Citation8]. Therefore, it is necessary to explore another technique to obtain Hp.
Figure 2. A typical elution profile of Hemoglobin-affinity column chromatography of plasma containing Haptoglobin and SDS-PAGE analysis on Hb-Sepharose FF affinity-purified human Hp. A: 30 ml PEG-treated human plasma fraction IV was applied to the Hb-Sepharose FF affinity column with a ligand density of 1.42 mg Hb/g gel, followed by extensive wash with Buffer B (20mM PB, pH 7.0),as P0. The bound materials were first eluted with 0.15M NaCl (pH 11.0) obtained P1, and then eluted with 5M Urea in 0.15M NaCl (pH 11) obtained P2. B: SDS-PAGE analysis of Hp purity. Lane 1: PEG precipitate of Human plasma IV fraction redissolved in buffer A (20 mM PB-citric acid, pH 5.0); M: Low-molecular-mass Marker. It's indicated that Hp2-2 was concentrated in P1 (lane 2) and P2 (lane 3), while the other high molecular-weight proteins, such as albumin, α-antitrypsin etc, and also few amount of Hp2-2 were in the flow through peak (P0, lane 4). 99 × 45mm (300 × 300 DPI).
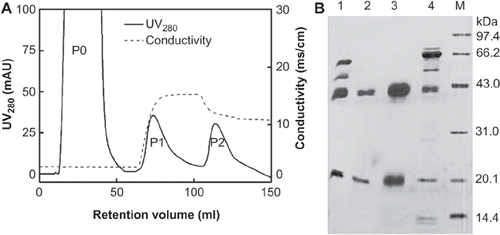
Table 1. Analytical recovery of haptoglobin 2-2 purified from 30 ml human plasma fraction IV.
Effect of pH on Elution of Hp by Ion-exchange Chromatography
The deviation of pIs between the target protein and impurities () offers a possibility to purify Hp from human plasma fraction IV. The effect of pH on the chromatography of Hp with DEAE Sepharose FF was studied. Initially, 7 ml of PEG precipitate of human plasma fraction IV redissolved in 20 mM PB-citric acid buffer of pH 5.0 was loaded onto the DEAE-Sepharose FF column, followed by extensive washing and elution. The flow rate was 1ml/min and detection wavelength was 280nm. The elution profiles are shown in and . Combined with the analysis of SDS-PAGE (), the results can be concluded as follows: (i) At pH 7.0, Hp was appeared both in P1 (, Lane 7) and P2 (Lane 8), but P1 only contains very few Hp while P2 has two bright bands at around 40 and 20 KDa, which were in accordance with the molecular weight of β and α2 subunits of Hp [Citation1], respectively. Also, there was a small amount of α1 subunits of Hp at around 14.4 kDa. (ii) The purity of Hp2-2 in P2 (Lane 8) was much lower compared to P1 (Lane 4) eluted with buffer of pH 5.0. Under the later condition, Hp2-2 was mostly eluted in P1 (Lane 4) and its purity achieved about 86.7%. So it is better to elute Hp from the ion-exchange column using a pH 5.0 buffer rather than pH 7.0.
Figure 3. Typical elution profiles and SDS-PAGE analysis of DEAE ion-exchange chromatography with buffers at different pH. A: Buffer A (20 mM PB-citric acid, pH 5.0) was used as basic mobile phase. A flow-through P0 peak (3C-lane 3) was obtained, and only one peak (P1; 3C, lane 4) appeared during the gradient elution. B: Buffer B (20 mM PB pH 7.0) was used in the purification process. The first peak (P0, flow through fraction) was obtained with buffer B and another two peaks (P1 and P2) were obtained from gradient elution with 0-0.4M NaCl in buffer B. C: SDS-PAGE analysis of Hp purity from ion-exchange chromatography. Lanes: 1. human plasma fraction IV; 2. PEG precipitate redissolved in Buffer A (20 mM PB-citric acid, pH 5.0); 5. PEG precipitate redissolved in Buffer B (20 mM PB, pH 7.0). The flow-though fractions were washed out by Buffer A (P0, lane 3) or Buffer B (P0, lane 6). And the target proteins were eluted in linear gradient with Buffer A (P1, lane 4) or Buffer B (P1, lane 7 and P2, lane 8) containing 0-0.4 M NaCl, respectively. M stands for the molecular markers. 150 × 97mm (300 × 300 DPI).
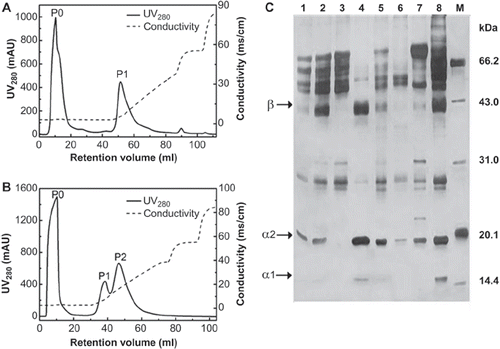
Table 2. Approximate masses, concentration, and pI of several polypeptides in plasma from healthy subjects, based on literature survey.
Optimization of Elution Gradients of Ion-exchange Chromatography
The purity of Hp (, Lane 4) eluted with a linear gradient (0-0.4M NaCl, 6 CV) was just about 90%, which needs further improving. To increase the purity of Hp, elution gradient, another important operation parameter in IEC, was optimized. The elution profile of IEC eluted with different NaCl concentrations (0, 0.05, 0.10, 0.15 and 0.50 M) is shown in . A flow through fraction (P0) and four elution fractions (P1, P2, P3 and P4) were eluted according to different NaCl concentrations, respectively.
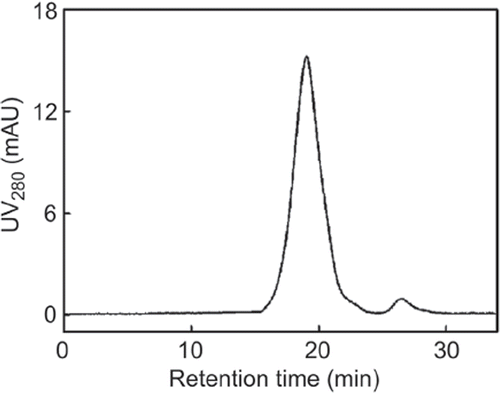
Most proteins flow through the column (, Lane 2), while Hp was pooled in P1, P2 and P3 (lane 3-5) with purity of Hp2-2 about 87.7%, 95.1% and 94.7%, respectively. There were also a few albumin and low- molecular weight proteins in P1-P3, and the main components of P4 were low-molecular-mass proteins. It meant that Hp2-2 was mainly eluted out between 0.05 and 0.15 M of NaCl. The purity of Hp in P2 was above 95% verified by the calculation of peak area in HPSEC (). The typical recovery of Hp in the whole process was 57.2-61.3%, which was relatively higher than that of Hb-affinity chromatography (30.3%).
Figure 4. A typical elution profile and SDS-PAGE analysis of DEAE-Sepharose IEC with elution with different NaCl concentration. A: The materials eluted with buffer A and different NaCl concentrations (0, 0.05, 0.10, 0.15 and 0.50 M) were as P0, P1, P2, P3 and P4. B: SDS-PAGE analysis. Lanes: M, LMW Markers; lane 1, PEG precipitate of Human plasma redissolved in 20 mM PB-citric acid buffer of pH 5.0, lane 2, P0; lane 3, P1; lane 4, P2; lane 5, P3; lane 6, P4. 99 × 151mm (300 × 300 DPI).
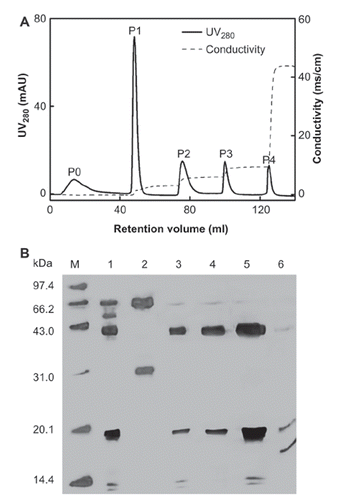
Figure 5. HPSEC analysis of Hp obtained from IEC. Conditions: Superdex 200 (300 mm × 10 mm I.D.); eluted by 20 mM PBS +0.1 M Na2SO4, pH 6.8, at room temperature (20−25°C) with a flow rate of 0.5 ml/min; 100 μl of injection volume with a protein concentration of 0.1−0.2mg/ml. 99 × 70 mm (300 × 300 DPI).
Scale-up of Ion-exchange Chromatography
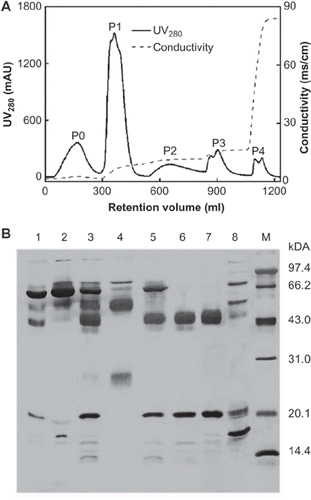
Comparing with gel filtration chromatography and affinity chromatography, it was easier to scale ion-exchange chromatography operation up to prepare Hp on a large scale with less cost. The sample volume loaded onto the IEC column was increased from 7 ml to 120 ml, and the size of the column was scaled up to 130ml, with the flow rate increased accordingly to 10ml/min. The DEAE- Sepharose FF column (6.5 cm × 5.0 cm I.D., CV = 128 ml) was pre-equilibrated by 5 CV of Buffer A (20mM PB-Citric acid, pH 5.0). The elution profile is as shown in . The analysis of SDS-PAGE () indicated that Hp was mainly eluted in P1 (Lane 5), P2 (Lane 6), and P3 (Lane 7). The purities of Hp2-2 in the three peaks were about 73.3%, 92.8%, and 95.7%, respectively. The total recovery of Hp2-2 is about 57%. Using the IEC method, around 175 mg high-purity Hp2-2 was prepared per batch and the whole process took only 4–5 hours.
Figure 6. A typical elution profile and SDS-PAGE analysis of DEAE-Sepharose ion-exchange chromatography at an amplified scale. 120 ml PEG-treated samples were loaded on a 130 ml DEAE IEC column and the flow rate was 10 ml/min, detected at 280 nm. B: SDS-PAGE analysis of Hp2-2 eluted from DEAEIEC column. Lanes: Lane 1, Human plasma fraction IV; lane 2, Supernatant of PEG precipitation; lane 3, dissolved precipitate of PEG-precipitation in buffer of pH 5.0; lane 4, flow though peak, P0; lane 5, elution peak, P1; lane 6, P2; lane 7, P3; lane 8, P4. M, LMW Markers. 99 × 149mm (300 × 300 DPI).
DISCUSSION
Human plasma fraction IV contains many proteins [Citation7], among which Hp is only 1–1.5 g/L [Citation6], while albumin is the richest one with the concentration of 35–55 g/L. Therefore, it is necessary to remove albumin from the plasma fraction IV first for the purification of Hp. Ammonium sulphate precipitation, acid precipitation, PEG precipitation [Citation16–20], etc., were commonly used as a pretreatment step prior to column chromatography. Based on our pre-experiments (data not shown), PEG precipitation was the most efficient one. After optimization, 10% (w/v) PEG 10 000 was selected for precipitation. The advantage of PEG precipitation over ammonium sulphate precipitation is its direct connection with ion exchange chromatography without the need of intermediate dialysis. Its superiority over acid precipitation is the high recovery of the product. Another advantage of using PEG is that the possible residual free PEG in the precipitate in PEG precipitation step would appear only in the go-through peak in ion- exchange chromatography ([Citation21] and private communication with Zhai, Y.), which avoids additional steps to remove it from the final products. In addition, the cost of PEG is only around 1.5 dollars per kilogram, which has economic advantage in large-scale applications.
Ion-exchange chromatography separates biomolecules on the basis of surface charge characteristics. Charged groups on the surface of a protein interact with oppositely charged groups immobilized on the ion- exchange medium [Citation22]. First, the effect of pH (7.0 and 5.0) on dissolving PEG precipitation and IEC was investigated. The reason for the optimal pH being 5.0 can be deduced from the pI of each protein existing in the human plasma fraction IV (). The pIs of most proteins are between 4 and 6; therefore their charges are similar, resulting in difficulty to separate them from each other when pH was set at 7.0. On the other hand, when pH was set at 5.0, the differences of proteins’ charges became distinguishable; for example, the pI of transferrin protein is 5.8 [Citation23], take the positive charge and can't bind to the anion-exchange column, while the pI of albumin is 4.7–5.1 [Citation24]. The solution ability of albumin in buffer of pH 5.0 is very low and binding ability to IEC column is very weak. In contrast, the pI of Hp is 4.1, which can easily re-dissolve in the buffer of pH 5.0 and bind the column strongly, resulting in easier to separate Hp from other proteins. Ion-exchange chromatography is easily scaled up to pilot or industry-scale and can significantly reduce process time and associated costs [Citation25].
The structural and functional relationships among the Hp phenotypes have not yet been well established [Citation9], but all of them can bind with Hb [Citation11], prompting a way of separating them from others. Liau et al. used a human hemoglobin affinity column to isolate three phenotypes of Hp and found that it was most suitable for the isolation of Hp1-1 in large quantities [Citation6]. Yueh et al. improved the purification method of human Hb-affinity chromatography by adding 0.1%SDS in the elution buffer and got 95% purity of Hp [Citation9], but the disadvantage is that the regeneration of the column using a large volume of PBS is time consuming.
From SDS-PAGE analyses (typically as shown in lane 1 and lane 1), Hp2-2 is the main phenotype in the human plasma fraction IV, while the percentage of Hp α1 subunit is very low, and only can be visualized after purification. The low recovery from Hb-affinity chromatography (30.3%) may be due to the least effective of Hp2-2 in binding Hb compared with Hp1-1 and Hp2-1, reviewed by Sadrzadeh [Citation26].
In conclusion, the basic principle of purification is using a simple, fast, and efficient process to get target proteins with high purity and high recovery. In our case, the target protein is haptoglobin (Hp) and the main phenotype of Hp in normally discarded human plasma fraction IV is Hp2-2. Here, PEG precipitation was introduced to remove most of the impurities to insure the high purity of the final product. The results of this study suggested that the novel procedure composed of PEG precipitation and ion-exchange chromatography is efficient, reproducible, and easily adaptable for process-scale operation in Hp purification. The application of this process will expand the usage of human blood plasma, which is especially significant when facing the shortage of human blood supply.
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
REFERENCES
- Saeed, S.A., Ahmad, N., Ahmed, S. (2007). Dual inhibition of cyclooxygenase and lipoxygenase by human haptoglobin: its polymorphism and relation to hemoglobin binding. Biochem Biophys Res Commun, 353: 915–920.
- Eckersall, P.D., Lawson, F.P., Kyle, C.E., Waterston, M., Bence, L., Stear, M.J., Rhind, S.M. (2008). Maternal undernutrition and the ovine acute phase response to vaccination. BMC Vet Res, 4: 1.
- Irons, L.I., MacLennan, A.P. (1979). Isolation of the lymphocytosis promoting factor-haemagglutinin of Bordetella pertussis by affinity chromatography. Biochim Biophys Acta, 580: 175–185.
- Poolman, J.T., Kuipers, B., Vogel, M.L., Hamstra, H.J., Nagel, J. (1990). Description of a hybridoma bank towards Bordetella pertussis toxin and surface antigens. Microb Pathog, 8: 377–382.
- Boros, B. (1979). Plasma fractionation: production and properties of albumin. Ann Immunol Hung, 19: 149–161.
- Liau, C.Y., Chang, T.M., Pan, J.P., Chen, W.L., Mao, S.J. (2003). Purification of human plasma haptoglobin by hemoglobin-affinity column chromatography. J Chromatogr B Analyt Technol Biomed Life Sci, 790: 209–216.
- Hortin, G.L., Sviridov, D., Anderson, N.L. (2008). High-abundance polypeptides of the human plasma proteome comprising the top 4 logs of polypeptide abundance. Clin Chem, 54: 1608–1616.
- Rademacher, B.E., Steele, W.J. (1987). A general method for the isolation of haptoglobin 1–1, 2–1, and 2–2 from human plasma. Anal Biochem, 160: 119–126.
- Yueh, S.C., Lai, Y.A., Chen, W.L., Hsu, H.H., Mao, S.J. (2007). An improved method for haptoglobin 1–1, 2–1, and 2–2 purification using monoclonal antibody affinity chromatography in the presence of sodium dodecyl sulfate. J Chromatogr B Analyt Technol Biomed Life Sci, 845: 210–217.
- Katnik, I., Jadach, J. (1993). Immunoaffinity purification of human haptoglobin using monoclonal antibodies. Arch Immunol Ther Exp (Warsz), 41: 303–308.
- Tseng, C.F., Huang, H.Y., Yang, Y.T., Mao, S.J. (2004). Purification of human haptoglobin 1–1, 2–1, and 2–2 using monoclonal antibody affinity chromatography. Protein Expr Purif, 33: 265–273.
- Akaiwa, S. (1982). Purification of haptoglobin from rat serum. Anal Biochem, 123: 178–182.
- Lu, X., Zhao, D., Ma, G., Su, Z. (2004). Polyethylene glycol increases purification and recovery, alters retention behavior in flow-through chromatography of hemoglobin. J Chromatogr A, 1059: 233–237.
- Zhang, Y., Luo, J., Bi, J., Wang, J., Sun, L., Liu, Y., Zhang, G., Ma, G., Su, Z. (2010). Efficient separation of homologous alpha-lactalbumin from transgenic bovine milk using optimized hydrophobic interaction chromatography. J Chromatogr A, 1217: 3668–3673.
- Sekura, R.D., Fish, F., Manclark, C.R., Meade, B., Zhang, Y.L. (1983). Pertussis toxin. Affinity purification of a new ADP-ribosyltransferase. J Biol Chem, 258: 14647–14651.
- Seddik, S.S., Wanas, S., Shehata, A., Fawaz, S., Helmy, M.H. (2002). Development of an improved method for production of antiscorpion F(ab’)2 fragment of IgG with high yield and potency. J Nat Toxins, 11: 123–132.
- Gulliver, B.S., Slabas, A.R. (1994). Acetoacyl-acyl carrier protein synthase from avocado: its purification, characterisation and clear resolution from acetyl CoA:ACP transacylase. Plant Mol Biol, 25: 179–191.
- Perosa, F., Carbone, R., Ferrone, S., Dammacco, F. (1990). Purification of human immunoglobulins by sequential precipitation with caprylic acid and ammonium sulphate. J Immunol Methods, 128: 9–16.
- Stampe, D., Wieland, B., Kohle, A. (1986). Isolation of factor IX concentrates for clinical use by ion-exchange chromatography and ammonium sulphate precipitation. J Chromatogr, 363: 101–103.
- Bjorling, H. (1985). A new protein fractionation method using ion-exchange chromatography and PEG precipitation. Vox Sang, 49: 240–243.
- Zhai, Y., Zhao, Y., Lei, J., Su, Z., Ma, G. (2009). Enhanced circulation half-life of site-specific PEGylated rhG-CSF: optimization of PEG molecular weight. J Biotechnol, 142: 259–266.
- Williams, A., Frasca, V. (2001). Ion-exchange chromatography. Curr Protoc Protein Sci, Chapter 8: Unit8 2.
- Thymann, M. (1978). Identification of a new serum protein polymorphism as transferrin. Hum Genet, 43: 225–229.
- Bagdasar'ian, S.N., Malyi, K.D., Zarechnaia, I.P., Azhitskii, G., Akhmed, M. (1981). Characteristics of isoelectric fractions of human serum albumin. Ukr Biokhim Zh, 53: 15–18.
- Levison, P.R., Koscielny, M.L., Butts, E.T. (1990). A simplified process for large-scale isolation of IgG from goat serum. Bioseparation, 1: 59–67.
- Sadrzadeh, S.M., Bozorgmehr, J. (2004). Haptoglobin phenotypes in health and disorders. Am J Clin Pathol, 121 Suppl: S97–104.