Abstract
Context: Lymphocytes predominantly express delayed rectifier K+-channels (Kv1.3) in their plasma membranes, and the channels play crucial roles in the lymphocyte activation and proliferation. Since macrolide antibiotics, such as clarithromycin and azithromycin, exert immunomodulatory effects, they would affect the Kv1.3-channel currents in lymphocytes.
Objective: This study determined the physiological involvement in the mechanisms of immunomodulation by these antibiotics.
Materials and methods: Employing the standard patch-clamp whole-cell recording technique in murine thymocytes, we examined the effects of 30 and 100 µM clarithromycin and azithromycin on the Kv1.3-channel currents and the membrane capacitance.
Results: Clarithromycin significantly suppressed the peak currents (30 µM, 178 ± 5.6 to 111 ± 2.0 pA/pF; 100 µM, 277 ± 4.4 to 89.6 ± 10 pA/pF) and the pulse-end currents (30 µM, 47.5 ± 2.2% to 15.5 ± 3.3%; 100 µM, 48.5 ± 1.4% to 15.8 ± 1.0%) of thymocyte Kv1.3-channels without significant effects on the membrane capacitance. In contrast, azithromycin did not affect the channel currents. However, it significantly decreased the membrane capacitance (30 µM, 4.68 ± 0.14 to 3.74 ± 0.13 pF; 100 µM, 4.47 ± 0.06 to 3.37 ± 0.08 pF), indicating its accumulation in the plasma membrane.
Discussion and conclusion: This study demonstrated for the first time that clarithromycin exerts inhibitory effects on thymocyte Kv1.3-channel currents, while azithromycin decreases the membrane capacitance without affecting the channel currents. These differences in the effects of the macrolide antibiotics may reflect differences in the mechanisms of immunomodulation by which they control the production of cytokines.
Introduction
Macrolide antibiotics are widely used for the treatment of a variety of bacterial infections, including respiratory and skin infections (Mazzei et al., Citation1993). In addition to such antimicrobial effects, their therapeutic efficacies as immunosuppressive agents have also been demonstrated in patients with diffuse panbronchiolitis (Kudoh et al., Citation1998), cystic fibrosis and inflammatory bowel diseases (Kanoh & Rubin, Citation2010). According to several in vitro studies, macrolide antibiotics, such as clarithromycin and azithromycin, suppress the production of pro-inflammatory cytokines from leukocytes and thus exert immunomodulatory effects (Khan et al., Citation1999; Morikawa et al., Citation2002; Sugiyama et al., Citation2007). However, we know little about the physiological involvement in the mechanisms by which these antibiotics control the production of cytokines. Leukocytes, including macrophages and lymphocytes, predominantly express delayed rectifier K+-channels (Kv1.3) in their plasma membranes, and the channels play crucial roles in the activation and proliferation of lymphocytes (Chandy et al., Citation2004; Lewis & Cahalan, Citation1995; Matko, Citation2003; Villalonga et al., Citation2010). In our recent study, we demonstrated that nonsteroidal anti-inflammatory drugs (NSAIDs) suppress the Kv1.3-channel currents in thymocytes when they modulate the immune response (Kazama et al., Citation2012a). We also revealed that these drugs induce microscopic surface changes in the thymocyte membranes, as detected by the increase in the membrane capacitance. Since macrolide antibiotics exert immunomodulatory effects as NSAIDs do, they would affect the Kv1.3-channel currents or induce microscopic surface changes in lymphocytes. To test this, employing the standard patch-clamp whole-cell recording technique in murine thymocytes, we examined the effects of clarithromycin and azithromycin on Kv1.3-channel currents and the membrane capacitance. Despite their structural and pharmacological similarities (Stepanic et al., Citation2011; Zuckerman, Citation2004), these drugs exerted differential effects on the thymocyte plasma membranes. Here, we show for the first time that clarithromycin suppresses thymocyte Kv1.3-channel currents, while azithromycin decreases the membrane capacitance without affecting the channel currents. These differences in the effects of the macrolide antibiotics may reflect differences in the mechanisms of immunomodulation by which they influence the production of cytokines.
Materials and methods
Cell sources and preparation
Male ddy mice (4–5 weeks old), supplied by Japan SLC Inc. (Shizuoka, Japan), were deeply anaesthetized with isoflurane and then sacrificed by cervical dislocation. The protocol for animal use was approved by the Animal Care and Use Committee of the Tohoku University Graduate School of Medicine. Single thymocytes, isolated from murine thymus as described previously (Kazama et al., 2012a, b), were resuspended in standard external (bathing) solution containing (in mM): NaCl, 145; KCl, 4.0; CaCl2, 1.0; MgCl2, 2.0; Hepes, 5.0; bovine serum albumin, 0.01% (pH 7.2 adjusted with NaOH). They were maintained at room temperature (22–24 °C) for use within 4 h.
Electrical setup and patch-clamp recordings
We conducted standard whole-cell patch-clamp recordings using an EPC-9 patch-clamp amplifier system (HEKA Electronics, Lambrecht, Germany) as described previously (Kazama et al., 2012a, b). The patch pipette resistance was 4-6 MΩ when filled with internal (patch pipette) solution containing (in mM): KCl, 145; MgCl2, 1.0; EGTA, 10; Hepes, 5.0 (pH 7.2 adjusted with KOH). After a giga-seal formation, we applied suction briefly to the pipette to rupture the patch membrane. The series resistance of the whole-cell recordings was maintained below 10 MΩ during the experiments. Peak currents were normalized by the membrane capacitance and expressed as the current densities (pA/pF). All experiments were carried out at room temperature.
Drug delivery
Clarithromycin, purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan) and azithromycin, from LKT Laboratories, Inc. (St. Paul, MN), were separately dissolved in the external solution at the final concentrations of 30 or 100 µM. We delivered one of the drugs to the cells by the standing hydrostatic pressure of 3 cm H2O from a nearby pipette as described previously (Kazama et al., 2012a, b). Then, whole-cell membrane currents were recorded before and after 30 s exposure to the drug. To rule out the possibility that the observed effect just resulted from the procedure of the reagent application, we simply applied the external solution to the cells and confirmed the absence of any significant changes in the channel currents.
Membrane capacitance measurements
To measure the membrane capacitance of the thymocytes, we employed a sine plus DC protocol using the Lock-in amplifier of the EPC-9 Pulse program. An 800-Hz sinusoidal command voltage was superimposed on the holding potential of −80 mV. The membrane capacitance (Cm), as well as membrane conductance (Gm) and series conductance (Gs), was continuously recorded before and after 30 s exposure to the drugs during the whole-cell recording configuration.
Statistic analyses
Data were analyzed using PulseFit software (HEKA Electronics, Lambrecht, Germany), IGOR Pro (WaveMetrics, Lake Oswego, OR) and Microsoft Excel (Microsoft Corporation, Redmond, WA) and reported as means ± SEM. Statistical significance was assessed by two-way ANOVA followed by Dunnett’s or Student’s t-test. A value of p < 0.05 was considered significant.
Results and discussion
Effects of clarithromycin and azithromycin on Kv1.3-channel currents in murine thymocytes
Stepwise changes in the membrane potential, from the holding potential of −80 mV to the various depolarizing potential levels, evoked membrane currents in the thymocytes, showing voltage-dependent activation and inactivation patterns characteristic to Kv1.3 () (Kazama et al., 2012a, b). To investigate the effects of clarithromycin and azithromycin on the channel currents, we applied external solution, containing 30 or 100 µM of these drugs, to the thymocytes and examined the changes in the whole-cell currents ( and ). In previous in vivo studies, the physiological concentrations of both drugs in lung tissues were between 10 and 15 µM (McCarty, Citation2000; Morris et al., Citation1991). In the following in vitro studies, however, single cells required as much as 30 µM of these drugs to effectively elicit their pharmacological effects on delayed rectifier Kv1.3 currents in cardiomyocytes (Gluais et al., Citation2003; Volberg et al., Citation2002). In those studies, the drugs were previously dissolved in whole culture media, or continuously infused into the bathing solution. In the present study, since we delivered the drugs to single, isolated cells by a puff application method, there was the possibility of partial and insufficient exposure of the drugs to the cells. Therefore, we applied the drugs at a dose significantly higher than 30 µM. However, since doses higher than 100 µM were considered to be superphysiological (Sugiyama et al., Citation2007), we tested at 30 and 100 µM for each drug.
Figure 1. Effects of clarithromycin on Kv1.3 channel currents in murine thymocytes. The effects of 30 µM (A) and 100 µM (B) clarithromycin. (a) Typical whole-cell current traces at different voltage-steps recorded before and after either drug application. The currents were elicited by voltage-steps from the holding potentials of −80 mV to −40, 0, 40 and 80 mV, as shown in the voltage protocol. Each pulse was applied for a 200-ms duration between 10 s intervals. (b) Peak current densities (peak currents normalized by the membrane capacitance) obtained from the records in (a) at the voltage-step of 80 mV. (c) Percentages of the pulse-end currents relative to the peak currents (100 * I/Ipeak) obtained from the records in (a) at the voltage-step of 80 mV. #p < 0.05 versus before the drug application. Values are means ± SEM (n = 5). Differences were analyzed by ANOVA followed by Dunnett’s or Student’s t test.
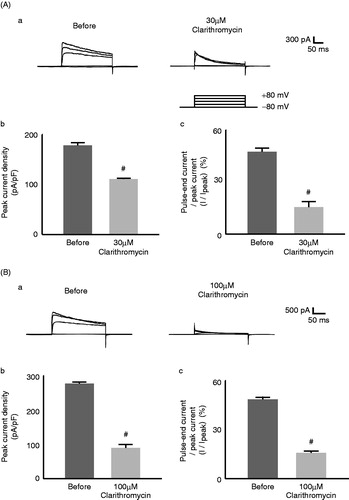
Figure 2. Effects of azithromycin on Kv1.3 channel currents in murine thymocytes. The effects of 30 µM (A) and 100 µM (B) azithromycin. (a) Typical whole-cell current traces at different voltage-steps recorded before and after either drug application. The currents were elicited by voltage-steps from the holding potentials of −80 mV to −40, 0, 40 and 80 mV, as shown in the voltage protocol. Each pulse was applied for a 200-ms duration between 10 s intervals. (b) Peak current densities (peak currents normalized by the membrane capacitance) obtained from the records in (a) at the voltage-step of 80 mV. (c) Percentages of the pulse-end currents relative to the peak currents (100 * I/Ipeak) obtained from the records in (a) at the voltage-step of 80 mV. Values are means ± SEM (n = 5). Differences were analyzed by ANOVA followed by Dunnett’s or Student’s t test.
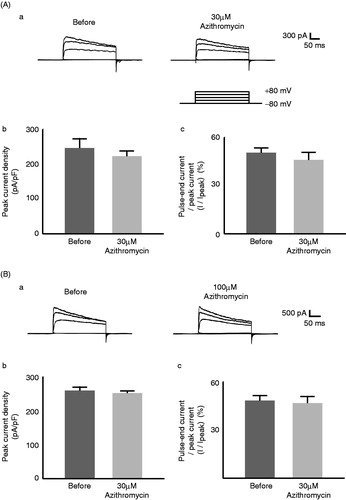
As previously demonstrated with voltage-dependent Kv1.3 currents in cardiomyocytes (Gluais et al., Citation2003; Volberg et al., Citation2002), clarithromycin suppressed Kv1.3-channel currents in thymocytes (). Thirty µM of this drug significantly lowered the peak currents (from 178 ± 5.6 to 111 ± 2.0 pA/pF, n = 5, p < 0.05, ) and more markedly suppressed the pulse-end currents expressed as percentages of the peak currents (I/Ipeak) (from 47.5 ± 2.2% to 15.5 ± 3.3%, n = 5, p < 0.05, ). One-hundred µM clarithromycin lowered the pulse-end currents to the same extent as 30 µM (from 48.5 ± 1.4% to 15.8 ± 1.0%, n = 5, p < 0.05, ). However, it suppressed the peak currents more markedly than 30 µM (from 277 ± 4.4 to 89.6 ± 10 pA/pF, n = 5, p < 0.05, ). These results were consistent with previous findings that clarithromycin exerts dose-dependent attenuating effects on the activation curves of voltage-dependent Kv1.3 currents (Gluais et al., Citation2003). In contrast, azithromycin failed to affect the currents (). Thirty µM azithromycin did not significantly alter the peak (from 246 ± 27 to 223 ± 15 pA/pF, n = 5, ) and the pulse-end currents (from 50.4 ± 3.1% to 46.0 ± 4.8%, n = 5, ). Doses as high as 100 µM did not affect the currents either ().
Of all macrolides, clarithromycin most potently suppresses the production of interleukin 2 (IL-2) (Morikawa et al., Citation2002; Sugiyama et al., Citation2007), which is essential for lymphocyte activation and proliferation (Price et al., Citation1989; Villalonga et al., Citation2010). In the present study, we showed for the first time that clarithromycin inhibits the Kv1.3-channel currents in thymocytes (). Since the channels trigger Ca2+ influx, which is necessary for the IL-2 synthesis (Chandy et al., Citation2004), and since channel blockade by highly selective inhibitors, including margatoxin and ShK-Dap22, actually repressed the immune response in lymphocytes (Kalman et al., Citation1998; Villalonga et al., Citation2010), such effects of clarithromycin on the channel currents were thought to contribute to their immunomodulatory properties, which have also been demonstrated with NSAIDs and Ca2+ channel blockers in our recent studies (Kazama et al., Citation2012a, Citation2013). In contrast, azithromycin, which was not demonstrated to suppress the production of IL-2 in murine T-lymphocytes (Sugiyama et al., Citation2007), did not affect the channel currents in the present study (). Notably, clarithromycin induced the current inactivation on a faster time scale than that before the drug application (). This represents a “N-type inactivation” pattern in kinetic studies (Yellen, Citation1998), suggesting that this drug plugs into the open-pores of the channel to inhibit the currents.
Effects of clarithromycin and azithromycin on whole-cell membrane capacitance in murine thymocytes
In thymocytes, microscopic changes in the cell membrane surface area were best monitored by the measurement of whole-cell membrane capacitance (Cm) (Kazama et al., Citation2012a, Citation2013). Therefore, in the present study, we employed this electrophysiological approach to detect the structural changes in the thymocyte plasma membranes induced by the macrolides (). Numerical changes in the parameter are summarized in . By simply applying the external solution alone to thymocytes (), we confirmed that our procedure of reagent application, a puff application by constant hydrostatic pressure with a nearby pipette, was without significant effects on the Cm and the other parameters, such as Gm and Gs (, ). The inclusion in the pipette of 30 µM clarithromycin did not change the Cm or other parameters (, ). A concentration as high as 100 µM did not change them, either (). However, the inclusion of 30 µM azithromycin induced a significant decrease in Cm immediately after the application (, ), with minimal changes in Gm and Gs. One-hundred µM azithromycin also induced a significant decrease in these parameters (), although ΔCm was not significantly different from that at 30 µM. These results indicated that azithromycin alone induced structural changes in the thymocyte plasma membranes and that the effect was not dose dependent.
Figure 3. Clarithromycin or azithromycin-induced changes in thymocyte membrane capacitance, series and membrane conductance. After establishing the whole-cell configuration, external solutions containing no drugs (A), 30 µM clarithromycin (B) or azithromycin (C) were delivered for 30 s to single thymocytes. Membrane capacitance, series and membrane conductance were monitored for at least 2 min. n = 5 for each trace.
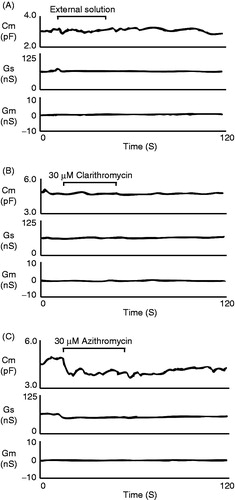
Table 1. Summary of changes in membrane capacitance after application of clarithromycin and azithromycin.
The whole-cell Cm is mathematically calculated from a parallel-plate capacitor formula: Cm = εA/d, where ε is the dielectric modulus of the plasma membrane; A, the membrane surface area and d, the membrane thickness (Fisher et al., Citation2004). Assuming that ε and A are relatively constant before and after the drug application, the increase in d is primarily considered to be responsible for the decrease in Cm (Morris & Homann, Citation2001). Since azithromycin is much more lipophilic than clarithromycin (Gladue et al., Citation1989; Zuckerman, Citation2004), it would disperse more easily into the lipid bilayers of the plasma membrane. Therefore, the azithromycin-induced decrease in the Cm was thought to represent increased membrane thickness (d) as a result of the accumulation of the drug in the plasma membrane. The lack of difference between the doses indicated that the accumulation had already reached a plateau at the concentration of 30 µM. Then, as previously demonstrated in human and mouse polymorphonuclear leukocytes (Gladue et al., Citation1989; Jain & Danziger, Citation2004), the highly lipophilic azithromycin would easily penetrate into the cytoplasm, facilitating its cellular uptake and achieving a higher cellular concentration of this drug.
When it exerts immunomodulatory effects, azithromycin stimulates the production of an inhibitory cytokine, IL-10, and thus suppresses the production of pro-inflammatory cytokines (Conti et al., Citation2003; Sugiyama et al., Citation2007). In contrast to the production of IL-2, which is dependent on the activity of the Kv1.3-channels (Chandy et al., Citation2004; Price et al., Citation1989; Villalonga et al., Citation2010), that of IL-10 is highly dependent on the activity of intracellular signaling molecules, such as signal transducer and activator of transcription 3 (STAT3) (Saraiva & O'Garra, Citation2010). Although additional studies are required to address the issue, we could speculate that the intracellularly accumulated azithromycin may directly activate STAT3 from inside the cells, as other macrolides do in macrophages (Yamauchi et al., Citation2008). Consequently, the production of IL-10 may be stimulated in lymphocytes, ultimately leading to immunomodulation.
Conclusion
In summary, this study demonstrated for the first time that clarithromycin exerts inhibitory effects on thymocyte Kv1.3-channel currents, while azithromycin decreases the membrane capacitance without affecting the channel currents. These effects of the macrolide antibiotics may be associated with differences in their mechanisms of immunomodulation by which they control the production of cytokines.
Declaration of interest
The authors declare no conflicts of interest.
Acknowledgements
We thank Mr. Brent Bell for proofreading the manuscript.
References
- Chandy KG, Wulff H, Beeton C, et al. (2004). K+ channels as targets for specific immunomodulation. Trends Pharmacol Sci 25:280–9
- Conti P, Kempuraj D, Kandere K, et al. (2003). IL-10, an inflammatory/inhibitory cytokine, but not always. Immunol Lett 86:123–9
- Fisher JL, Levitan I, Margulies SS. (2004). Plasma membrane surface increases with tonic stretch of alveolar epithelial cells. Am J Respir Cell Mol Biol 31:200–8
- Gladue RP, Bright GM, Isaacson RE, Newborg MF. (1989). In vitro and in vivo uptake of azithromycin (CP-62,993) by phagocytic cells: Possible mechanism of delivery and release at sites of infection. Antimicrob Agents Chemother 33:277–82
- Gluais P, Bastide M, Grandmougin D, et al. (2003). Clarithromycin reduces Isus and Ito currents in human atrial myocytes with minor repercussions on action potential duration. Fundam Clin Pharmacol 17:691–701
- Jain R, Danziger LH. (2004). The macrolide antibiotics: A pharmacokinetic and pharmacodynamic overview. Curr Pharm Des 10:3045–53
- Kalman K, Pennington MW, Lanigan MD, et al. (1998). ShK-Dap22, a potent Kv1.3-specific immunosuppressive polypeptide. J Biol Chem 273:32697–707
- Kanoh S, Rubin BK. (2010). Mechanisms of action and clinical application of macrolides as immunomodulatory medications. Clin Microbiol Rev 23:590–615
- Kazama I, Maruyama Y, Matsubara M. (2013). Benidipine persistently inhibits delayed rectifier K(+)-channel currents in murine thymocytes. Immunopharmacol Immunotoxicol 35:28--33
- Kazama I, Maruyama Y, Murata Y. (2012a). Suppressive effects of nonsteroidal anti-inflammatory drugs diclofenac sodium, salicylate and indomethacin on delayed rectifier K(+)-channel currents in murine thymocytes. Immunopharmacol Immunotoxicol 34:874–8
- Kazama I, Maruyama Y, Murata Y, Sano M. (2012b). Voltage-dependent biphasic effects of chloroquine on delayed rectifier K(+)-channel currents in murine thymocytes. J Physiol Sci 62:267–74
- Khan AA, Slifer TR, Araujo FG, Remington JS. (1999). Effect of clarithromycin and azithromycin on production of cytokines by human monocytes. Int J Antimicrob Agents 11:121–32
- Kudoh S, Azuma A, Yamamoto M, et al. (1998). Improvement of survival in patients with diffuse panbronchiolitis treated with low-dose erythromycin. Am J Respir Crit Care Med 157:1829–32
- Lewis RS, Cahalan MD. (1995). Potassium and calcium channels in lymphocytes. Annu Rev Immunol 13:623–53
- Matko J. (2003). K+ channels and T-cell synapses: The molecular background for efficient immunomodulation is shaping up. Trends Pharmacol Sci 24:385–9
- Mazzei T, Mini E, Novelli A, Periti P. (1993). Chemistry and mode of action of macrolides. J Antimicrob Chemother 31:1–9
- Mccarty JM. (2000). Clarithromycin in the management of community-acquired pneumonia. Clin Ther 22:281–94; discussion 265
- Morikawa K, Zhang J, Nonaka M, Morikawa S. (2002). Modulatory effect of macrolide antibiotics on the Th1- and Th2-type cytokine production. Int J Antimicrob Agents 19:53–9
- Morris DL, De Souza A, Jones JA, Morgan WE. (1991). High and prolonged pulmonary tissue concentrations of azithromycin following a single oral dose. Eur J Clin Microbiol Infect Dis 10:859–61
- Morris CE, Homann U. (2001). Cell surface area regulation and membrane tension. J Membr Biol 179:79–102
- Price M, Lee SC, Deutsch C. (1989). Charybdotoxin inhibits proliferation and interleukin 2 production in human peripheral blood lymphocytes. Proc Natl Acad Sci USA 86:10171–5
- Saraiva M, O'garra A. (2010). The regulation of IL-10 production by immune cells. Nat Rev Immunol 10:170–81
- Stepanic V, Kostrun S, Malnar I, et al. (2011). Modeling cellular pharmacokinetics of 14- and 15-membered macrolides with physicochemical properties. J Med Chem 54:719--33
- Sugiyama K, Shirai R, Mukae H, et al. (2007). Differing effects of clarithromycin and azithromycin on cytokine production by murine dendritic cells. Clin Exp Immunol 147:540–6
- Villalonga N, David M, Bielanska J, et al. (2010). Immunomodulatory effects of diclofenac in leukocytes through the targeting of Kv1.3 voltage-dependent potassium channels. Biochem Pharmacol 80:858–66
- Volberg WA, Koci BJ, Su W, et al. (2002). Blockade of human cardiac potassium channel human ether-a-go-go-related gene (HERG) by macrolide antibiotics. J Pharmacol Exp Ther 302:320–7
- Yamauchi K, Ishikawa T, Shibata Y, et al. (2008). Enhanced interleukin-10 signaling with 14-member macrolides in lipopolysaccharide-stimulated macrophages. EXCLI J 7:169–76
- Yellen G. (1998). The moving parts of voltage-gated ion channels. Q Rev Biophys 31:239–95
- Zuckerman JM. (2004). Macrolides and ketolides: Azithromycin, clarithromycin, telithromycin. Infect Dis Clin North Am 18:621–49
