Abstract
Objective:
Around one-third of patients with bipolar I disorder (BD-I) experience mixed episodes, characterized by both mania and depression, which tend to be more difficult and costly to treat. Atypical antipsychotics are recommended for the treatment of mixed episodes, although evidence of their efficacy, tolerability, and cost in these patients is limited. This study evaluates, from a UK National Health Service perspective, the cost-effectiveness of asenapine vs olanzapine in BD-I patients with mixed episodes.
Methods:
Cost-effectiveness was assessed using a Markov model. Efficacy was informed by a post-hoc analysis of two short-term clinical trials, with response measured as a composite Young Mania Rating Score and Montgomery-Åsberg Depression Rating Scale end-point. Probabilities of discontinuation and relapse to manic, mixed, and depressive episodes were sourced from published meta-analyses. Direct costs (2012–2013 values) included drug acquisition, monitoring, and resource use related to bipolar disorder as well as selected adverse events. Benefits were measured as quality-adjusted life years (QALYs).
Results:
For treating mixed episodes, asenapine generated 0.0187 more QALYs for an additional cost of £24 compared to olanzapine over a 5-year period, corresponding to a £1302 incremental cost-effectiveness ratio. The higher acquisition cost of asenapine was roughly offset by the healthcare savings conferred through its greater efficacy in treating these patients. The model shows that benefits were driven by earlier response to asenapine during acute treatment and were maintained during longer-term follow-up. Results were sensitive to changes in key parameters including short and longer-term efficacy, unit cost, and utilities, but conclusions remained relatively robust.
Conclusions:
Results suggest that asenapine is a cost-effective alternative to olanzapine in mixed episode BD-I patients, and may have specific advantages in this population, potentially leading to healthcare sector savings and improved outcomes. Limitations of the analysis stem from gaps in clinical and economic evidence for these patients and should be addressed by future clinical trials.
Introduction
Bipolar disorder is a complex, recurrent mood disorder, which has a significant impact on a patient’s quality-of-lifeCitation1,Citation2 and places a considerable economic burden on societyCitation3–6. Patients diagnosed with bipolar I disorder (BD-I) may experience mixed episodes, which are characterized by the presence of both mania and depressionCitation7. Studies investigating the burden of illness in bipolar disorder (BD) have found that patients with mixed episodes tend to have more frequent episodes of illness, with episodes of longer durationCitation8, greater functional severityCitation9, and larger impact on quality-of-lifeCitation10. They have found them to be associated with the highest average length of hospital stay of any BD episode typeCitation11,Citation12. The cause of this extra burden on inpatient services is driven by a lower likelihood of and longer time to recovery for patients with mixed episodes compared to patients with only manic or depressive episodesCitation13. Additionally, these patients tend to experience less inter-episodic remission, have higher risks of relapse, and have higher rates of suicide and co-morbid conditions such as substance abuseCitation14. Thus, the consequent complexity of mixed episodes can make them harder and more costly to manage than the more commonly-occurring manic or depressive episodes.
Published clinical guidanceCitation15,Citation16 currently recommends treating mixed episodes in much the same way as one would treat acute manic episodes, with atypical antipsychotics (AAPs), often olanzapine (OLA). However, the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5)Citation7 has refined the diagnosis of mixed episodes with a mixed-feature specifierCitation17, a move that will allow clinicians to more accurately identify patients who may be suffering from concurrent symptoms of depression and mania/hypomania. This increased diagnostic accuracy should enable clinicians to better tailor treatment to a patient’s behaviours.
Asenapine (ASE) is a new AAP developed for the treatment of schizophrenia and bipolar disorder. In Europe, asenapine is licensed for the treatment of moderate-to-severe manic episodes associated with BD-I in adults. In the US and other countries, where it is approved as both monotherapy and adjunctive therapy with lithium or valproate, asenapine is approved for the acute treatment of manic or mixed episodes associated with BD-I. In the US, it is also approved for the treatment of adults with schizophrenia.
Short-term efficacy, tolerability, and safety of asenapine in treating acute mania in BD-I was assessed via two identically designed 3-week, randomized, placebo- and OLA-controlled trials as well as a 9-week, double-blind OLA-controlled extension study. In this trial program (called ARES), ASE was statistically significantly more effective than placebo and statistically non-inferior to OLA in reducing both mania as measured on the Young Mania Rating Score (YMRS) and depression measured on the Montgomery-Åsberg Depression Rating Scale (MADRS).
A recent post-hoc analysis by Azorin et al.Citation18 used datasets from ARES to evaluate efficacy in the sub-set of BD-I patients who had enrolled with a mixed episode as defined by the DSM-IV-TRCitation19. The authors found that ASE produced a statistically significantly greater improvement on YMRS and MADRS total scores and a higher composite response (≥50% reduction from baseline in YMRS and MADRS scores) rate at week 3 compared to placebo. In contrast, no statistically significant difference between OLA and placebo at week 3 was found. At week 12, during the extension phase, YMRS and MADRS total scores further decreased. The composite response rate increased with ASE and to a lesser extent with OLA. Although no statistically significant difference was observed between asenapine and olanzapine in composite response rate or mean reduction on either score, the findings from the Azorin et al.Citation18 analysis do suggest a potential difference in the way the two drugs work in mixed episodes.
Second-generation AAPs are cost-effective drugs for managing the manic episodes associated with BD-I, as numerous analyses have demonstratedCitation20–26. However, no study has tried to estimate the cost-effectiveness of AAPs specifically in managing mixed episodes in BD-I patients.
Methods
Model structure
This study presents a modeled evaluation to estimate the relative costs and benefits of using ASE compared to OLA for the treatment of mixed episodes in BD-I patients. Comparison against other second-generation AAPs was not possible due to differences in the ways outcomes were reported in the clinical trials. Benefits of treatment were measured as quality-adjusted life years (QALYs), and costs were considered from a UK National Health Service and Personal Social Services perspective. Hypothetical patients included in the model have an average age of 38.7 years and are 54.6% male, reflective of the mixed episode sub-group from the ARES trial program.
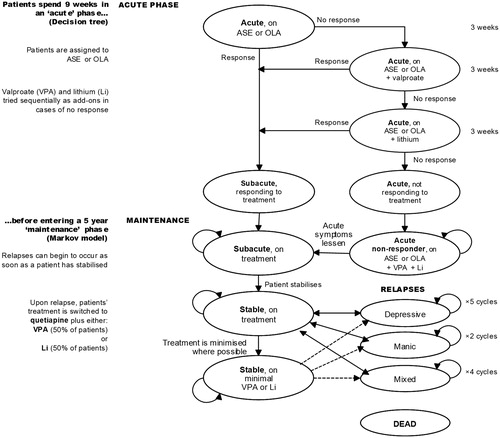
The model is divided into two phases: a 9-week acute phase and a 5-year maintenance phase. The acute phase covers the initial treatment of a mixed episode and the maintenance phase uses a Markov model to follow patients as their condition stabilizes and the goal of treatment shifts into relapse prevention. presents a graphical illustration of the model’s main attributes. Costs and QALYs are discounted at 3.5% per annum during the maintenance phase, in accordance with recommended methodsCitation27.
Hypothetical patients enter the model presenting with an acute mixed episode, and are allocated either ASE or OLA. If they respond to their allocated treatment at week 3, treatment is continued and they enter a ‘sub-acute’ health state. If a patient does not respond at week 3, valproate (VPA) is added to their starting treatment; if this combination produces a response by week 6, it is continued and the patient enters the ‘sub-acute’ health state. If the addition of VPA fails to induce a response by week 6, the VPA ‘add-on’ is replaced with lithium (Li) for the final 3 weeks of the acute phase at which time response is assessed again. A recent survey of treatment guidelinesCitation28 demonstrated a lack of consensus regarding the appropriate time to change treatment strategies in the case of partial or non-response during an acute manic episode, with recommendations ranging from 2–12 weeks. In the absence of firm evidence and given the high risk of suicide in mixed episodes, a shorter duration (3 weeks) during which to assess response to treatment was considered appropriate as it may be dangerous to wait longer before changing.
The maintenance phase of the economic model is designed to follow patients for 5 years after their qualifying mixed episode was first treated. Five years was deemed a sufficient length of time by clinical experts to capture the longer-term aspects of treating bipolar disorder, including stabilization after an acute episode, possible discontinuation of AAP therapy, management and prevention of relapse episodes, and mortality. Shorter and longer time horizons were tested in sensitivity analysis.
‘Responders’ from the acute phase enter the maintenance phase in a sub-acute health state, during which the treatment to which they responded is continued, and the lingering (but greatly diminished) effects of the initial episode continue to have some impact on costs and quality-of-life.
‘Non-responders’ from the acute phase are allocated to triple therapy (ASE or OLA + VPA + Li) in the maintenance phase, an assumption informed by clinical expert opinion. They remain in an acute mixed episode state until they eventually experience a diminution of their acute symptoms and move to the sub-acute health state or die.
Whilst still on treatment, sub-acute patients’ symptoms are expected to further diminish, eventually resolving in a state of euthymia— - reflected by the ‘Stable’ health state. At this point patients may discontinue treatment with the AAP they were assigned, although based on expert opinionCitation29 we assumed that patients would not come off treatment entirely. Instead we assumed that patients discontinuing their AAP were most likely to remain on a minimal mood stabilizer therapy in the form of either VPA (50% of patients) or Li (50% of patients).
Stable patients (either on regular antipsychotic treatment or on the minimal treatment described above) can relapse and experience a manic, mixed, or depressive episode. Following a relapse, patients switch to treatment with quetiapine (QTP) in combination with either VPA or Li (a 50/50 split is assumed). The duration of episodes depends on the type of event, as described in the next section.
At any point in the model, patients can die. Due to the increased risk of suicide, risk of death is assumed to be higher during an acute episode and immediately afterwards (in the sub-acute health states). Mortality risk is reduced, although still higher than in the general population, during periods of stable euthymia.
Clinical inputs
Response to treatment (acute phase)
Response to the starting therapy (ASE or OLA) at week 3 was sourced from the post-hoc analysis of ARESCitation18, and defined as a reduction (improvement) of 50% or more from baseline in both YMRS and MADRS scores. Comparative response rates for ASE and OLA in combination with VPA or Li on the composite YMRS and MADRS outcome are not available from clinical trial data, so we used the upper limit of the 95% confidence intervals for ASE or OLA, as observed in the post-hoc analysis. This assumption was deemed reasonable by clinical expert opinion, given that combination therapy is expected to be more efficacious than monotherapy; it is explored in sensitivity analysis. Probabilities of response to each treatment are presented in .
Table 1. Clinical input data for acute and maintenance phases.
Stabilization, discontinuation, and relapse (maintenance phase)
Patients who failed to achieve a response in the acute phase enter the maintenance phase as ‘non-responders’ receiving triple therapy (ASE or OLA + VPA + Li). The probability of transitioning from this state to ‘sub-acute’ was assumed to be 10% per week (corresponding to a 35% per cycle probability). Although no hard clinical data could be found to support this assumption, it has been validated through expert opinionCitation29.
The sub-acute health state was implemented in the Markov model as a temporary tunnel state, through which responding patients transition before achieving euthymia. A 60% monthly probability of achieving euthymia was applied estimated based on the assumptions that the average duration of a mixed episode is 16 weeks, and that 90% of all patients will reach euthymia within 16 weeks of the start of the mixed episode.
The probability of discontinuing treatment with the initial AAP during periods of disease stability was calculated by applying a risk ratio from a published meta-analysis of BD trialsCitation30 to an underlying baseline probability of discontinuing on placebo. The placebo discontinuation rates are based on a published Discrete Event Simulation (DES) model for patients with acute bipolar depressionCitation31. The per-cycle probabilities of moving off treatment onto minimal VPA or Li are reported in .
Probabilities of relapse for most modeled treatments were sourced from a health technology assessment (HTA) conducted by Soares-Weiser et al.Citation32 in the maintenance setting. Patients who relapse in the maintenance phase are switched to QTP + Li or VPA and, whilst on this treatment, they can relapse again into manic, mixed, or depressive episodes. For this group of patients, the annual risk of relapse for each type of acute episode was extracted from Kaplan-Meier curves reported in Vieta et al.Citation33.
Relapse probabilities for ‘manic/mixed episode’ and ‘depressive episodes’ were reported in the Soares-Weiser et al.Citation32 HTA. In order to disaggregate the ‘manic/mixed’ relapse type we replicated the observed balance of relapse episodes from the placebo arm of Vieta et al.Citation33 to assume that 37% of those relapses would be manic and 63% would be mixed. All probabilities were calculated as per-cycle values and reported in .
The Markov model implements relapsed episodes using tunnel states, each with a fixed number of cycles. The length of an acute episode was defined using data from Soares-Weiser et al.Citation32 and the 2011/12 NHS Hospital Episode Statistics (HES)Citation34, and validated by clinical expert opinion.
Soares-Weiser et al.Citation32 estimated that, in cases where an acute episode required hospitalization, the inpatient length of stay (LOS) represented ∼60% of the total episode duration in the case of manic episodes and 38% in the case of depressive episodes. To estimate updated figures for total episode duration, the same proportions were applied to the most recently published HES data. In the absence of a defined relationship between LOS and episode duration for mixed episodes, it was assumed to be the same as for manic episodes. See for the resulting total episode duration and corresponding number of cycles assumed for each episode.
Table 2. Relapse episode duration and length of associated inpatient stay in patients admitted to hospital.
Mortality
Base mortality rates were sourced from life tables of the UK general populationCitation35 and general population risks of suicide were estimated from UK Mortality StatisticsCitation36. Based on the published literatureCitation37, all patients with bipolar disorder are reported to have an increased risk of death due to non-suicidal causes (standardized mortality ratio = 1.69). During an acute episode and whilst in the sub-acute health state, patients also have an increased risk of death due to suicide, which was estimated to be 11.53 (SMR).
Adverse events
Weight gain is expected to be associated with all drugs given in this model. For combination drug regimens, the weight gain incidence is set to equal that of the drug most highly associated with weight gain. Extra-pyramidal symptoms (EPS) are assumed to occur in all patients receiving AAPs in the model (i.e., OLA, ASE, or QTP). Both adverse events are assumed to occur independently.
Weight gain is assumed to occur soon after starting treatment, and once gained the extra weight (and associated health effects) persists for the remainder of the model. EPS are also assumed to occur early after starting antipsychotic treatment, but with appropriate medication the symptoms are brought under control and, therefore, EPS-related health detriment is only assumed to occur in the acute phase. The incidence values for weight gain and EPS for each drug are reported in .
Quality-of-life inputs
Utility values for model health states and adverse events (AEs) were extracted from published literature and from post-hoc analyses of patient-level data in ARESCitation38. Mixed episode utilities for the model were estimated by converting responses from version 2 of the 36-item short form health survey (SF-36v2) gathered during the ARES trial program into EuroQol-5D utility scores using a published algorithmCitation39. All utilities were converted to multipliers using Ara and Brazier’sCitation40 formulae so that they could be age- and sex-adjusted relative to the study population and then combined with disutility figures for EPS and weight gain using a multiplicative method ().
Table 3. Utility values.
Resource use and cost inputs
Four relevant cost categories were identified: drug acquisition costs, monitoring costs, other health care services costs, and adverse event costs.
Drug acquisition
Drug acquisition costs (see ) are based on 2013 list prices from the British National FormularyCitation41 and dosing information from trial data on fileCitation38. The costs of generic olanzapine and quetiapine were used to reflect UK clinical practice and a more conservative costing scenario.
Table 4. Drug dosages, acquisition, and monitoring costs.
Monitoring
We assumed monitoring would involve laboratory tests, which differed according to drug regimen. We drew on evidence from Soares-Weiser et al.Citation32 as well as the NICE Clinical Guideline for bipolar disorderCitation15, with NHS unit costsCitation42–44 inflated where appropriate to 2012 values. Acute and maintenance phase monitoring costs are presented in .
Healthcare services consumed (hereafter ‘health state costs’)
Costs associated with manic, mixed, and depressive episodes as well as with periods of disease stability (see ) were sourced largely from the costing estimates and assumptions of Soares-Weiser et al.Citation32 and the NICE guideline on bipolar disorderCitation15. Depending upon the episode type, patients could be managed on an inpatient or outpatient basis or with a crisis resolution team. Apart from inpatient hospital stays, which were sourced from NHS hospital episode statisticsCitation34, we conservatively assumed that manic and mixed episodes consume the same level of healthcare resources.
Table 5. Healthcare resources consumed (source: Soares-Weiser et al.Citation32).
Combining all assumptions we estimated overall per cycle health state costs to be £86 for stable BD-I and £4945, £4645, and £594 during manic, mixed, and depressive episodes, respectively. Based on the expected duration of each episode type (see ), the total healthcare cost for managing an acute manic, mixed, and depressive episode is estimated to be £9890, £18,580, and £2970, respectively.
Adverse event costs (weight gain and EPS)
The current model assumes that weight gain will occur and be actively managed during the 9-week acute phase. Although some further weight gain may occur as patients continue treatment during the maintenance phase, gains are assumed to plateau and, thus, no further intervention or active management is considered necessary. This conservative assumptions means that weight gain-associated costs only apply in the acute phase, as shown in . Regarding EPS, patients are expected to need a psychiatric consultant visit as well as medication (procyclidine) during the acute phase, but during the maintenance we have only applied the ongoing medication cost and no further consultant visits specifically to deal with EPS.
Table 6. Adverse event (weight gain and EPS) costs.
Sensitivity analysis
The model’s primary outcomes were the number of life-years and QALYs. Total QALYs were estimated both when the impact of AEs on quality-of-life was included and excluded. One-way sensitivity analyses and probabilistic sensitivity analysis (PSA) were performed to assess the effect of variation in key inputs on the model results. For PSA we assigned statistical distributions to all main parameters: a beta distribution was used for probabilities and utility parameters, whilst risk ratios and costs were assumed to have a log-normal distribution. The results of 2000 iterations are presented as mean costs and QALYs for each comparator and their cost-effectiveness is compared using standard decision rules.
Results
Base case analysis
Over 5 years and 9 weeks, patients with mixed episodes treated with ASE enjoyed 0.0187 more QALYs than OLA for an additional cost to the healthcare system of only £24. These results correspond to an incremental cost-effectiveness ratio (ICER) of £1302 per QALY gained. Results presented in indicate that, although drug costs for asenapine are greater during both acute and maintenance phases (incremental differences are £186 and £2128, respectively), the savings achieved by avoiding prolonged hospitalization (∼£2000 over 5 years) are sufficient to offset virtually all of the extra drug cost among mixed episode patients. Earlier response to treatment and discharge from hospital during the acute and early maintenance phases also generates gains in health-related quality-of-life (0.0021 QALYs in the first 9 weeks and 0.0166 QALYs over the subsequent 5 years).
Table 7. Base case analysis results (5 years and 9 weeks).
Sensitivity analyses
Deterministic sensitivity analyses
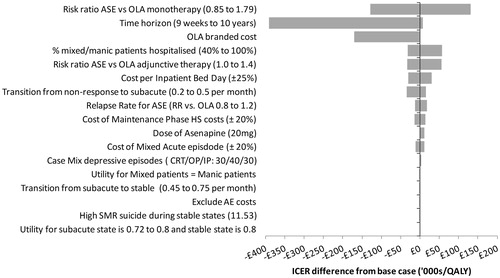
presents the results of extensive deterministic sensitivity analyses in the form of a tornado diagram. The figure illustrates the degree to which the base case ICER is sensitive to changes across a given range for each parameter.
An analysis in which the modeled time horizon is varied suggests that the base case results are sensitive to changes in the length of time the model runs. Limiting analysis to the 9-week acute phase, ASE dominates OLA, that is it generates 0.0021 additional QALYs at a savings of £820. ASE continues to dominate OLA up until the time horizon extends to just under 5 years. When the time horizon is extended out to 10 years, the incremental costs are still modest relative to the health gains generated, producing an ICER of £8493 per QALY. This value is still well below conventional willingness-to-pay thresholds in the UK (£30,000).
The model results were most sensitive to changes in the relative efficacy of ASE and OLA as first line treatments in the acute model phase. In the base case, ASE has a 23% higher probability of obtaining a response than OLA (Risk Ratio = 1.234, 95% CI = 0.85–1.79). When this advantage was reduced to the lower limit of the 95% confidence interval the ICER for ASE jumped to £132,951. However, when the risk ratio was increased to the upper limit of the 95% confidence interval, ASE dominated OLA, generating more QALYs for an overall savings of £2955. Indeed, threshold analyses indicated that, for any value of the risk ratio greater than 1.238, ASE dominates OLA and for any value greater than 1.138, ASE was most cost-effective at a willingness-to-pay threshold of £30,000 per QALY gained.
Further to this, we tested assumptions under which adding Li or VPA (during acute phase) altered or eliminated any difference in efficacy between ASE and OLA. In the base case, ASE in combination with Li or VPA was 21% more effective. When ASE was assumed to be 40% more effective, it dominated OLA. However, when the efficacy difference was eliminated and both drugs were assumed to be equally effective in combination therapy, the ICER for ASE increased to £57,491.
Variation in the assumed longer-term efficacy of ASE (i.e., its ability to prevent relapse during the maintenance phase) had an impact on its relative cost-effectiveness. Threshold analyses indicate that ASE dominates OLA when its associated risk of relapse is at least 2% lower and that ASE is more cost-effective up to a £30,000 WTP threshold so long as its risk of relapse is not more than 29% higher.
Although the ICER varied widely with changes to the acquisition cost of OLA, the dose of ASE and the healthcare costs associated with managing acute episodes, the conclusion that ASE is more cost-effective than OLA at a £30,000 threshold was relatively robust. ASE remained cost-effective compared to OLA when the assumed cost per inpatient bed-day was reduced by nearly 25% and became dominant when the cost was increased by just 2%. When the cost of non-proprietary OLA was replaced by that of brand-name Zyprexa (Eli Lilly & Co Ltd, Indianapolis, IN), ASE was dominant, saving the healthcare system £3169 over 5 years. The model was also sensitive to the total proportion of manic/mixed episodes which require hospitalization. The likelihood that ASE was cost-effective compared to OLA decreased as more patients were assumed to be managed by a crisis resolution team than as inpatients.
Health state utility values had very little impact on the main result. Under all variations tested in sensitivity analysis, ASE produced ICERs of under £2000 per QALY gained compared to OLA.
Probabilistic sensitivity analysis
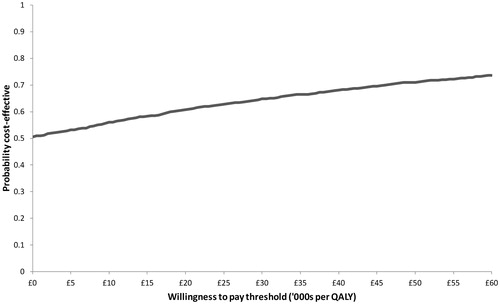
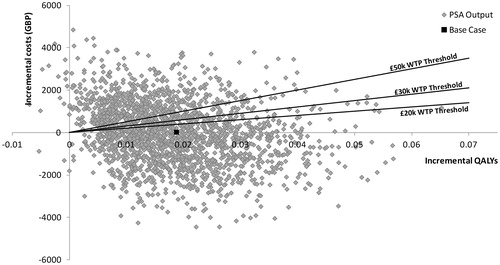
In the PSA, uncertainty around total and incremental costs and QALYs was quantified. Asenapine generated 3.869 (95% CI = 3.695–4.034) QALYs at a total cost of £32,283 (28,932–36,069), whilst OLA generated 3.851 (3.676–4.017) QALYs for a total cost of £32,302 (28,640–36,069). Compared to OLA, ASE was dominant, producing an incremental benefit of 0.018 (−0.222–0.258) QALYs and costing on average £19 less (£5308 less to £5270 more). Mean incremental differences are very close to zero and the confidence intervals cross zero for both costs and QALYs indicating no statistically significant difference between the two treatments. However, results of individual iterations indicated that ASE had a 60.8% and an 64.8% probability of being optimal at willingness-to-pay (WTP) thresholds of £20,000 and £30,000 per QALY, respectively (see ). Furthermore, at a £0 per QALY WTP threshold, ASE had a 50.5% probability of being optimal, indicating that, in half of all iterations, it was dominant, i.e., both less costly and more effective. presents a scatter plot on the cost-effectiveness plane for all 2000 simulations.
Discussion
Our economic evaluation was designed to model BD-I patients presenting with a mixed episode, and estimate the costs and benefits of starting treatment with either olanzapine or asenapine. Short-term costs and benefits during the 9-week acute phase were informed by post-hoc analyses of the ARES trial program. For longer-term outcomes we used a 5-year Markov model driven by data in the literature, expert opinion, and further analysis. Health benefits were estimated as QALYs and costs measured from a UK NHS and Personal Social Services perspective.
Previous economic analyses in the BD-I population have either combined manic and mixed episode patients as one group (consistent with the FDA label for most AAPs), or they have excluded mixed episodes to focus solely on patients with manic or depressive episodes (more in line with the EU labels for AAPs). This is the first economic evaluation specifically considering the management of BD-I patients who present with mixed episodes, a group now more clearly defined in the new DSM-5Citation7. This is also the first economic model that has attempted to disaggregate the costs and health effects of mixed and manic episodes in BD-I, during initial acute management and during relapse prevention/maintenance. This distinction between BD-I patients is especially important given that studies have shown that patients with predominantly mixed episodes experience episodes with greater frequency, longer duration, and greater functional severity, and have higher rates of suicide and co-morbid conditions. As a consequence, mixed episodes tend to be more complex and costly to manage than the more commonly-occurring manic or depressive episodes.
The 5-year and 9-month deterministic model base case found ASE to be associated with marginally more QALYs and very similar total healthcare costs compared with OLA. Total costs were so similar that the probabilistic sensitivity analysis found that, on average, ASE was cost-saving compared to OLA and dominant in ∼51% of iterations. The higher drug acquisition cost of ASE was roughly offset by the healthcare cost savings conferred through its potentially greater efficacy in treating BD-I mixed episode patients. In fact, considering the small quality-of-life gains modeled (less than 2% of one QALY over 5 years), this model is primarily a demonstration of how achieving a quicker response on ASE is cost neutral or could potentially lead to healthcare cost savings compared to using OLA. Indeed, the base case results, for which the cost of generic OLA was used, were very sensitive to variation in the unit cost of drugs, with ASE expected to generate more than £3000 in savings when compared to proprietary OLA.
Clinically, the model was most sensitive to short-term efficacy of the initial treatment with ASE or OLA during the acute phase. ASE was dominant as long as its response rate in the acute phase was at least 23.8% better than that of OLA, and cost-effective up to a £30,000 per QALY WTP threshold providing it was at least 13.8% better. In longer term use, sensitivity analyses indicate that ASE is dominant when the risk of relapse is at least 2% lower than OLA and remains cost-effective at £30,000 per QALY when the risk of relapse is up to 29% higher. The results were sensitive to the modeled time horizon, with ASE expected to be dominant over time horizons up to 5 years and then cost-effective out to 10 years.
Although not all patients experiencing an acute manic or mixed episode are admitted to hospitalCitation32, those who are typically have lengthy inpatient stays. Relevant publications have emphasized both the high cost and the high clinical necessity of prolonged inpatient care in hospitalized BD-I patientsCitation45. Our model assumptions around the average duration of hospitalization are in line with previous economic evaluations in bipolar disorderCitation15,Citation20–26,Citation32,Citation46,Citation47. The cost per bed-day had some effect on the model, but a more significant driver of cost was the expected proportion of patients whose episode would cause them to be admitted to hospital instead of managed by a CRT. Our assumed case mix of hospitalization to CRT management was 80%/20% in the base case. A threshold analysis suggests that ASE ceases to be cost-effective only once fewer than 60% of patients are managed as inpatients. Given the severity and complexity of treating patients experiencing mixed episodes, the majority are likely to be managed within a hospital setting.
Probabilistic sensitivity analysis results were encouraging because, although they reflect the statistical uncertainty inherent in the post-hoc analysis of the ARES data, they also demonstrate that ASE has a high probability of being the optimal therapy option in BD-I mixed patients. In up to 65% of model iterations ASE was expected to be either dominant or cost-effective at established willingness to pay thresholds.
Several limitations are worthy of consideration—the foremost of which relates to the absence of a contiguous set of data to model outcomes and costs for mixed episode BD-I patients. We made extensive use of post-hoc analyses of the ARES trial data to identify, wherever possible, statistics specifically relevant to the sub-population of BD-I patients who had entered treatment with a mixed episode. Whilst important basic values, such as efficacy and safety of OLA and ASE, were available from these post-hoc analyses, evidence for other important parameters—short-term efficacy of adjunctive therapies, longer-term efficacy of AAPs in relapse prevention and healthcare resource use—was not available for a distinct sub-group of patients with mixed episodes. As other authors have already highlightedCitation14, the revision of the DSM-5 to capture sub-threshold non-overlapping symptoms of the opposite pole using a ‘mixed features’ specifier will have a substantial effect across several fields, including epidemiology, diagnosis, treatment, and research. Future clinical trials that prospectively evaluate the comparative efficacy of treatments according to the presence or absence of mixed features will help to fill in these important gaps in the model’s data set.
In the absence of such sub-group evidence at present, assumptions were made, based on data in which mixed and manic episodes were conflated, and then tested in sensitivity analysis. Given what we know about the additional burden of mixed episodes, for example in terms of hospitalizationCitation11,Citation12, this may have led us to report more conservative results. Consequently the model results provide a valuable insight into the kinds of savings and potential health benefits that might be possible if the trends observed in post-hoc analysisCitation18 hold in clinical practice.
Conclusion
Asenapine represents a highly cost-effective alternative to olanzapine in the management of mixed episode BD-I patients in the UK setting, and may have specific advantages in this population which could lead to substantial healthcare sector savings and better clinical outcomes. Future research in this area could focus on designing an adequately-powered study investigating the real-life efficacy of asenapine vs standard AAPs in mixed episode patients as well as measuring levels of healthcare resource consumption in both acute management settings and longer term maintenance.
Transparency
Declaration of funding
The development of the study and this manuscript was funded by Lundbeck SAS, the manufacturer of asenapine.
Declaration of financial/other relationships
A. Guiraud-Diawara, C. Marre, and K. Hansen are employees of Lundbeck SAS. L. Sawyer, S. Chang, and C. Rinciog are employees of Symmetron Limited, a contract research organization in health economic and outcomes research, and provided consultant services to Lundbeck SAS. JM Azorin served as a consultant to Lundbeck SAS with respect to this study and has also served as consultant, advisor, or speaker for the following entities: AstraZeneca, Otsuka, Roche, and Servier.
Acknowledgments
L. Sawyer was the principal author of the study. L. Sawyer, S. Chang, C. Rinciog, A. Guiraud-Diawara, C. Marre, and K. Hansen contributed to the study concept and design with input from the co-authors. Data collection was the work of L. Sawyer, with input from J. Azorin, S. Chang, C. Rinciog, A. Guiraud-Diawara, C. Marre, and K. Hansen. Model development was the work of S. Chang, with input from L. Sawyer and C. Rinciog. Drafting the manuscript and its revision were primarily the work of L. Sawyer and C. Rinciog with input from the co-authors and editorial support from Jana Tillotson, independent medical writer.
References
- Hirschfeld RM, Vornik LA. Bipolar disorder–costs and comorbidity. Am J Manag Care 2005;11:S85-90
- Muller-Oerlinghausen B, Berghofer A, et al. Bipolar disorder. Lancet 2002;359:241-7
- Das GR, Guest JF. Annual cost of bipolar disorder to UK society. Br J Psychiatry 2002;180:227-33
- Fajutrao L, Locklear J, Priaulx J, et al. A systematic review of the evidence of the burden of bipolar disorder in Europe. Clin Pract Epidemiol Ment Health 2009;5:3
- Kleine-Budde K, Touil E, Moock J, et al. Cost of illness for bipolar disorder: a systematic review of the economic burden. Bipolar Disord 2013; epub ahead of print. Available at: http://onlinelibrary.wiley.com/doi/10.1111/bdi.12165/abstract [Last accessed 20 December 2013]
- Kleinman L, Lowin A, Flood E, et al. Costs of bipolar disorder. Pharmacoeconomics 2003;21:601-22
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5). 5th edn. Arlington, VA: American Psychiatric Publishing, 2013
- Martin-Carrasco M, Gonzalez-Pinto A, Galan JL, et al. Number of prior episodes and the presence of depressive symptoms are associated with longer length of stay for patients with acute manic episodes. Ann Gen Psychiatry 2012;11:7
- Rosa AR, Reinares M, Franco C, et al. Clinical predictors of functional outcome of bipolar patients in remission. Bipolar Disord 2009;11:401-9
- Michalak EE, Guiraud-Diawara A, Sapin C. Asenapine treatment and health-related quality of life in patients experiencing bipolar I disorder with mixed episodes: post-hoc analyses of pivotal trials. Curr Med Res Opin 2014;30:711-18
- Gonzalez-Pinto A, Barbeito S, Alonso M, et al. Poor long-term prognosis in mixed bipolar patients: 10-year outcomes in the Vitoria prospective naturalistic study in Spain. J Clin Psychiatry 2011;72:671-6
- Osby U, Tiainen A, Backlund L, et al. Psychiatric admissions and hospitalization costs in bipolar disorder in Sweden. J Affect Disord 2009;115:315-22
- Kupfer DJ, Frank E, Grochocinski VJ, et al. Stabilization in the treatment of mania, depression and mixed states. Acta Neuropsychiatrica 2000;12:110-14
- Vieta E, Valenti M. Mixed states in DSM-5: implications for clinical care, education, and research. J Affect Disord 2013;148:28-36
- National Collaborating Centre for Mental Health. Bipolar disorder. The management of bipolar disorder in adults, children and adolescents, in primary and secondary care. National Clinical Practice Guideline Number 38. London, UK: The British Psychological Society and Gaskell, 2006
- Yatham LN, Kennedy SH, Parikh SV, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) collaborative update of CANMAT guidelines for the management of patients with bipolar disorder: update 2013. Bipolar Disord 2013;15:1-44
- American Psychiatric Association. Mixed Features Specifier. 2013. http://www.dsm5.org/Documents/Mixed%20Features%20Specifier%20Fact%20Sheet.pdf. [Last accessed 16 December 2013]
- Azorin JM, Sapin C, Weiller E. Effect of asenapine on manic and depressive symptoms in bipolar I patients with mixed episodes: results from post hoc analyses. J Affect Disord 2013;145:62-9
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders Text Revision (DSM-IV-TR). 5th edn. Arlington, VA: American Psychiatric Publishing, Inc., 2000
- All Wales Medicines Strategy Group. Aripiprazole (Abilify) tablets for the treatment of moderate to severe manic episodes in Bipolar I Disorder and for the prevention of a new manic episode in patients who experienced predominantly manic episodes and whose manic episodes responded to aripiprazole treatment. 2009
- Chisholm D, van OM, Ayuso-Mateos JL et al. Cost-effectiveness of clinical interventions for reducing the global burden of bipolar disorder. Br J Psychiatry 2005;187:559-67
- Fajutrao L, Paulsson B, Liu S, et al. Cost-effectiveness of quetiapine plus mood stabilizers compared with mood stabilizers alone in the maintenance therapy of bipolar I disorder: results of a Markov model analysis. Clin Ther 2009;31:1456-68
- Klok RM, Al Hadithy AF, van Schayk NP, et al. Pharmacoeconomics of quetiapine for the management of acute mania in bipolar I disorder. Expert Rev Pharmacoecon Outcomes Res 2007;7:459-67
- Lebmeier M, Dudley E, Pericleous L, et al. The cost effectiveness of aripiprazole in patients with bipolar I disorder in the UK. ISPOR 13th Annual European Congress. ISPOR 13th Annual European Congress. Prague. ISPOR 13th Annual European Congress, 2010
- McKendrick J, Cerri KH, Lloyd A, et al. Cost effectiveness of olanzapine in prevention of affective episodes in bipolar disorder in the United Kingdom. J Psychopharmacol 2007;21:588-96
- Ramirez de Arellano A, Rubio-Terres C, Baca E, et al. Economic evaluation analysis in the treatment of bipolar disorder with aripiprazole and other atypical antipsychotic drugs in Spain. ISPOR 13th Annual European Congress. ISPOR 13th Annual European Congress. Prague. ISPOR 13th Annual European Congress, 2010
- National Institute for Health and Clinical Excellence. Guide to the methods of technology appraisal. London: National Institute for Health and Clinical Excellence, 2008
- Nivoli AM, Colom F, Murru A, et al. New treatment guidelines for acute bipolar depression: a systematic review. J Affect Disord 2011;129:14-26
- Azorin JM. Personal Communication. 2013
- Vieta E, Gunther O, Locklear J, et al. Effectiveness of psychotropic medications in the maintenance phase of bipolar disorder: a meta-analysis of randomized controlled trials. Int J Neuropsychopharmacol 2011;14:1029-49
- Ekman M, Lindgren P, Miltenburger C, et al. Cost effectiveness of quetiapine in patients with acute bipolar depression and in maintenance treatment after an acute depressive episode. Pharmacoeconomics 2012;30:513-30
- Soares-Weiser K, Bravo VY, Beynon S, et al. A systematic review and economic model of the clinical effectiveness and cost-effectiveness of interventions for preventing relapse in people with bipolar disorder. Health Technol Assess 2007;11:iii-206
- Vieta E, Suppes T, Ekholm B, et al. Long-term efficacy of quetiapine in combination with lithium or divalproex on mixed symptoms in bipolar I disorder. J Affect Disord 2012;142:36-44
- Department of Health. Hospital Episode Statistics, Admitted Patient Care - England 2011–12. 2013. http://www.hscic.gov.uk/searchcatalogue?productid=9161&q=title%3a%22Hospital+Episode+Statistics%3a+Admitted+patient+care%22&sort=Relevance&size=10&page=1#top. [Last accessed 16 December 2013]
- Office for National Statistics. UK Interim Life tables, England and Wales, 2009–2011. 2013. http://www.ons.gov.uk/ons/taxonomy/index.html?nscl=Interim+Life+Tables#tab-data-tables. [Last accessed 16 December 2013]
- Office of National Statistics. Mortality statistics: deaths registered in England and Wales. 2012. http://www.ons.gov.uk/ons/rel/vsob1/mortality-statistics–deaths-registered-in-england-and-wales–series-dr-/2011/index.html. [Last accessed 16 December 2013]
- Angst F, Stassen HH, Clayton PJ, et al. Mortality of patients with mood disorders: follow-up over 34–38 years. J Affect Disord 2002;68:167-81
- Post-hoc analysis of ARES trial dataset. Data on File. 2013
- Ara R, Brazier J. Deriving an algorithm to convert the eight mean SF-36 dimension scores into a mean EQ-5D preference-based score from published studies (where patient level data are not available). Value Health 2008;11:1131-43
- Ara R, Brazier JE. Populating an economic model with health state utility values: moving toward better practice. Value Health 2010;13:509-18
- Joint Formulary Committee. British national formulary (BNF) 66, 66th edn. London: British Medical Association and The Royal Pharmaceutical Society of Great Britain, 2013
- Curtis L. Unit costs of social health care. Canterbury, UK: Personal Social Services Research Unit, University of Kent, 2012
- Department of Health. NHS reference costs 2010–2011. 2012. https://www.gov.uk/government/publications/2010-11-reference-costs-publication. [Last accessed 16 December 2013]
- Department of Health. NHS reference costs 2011–2012. 2013. https://www.gov.uk/government/publications/nhs-reference-costs-financial-year-2011-to-2012. [Last accessed 16 December 2013]
- Brunelle J, Consoli A, Tanguy ML, et al. Phenomenology, socio-demographic factors and outcome upon discharge of manic and mixed episodes in hospitalized adolescents: a chart review. Eur Child Adolesc Psychiatry 2009;18:185-93
- Bridle C, Palmer S, Bagnall AM, et al. A rapid and systematic review and economic evaluation of the clinical and cost-effectiveness of newer drugs for treatment of mania associated with bipolar affective disorder. Health Technol Assess 2004;8:iii-187
- Lam DH, McCrone P, Wright K, et al. Cost-effectiveness of relapse-prevention cognitive therapy for bipolar disorder: 30-month study. Br J Psychiatry 2005;186:500-6
- Revicki DA, Hanlon J, Martin S, et al. Patient-based utilities for bipolar disorder-related health states. J Affect Disord 2005;87:203-10
- Hayhurst H, Palmer S, Abbott R, et al. Measuring health-related quality of life in bipolar disorder: relationship of the EuroQol (EQ-5D) to condition-specific measures. Qual Life Res 2006;15:1271-80
- Briggs A, Wild D, Lees M, et al. Impact of schizophrenia and schizophrenia treatment-related adverse events on quality of life: direct utility elicitation. Health Qual Life Outcomes 2008;6:105