Abstract
Breast cancer is a major form of cancer, with a high mortality rate in women. It is crucial to achieve more efficient and safe anticancer drugs. Recent developments in medical nanotechnology have resulted in novel advances in cancer drug delivery. Cisplatin, doxorubicin, and 5-fluorouracil are three important anti-cancer drugs which have poor water-solubility. In this study, we used cisplatin, doxorubicin, and 5-fluorouracil-loaded polycaprolactone-polyethylene glycol (PCL-PEG) nanoparticles to improve the stability and solubility of molecules in drug delivery systems. The nanoparticles were prepared by a double emulsion method and characterized with Fourier Transform Infrared (FTIR) spectroscopy and Hydrogen-1 nuclear magnetic resonance (1HNMR). Cells were treated with equal concentrations of cisplatin, doxorubicin and 5-fluorouracil-loaded PCL-PEG nanoparticles, and free cisplatin, doxorubicin and 5-fluorouracil. The 3-[4,5-dimethylthiazol-2yl]-2,5-diphenyl tetrazolium bromide (MTT) assay confirmed that cisplatin, doxorubicin, and 5-fluorouracil-loaded PCL-PEG nanoparticles enhanced cytotoxicity and drug delivery in T47D and MCF7 breast cancer cells. However, the IC50 value of doxorubicin was lower than the IC50 values of both cisplatin and 5-fluorouracil, where the difference was statistically considered significant (p˂0.05). However, the IC50 value of all drugs on T47D were lower than those on MCF7.
Introduction
The synthesis, characterization, engineering, and use of materials and devices of 100 nanometers or less is called nanotechnology (CitationWang et al. 2012). In cancer therapy, nanotechnology has become a potential and powerful application for the development and progress of nanoparticles as drug delivery systems. Nowadays, nanoparticles have become extremely attractive and applicable for their applications in the fields of medicine and biology (CitationXu et al. 2007, CitationMahapatro and Singh 2011). Nanoparticles are spherical and solid structures prepared from synthetic or natural polymers, and range around 100 nm in size. The delivery of a large group of drugs, like vaccines, biological macromolecules, hydrophobic small drugs, and hydrophilic small drugs, can be managed using nanoparticles . Targeted delivery to particular organs or cells can also be enhanced using nanoparticles (CitationKumari et al. 2010, CitationHillaireau and Couvreur 2009, CitationDanhier et al. 2012). Targeted anticancer drug delivery has the advantages of few side effects and low toxicity, which enhance therapeutic efficacy rates for cancer chemotherapy (CitationFarokhzad and Langer 2009, CitationBailey and Berkland 2009). Among a range of drug carriers, polymer nanoparticles have been more popular, because of their potential and ability for targeted and preferential delivery of drugs to cancer tissues (CitationGhosh et al. 2008, CitationMoghimi et al. 2010). Nanoparticles with a hydrophilic shell, such as polymer micelles with polyethylene glycol (PEG) outer layers, have “stealth” properties, and can thus escape detection by the reticuloendothelial system (RES). They have a delayed circulatory time in the bloodstream, and have enhanced permeation and retention (EPR) effects, which allow their accumulation in solid tumors (CitationTorchilin 2007, CitationBogdanov et al. 1999). The development of a molecular target that will adjust mechanisms of several signaling pathways would be appropriate for anticancer therapy. Cisplatin, doxorubicin, and 5-fluorouracil are three main and important drugs in chemotherapy, which nowadays attract global attention because of their powerful potential in cancer therapy. Cisplatin is one of the chemotherapeutic drugs which are well-known to crosslink DNA molecules in different ways to hinder cell division via mitosis. The main disadvantage of cisplatin is its toxicity in healthy tissues after the full therapeutic exploitation (CitationRosenberg 1985). On other hand, doxorubicin, an anthracycline antibiotic, is one of the most broadly used anticancer agents and shows high anticancer activity. The main disadvantage of doxorubicin is its dependent cardio toxicity and myelosuppression (CitationMisra and Sahoo 2010). 5-fluorouracil is a chemotherapeutic drug, which is known as a pyrimidine analog. In this study, we compare the cell toxicity of cisplatin and doxorubicin in free and polycaprolactone-polyethylene glycol (PCL-PEG)-encapsulated forms.
Materials and methods
PCL (1000), PEG (4000), Polyvinyl alcohol (PVA), stannous octoate (Sn (Oct) 2: stannous 2-ethylhexanoate) and dichloromethane (DCM) were purchased from Sigma-Aldrich (USA). Scanning electron microscopy (SEM) measurements were recorded using the KYKY model EM3200. The drug-loading (DL) capacity was determined using a UV-Vis 2550 spectrometer (Shimadzu). Infrared spectra were recorded at room temperature with Fourier transform infrared spectroscopy (FTIR, Perkin Elmer Series). The Hydrogen-1 nuclear magnetic resonance spectra (1H NMR) were recorded at room temperature (RT) with a Brucker DRX 300 spectrometer operating at 300.13 MHz. The organic phase was vaporized using a rotary (Rotary Evaporator, Heidolph Instruments, Hei-VAP series). A homogenizer (Silent Crusher M, Heidolph Instruments GmbH, Schwabach, Germany) was used to homogenize the samples. MCF7 and T47D breast cell lines were purchased from the Pasteur Institute of Iran. Trypsin-EDTA, RPMI-1640, and fetal bovine serum (FBS) were from Gibco, Invitrogen (UK). We also purchased 0.08 mg penicillin-G (Serva co, Germany), and 2 mg sodium bicarbonate (Merck, Germany), Sigma-Aldrich (USA) supplied 0.2 mg amphotericin B, 10% heat-inactivated fetal bovine serum (FBS) (Gibco, Invitrogen, UK), and 50 mg streptomycin (Merck, Germany) which were was supplemented with one liter of RPMI 1640, MTT (3(4, 5-dimethylthiazol-2-yl) 2, 5-diphenyl-tetrazolium bromide), and DMSO (dimethyl sulfoxide).
Cell culture and cell line
MCF7 and T47D breast cell lines were cultured in RPMI1640 complemented with 0.08 mg/ml of streptomycin (Merck, Germany), 0.05 mg/ml penicillin-G, 10% heat-inactivated FBS, and 2 mg/ml sodium bicarbonate, and the cells were grown at 37°C in an incubator with 55% humidity and 5% CO2.
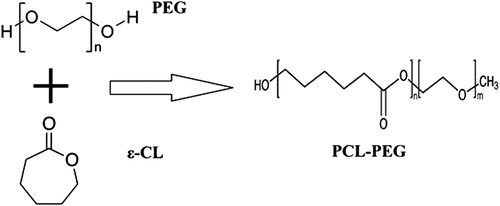
Synthesis and characterization of PCL (1000)-PEG (4000)-PCL (1000) triblock copolymers
The PCL (1000)-PEG (4000)-PCL (1000) triblock copolymers were synthesized by an already reported method, with minor modifications (CitationFeng et al. 2012). The chemical structure is shown in . Briefly, ring- opening polymerization of ε-caprolactone was induced by using PEG as the initiator, and Tin(II) 2-ethylhexanoate or stannous octoate (Sn (Oct)2) as the catalyst. 3gr ethylene glycol and 7.4 gr ε-caprolactone were heated in a dry three-necked flask and under vacuum for 10 min, to a temperature of 130°C and under nitrogen atmosphere, until melting was completed (). The molar proportion of ethylene glycol and ε-caprolactone was 1/2.5. Then, 0.05% (w/w) (about 40λ) of Sn (Oct)2 was added into a three-necked flask under a nitrogen atmosphere, and the temperature of the reaction mixture was increased to 180°C. A heater device was used to reach to 180°C, with the stirring condition continued for 6 h, and polymerization was carried out (CitationAkbarzadeh et al. 2012, CitationValizadeh et al. 2012, CitationAkbarzadeh et al. 2013, CitationMollazade et al. 2013, CitationNejati-Koshki et al. 2013).
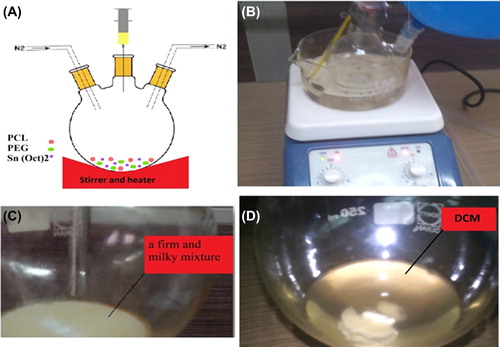
After 12 h of cooling at room temperature, a firm and milky mixture was obtained (). The copolymer was dissolved by dichloromethane (). To achieve precipitation, after 1 h, ice-cold diethyl ether was added under stirring conditions (CitationRezaei-Sadabady et al. 2013, CitationFallahzadeh et al. 2010, CitationEbrahimnezhad et al. 2013, CitationPourhassan-Moghaddam et al. 2013, CitationAhmadi et al. 2014, CitationDavaran et al. 2013, CitationGhasemali et al. 2013, CitationSadat et al. 2014, CitationDavaran et al. 2014). The solution was precipitated after 24 h. The precipitate was stored in a desiccator until it was used (CitationRezaei-Sadabady et al. 2013, CitationFallahzadeh et al. 2010, CitationEbrahimnezhad et al. 2013, CitationPourhassan-Moghaddam et al. 2013, CitationAhmadi et al. 2014, CitationDavaran et al. 2013, CitationGhasemali et al. 2013, CitationSadat et al. 2014, CitationDavaran et al. 2014). The morphology of the nanoparticles was examined using SEM. A Brucker DRX 300 MHz spectrometer was used and the 1H NMR spectra were recorded in CDCI3. The samples were homogenized using a homogenizer (Heidolph Instruments GmbH and Co. KG, SilentCrusher M). The organic phase was evaporated by rotary (Rotary Evaporators, Heidolph Instruments, Hei-VAP Series). The FTIR spectrum was gained from a neat film cast of the chloroform copolymer solution between KBr tablets (CitationKouhi et al. 2014, CitationAbbasi et al. 2014, CitationPourhassan-Moghaddam et al. 2014, CitationHosseininasab et al. 2014, CitationDavoudi et al. 2014, CitationTaheri et al. 2014, CitationAlimirzalu et al. 2014, CitationEatemadi et al. 2014).
Drug loading
Nowadays, there are different techniques for loading the drug into nanoparticles. Combinations of different techniques were used to prepare for the loading. These were methods such as double emulsion (w/o/w), probe type sonication, and solvent evaporation (). PCL-PEG nanoparticles loaded with cisplatin were prepared using the double emulsion method (CitationKarnoosh-Yamchi et al. 2014, CitationAlizadeh et al. 2014, CitationNejati-Koshki et al. 2014, CitationEbrahimi et al. 2014, CitationGhalhar et al. 2014, CitationDaraee et al. 2014, CitationSadat et al. 2014, CitationDaraee et al. 2014). An aqueous solution of cisplatin (0.4 ml) was added and emulsified in 2 ml of dichloromethane (CitationKarnoosh-Yamchi et al. 2014, CitationAlizadeh et al. 2014, CitationNejati-Koshki et al. 2014, CitationEbrahimi et al. 2014, CitationGhalhar et al. 2014, CitationDaraee et al. 2014, CitationSadat et al. 2014, CitationDaraee et al. 2014). Thus, 100 mg of the copolymer was emulsified in the mixture, using probe sonication at 10 W for 45 s. The mixture obtained (w/o emulsion) was added to an aqueous solution of sodium cholate (6 ml), and was then probe sonicated at 18 W for 1 min. The mixture obtained (w/o/w emulsion) was gently stirred at room temperature until the evaporation of the organic phase was complete (CitationNasrabadi et al. 2014, CitationChung et al. 2014, CitationFekri Aval et al. 2014, CitationZohre et al. 2014, CitationValizadeh et al. 2014, CitationMellatyar et al. 2014, CitationDadashzadeh et al. 2014, CitationRahimzadeh et al. 2014, CitationBadrzadeh et al. 2014, CitationHerizchi et al. 2014, CitationKafshdooz et al. 2014, CitationSohrabi et al. 2014, CitationTozihi et al. 2014, CitationShafiei et al. 2014, CitationKordi et al. 2014, CitationBarkhordari et al. 2014, CitationDadashzadeh et al. 2014, CitationZare et al. 2014, CitationKordi et al. 2014, CitationMajidi et al. 2014). To prepare doxorubicin-loaded PEG-PCL, 3 mg of doxorubicin hydrochloride was emulsified in 2 ml of dichloromethane (CH2Cl2) in the presence of a triple molar ratio of triethylamine, at room temperature, and stirred at 400 rpm on a magnetic/heater stirrer for 6 h (CitationNasrabadi et al. 2014, CitationChung et al. 2014, CitationFekri Aval et al. 2014, CitationZohre et al. 2014, CitationValizadeh et al. 2014, CitationMellatyar et al. 2014, CitationDadashzadeh et al. 2014, CitationRahimzadeh et al. 2014, CitationBadrzadeh et al. 2014, CitationHerizchi et al. 2014, CitationKafshdooz et al. 2014, CitationSohrabi et al. 2014, CitationTozihi et al. 2014, CitationShafiei et al. 2014, CitationKordi et al. 2014, CitationBarkhordari et al. 2014, CitationDadashzadeh et al. 2014, CitationZare et al. 2014, CitationKordi et al. 2014, CitationMajidi et al. 2014). Then, 10 mg of PEG-PCL copolymer was added and emulsified into the mixture obtained, and stirred at 400 rpm until it was completely emulsified. Afterwards, the obtaining solvent was added and emulsified drop wise in 20 mL of distilled water. The mixture was sonicated 16 times at intervals, and the 1-min pulse was turned off for 1 s at 15-s intervals. For solvent evaporation, micelle formation, and removal of the residual CH2Cl2, the beaker was left open to air during the night, and using a rotary evaporator, and the residual solvent was removed. The procedure of preparation of 5-flurouracil was the same as that for the preparation of the doxorubicin-loading method, but we used 10 mg of 5-fluorouroacil and 100 mg of polymer. For the elimination of cisplatin, doxorubicin and 5-fluorouracil aggregates, all solution was filtered with a syringe filter (pore size 0.22 m). All the procedures were carried out under vacuum and in a nitrogen atmosphere.
![Figure 3. Preparation of encapsulated drugs. The drugs were loaded in nanoparticles with a combination of different techniques, which are known as w/o/w or water-in-oil-in-water (double emulsion). (A) Water [W], (B) Oil [O], (C) W/O emulsion, (D) W/O/W emulsion, and (E) Schematic structure of encapsulated drugs.](/cms/asset/3ac32b2c-c46d-440b-9370-65cfa48b2fa0/ianb_a_1008510_f0003_oc.jpg)
Determination of entrapment efficiency (EE) and drug loading (DL)
The solution obtained in the last step was centrifuged for 30 min at 10,000 rpm. To calculate the encapsulation efficiency (EE) and the drug-loading (DL) rate of the nano particles, for cisplatin, doxorubicin, and 5-fluorouracil, the supernatant was cut off and used for comparison with the total amount of cisplatin, doxorubicin, and 5-fluorouracil. The amount of non-encapsulated cisplatin, doxorubicin, and 5-fluorouracil in the supernatant was calculated by using an ultraviolet 2550 spectrophotometer (Shimadzu), with an absorbance peak at 301 ± 2 (cisplatin), 254 (doxorubicin), and 266 (5-fluorouracil). The following formula was used to measure the percent of cisplatin, doxorubicin and 5-fluorouracil encapsulated in the nanoparticles (Eq. 1), and the rate of DL (Eq. 2).
Cell lines and culture cmonditions
The human breast cancer cell lines MCF7 and T47D were cultured in a RPMI 1640 culture medium supplemented with 2 mg of sodium bicarbonate, 2 mM L-glutamine, 10% FBS, 0.2 mg of amphotericin B, penicillin-G (80 mg/mL), and streptomycin (50 mg/mL) at 37°C in a 5% humidified CO2 incubator.
Cell viability and MTT-based cytotoxicity test
Cells were exposed to free cisplatin, doxorubicin, and 5-fluorouracil, and cisplatin, doxorubicin, and 5-fluorouracil-loaded PCL-PEG, during the exponential phase of growth, and cytotoxicity was measured at 24-h after treatment using the MTT assay. First, in a 96-well plate (Costar® from Corning, NY) 10000 cells/well were seeded and kept for 24 h in the incubator, to promote cell attachment. Then, both T47D and MCF7 cell lines were treated, in triplicate, with different concentrations of free cisplatin, doxorubicin and 5-fluorouracil, and cisplatin, doxorubicin, and 5-fluorouracil-loaded PCL-PEG (0.312–20 μM for doxorubicin, 5–320 μM for cisplatin, and 6–344 μM for 5-fluorouracil), for 24 h. Three controls were used, namely 1% DMSO and PCL-PEG control, for estimation of nanoparticle effect, and a control of the cells alone. After 24 h of drug exposure, the medium was replaced with 200 μl fresh medium for 24 h. After that, the cells were incubated with 50 μL of 2 mg/ml MTT, which was dissolved in PBS, for 4 h. By this method, the plates were covered with aluminum foil. The content of all wells was removed, and 200 μL of pure DMSO and 25 μL of Sorensen's glycine buffer were added to each well. Finally, an ELISA reader (with a reference wavelength of 630 nm) was used to calculate the absorbance measurement at 570 nm.
Results
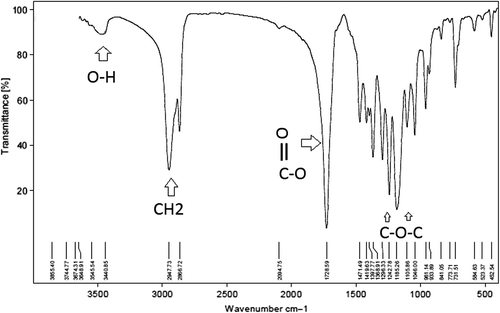
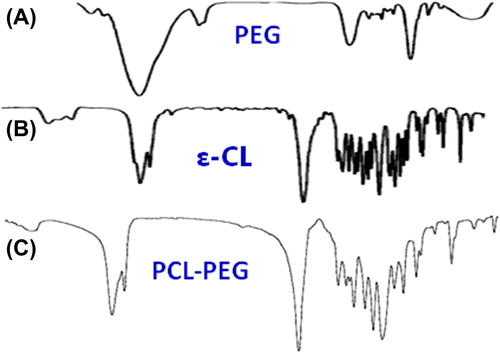
FTIR spectrum of poly caprolactone 1000- poly ethylene glycol 4000- poly caprolactone 1000 (PCL-PEG-PCL) copolymer
The PCL-PEG-PCL co-polymer nanoparticles were successfully perpetrated by ring-opening copolymerization of PEG and ε-CL (). FTIR spectroscopy was done using a Shimadzu spectrophotometer. The FTIR spectrum is compatible with the structure of the presupposed PCL-PEG copolymer. FTIR spectroscopy was used to show the original and main structure of the PCL-PEG copolymer nanoparticle. As illustrated in , due to the coupling reaction of –NCO with the –OH group, there is no absorption at 2250–2270 cm− 1, which means that the –NCO groups of hexamethylene diisocyanate disappeared completely. The ester bond appeared at 1721 cm− 1 with a strong C = O stretching band. The absorption bands at 1528 cm− 1 are attributed to the N–H bending vibrations, which proved the construction of PCL-PEG-PCL triblock copolymers. The terminal hydroxyl groups in the copolymer, from which the PEG homopolymer was removed, was attributed to the appearance of the absorption band at 3509.9 cm− 1. Our FTIR results were compared with other studies, and showed strong similarity of data (CitationFeng et al. 2012). These absorption bands indicated that the PCL-PEG-PCL block copolymer was successfully synthesized (, ).
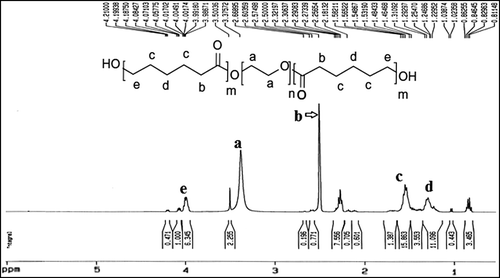
1H NMR spectrum of the PCL- PEG co-polymer
To confirm the basic chemical structure of PCL-PEG-PCL triblock nanoparticles, the 1H-NMR spectrum was recorded at RT, with a Brucker DRX 300 spectrometer operating at 400 MHz (). One of the important features was a large and sharp single peak at 3.65 ppm, corresponding to the methylene groups of the PEG segments. The very weak peak at 4.23 ppm was attributed to the methylene proton of the PEG end unit. In addition, peaks at 4.06, 2.31, 1.65 and 1.38 ppm were corresponding to the methylene protons of OCOCH2CH2CH2CH2–CH2, OCO-CH2–CH2)4, OCOCH2–CH2–CH2–CH2–CH2, and OCOCH2CH2–CH2–CH2CH2 in the PCL segments, respectively. Our data was very similar to the reported spectra (CitationFeng et al. 2012, CitationJia et al. 2008). All the FTIR and 1H-NMR results showed that the PCL-PEG-PCL triblock copolymer was synthesized successfully.
Entrapment efficiency (EE) and drug loading (DL)
The cisplatin, doxorubicin and 5-fluorouracil content in the drug-loaded nanoparticles were determined using the ultraviolet spectrophotometer at 301 ± 2, 254 and 266 nm, respectively. The DL content and drug EE were calculated based on the Equations 1 and 2, by which the values for the EE for cisplatin, doxorubicin, and 5-fluorouracil were 98.8%, 99.1%, and 98.9%, respectively. The DL for cisplatin, doxorubicin, and 5-fluorouracil were 13.53%, 16.12%, and 14.24%, respectively.
Cell cytotoxicity (MTT assay)
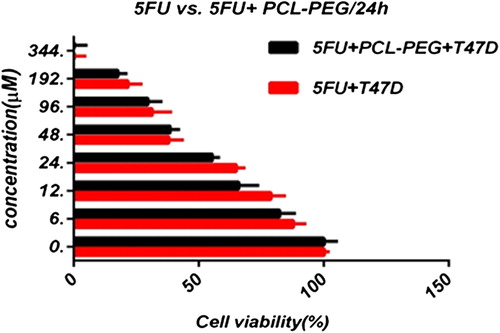
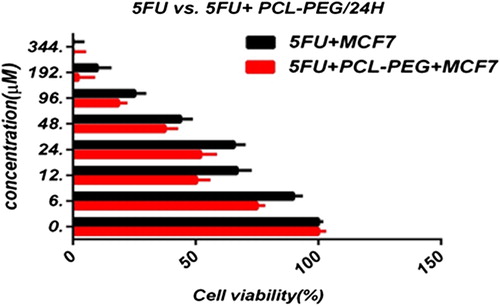
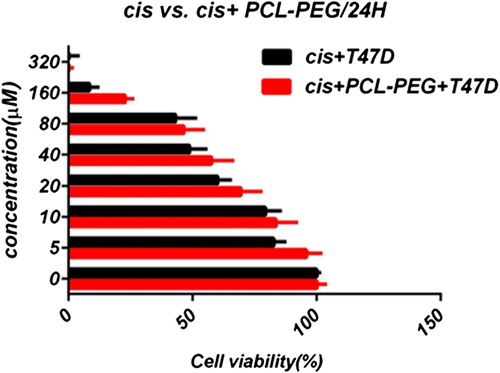
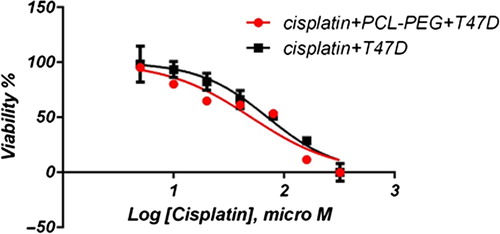
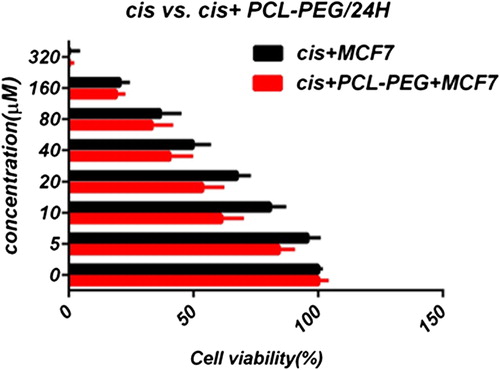
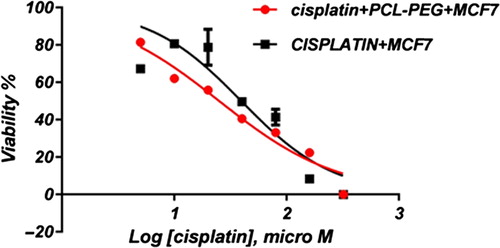
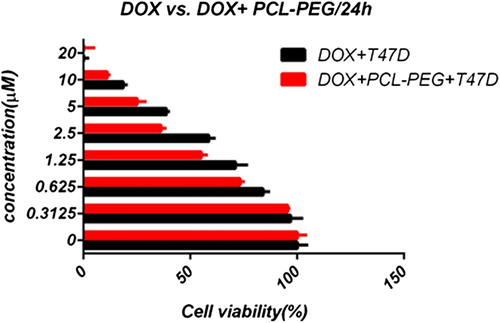
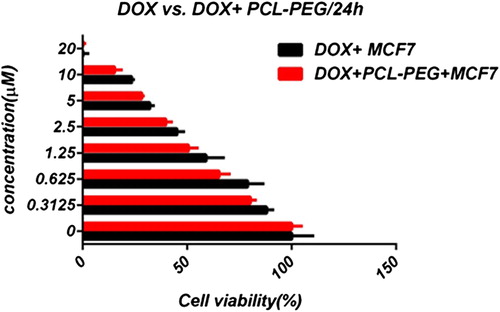
In this study, cell viability was evaluated by the MTT assay, by exposing the T47D and MCF7 cell lines to different concentrations of pure cisplatin, doxorubicin, and 5-fluorouracil, and cisplatin, doxorubicin, and 5-fluorouracil-loaded PCL-PEG, for 24 h. By increasing the amount of drug, the cell toxicity outcome was boosted, which demonstrates how cisplatin, doxorubicin, and 5-fluorouracil are dose-dependent and the cell viability is inversely proportional. The pure cisplatin, doxorubicin and 5-fluorouracil had a cytotoxic effect on T47D and MCF7 cell lines. The values for inhibitory concentration at 50% (IC50), for free cisplatin, doxorubicin, and 5-fluorouracil on T47D, were 70.91, 3.001 and 28.46 for 24 h, respectively. The IC50 values of encapsulated cisplatin, doxorubicin, and 5-fluorouracil on T47D were 50.32, 1.626 and 39.11 for 24 h, respectively (, , , , , and ). The IC50 values of free cisplatin, doxorubicin, and 5-fluorouracil on MCF7 were 44.17, 2.286 and 33.65 for 24 h, respectively (). The values for IC50 of encapsulated cisplatin, doxorubicin, and 5-fluorouracil on MCF7 were 26.39, 1.23 and 23.79 for 24 h, respectively (, , , , , and ).
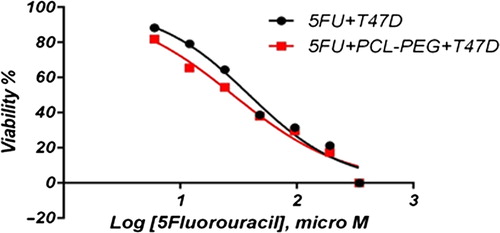
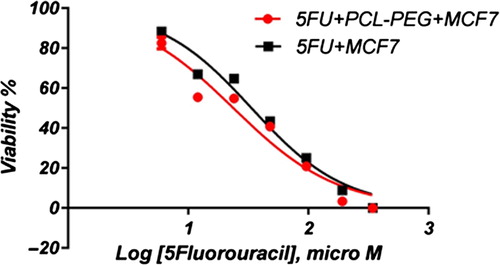
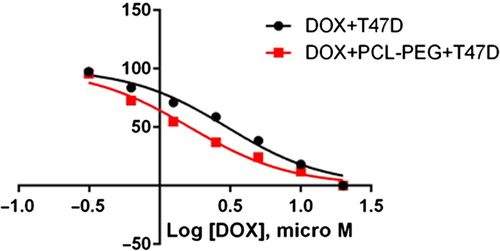
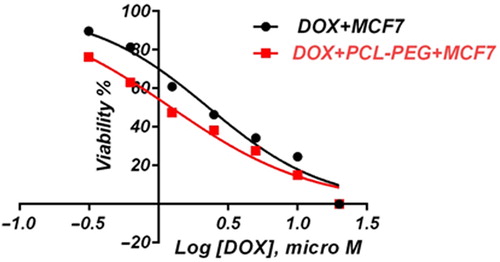
PCL-PEG and DMSO showed an absorbance value equivalent to 99% and 98% of control for MCF7, and 98% and 97% for T47D, respectively. It shows that DMSO and PCL-PEG have very low effect on the cells. The time-dependency of this drug was suggested by the lack of similarity between results for different periods. The graph was plotted using PRISM 4. The statistical significances were evaluated by the t-test, and a value of p ˂ 0.05 was considered significant.
Discussion
Breast cancer is the main cause of the high cancer-related mortality rate in women. The delivery of anticancer drugs for cancer therapy may involve direct delivery into the tumor. Nowadays, chemotherapy is the main method for cancer therapy, but it always has many different side effects. While in the treatment of human cancer, chemotherapy has toxic side effects in healthy and cancerous tissues, nanotechnology attempts to resolve these problems by encapsulating or loading drugs in material which can deliver drugs directly to cancerous tissues (CitationEatemadi et al. 2014, CitationEatemadi et al. 2014, CitationDaraee et al. 2014, CitationDaraee et al. 2014, CitationSeidi et al. 2014). By synthesis of a core-shell nanoparticle formulation of chemotherapeutic agents, the delivery of an efficient agent to where it is needed can be easier and the efficiency of treatment can be enhanced, and it may also be helpful for the treatment of tumors. By this method, the selective delivery of cisplatin, doxorubicin, and 5-fluorouracil to tumor cells would considerably reduce drug toxicity, and improve their therapeutic index. As discussed above, co-polymers (such as PCL-PEG, PLGA-PEG, and PLA-PEG) promise to be a successful system for the targeted and controlled release of cisplatin, doxorubicin, and 5-fluorouracil, with decreased systemic toxicity, increased therapeutic efficiency, and patient compliance. This study explored the cell toxicity effect of the three main chemotherapeutic drugs (cisplatin, doxorubicin, and 5-fluorouracil) on two cell lines attributed to breast cancer (T47D and MCF7) to obtain appropriate data for comparison. The results suggest that drug-loaded PCL-PEG is a suitable candidate for chemotherapy. The entrapment of cisplatin, doxorubicin, and 5-fluorouracil within a nanoparticulate carrier noticeably improved the IC50 and cell toxicity parameters of cisplatin, doxorubicin, and 5-fluorouracil. Also, either free or encapsulated doxorubicin had a cytotoxic effect at a minimum amount on MCF7 and T47D, as compared with free and encapsulated cisplatin and 5-fluorouracil, where the difference was statistically considered significant (p ˂ 0.05). The free and encapsulated cisplatin had higher IC50 values than doxorubicin and 5-fluorouracil, where the difference was statistically considered significant (p ˂ 0.05). Also, all IC50 values of the free and encapsulated drugs in T47D were lower than those for MCF7, and this result suggests that T47D is more sensitive than MCF7, where the difference was statistically considered significant (p ˂ 0.05).
Authors’ contributions
AE conceived of the study and participated in its design and coordination. AA participated in the sequence alignment and drafted the manuscript. All authors read and approved the final manuscript.
Acknowledgments
The authors thank the Department of Medical Nanotechnology, Faculty of Advanced Medical Science of Tabriz University, for all support provided. This work is funded by a 2014 grant by the Drug Applied Research Center, Tabriz University of Medical Sciences. This work is funded by a 2014 grant by the Drug Applied Research Center, Tabriz University of Medical Sciences, and Student Research Committee.
Declaration of interest
The authors have no declaration of interest. The authors alone are responsible for the content and writing of the paper.
References
- Abbasi E, Akbarzadeh A, Kouhi M, Milani M. 2014. Graphene: Synthesis, Bio-Applications, and Properties. Artif Cells Nanomed Biotechnol. 30:1–7.
- Abbasi E, Fekri Aval S, Akbarzadeh A, Milani M, Nasrabadi HT, Hanifepour Y, Nejati-Koshki K, Pashaei-Asl R. 2014. Dendrimers: synthesis, applications, and properties. Nanoscale Res Lett. 9:247.
- Abbasi E, Milani M, Fekri Aval S, Kouhi M, Akbarzadeh A, Nasrabadi HT. 2014. Silver nanoparticles: synthesis, properties, bio-applications and limitations. Crit Rev Microbiol. 17:1–8.
- Ahmadi A, Shirazi H, Pourbagher N, Akbarzadeh A, Omidfar K. 2014. An electrochemical immunosensor for digoxin using core-shell gold coated magnetic nanoparticles as labels. Mol Biol Rep. 41: 1659–1668.
- Akbarzadeh A, Mikaeili H, Asgari D, Zarghami N, Mohammad R, Davaran S. 2012. Preparation and in-vitro evaluation of doxorubicin-loaded Fe3O4 magnetic nanoparticles modified with biocompatible copolymers, Int J Nanomedicine. 7 511–526.
- Akbarzadeh A, Mohamad S, Davaran S. 2012. Magnetic Nanoparticles: Preparation, Physical Properties and Applications in biomedicine, Nanoscale Res Lett. 7:144.
- Akbarzadeh A, Rezaei-Sadabady R, Davaran S, Joo SW, Zarghami N, Hanifehpour Y, et al. 2013. Liposome: Classification, Preparation, and Applications. Nanoscale Res Lett. 8:102.
- Akbarzadeh A, Samiei M, Joo SW, Anzaby M, Hanifehpour Y, Nasrabadi HT, Davaran S. 2012. Synthesis, characterization and in vitro studies of doxorubicin-loaded magnetic nanoparticles grafted to smart copolymers on A549 lung cancer cell line, J Nanobiotechnology. 10:46.
- Akbarzadeh A, Zarghami N, Mikaeili H, Asgari D, Goganian AM, Khiabani HK, Davaran S. 2012. Synthesis, characterization and in vitro evaluation of novel polymer-coated magnetic nanoparticles for controlled delivery of doxorubicin. Nanotechnol Sci Appl. 5 13–25.
- Alimirzalu S, Akbarzadeh A, Nejati-Koshki K, Nasrabadi HT, Akbarzadeh A, Khandaghi AA, Abbasian M, Alimohammadi S. 2014. Synthesis and study of physicochemical characteristics of Fe3O4 magnetic nanocomposites based on poly (Nisopropylacrylamide)for anti-cancer drugs delivery. Asian Pac J Cancer Prev. 15:49–54.
- Alizadeh E, Akbarzadeh A, Zarghami N, Eslaminejad MB, Hashemzadeh S, Nejati-Koshki K. 2014. Up-regulation of Liver enriched Transcription Factors (HNF4a and HNF6) and Liver Specific MicroRNA (miR-122) by Inhibition of Let-7b in Mesenchymal Stem Cells. Chem Biol Drug Des. 85:600–608.
- Alizadeh E, Zarghami N, Eslaminejad MB, Akbarzadeh A, Barzegar A, Mohammadi SA. 2014. The Effect of Dimethyl Sulfoxide (DMSO) on Hepatic Differentiation of Mesenchymal Stem Cells. Artif Cells Nanomed Biotechnol. 30:1–8.
- Badrzadeh F, Akbarzadeh A, Zarghami N, Yamchi MR, Zeighamian V, Tabatabae FS, Taheri M, Kafil HS. 2014. Comparison between Effects of Free Curcumin and Curcumin Loaded NIPAAm-MAA Nanoparticles on Telomerase and PinX1 Gene Expression in Lung Cancer Cells. Asian Pac J Cancer Prev. 15:8931–8936.
- Badrzadeh F, Rahmati-Yamchi M, Badrzadeh K, Valizadeh A, Zarghami N, Farkhani SM, Akbarzadeh A. 2014. Drug delivery and nanodetection in lung cancer. Artif Cells Nanomed Biotechnol. 11:1–17.
- Bailey MM, Berkland CJ. 2009. Fluorinated copolymer nanoparticles for multimodal imaging applications. Med Res Rev. 29:196–212.
- Barkhordari A, Rahmati Yamchi M, Fekri S, Pourhassan-Moghaddam M, Nejati Koshki K, Davaran S, Akbarzadeh A, Nasiri M, Zarghami N. 2014. Study of inhibitory effect of Helenalin on hTERT gene expression in breast cancer cell line by Real-time PCR, Bio infopublications. In Press
- Bogdanov Jr, A, Marecos E, Cheng H, Chandrasekaran L, Krutzsch H, Roberts D, Weissleder R. 1999. Urinary 8-hydroxy-2’-deoxyguanosine, a metabolite of oxidized DNA, is not elevated in HIV patients on combination antiretroviral therapy. Neoplasia. 1:438–445.
- Chung JH, Kim YK, Kim KH, Kwon TY, Vaezmomeni SZ, Samiei M, et al. 2014. Synthesis, characterization, biocompatibility of hydroxyapatite-natural polymers nanocomposites for dentistry applications. Artif Cells Nanomed Biotechnol. 11:1–8.
- Dadashzadeh K, Milani M, Rahmati M, Akbarzadeh A. 2014. Real-Time PCR Detection of 16S rRNA Novel Mutations Associated with Helicobacter pylori Tetracycline Resistance in Iran. Asian Pac J Cancer Prev. 15:8883–8886.
- Danhier F, Ansorena E, Silva JM, Coco R, Le Breton A, Préat V. 2012. PLGA-based nanoparticles: an overview of biomedical applications. J Control Release. 161:505–522.
- Daraee H, Eatemadi A, Abbasi E, Fekri Aval S, Kouhi M, Akbarzadeh A. 2014. Application of gold nanoparticles in biomedical and drug delivery. Artif Cells Nanome Biotechnol. 1–13.
- Daraee H, Eatemadi A, Abbasi E, Fekri Aval S, Kouhi M, Akbarzadeh A. 2014. Application of gold nanoparticles in biomedical and drug delivery. Artif Cells Nanomed Biotechnol. 1–13.
- Daraee H, Etemadi A, Kouhi M, Alimirzalu S, Akbarzadeh A. 2014. Nanofiber: Synthesis and biomedical applications. Artif Cells Nanomed Biotechnol. 1–11.
- Daraee H, Etemadi A, Kouhi M, Alimirzalu S, Akbarzadeh A. 2014. Application of liposomes in medicine and drug delivery. Artif Cells Nanomed Biotechnol. 15:1–11.
- Davaran S, Akbarzadeh A, Nejati-Koshki K, Alimohammadi S, Ghamari MF, Soghrati MM, Rezaei A, Khandaghi AA. 2013. In vitro studies of NIPAAM-MAA-VP copolymer-coated magnetic nanoparticles for controlled anticancer drug release, JEAS. 3:108–115.
- Davaran S, Rezaei A, Alimohammadi S, Khandaghi AA, Nejati-Koshki K, Nasrabadi HT, Akbarzadeh A. 2014. Synthesis and Physicochemical Characterization of Biodegradable star-shaped poly lactide-co-glycolide– β -cyclodextrin copolymer Nanoparticles Containing Albumin. Advances in Nanoparticles. 3:14–22.
- Davoudi Z, Akbarzadeh A, Rahmatiyamchi M, Movassaghpour AA, Alipour M, Nejati-Koshki K, et al. 2014. Molecular Target Therapy of AKT and NF-kB Signaling Pathways and Multidrug Resistance by Specific Cell Penetrating Inhibitor Peptides in HL-60 Cells. Asian Pac J Cancer Prev. 15:4353–4458.
- Eatemadi A, Daraee H, Karimkhanloo H, Kouhi M, Zarghami N, Akbarzadeh A, Abasi M, Hanifehpour Y, Joo SW. 2014. Nanoscale Res Lett. 9:1–13.
- Eatemadi A, Daraee H, Karimkhanloo H, Kouhi M, Zarghami N, Akbarzadeh A, Hanifehpour Y, Joo SW. 2014. Carbon Nanotubes: Properties, Synthesis, Purification, and Medical Applications. Nanoscale Res Lett. 9:393.
- Eatemadi A, Daraee H, Zarghami N, Melat Yar H, Akbarzadeh A. 2014. Artif Cells Nanomed Biotechnol. 1–11.
- Eatemadi A, Daraee H, Zarghami N, Yar HM, Akbarzadeh A, Hanifehpour Y. 2014. Nanofiber: Synthesis and Biomedical Applications. Artif Cells Nanomed Biotechnol. 6:1–11.
- Ebrahimi E, Abbasi E, Akbarzadeh A, Khandaghi AA, Davaran S. 2014. Novel Drug Delivery System Based on Doxorubicin-Encapsulated Magnetic Nanoparticles modified with PLGA-PEG1000 copolymer. Artif Cells Nanomed Biotechnol. 11:1–8.
- Ebrahimi E, Khandaghi AA, Valipour F, Babaie S, Asghari F, Motaali S, et al. 2014. In vitro study and characterization of doxorubicin-loaded magnetic nanoparticles modifi ed with biodegradable copolymers. Artif Cells Nanomed Biotechnol. Early Online: 1–9.
- Ebrahimnezhad Z, Zarghami N, Keyhani M, Amirsaadat S, Akbarzadeh A, Rahmati M, Mohammad Taheri Z, Nejati-Koshki K. 2013. Inhibition of hTERT Gene Expression by Silibinin-Loaded PLGA-PEG-Fe3O4 in T47D Breast Cancer Cell Line. BioImpacts. 3:67–74.
- Fallahzadeh S, Bahrami H, Akbarzadeh A, Tayarani M. 2010. High-isolation dual-frequency operation patch antenna using spiral defected microstrip structure. Antennas and Wireless Propagation Letters, IEEE. 9:122–124.
- Farokhzad OC, Langer R. 2009. Formulation/preparation of functionalized nanoparticles for in vivo targeted drug delivery. ACS Nano. 3:16–20.
- Fekri Aval S, Akbarzadeh A, Yamchi MR, Zarghami F, Nejati-Koshki K, Zarghami N. 2014. Gene silencing effect of SiRNA-magnetic modified with biodegradable copolymer nanoparticles on hTERT gene expression in lung cancer cell line. Artif Cells Nanomed Biotechnol. 14:1–6.
- Feng R, Song Z, Zhai G. 2012. Exploration on the underlying mechanism of female predominance in spasmodic dysphonia: an anatomical study of nodose ganglion in rats. Int J Nanomed. 7:4089.
- Ghalhar MG, Akbarzadeh A, Rahmati M, Mellatyar H, Dariushnejad H, Zarghami N, Barkhordari A. 2014. Comparison of Inhibitory Effects of 17-AAG Nanoparticles and Free 17-AAG on HSP90 Gene Expression in Breast Cancer. Asian Pac J Cancer Prev. 15:7113–7118.
- Ghasemali S, Nejati-Koshki K, Akbarzadeh A, Tafsiri E, Zarghami N, Rahmati-Yamchi M, et al. 2013. Study of Inhibitory Effect of β-Cyclodextrin-HelenalinComplex on HTERT Gene Expression in T47D Breast Cancer Cell Line by Real TimeQuantitative PCR (q-PCR), Asian Pac J Cancer Prev. 14:6949–6953.
- Ghosh P, Han G, De M, Kim CK, Rotello VM. 2008. Nanoparticulated quercetin in combating age related cerebral oxidative injury. Adv Drug Deliv Rev. 60:1307–1315.
- Herizchi R, Abbasi E, Milani M, Akbarzadeh A. 2014. Current methods for synthesis of gold nanoparticles. Artif Cells Nanomed Biotechnol. 3:1–7.
- Hillaireau H, Couvreur P. 2009. Nanocarriers’ entry into the cell: relevance to drug delivery. Cell Mol Life Sci. 66:2873–2896.
- Hosseininasab S, Pashaei-Asl R, Khandaghi AA, Nasrabadi HT, Nejati-Koshki K, Akbarzadeh A, et al. 2014. Synthesis, characterization, and In vitro studies of PLGA-PEG nanoparticles for oral Insulin delivery. Chem Biol Drug Design. 84:307–315.
- Jia WJ, Gu YC, Gou ML, Dai M, Li XY, Kan B, Yang JL, Song QF, Wei YQ, Qian ZY. 2008. Trends in the exploration of anticancer targets and strategies in enhancing the efficacy of drug targeting. Drug Deliv. 15:409–416.
- Kafshdooz T, Kafshdooz L, Akbarzadeh A, Hanifehpour Y, Joo SW. 2014. Applications of nanoparticle systems in gene delivery and gene therapy. Artif Cells Nanomed Biotechnol. 3:1–7.
- Karnoosh-Yamchi J, Mobasseri M, Akbarzadeh A, Davaran S, Ostad-Rahimi AR, Hamishehkar H, et al. 2014. Preparation of pH sensitive insulin-loaded Nano hydrogels and evaluation of insulin releasing in different pH conditions. Molecular Biol Rep. 41: 6705–6712.
- Kordi S, Zarghami N, Akbarzadeh A, Rahmati YM, Ghasemali S, Barkhordari A, Tozihi M. 2014. A comparison of the inhibitory effect of nano-encapsulated helenalin and free helenalin on telomerase gene expression in the breast cancer cell line, by real-time PCR. Artif Cells Nanomed Biotechnol. 1:1–9.
- Kordi S, Zarghami N, Rahmati YM, Ghasemali S, Tozihi M, Nejati- Koshki K, Akbarzadeh A. 2014. The Comparison of inhibitory effect of nanocapsolated Helenalin and free Helenalin on Telomerase gene expression in breast cancer cell line by Real-time PCR. Molecular Biology Reports. Artif Cells Nanomed Biotechnol. 2014; Early Online: 1–17.
- Kouhi M, Vahedi A, Akbarzadeh A, Hanifehpour Y, Joo SW. 2014. Investigation of Quadratic Electro-Optic Effects and Electro Absorption Process in GaN/AlGaN Spherical Quantum Dot. Nanoscale Res Lett. 9:131
- Kumari A, Yadav SK, Yadav SC. 2010. Metabolic Syndrome and Altered Gut Microbiota in Mice Lacking Toll-Like Receptor 5. Colloids Surf B Biointerfaces. 75:1–18.
- Mahapatro A, Singh DK. 2011. Biodegradable nanoparticles are excellent vehicle for site directed in-vivo delivery of drugs and vaccines. J. Nanobiotechnol. 9:1–11.
- Majidi S, Zeinali Sehrig F, Farkhani SM, Soleymani Goloujeh M, Akbarzadeh A. 2014. Current methods for synthesis of magnetic nanoparticles. Artif Cells Nanomed Biotechnol. 1:1–13.
- Mellatyar H, Akbarzadeh A, Rahmati M, Ghalhar MG, Etemadi A, Nejati-Koshki K, Zarghami1 N, Barkhordari A. 2014. Comparison of Inhibitory Effect of 17-DMAG Nanoparticles and Free 17-DMAG in HSP90 Gene Expression in Lung Cancer. Asian Pac J Cancer Prev. 15(20):8693–8698.
- Misra R, Sahoo SK. 2010. Cancer nanotechnology: application of nanotechnology in cancer therapy. Eur J Pharm Sci. 39:152–163.
- Moghimi SM, Andersen AJ, Hashemi SH, Lettiero B, Ahmadvand D, Hunter A, Andresen TL, Hamad I, Szebeni J. 2010. Distinct polymer architecture mediates switching of complement activation pathways at the nanosphere-serum interface: implications for stealth nanoparticle engineering. J Control Release. 146: 175–181.
- Mollazade M, Nejati-Koshki K, Akbarzadeh A, Hanifehpour Y, Zarghami N, Joo SW. 2013. PAMAM Dendrimers Arugment Inhibitory Effect of Curcumin on Cancer Cell Proliferation: Possible Inhibition of Telomerase. Asian Pac J Cancer Prev. 14:6925–6928.
- Nasrabadi HT, Abbasi E, Davaran S, Kouhi M, Akbarzadeh A. 2014. Bimetallic nanoparticles: Preparation, properties, and biomedical applications. Artif Cells Nanomed Biotechnol. 9:1–5.
- Nejati-Koshki K, Akbarzadeh A, Pourhasan-Moghadam M, Abhari A, Dariushnejad H. 2013. Inhibition of Leptin and Leptin Receptor Gene Expression by Silibinin- Curcumin Combination. Asian Pac J Cancer Prev. 14:6595–6599.
- Nejati-Koshki K, Mesgari M, Ebrahimi E, Abbasalizadeh F, Fekri Aval S, Khandaghi AA, Abasi M, Akbarzadeh A. 2014. Synthesis and In-vitro study of Cisplatin-loaded Fe3O4 Nanoparticles Modified with PLGA-PEG6000 Copolymers in Treatment of Lung Cancer. J Microencapsul. 31:815–823.
- Pourhassan-Moghaddam M, Rahmati-Yamchi M, Akbarzadeh A, Daraee H, Nejati-Koshki K, Hanifehpour Y, Joo SW. 2013. Protein detection through different platforms of immuno-loop-mediated isothermal amplification. Nanoscale Res Lett. 8:485.
- Pourhassan-Moghaddam M, Zarghami N, Mohsenifar A, Rahmati-Yamchi M, Gholizadeh D, Akbarzadeh A, de la Guardia M, Nejati-Koshki K. 2014. Watercress-based gold nanoparticles: biosynthesis, mechanism of formation and study of their biocompatibility in vitro. IET Digital Library. 9:345–350.
- Rahimzadeh A, Sadat TMF, Movassaghpour A, Shamsasenjan K, Kariminekoo S, Talebi M, et al. 2014. Biotechnological and biomedical applications of mesenchymal stem cells as a therapeutic system. Artif Cells Nanomed Biotechnol. 23:1–12.
- Rezaei-Sadabady R, Zarghami N, Barzegar A, Eidi A, Akbarzadeh A, Rezaei-Tavirani M. 2013. Studies of the Relationship between Structure and Antioxidant Activity in Interesting Systems, Including Tyrosol, Hydroxytyrosol Derivatives Indicated by Quantum Chemical Calculations. Soft. 2:13–18.
- Rosenberg B. 1985. How nonprofit organizations can develop comprehensive in-home support services by utilizing various funding streams. Cancer. 55:2303–2316.
- Sadat TMF, Akbarzadeh A, Milani M, Zarghami N, Taheri-Anganeh M, Zeighamian V, Badrzadeh F, Rahmati-Yamchi M. 2014. A Comparison between the cytotoxic effects of pure curcumin and curcumin-loaded PLGA-PEG nanoparticles on the MCF-7 human breast cancer cell line. Artif Cells Nanomed Biotechnol. 17:1–8.
- Sadat TMF, Nejati-Koshki K, Akbarzadeh A, Yamchi MR, Milani M, Zarghami N, et al. 2014. PLGA-Based Nanoparticles As Cancer Drug Delivery Systems, APJCP. Asian Pac J Cancer Prev.15:517–535.
- Seidi K, Eatemadi A, Mansoori B, Jahanban-Esfahlan R, Farajzadeh D. 2014. J AIDS Clin Res. 5.
- Shafiei A, Hosseini RH, Boroumand MA, Ziaee S, Zarghami N, Akbarzadeh A. 2014. The Association between Serum KALRN Levels with Polymorphism Gene KALRN (rs9289231) with Risk of Early-Onset Coronary Artery Disease (CAD). Mol Biol Rep.
- Sohrabi N, Valizadeh A, Farkhani SM Akbarzadeh A. 2014. Basics of DNA biosensors and cancer diagnosis. Artif Cells Nanomed Biotechnol. 13:1–10.
- Taheri M, Sadat TMF, Izadi M, Zeighamian V, Badrzadeh F, Salehi R, et al. 2014. The comparison between effects of free curcumin and curcumin loaded PLGA-PEG on telomerase and TRF1 expressions in calu-6 lung cancer cell line. Int J Biosci. 4:134–145.
- Torchilin VP. 2007. Tat peptide-mediated intracellular delivery of pharmaceutical nanocarriers. Pharm Res. 24:1–16.
- Tozihi M, Zarghami N, Rahmati M, Nejati-Koshki K, Akbarzadeh A, Mohamadian J, Frahadi B, Kordi Sh. 2014. CDH1 and FGFR2 gene Polymorphisms in Breast Cancer Patients. Int J Biosci (IJB). InPress
- Valizadeh A, Bakhtiary M, Akbarzadeh A, Salehi R, Frakhani SM, Ebrahimi O, Rahmati-yamchi M, Davaran S. 2014. Preparation and characterization of novel electrospun poly(e-caprolactone)-based nanofibrous scaffolds. Artif Cells Nanomed Biotechnol.Early Online: 1–6.
- Valizadeh A, Mikaeili H, Samiei M, Farkhani SM, Zarghami N, Kouhi M, Akbarzadeh A, Davaran S. 2012. Quantum dots: synthesis, bioapplications, and toxicity. Nanoscale res lett. 7:480.
- Wang AZ, Langer R, Farokhzad OC. 2012. 2b-RAD: a simple and flexible method for genome-wide genotyping. Annu Rev Med. 63:185–198.
- Xu T, Zhang N, Nichols HL, Shi D, Wen X. 2007. Zinc, cadmium, and cobalt interreplacement and relative use efficiencies in the coccolithophore Emiliania huxleyi. Mater Sci Eng C. 27: 579–594.
- Zare M, Soleimani M, Akbarzadeh A, Bakhshandeh B, Aghaee-Bakhtiari SH, Mohammadian M, Zarghami N. 2014. A Novel Protocol to Differentiate Induced Pluripotent Stem Cells by Neuronal microRNAs to Provide a Suitable Cellular Model. Chem Biol Drug Des. doi: 10.1111/cbdd.12485.
- Zare M, Soleimani M, Mohammadian M, Akbarzadeh A, Havasi P, Zarghami N. 2014. Efficient biotechnological approach for lentiviral transduction of induced pluripotent stem cells. Artif Cells Nanomed Biotechnol. 25:1–6.
- Zohre S, Kazem NK, Abolfazl A, Mohammad RY, Aliakbar M, Effat A, et al. 2014. Trichostatin A-induced Apoptosis is Mediated by Kruppel-like Factor 4 in Ovarian and Lung Cancer. Asian Pac J Cancer Prev. 15:6581–6.