Abstract
Objective:
To examine the effect of antipsychotic medication half-life on the risk of psychiatric hospital admission and emergency department (ED) visits among adults with schizophrenia.
Methods:
Retrospective claims-based cohort study of adult Medicaid patients with schizophrenia who were prescribed second-generation antipsychotic monotherapy following hospital discharge between 1/1/04 and 12/31/06. Cox proportional hazards models were applied to compare adjusted hazards of mental disorder admission among patients treated with oral antipsychotics that have either a long [risperidone (t1/2 = 20 h), olanzapine (t1/2 = 30 h), aripiprazole (t1/2 = 75 h)] (n = 1479) or short [quetiapine (t1/2 = 6 h), ziprasidone (t1/2 = 7 h)] (n = 837) half-life. Day-level models controlled for baseline background characteristics and antipsychotic adherence over time as measured by gaps in the prescription record. Similar analyses examined either hospitalization or ED visits as separate endpoints.
Results:
A significantly lower rate of hospitalization/ED visits was evident for long (0.74/patient-year) vs short (1.06/patient-year) half-life antipsychotics (p < 0.001). The unadjusted rate of hospitalization alone was significantly lower for long (0.38/patient-year) vs short (0.52/patient-year) half-life antipsychotics (p = 0.005). Compared with short half-life antipsychotic drugs, the adjusted hazard ratio associated with long half-life medications was 0.77 (95% CI = 0.67–0.88) for combined hospitalization/ED visits and 0.80 (95% CI = 0.67–0.96) for hospitalization. The corresponding number needed to treat with long, rather than short, half-life medications to avoid one hospitalization was 16 patients for 1 year and to avoid one hospitalization or ED visit was 11 patients for 1 year.
Limitations:
This study demonstrated an association between antipsychotic medication half-life and hospitalization, not a causal link. Patients using long half-life medications had fewer comorbid mental health conditions and took fewer psychiatric medications at baseline. Other unmeasured differences may have existed between groups and may partially account for the findings.
Conclusions:
In schizophrenia management, longer-acting second-generation antipsychotics were associated with a lower risk of hospital admission/ED visits for mental disorders.
Introduction
Despite the introduction of multiple therapeutic options to treat schizophrenia, medication non-adherence remains a serious problem. Nearly half of patients with schizophrenia become non-adherent to their medication regimen during the course of 2 yearsCitation1. Medication adherence is a key determinant of clinical outcome, with lower adherence correlating with higher relapse rates, greater use of inpatient services, poorer function, and higher risks of substance abuse and criminal arrestCitation2–7.
A wide variety of programmatic, psychosocial, and pharmacologic interventions have been developed to improve medication adherence in patients with schizophrenia. Although there are successful programmatic interventions, they are resource-intensive and pose organizational challenges to implementCitation8. Psychosocial interventions, such as cognitive-behavioral therapy, have also shown promise, but require expert training that is in short supplyCitation9. To date, pharmacologic approaches to improving adherence have focused primarily on the use of long-acting parenteral antipsychotic formulations. Because of attitudinal and logistical barriers, however, their use in the US has been limitedCitation10. Longer-acting oral formulations of antipsychotics may share some of the adherence advantages of long-acting parenteral medications, while posing fewer obstacles to acceptance.
Second-generation antipsychotics (SGAs) differ from first-generation antipsychotics in their mechanisms of action as well as their pharmacokinetics, with half-lives that vary from 6–75 hCitation11–15. The effects of antipsychotics on brain chemistry are complex, and half-life is only one component of a complex interplay of factors controlling the onset and offset of these effects. Indeed, as a result of changes at the receptor level in the brain, therapeutic effects may occur over weeks to months. However, a drug with a longer half-life will remain in the circulation longer after the last dose, potentially resulting in oral antipsychotics with longer half-lives being less impacted by partial non-adherence than those with shorter half-lives. The drop in SGA blood levels caused by missed doses might be bridged more effectively by longer half-life SGAs. As a result, in partially adherent schizophrenia patients, long half-life (LHL) SGAs might prevent hospitalization more effectively than short half-life (SHL) drugs. This study tested this hypothesis using a Medicaid database from several US states.
Methods
Study design and population
The authors conducted a retrospective cohort study of adult schizophrenia patients hospitalized between January 1, 2004 and December 31, 2006, using a partial likelihood Cox proportional hazards model with day-level therapy gap as a time-dependent variable. Time-dependent variables are those that vary over an observation period. In this case, on any given day the patient may have gone a different number of days since their last medication dose. A ‘day-level’ variable (e.g., one that may be different on every day) is used to address this situation in a multivariate model. The data were from MedStat’s MarketScan Medicaid database, a Health Insurance Portability and Accountability Act (HIPAA)-compliant administrative claims data set that aggregates Medicaid data from multiple states, making the study exempt from institutional review board review. The data contain claims for prescriptions, inpatient services, and outpatient services. Pharmacy claims are available only for outpatients; medical claims cover both the inpatient and outpatient settings. Enrollment and limited patient demographic information are also available.
The study included patients aged 18–64 years who were discharged while receiving SGA monotherapy following a schizophrenia hospitalization that ended during the study period. Patients who were hospitalized for schizophrenia had either a primary diagnosis of schizophrenia (International Classification of Diseases, 9th Revision, Clinical Modification [ICD-9-CM] 295) or a secondary diagnosis of schizophrenia with a primary diagnosis of other mental disorders (ICD-9-CM 290–294, 296–319). If there were multiple admissions, the first qualifying one was selected.
Cohorts were defined by the SGA monotherapy used at discharge. Patients receiving medications requiring once-daily dosing (risperidone, t1/2 20 h; olanzapine, t1/2 30 h; or aripiprazole, t1/2 75 h) were included in the LHL cohort; those taking medications requiring twice-daily dosing (quetiapine, t1/2 6 h; ziprasidone, t1/2 7 h) were included in the SHL cohortCitation11–15. The study followed patients until the first of four events was seen: (1) fill of a prescription for a second, different SGA, (2) discontinuation of the index SGA (refill gap > 30 days), (3) the end of enrollment, or (4) the end of the study period (12/31/2007).
Participants were excluded from the study if they were hospitalized with a primary diagnosis of a mental disorder in the 6 months before the baseline hospitalization or if they were not continuously enrolled for at least 6 months before the index date. To ensure a minimum amount of SGA use before outcomes were measured, patients with <2 prescriptions for the index medication in the 90 days after discharge were excluded. The date of the second SGA fill following discharge was the study index date.
Study measures
The primary outcome was the rate of hospitalization or emergency department (ED) visit for any mental disorder. Secondary outcomes included hospitalization/ED visits for schizophrenia, outpatient contacts for mental disorders, initiation of additional antipsychotic pharmacotherapy, and discontinuation of the index medication.
Baseline variables included demographics (age, race, gender), benefit information, length of stay of baseline admission, selected comorbid mental disorders and other chronic conditionsCitation16, use of psychiatric medications, and ED and outpatient contacts for mental disorders in the pre-index period. The database did not include data on the participants’ geographic region or symptoms.
Many measures of therapy adherence have been testedCitation17. The authors chose therapy gap, defined as the number of days the patient was expected to be without a supply of the index medication, as the primary adherence measure in this study. Therapy gap has been used in other studies in a similar populationCitation2, and an expert panel reported it to be an acceptable and interpretable measureCitation3. The gap was 0 if patients were currently receiving therapy. If a patient filled the prescription before its supply should have been exhausted, the number of days of extra supply was added to the next fill. Therapy gap was updated for each day of the follow-up period. A secondary adherence measure, medication possession ratio over the previous 90 days (MPR-90), was also used in a sensitivity analysis. MPR-90 was defined as a ratio of the number of days of medication supplied over the number of days in the period, and this ratio was updated daily. While quetiapine is indicated on a once-daily schedule for bipolar depression, it is indicated twice daily for schizophrenia (although physicians can and do prescribe it daily for this indication). Both therapy gap and MPR were calculated using the ‘days of supply’ field in the pharmacy claims, rather than the number of pills supplied, and therefore should not be affected by the use of different dosing schedules.
Statistical analysis
The authors conducted univariate comparisons between the LHL and SHL SGA cohorts for all variables. For continuous variables, means and standard deviations were reported, and t-tests were conducted to compare cohorts. For categorical variables, counts and percentages were reported and chi-square tests were conducted to compare cohorts. Outcomes were reported per patient-year (PY) of follow-up. For hospitalization and composite hospitalization/ED outcomes, the observation period ended after the first such event; for outpatient contacts and medication use, observation continued. Crude event rates were compared using the negative binomial distribution rather than the Poisson distribution to account for the over-dispersion observed in the dataCitation18,Citation19. To adjust for multiple comparisons, p-values for pairwise comparisons were adjusted using Holm’sCitation20 step-down Bonferroni method. With this method, 0.05 is still considered the threshold for statistical significance.
To control for baseline differences and adjust for real-time adherence to therapy, the authors conducted a partial likelihood Cox proportional hazards model with day-level therapy gap as a time-dependent variable. Therapy gap was updated at each follow-up day and was modeled as a continuous variable. To test if rehospitalization rates differed between cohorts at certain therapy gaps, an interaction term for therapy gap and medication cohort was evaluated; the result was not statistically significant and was dropped. A quadratic term was similarly tested and dropped. In a sensitivity analysis, the authors conducted a Cox proportional hazards model, replacing gap length with MPR-90 as the adherence measure. The results were virtually identical to the base model and are not presented.
The authors calculated the number of patients who would need to be treated with LHL medications instead of SHL medications for 6 months, 1 year, and 2 years to prevent one additional event. At each time point (t), the number needed to treat (NNT) was determined using the survival probability of the SHL cohort (Ss(t)) and the adjusted hazard ratio (h; LHL vs SHL) with the equation: NNT = 1/{[Ss(t)]h − Ss(t)Citation21. All data transformations and statistical analyses were performed using SAS© version 9.2 (SAS Institute, Cary, NC, USA).
Results
The study identified 36,736 schizophrenia hospitalizations between January 1, 2004, and December 31, 2006, representing 17,881 unique Medicaid patients. Excluding 2,902 patients who were hospitalized for mental disorders in the 6 months before the index hospitalization, 10,245 who were not treated with SGA monotherapy after discharge, and 1491 with only one fill of SGA monotherapy, left 3243 eligible patients. A further 927 were excluded because they were younger than 18, older than 64, or were not continuously enrolled for 6 months before the index date, leaving 2316 unique patients.
There were 1479 (63.9%) participants in the LHL group, comprising 258 aripiprazole, 433 olanzapine, and 788 risperidone users; and 837 (36.1%) in the SHL group, comprising 525 quetiapine and 312 ziprasidone users. The LHL and SHL groups were similar with regard to age (mean, 40.5 years for LHL and 40.6 for SHL, p = 0.82) but differed on a variety of background characteristics and in their baseline mental health service use. Sixty percent of the SHL group was female, compared with 51% of the LHL group (p < 0.001). Racial distribution differed between groups: 51.5% of the LHL group was White compared with 59.9% of SHL (p < 0.001).
The groups differed significantly with regard to their comorbid conditions. The LHL group had a median of two chronic conditions, compared with three for SHL (p < 0.001) (). Patients in both groups had similar lengths of stay at the index hospitalization (mean, 7.8 days for LHL and 7.3 days for SHL, p = 0.16). There were fewer patients in the LHL group that used drugs to treat depression, anxiety, and mania in the 6 months before the index hospitalization, and this difference was statistically significant. There were fewer patients in the LHL group that had claims for bipolar disorder, schizoaffective disorder, substance use disorder, anxiety disorder, and depression compared with the SHL group during the 6 months before the index hospitalization, and this difference was statistically significant (). The discontinuation rate for SGA monotherapy was the same in both groups (0.97/PY for LHL and 0.94 for SHL, p = 0.99).
Table 1. Background characteristics of schizophrenia patients treated with long and short half-life antipsychotic medications.
Table 2. Baseline mental health service use for schizophrenia patients treated with long and short half-life antipsychotic medications.
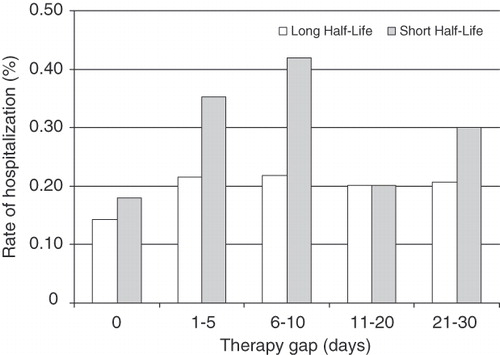
Patients were followed until they stopped the index drug, filled a prescription for a second SGA, disenrolled, or reached the end of the study period. There were a total of 959.9 PYs of follow-up in the LHL group (mean, 0.65 years) and 518.8 PYs (mean, 0.62 years) in the SHL group. The unadjusted rate of hospitalization/ED visits for mental disorders was significantly lower for LHL (0.74/PY) compared with SHL (1.06/PY) SGAs (p < 0.001) (). There was a trend toward lower unadjusted schizophrenia hospitalization/ED visit rates for LHL drugs, but the difference was not statistically significant. The unadjusted rate of hospitalization for mental disorders was significantly lower for LHL (0.38/PY) vs SHL (0.52/PY) antipsychotics (p = 0.005) (, ). The unadjusted rate of schizophrenia hospitalization was similar between groups (0.31/PY for LHL and 0.36/PY for SHL, p = 0.38). Significantly fewer patients in the LHL group initiated an additional antipsychotic medication during follow-up (0.34/PY compared with 0.42/PY, p = 0.02). There was a mean of 6.47 psychiatrist office visits/PY in the LHL group compared with 5.70/PY in the SHL group (p = 0.94). There was no difference in the overall rate of outpatient contacts with a diagnosis of mental disorder between groups ().
Table 3. Years of follow-up and unadjusted outcomes, stratified by long and short half-life cohorts.
A partial likelihood Cox proportional hazards model with day-level therapy gap as a time-dependent variable was used to adjust for baseline differences between groups and for adherence to medication. The adjusted hazard ratio for combined hospitalization/ED visits for mental disorders was 0.77 (95% CI: 0.67–0.88, p < 0.001) for LHL compared with SHL SGAs. The corresponding hazard ratio for combined hospitalization/ED visits for schizophrenia was 0.84 (95% CI: 0.71–0.997, p < 0.05). For hospitalization alone the hazard ratios were 0.80 (95% CI: 0.67–0.96, p < 0.05) for any mental disorder and 0.86 (95% CI: 0.70–1.05) for schizophrenia (). In a sensitivity analysis the authors replaced gap length with MPR in the regression model; the magnitude and statistical significance of the results were unchanged in that model.
Table 4. Adjusted* hazard ratio for hospitalization and emergency department visits among schizophrenia patients using long vs short half-life antipsychotics.
To estimate the clinical value of the identified difference in hospitalization, the authors calculated the NNT to prevent one hospitalization. Treating 11 patients for 1 year with LHL instead of SHL medications would prevent one hospitalization or ED visit for mental disorders. Treating 16 patients for 1 year would prevent one hospitalization.
In a post-hoc analysis designed to test whether the results differed dramatically from one specific drug to another, unadjusted outcomes were compared across individual drugs. For the primary outcome of hospitalization or ED visit for mental disorders, the range of rates within the groups was narrow, with the rate for the LHL group ranging from 0.74–0.76/PY and for SHL from 1.05–1.08/PY (p < 0.001). None of the hospitalization or ED outcomes occurred at higher rates in users of LHL medications than in users of SHL medications.
Discussion
Following inpatient discharge, Medicaid patients with schizophrenia who were treated with LHL SGAs were less likely to require mental healthcare in the ED or hospital than those treated with SHL antipsychotic medications. This effect persisted after controlling for medication adherence, suggesting, but not proving, that half-life may mediate the difference in effectiveness, with longer half-life reducing the impact of missed doses. The reduction in emergency or hospital care did not appear to be offset by an increase in psychiatrist office visits or other outpatient contacts. For every 11 schizophrenia patients who use LHL instead of SHL medications, the model suggests one fewer mental health hospital admission or ED visit over the course of a year. This study identified an association between differences in antipsychotic medication half-life and hospitalization, but does not prove a causal link. As such, the NNT should be interpreted cautiously.
Much of the research into adherence to antipsychotic treatment has dichotomized patients into adherent or non-adherent groups. This analytic approach and clinical orientation risks obscuring the most common pattern of non-adherence that involves intermittent use of antipsychotic medications, or ‘partial’ non-adherenceCitation8. The effect of adherence on outcomes appears to be continuous: incremental declines in adherence incrementally worsen outcomesCitation2,Citation5.
The current study may be most relevant to the large group of partially non-adherent patients. Medication half-life cannot affect outcomes in patients who completely stop treatment, but among those who continue treatment but intermittently miss doses, half-life appears to be an important determinant of acute service utilization. For a given number of missed doses, schizophrenia patients using LHL SGAs had better outcomes than those using SHL medications. It appears that longer half-life medications may help bridge the gap between missed doses.
In some previous studies examining adherence as a predictor of outcome, results have been aggregated across medicationsCitation2,Citation5. The study hypothesis was that pharmacokinetic differences among SGAs would have an impact on imperfect adherence and would be different for LHL and SHL medications. The authors expected that schizophrenia patients using SGAs with longer half-lives would be less affected by imperfect adherence. The results support this hypothesis. Although this study’s sample size was inadequate for looking at adjusted outcomes for specific medications, the unadjusted results were consistent within groups, with each LHL medication having lower rehospitalization risk than each SHL medication. Larger studies will be needed to determine whether there are statistically significant differences among individual drugs.
Many factors influencing outcomes in schizophrenia, such as age, gender, age of illness onset, drug abuse, and the presence of other comorbidities, are difficult or impossible to modifyCitation3. To improve outcomes, intensive efforts to change modifiable factors have been made, many of them focused on improving adherence. The study also found that going from a SHL to a LHL medication reduced the risk of hospitalization by 77%, a substantial difference in light of the difficulty of improving medication adherence among patients with schizophrenia. Modest effects reported from interventions to improve adherence underscore the complexity of the clinical challenge posed by medication non-adherence in patients with schizophreniaCitation22.
This study has several limitations. First, it was conducted in a Medicaid population and may not be generalizable to other settings. Second, there were many unmeasured covariates that might have influenced hospitalization risk, such as homelessness or social supportCitation23,Citation24. In unadjusted comparisons, the patients in the SHL group had more comorbid mental health conditions and took more psychoactive medications than those in the SHL group. These differences might be expected to bias the unadjusted comparisons to show a benefit of LHL medications, when in fact the differences in outcome simply reflected differences in group composition at baseline. In multivariate comparisons, adjusting for these baseline differences, the LHL group still had fewer hospitalizations for mental disorders. However, if there were unmeasured differences in disease severity between groups that were not colinear with the observed differences, they would not have been controlled for in the multivariate analysis, and these findings could in part be accounted for by these baseline differences. Third, Medicaid files are generated for administrative and reimbursement rather than clinical or research purposes, and the information is subject to coding errors and omissions. Fourth, medication use is assumed when prescription fills are observed, but such use cannot be confirmed. Fifth, the study used a recently discharged group of Medicaid patients who were expected to have a higher rate of rehospitalization than the general schizophrenia population, and the findings may not extend to a less severely affected population. Sixth, in multivariate analyses there was no difference between the rates of hospitalization for schizophrenia between groups. In keeping with prior studies of resource use in patients with schizophreniaCitation2,Citation5,Citation6, hospitalizations for any mental disorder were included in the primary outcome because patients diagnosed with schizophrenia presenting to a hospital may be given a number of different diagnoses. The similarity of the point estimates for both the composite (i.e., all mental disorders) and individual (i.e., schizophrenia) multivariate comparisons suggests that this study may have been under-powered to detect a difference in schizophrenia hospitalization alone. Seventh, brain biology in schizophrenia is complex, and changes at the receptor level may follow a different time course than circulating drug levels, making the groupings of LHL and SHL potentially less relevant. Finally, the study did not include depot preparations; although such preparations may have adherence advantages, their use in the US has been limitedCitation10.
Conclusions
The authors hypothesized that clinical differences among the SGAs reflect underlying differences among the agents and that pharmacokinetic differences might mediate the impact of adherence on acute mental health service utilization in patients with schizophrenia. Consistent with this hypothesis, users of LHL compared with SHL antipsychotics were less likely to use acute mental health services. Longer half-life SGAs may have been more forgiving of missed doses than shorter half-life medications.
Although it would be unreasonable to expect that simply selecting a LHL SGA over one with a SHL will address the complex and often stubborn problem of partial non-adherence, the pharmacokinetic properties of SGAs may have a discernible impact on clinical outcomes in patients with schizophrenia. Beyond potential clinical implications, the results will also hopefully help motivate development of even longer acting oral antipsychotic medications.
Transparency
Declaration of funding
This research was funded by the Bristol-Myers Squibb Company and Otsuka Pharmaceuticals.
Declaration of financial/other relationships
M.B. and E.C. are employees of Partnership for Health Analytic Research, LLC, which was paid by Bristol-Myers Squibb to conduct the research. J.A.B., Y.J., and T.H. are employees of Bristol-Myers Squibb Company. R.A.F. is an employee of Otsuka Pharmaceuticals.
Acknowledgments
No assistance in the preparation of this article is to be declared.
References
- Lacro JP, Dunn LB, Dolder CR, et al. Prevalence of and risk factors for medication nonadherence in patients with schizophrenia: a comprehensive review of recent literature. J Clin Psychiatry 2002;63:892-909
- Weiden PJ, Kozma C, Grogg A, et al. Partial compliance and risk of rehospitalization among California Medicaid patients with schizophrenia. Psychiatr Serv 2004;55:886-91
- Velligan DI, Diamond PM, Mintz J, et al. The use of individually tailored environmental supports to improve medication adherence and outcomes in schizophrenia. Schizophr Bull 2008;34:483-93
- Knapp M, King D, Pugner K, et al. Non-adherence to antipsychotic medication regimens: associations with resource use and costs. Br J Psychiatry 2004;184:509-16
- Valenstein M, Copeland LA, Blow FC, et al. Pharmacy data identify poorly adherent patients with schizophrenia at increased risk for admission. Med Care 2002;40:630-9
- Ascher-Svanum H, Faries DE, Zhu B, et al. Medication adherence and long-term functional outcomes in the treatment of schizophrenia in usual care. J Clin Psychiatry 2006;67:453-60
- Svarstad BL, Shireman TI, Sweeney JK. Using drug claims data to assess the relationship of medication adherence with hospitalization and costs. Psychiatr Serv 2001;52:805-11
- Velligan DI, Weiden PJ, Sajatovic M, et al. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry 2009;70(4 Suppl):1-46
- Weissman MM, Verdeli H, Gameroff MJ, et al. National survey of psychotherapy training in psychiatry, psychology, and social work. Arch Gen Psychiatry 2006;63:925-34
- Chue P, Emsley R. Long-acting formulations of atypical antipsychotics: time to reconsider when to introduce depot antipsychotics. CNS Drugs 2007;21:441-8
- Abilify [package insert]. Tokyo, Japan: Otsuka Pharmaceutical Co, Ltd, 2009. http://www.abilify.com/pdf/pi.aspx?q=aripiprazole+prescribing+information&ie=utf-8&oe=utf-8&aq=t&rls=org.mozilla:en-US:official&client=firefox-a. Accessed August 3, 2010
- Seroquel [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP, 2010. http://www1.astrazeneca-us.com/pi/Seroquel.pdf. Accessed August 3, 2010
- Risperdal [package insert]. Raritan, NJ: Ortho-McNeil-Janssen Pharmaceuticals, Inc, 2010. http://www.risperdal.com/sites/default/files/shared/pi/risperdal.pdf. Accessed August 3, 2010
- Zyprexa [package insert]. Indianapolis, IN: Eli Lilly and Company, 2010 http://pi.lilly.com/us/zyprexa-pi.pdf. Accessed August 3, 2010
- Geodon [package insert]. New York, NY: Roerig Division of Pfizer, 2009. http://media.pfizer.com/files/products/uspi_geodon.pdf. Accessed August 3, 2010
- Hwang W, Weller W, Ireys H, et al. Out-of-pocket medical spending for care of chronic conditions. Health Aff (Millwood) 2001;20:267-78
- Karve S, Cleves MA, Helm M, et al. An empirical basis for standardizing adherence measures derived from administrative claims data among diabetic patients. Med Care 2008;46:1125-33
- Gardner W, Mulvey EP, Shaw EC. Regression analyses of counts and rates: poisson, overdispersed poisson, and negative binomial models. Psychol Bull 1995;118:392-404
- Agresti A. Categorical data analysis. Hoboken, NJ: John Wiley & Sons, Inc., 1990
- Holm S. A simple sequentially rejective Bonferroni test procedure. Scand J Statist 1979;6:65-70
- Altman DG, Andersen PK. Calculating the number needed to treat for trials where the outcome is time to an event. Br Med J 1999;319:1492-5
- Dolder CR, Lacro JP, Leckband S, et al. Interventions to improve antipsychotic medication adherence: review of recent literature. J Clin Psychopharmacol 2003;23:389-99
- Hendryx MS, Russo JE, Stegner B, et al. Predicting rehospitalization and outpatient services from administration and clinical databases. J Behav Health Serv Res 2003;30:342-51
- Olfson M, Mechanic D, Hansell S, et al. Predicting medication noncompliance after hospital discharge among patients with schizophrenia. Psychiatr Serv 2000;51:216-22