Abstract
Objective:
To evaluate the cost-benefit of using levosimendan compared with dobutamine, in the perioperative treatment of patients undergoing cardiac surgery who require inotropic support.
Methods:
A two-part Markov model was designed to simulate health-state transitions of patients undergoing cardiac surgery, and estimate the short- and long-term health benefits of treatment. Hospital length of stay (LOS), mortality, medication, and adverse events were key clinical- and cost-inputs. Cost-benefits were evaluated in terms of costs and bed stays within the German healthcare system. Drug prices were calculated from the German Drug Directory (€/2014) and published literature, with a 3% annual discount rate applied. The base case analysis was for a 1-year time horizon.
Results:
The use of levosimendan vs dobutamine was associated with cost savings of €4787 per patient from the German hospital perspective due to reduced adverse events and shorter hospital LOS, leading to increased bed capacity and hospital revenue.
Limitations:
A pharmacoeconomic calculation for the specific situation of the German healthcare system that is based on international clinical trial carries a substantial risk of disregarding potentially relevant but unknown confounding factors (i.e., ICU-staffing, co-medications, standard-ICU care vs fast-tracking, etc.) that may either attenuate or increase the outcome pharmacoeconomic effects of a drug; however, since these conditions would also apply for patients treated with comparators, their net effects may not necessarily influence the conclusions.
Conclusions:
The use of levosimendan in patients undergoing cardiac surgery who require inotropic support appears to be cost-saving. The results of the analysis provide a strong rationale to run local clinical studies with pharmacoeconomic end-points which would allow a much more precise computation of the benefits of levosimendan.
Introduction
Cardiac surgery is associated with a number of outcomes, which can lead to organ dysfunctionCitation1,Citation2. Acute cardiovascular dysfunction represents a concern for the ongoing safety of cardiac surgical proceduresCitation3. Low cardiac output syndrome (LCOS) poses a particular risk in cardiac surgery, affecting ∼3–14% of patients undergoing coronary artery bypass graft (CABG) surgery, and increases the risk of mortality by 10–15-foldCitation3,Citation4. LCOS following CABG can also cause pulmonary complications, myocardial infarction, stroke, and renal failure, which ultimately increase the need for further operationsCitation4,Citation5. These complications may lead to longer intensive care unit (ICU) and hospital length of stay (LOS), resulting in increased costsCitation5,Citation6.
Preserving cardiac function during cardiac surgery is, therefore, a major goal for patient safety, and pharmacological support with inotropes is frequently initiated in the perioperative period to improve post-surgical outcomes, being also important after weaning from cardiopulmonary bypass (CPB) or CABG surgeryCitation7.
Inotropes’ use is usually reserved for patients with low systolic blood pressure or a low cardiac index in the presence of signs of hypoperfusion or congestionCitation8,Citation9. A major drawback of classical inotropic agents such as dobutamine, milrinone, or enoximone, is that they increase myocardial oxygen consumption, which can result in ischemia and contribute to increased mortality and major post-operative morbidityCitation10.
Levosimendan is a calcium sensitizer and potassium channel opener which has been in clinical use for over 15 years. Levosimendan acts through three complementary mechanisms: calcium sensitization, which results in a positive inotropic effect without increase in oxygen consumption; the opening of KATP channels in vascular smooth muscle cells, which results in increased blood perfusion in key organs; and the opening of KATP channels in cardiomyocyte mitochondria, which has a cardioprotective effectCitation11,Citation12.
Consequently, treatment with levosimendan does not only lead to improved hemodynamics, but is associated with less organ dysfunction and improved recovery from the surgical insultCitation13–16.
A number of meta-analyses have shown that the use of levosimendan during cardiac surgery results in reduced mortality and post-operative complicationsCitation17–19, the clinical benefits being particularly evident in patients with severely compromised left ventricular functionCitation15–17.
Economic analyses have demonstrated that levosimendan is cost-effective for acute heart failure, largely due to a reduction in hospital LOS and rate of re-hospitalizationsCitation20–23. However, the cost-effectiveness of levosimendan has not been explored in a cardiac surgery setting in patients with severely compromised left ventricular function. Given the previous results on reduced mortality with levosimendan compared with dobutamine and standard of careCitation17–19, the aim of the current study was to assess the potential cost-savings associated with levosimendan treatment compared with dobutamine for patients in need of inotropic support following cardiac surgery. To this end, a pharmacoeconomic model was built, with Germany as the base case, and for two different perspectives: hospital and health insurance.
Methods
Model overview
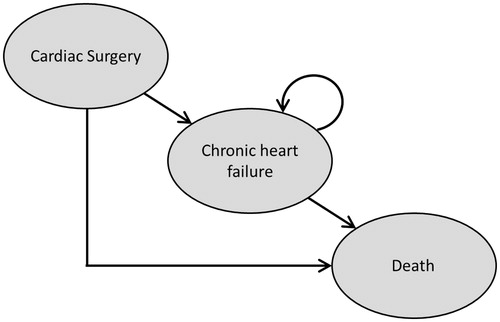
The pharmacoeconomic model was developed in Microsoft Excel 2010. The model structure consisted of a two-part Markov model with three health states: ‘cardiac surgery’, ‘chronic heart failure’ (CHF), and ‘death’ (). The Markov model was split into two distinct timeframes. For the first 6 months after cardiac surgery, monthly cycles were used to capture the differences among the treatments in transitions between the health states. After 6 months, a time horizon of 1 year was chosen for the analysis. Dobutamine was identified as the most relevant comparator due to its extensive use in clinical practice. In the base case analysis, both costs and health outcomes were discounted at a rate of 3.0% per annum in line with German healthcare guidelinesCitation24.
The model was designed primarily to assess the hospital perspective within the German healthcare system. The German reimbursement system pays hospitals through a dual-financing system, in which infrastructure investments are paid by tax-funded state budgets and operating costs are reimbursed through sickness funds and private health insurers. Operating costs are paid through diagnostic-related group- (DRG) type hospital payment, with the hospital receiving a reimbursement fee per admitted patient. Additional payment for serious complications or adverse events (such as post-operative renal failure, myocardial infarction, and treatment with an intra-aortic balloon pump) is also available. In the pharmacoeconomic model, the reimbursement fee was used as a proxy for the costs incurred by the hospital whilst the patient was in their care.
Further costs incurred by the hospital included the cost of inotropic support and the treatment of atrial fibrillation. The analyses also considered costs from the health insurance perspective. The health insurance perspective assumed that the insurer incurred the cost of reimbursement whilst the patient was in hospital, as well as additional outpatient and medication costs once the patient had been discharged.
Revenue generation was calculated based on reduced hospital LOS. Levosimendan is associated with a shorter hospital LOS than dobutamineCitation19. This allows hospital beds to be used for more hospital admissions. In order to calculate the extra revenue gained, mean revenue per day was calculated by dividing the mean reimbursement for the patients in the cardiac surgery department by the mean LOS. The DRG codes used to calculate mean revenue per day are reflective of the additional patients who could be admitted due to freed capacity. Bed occupancy was assumed to be 100% in the model base case, but was reduced for sensitivity analyses.
Model inputs
Clinical data
A number of key clinical data inputs were used in the model, including hospital LOS, mortality, and adverse event rates. Three key meta-analyses were identified which evaluated the efficacy of levosimendan in patients undergoing cardiac surgery and requiring inotropes, as shown in . Data from additional clinical studies were also used to estimate model parametersCitation16,Citation25.
Table 1. Data input in the model. Key clinical data sources for the pharmaco-economic model.
Hospital LOS
Only one relevant study of patients undergoing cardiac surgery was identified that reported relevant LOS data in terms of days spent in the hospital ICUCitation25. As data on LOS in routine care were not available, an assumption was made that the overall LOS for patients who receive dobutamine is 13.10 days, in line with the average LOS for the relevant DRG codesCitation26. Based on an overall stay of 13.10 days for patients who receive comparator treatments, LOS in routine care was calculated as 6.52 days. The model conservatively assumed that the duration in routine care was the same for all treatments ().
Table 2. Data input in the model. Data inputs for hospital length of Stay; mean (SD).
Probabilities of death
All-cause mortality data was based on life tables from the German Federal Office of StatisticsCitation27. HF-related deaths in the CHF state were based on a cost-effectiveness analysis for natriuretic peptide testing in patients with acute heart failureCitation28. The data used in this study were part of a secondary analysis of national heart failure audits from 2009–2013.
Probabilities of adverse events
Adverse events of cardiac surgery are an important consideration, and have a great impact on the overall cost of treatment. Given that adverse event rates were not available for levosimendan compared with dobutamine, rates for standard of care were used for both comparator arms, as shown in (see Harrison et al.Citation17). The probability of a patient requiring treatment with an intra-aortic balloon pump for levosimendan compared with dobutamine was taken from a randomized controlled trial comparing levosimendan and dobutamine in patients with LCOS following cardiac surgeryCitation25. The probability of a patient requiring treatment with an intra-aortic balloon pump for levosimendan compared with standard of care was taken from a randomized controlled trial comparing levosimendan with placeboCitation16. A similar profoundly decreased need for intra-aortic balloon pump was observed in a 200-patient cardiac surgery study against placebo; one patient (1%) in the levosimendan vs nine patients (9%) in the placebo group (risk ratio = 0.11; 95% CI = 0.01–0.87), p = 0.018Citation29.
Table 3. Data input in the model. Probability of adverse events.
Cost data
Medication costs
The costs of levosimendan and dobutamine were calculated using dosing information, average patient weight, and unit costs from the summary of product characteristics for each drugCitation30,Citation31. The cost per vial (€745.00) and vial size (12.50 mg) for levosimendan as well as the cost per vial (€10.35) and vial size (250.00 mg) for dobutamine were taken from the LAUER-Taxe online German pharmaceutical pricing databaseCitation32.
Hospitalization costs
Reimbursement for cardiac surgery patients used in the model was calculated by identifying the DRG codes that are relevant to cardiac surgery from the Fallpauschalen-Katalog and taking the average weighted by the number of cases for each code in 2013–2014Citation26; the weighted average price was €18 609.84.
Adverse event costs
The model assumed that atrial fibrillation is treated with an intravenous infusion of 10–15 mg of metoprolol (unit cost = €1.54). Medication costs were incurred by the hospital, and priced according to the LAUER-Taxe online German pharmaceutical pricing databaseCitation32. Costs associated with renal failure, myocardial injury, or treatment with an intra-aortic balloon pump were reimbursed by the insurer. If cardiac surgery patients experienced a myocardial injury whilst in hospital, the hospital would receive a higher base DRG reimbursement. The weighted mean reimbursement was calculated at €20 396.33. The additional reimbursement received by the hospital was calculated by taking the difference between the DRG reimbursement for patients who experienced a myocardial injury (€20 396.33) and the base case DRG reimbursement (€18 609.84). This difference (€1786.50) represented an additional cost to the insurer.
Costs associated with renal failure led to similar additional reimbursement for the hospital. A number of DRG codes were applicable; the default code used (ZE120.04 8-854.63, Hemodialysis, continuously: venovenous, driven pumps [CVVHD]; Anticoagulation with heparin or without anticoagulation: more than 144 hours to 264 hours) had a value of €2672.94Citation26. Similarly, costs associated with intra-aortic balloon pump treatment can led to an additional reimbursement of €2400.00 to cover the cost of treatment.
CHF costs
Resource use for patients in the CHF state were taken from German-specific guidelines for the management of CHFCitation28,Citation33. Unit costs for medication were taken from the LAUER-Taxe online German pharmaceutical pricing databaseCitation32. Resource use associated with outpatient costs were taken from the National Clinical Guideline Centre cost-effectiveness model for natriuretic peptide testing in AHF patientsCitation28.
Although this analysis was for a population of patients with acute heart failure, the model assumed that resource use for the CHF state would be the same. This assumption was validated by a clinical expert. German-specific unit costs were sourced from the German Einheitlicher Bemessungsmaβstab (EBM catalog)Citation34 and validated by a German clinical expertCitation35.
Sensitivity analyses
Sensitivity analyses were conducted to assess the impact of varying model assumptions and parameters on model results in terms of clinical parameters, transition probabilities, and drug costs.
Results
Hospital cost savings
Patient outcomes in the base case analysis, as obtained by using the Markov model with a time horizon of 1 year, are shown in . Treatment with levosimendan resulted in a greater survival benefit (71.93% of patients alive) at 1 year after surgery compared with dobutamine (64.97% of patients alive). Treatment with levosimendan also led to hospital bed savings, with a mean of 1.915 bed days saved per patient compared with dobutamine.
Table 4. Patient outcomes in the base case analysis (cohort size = 1000; time horizon = 1 year).
A comprehensive breakdown of costs from the hospital perspective is provided in . The hospital perspective included the cost of inotrope and costs for adverse events not associated with a DRG code (such as atrial fibrillation). Foregone revenue was also taken into account to provide the total net costs. These results are presented for levosimendan and dobutamine, alongside the total incremental cost and the incremental cost per patient.
Table 5. Costs breakdown from the hospital perspective (cohort size = 1000; time horizon = 1 year; costs in €).
The cost breakdown shows that, although the cost of levosimendan is higher than that of dobutamine, the savings made in terms of adverse events (not associated with a DRG code) and forgone revenue led to an overall cost saving made by the hospital when using levosimendan. This was calculated as total net cost savings of €4787 per patient, in the base case setting. Cost savings were also calculated from the health insurance perspective, as shown in .
Table 6. Costs breakdown from the health insurance perspective (cohort size = 1000; time horizon = 1 year; costs in €).
Health insurance cost savings
From the health insurance perspective, the use of levosimendan remained cost-saving as well, leading to savings of €432 per patient.
Sensitivity analyses
When bed occupancy was assumed to be 80%, instead of 100% as in the base case analysis, the foregone revenue and total net costs were slightly decreased (see ). This is to be expected, as the number of additional admissions that the hospital would be able to process would be lower. A threshold analysis indicated that it would take the hospital to have bed occupancy as low as 12% to break-even.
Table 7. Costs breakdown from the hospital perspective, with bed occupancy assumed at 80% (costs in €).
A cost-effectiveness plane for incremental costs vs incremental bed days (data not shown) suggests that hospital costs tend to be stable and not subject to uncertainty, whilst the incremental bed days capture uncertainty in the hospital length of stay associated with levosimendan and dobutamine. However, the conclusions of the analysis do not change and levosimendan remains cost saving for a range of LOS values.
Sensitivity analysis showed that, as the time horizon of the analysis is increased, the cost savings of levosimendan tend to decrease. This is due to the lower mortality rates for levosimendan and the associated chronic heart failure medication and outpatient costs of patients who survive. However, even when a lifetime horizon is considered, the cost savings of levosimendan remain positive (see ).
Table 8. Cost-effectiveness results (cost savings per patient) presented over a variety of time horizons.
Discussion
The clinical benefits of using levosimendan in cardiac surgery have been demonstrated in a number of studiesCitation1,17,36–40. In the current economic analysis, which evaluated the costs of levosimendan compared with dobutamine in patients requiring inotropic support following cardiac surgery in Germany, levosimendan was found to be cost saving from the hospital and health insurance perspectives. While the drug acquisition costs were higher for levosimendan compared with dobutamine, costs were saved by the lower incidence of adverse events and shorter hospital LOS, potentially leading to increased hospital revenue.
Very few economic analyses of pharmacological interventions in cardiac surgery are available; this study, therefore, provides a useful platform upon which further economic analyses can be performed. The present study compliments economic modelling studies that demonstrate the cost-effectiveness of levosimendan in the treatment of acute heart failure. It has been shown that levosimendan is cost-effective compared with dobutamine in patients who experience acute heart failure in European countries, based on data from the Survival of Patients with Acute Heart Failure in Need of Intravenous Inotropic Support (SURVIVE) studyCitation41. An Italian observational study also found levosimendan to be cost-effective compared with dobutamine in patients treated for acute heart failureCitation22. Similarly, an analysis of data from the LIDO study showed that levosimendan was a cost-effective alternative to dobutamine in patients who experience severe low-output heart failureCitation20. Finally, an American study of patients who experience acute heart failure found that levosimendan would likely represent a cost-effective alternative to standard of care (placebo), based on data from the Randomized EValuation of Intravenous Levosimendan Efficacy (REVIVE II) studyCitation21.
Levosimendan has also demonstrated benefits in ventilatory function. A randomized study in healthy volunteers showed that subjects receiving levosimendan had a 21% increase in neuromechanical efficiency of the diaphragm (p < 0.05) compared with no change in the placebo groupCitation42. Levosimendan was compared to dobutamine in difficult-to-wean patients with chronic obstructive pulmonary diseaseCitation43. Levosimendan resulted in significantly greater inhibition of spontaneous ventilation induced increase in pulmonary artery occlusion pressure. Similarly, mean pulmonary artery pressure increased to a lesser extent with levosimendan than with dobutamine.
Finally, an observational study of ventilator-dependent patients in the ICU after critical illness found that those treated with levosimendan exhibited significant improvement in weaning from mechanical ventilation compared with patients who received placebo (p < 0.02)Citation44. This would lead to increased likelihood of separation from mechanical ventilation, which represents a substantial expense for hospitals. While it was not possible to include these costs in the current economic model due to a lack of available data in cardiac surgery, it is likely that further savings would be achieved in a real-world setting in hospitals using levosimendan as a result of a lowered requirement for mechanical ventilation of patients.
Limitations
The present study is associated with a number of limitations, largely due to the scarce availability of data. First, it must be taken into account that the mode and the timing of levosimendan application may affect the pharmacoeconomic benefit of this drug compared with treatment with traditional beta-mimetic or phosphodiesterase inhibitors. In case of beta-mimetic drugs like dobutamine, it was shown that a peri- and post-operative use in cardiac surgical patientsCitation45,Citation46 is common. On the contrary, the available data show that the beneficial effects on mortality and organ dysfunction of levosimendan are most prominent upon pre-emptive and pre-operative use and in patients with reduced left ventricular functionCitation16,Citation17. Also, levosimendan had no effect on long-term mortality in a study in which the drug was started after induction of anesthesiaCitation29.
Second, limited evidence was available regarding the effect of levosimendan compared with comparator treatments on hospital LOS. The model used an ICU LOS that compared levosimendan with dobutamineCitation40, but, due to a lack of data on LOS in routine care it was necessary to assume that LOS was the same, regardless of treatment typeCitation40. This would prove a conservative assumption, as it is likely that in clinical practice patients who experience a shorter ICU stay also experience a shorter stay in routine care. On the other hand, since hospital LOS in the German healthcare system may be—at least in part—directed by DRG reimbursement (i.e., the hospital receives a deducted reimbursement if a patient is discharged earlier than a pre-specified hospital LOS), this assumption may reflect clinical reality.
Third, the costs associated to the administration of other inotropes on top of levosimendan as rescue therapy was not taken into account. Inotropes such as milrinone and enoximone, in fact, are costing significantly more than the relatively cheap dobutamine or norepinephrine, and their use concomitantly with levosimendan could have a strong impact on the economic and clinical results. Clear data, however, are available which show how in cardiac surgery the use of levosimendan is associated with a decrease of the use of milrinone or other rescue therapyCitation29. It is safe, therefore, to conclude that no additional costs for such additional drugs will be required, in a comparison of levosimendan vs other inotropes. Indeed, a more accurate pharmacoeconomic analysis could show that such costs should be allocated to the comparator arm, resulting is a more pronounced advantage for levosimendan.
Finally, it has to be taken into account that a pharmaco-economic calculation for the specific situation of the German healthcare system that is based on estimates derived from study cohorts treated outside Germany carries a substantial risk of disregarding potentially relevant but unknown confounding factors (i.e., ICU-staffing, co-medications, standard-ICU care vs fast-tracking, etc.) that may either attenuate or increase the outcome pharmaco-economic effects of a drug. However, since these conditions would also apply for patients treated with comparators, their net effects may not necessarily influence the conclusions.
Conclusions
Overall, taking into account all the limitations, the present analysis suggests that the treatment of cardiac surgery patients with levosimendan compared with dobutamine may be cost-effective and even facilitate cost-savings from a German hospital perspective. We warrant, however, that local clinical studies with pharmaco-economic end-points would allow a more precise computation of such benefits.
Transparency
Declaration of financial relationship
MK, PP, and MA are employees of Orion Pharma. SM, CS, and EB are employees of PAREXEL, which received funding for this project by Orion Pharma. MH did not receive any funding for this project, but has received lecture honoraria from Orion Pharma. JME peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
This paper was funded by Orion Pharma, the originator of levosimendan.
References
- Toller W, Heringlake M, Guarracino F, et al. Preoperative and perioperative use of levosimendan in cardiac surgery: European expert opinion. Int J Cardiol 2015;184:323-36
- O’Brien MM, Shroyer AL, Moritz TE, et al.; VA Cooperative Study Group on Processes, Structures, and Outcomes of Care in Cardiac Surgery. Relationship between processes of care and coronary bypass operative mortality and morbidity. Med Care 2004;42:59-70
- Mebazaa A, Pitsis AA, Rudiger A, et al. Clinical review: practical recommendations on the management of perioperative heart failure in cardiac surgery. Crit Care 2010;14:201
- Algarni KD, Elhenawy AM, Maganti M, et al. Decreasing prevalence but increasing importance of left ventricular dysfunction and reoperative surgery in prediction of mortality in coronary artery bypass surgery: trends over 18 years. J Thorac Cardiovasc Surg 2012;144:340-6
- Ding W, Ji Q, Shi Y, et al. Predictors of low cardiac output syndrome after isolated coronary artery bypass grafting. Int Heart J 2015;56:144-9
- Legare JF, Hirsch GM, Buth KJ, et al. Preoperative prediction of prolonged mechanical ventilation following coronary artery bypass grafting. Eur J Cardiothorac Surg 2001;20:930-6
- Gillies M, Bellomo R, Doolan L, et al. Bench-to-bedside review: Inotropic drug therapy after adult cardiac surgery – a systematic literature review. Crit Care 2005;9:266-79
- Teerlink JR, Metra M, Zacà V, et al. Agents with inotropic properties for the management of acute heart failure syndromes. Traditional agents and beyond. Heart Fail Rev 2009;14:243-53
- Carl M, Alms A, Braun J, et al.; German Society for Thoracic and Cardiovascular Surgery; German Society of Anaesthesiology and Intensive Care Medicine. Guidelines for intensive care in cardiac surgery patients: haemodynamic monitoring and cardio-circulatory treatment guidelines of the German Society for Thoracic and Cardiovascular Surgery and the German Society of Anaesthesiology and Intensive Care Medicine. Thorac Cardiovasc Surg 2007;55:130-48
- Nielsen DV, Hansen MK, Johnsen SP, et al. Health outcomes with and without use of inotropic therapy in cardiac surgery: results of a propensity score-matched analysis. Anesthesiology 2014;120:1098-108
- Papp Z, Édes I, Fruhwald S, et al. Levosimendan: molecular mechanisms and clinical implications: consensus of experts on the mechanisms of action of levosimendan. Int J Cardiol 2012;159:82-7
- Leprán I, Pollesello P, Vajda S, et al. Preconditioning effects of levosimendan in a rabbit cardiac ischemia-reperfusion model. J Cardiovasc Pharmacol 2006;48:148-52
- Nieminen MS, Fruhwald S, Heunks LM, et al. Levosimendan: current data, clinical use and future development. Heart Lung Vessels 2013;5:227-45
- Eriksson HI, Jalonen JR, Heikkinen LO, et al. Levosimendan facilitates weaning from cardiopulmonary bypass in patients undergoing coronary artery bypass grafting with impaired left ventricular function. Ann Thorac Surg 2009;87:448-54
- De Hert SG, Lorsomradee S, Cromheecke S, et al. The effects of levosimendan in cardiac surgery patients with poor left ventricular function. Anesth Analg 2007;104:766-73
- Levin R, Degrange M, Del Mazo C, et al Preoperative levosimendan decreases mortality and the development of low cardiac output in high-risk patients with severe left ventricular dysfunction undergoing coronary artery bypass grafting with cardiopulmonary bypass. Exp Clin Cardiol 2012;17:125-30
- Harrison RW, Hasselblad V, Mehta RH, et al. Effect of levosimendan on survival and adverse events after cardiac surgery: a meta-analysis. J Cardiothorac Vasc Anesth 2013;27:1224-32
- Landoni G, Biondi-Zoccai G, Greco M, et al. Effects of levosimendan on mortality and hospitalization. A meta-analysis of randomized controlled studies. Crit Care Med 2012;40:634-46
- Huang X, Lei S, Zhu MF, et al. Levosimendan versus dobutamine in critically ill patients: a meta-analysis of randomized controlled trials. J Zhejiang Univ Sci B 2013;14:400-15
- Cleland JG, Takala A, Apajasalo M, et al. Intravenous levosimendan treatment is cost-effective compared with dobutamine in severe low-output heart failure: an analysis based on the international LIDO trial. Eur J Heart Fail 2003;5:101-8
- de Lissovoy G, Fraeman K, Teerlink JR, et al. Hospital costs for treatment of acute heart failure: economic analysis of the REVIVE II study. Eur J Health Econ 2010;11:185-93
- Lucioni C, D’Ambrosi A, Mazzi S, et al. Economic evaluation of levosimendan versus dobutamine for the treatment of acute heart failure in Italy. Adv Ther 2012;29:1037-50
- Nieminen MS, Buerke M, Parissis J, et al. Pharmaco-economics of levosimendan in cardiology: A European perspective. Int J Cardiol 2015;199:337–41
- Institute for Quality and Efficiency in Health Care. Working Paper: Cost Estimation. 2009. http://www.ispor.org/peguidelines/source/Germany_WorkPaperCostEst.pdf, p. 12. Accessed September 4, 2015
- Levin R, Degrange M, Porcile R, et al. Preoperative use of calcium sensitizer levosimendan reduces mortality and low cardiac output syndrome in patients with aortic stenosis and left ventricular dysfunction. Circulation 2008;118:E217
- InEK. Fallpauschalen-Katalog 2014. 2014. http://www.g-drg.de/cms/G-DRG-System_2014/Fallpauschalen-Katalog/Fallpauschalen-Katalog_2014. Accessed September 4, 2015
- Statistische Bundesamt. Sterbetafeln 2005/2007, Früheres Bundesgebiet und neue Länder. 2009. https://www.destatis.de/DE/Publikationen/Thematisch/Bevoelkerung/Bevoelkerungsbewegung/PeriodensterbetafelnPDF_5126202.pdf?__blob=publicationFile, p. 359–70. Accessed September 4, 2015
- National Clinical Guideline Centre. Acute heart failure: diagnosing and managing acute heart failure in adults; Commissioned by the National Institute for Health and Care Excellence. 2014. https://www.nice.org.uk/guidance/cg187/resources/guidance-acute-heart-failure-pdf. Accessed September 4, 2015
- Lahtinen P, Pitkänen O, Pölönen P, et al. Levosimendan reduces heart failure after cardiac surgery: a prospective, randomized, placebo-controlled trial. Crit Care Med 2011;39:2263-70
- Orion Pharma. Levosimendan − Summary of Product Characteristics. Orion Pharma. 2015. http://www.simdax.com/About-Simdax/SPC/. Accessed September 4, 2015
- Electronic Medicines Compendium. Summary of product characteristics: Dobutamine. 2013. http://www.evidence.nhs.uk/bnf/Search?om=[{%22srn%22:[%22electronic%20Medicines%20Compendium%20-%20eMC%22]}]&q=dobutamine&cid=5001. Accessed September 4, 2015
- LAUER-taxe. LAUER-taxe online pharmaceutical pricing database. 2014. http://www2.lauer-fischer.de/produkte/lauer-taxe/webapo-infosystem-english/. Accessed September 4, 2015
- Programm für Nationale VersorgungsLeitlinien. Nationale VersorgungsLeitlinie: Chronische Herzinsuffizienz. 2014. http://www.leitlinien.de/mdb/downloads/nvl/herzinsuffizienz/herzinsuffizienz-1aufl-vers7-lang.pdf. Accessed September 4, 2015
- German Einheitlicher Bemessungsmaβstab (EBM Katalog). 2015. http://www.kbv.de/html/online-ebm.php. Accessed September 4, 2015
- Kassenärztliche Bundesvereinigung. EBM Katalog (Einheitlicher Bemessungsmaβstab). 2015. http://www.kbv.de/media/sp/EBM_Gesamt___Stand_1._Quartal_2015.pdf. Accessed September 4, 2015
- Baysal A, Yanartas M, Dogukan M, et al. Levosimendan improves renal outcome in cardiac surgery: A randomized trial. J Cardiothorac Vasc Anesth 2014;28:586-94
- Balzer F, Treskatsch S, Spies C, et al. Early administration of levosimendan is associated with improved kidney function after cardiac surgery inverted question mark a retrospective analysis. J Cardiothorac Surg 2014;9:167
- Alvarez J, Baluja A, Selas S, et al. A comparison of dobutamine and levosimendan on hepatic blood flow in patients with a low cardiac output state after cardiac surgery: a randomised controlled study. Anaesth Intensive Care 2013;41:719-27
- Ersoy O, Boysan E, Unal EU, et al. Effectiveness of prophylactic levosimendan in high-risk valve surgery patients. Cardiovasc J Afr 2013;24:260-4
- Levin RL, Degrange MA, Porcile R, et al. The calcium sensitizer levosimendan gives superior results to dobutamine in postoperative low cardiac output syndrome. Rev Esp Cardiol 2008;61:471-9
- de Lissovoy G, Fraeman K, Salon J, et al. The costs of treating acute heart failure: an economic analysis of the SURVIVE trial. J Med Econ 2008;11:415-29
- Doorduin J, Sinderby CA, Beck J, et al. The calcium sensitizer levosimendan improves human diaphragm function. Am J Respir Crit Care Med 2012;185:90-5
- Ouanes-Besbes L, Ouanes I, Dachraoui F, et al. Weaning difficult-to-wean chronic obstructive pulmonary disease patients: a pilot study comparing initial hemodynamic effects of levosimendan and dobutamine. J Crit Care 2011;26:15-21
- Sterba M, Banerjee A, Mudaliar Y. Prospective observational study of levosimendan and weaning of difficult-to-wean ventilator dependent intensive care patients. Crit Care Resusc 2008;10:182-6
- Kastrup M, Markewitz A, Spies C, et al. Current practice of hemodynamic monitoring and vasopressor and inotropic therapy in post-operative cardiac surgery patients in Germany: results from a postal survey. Acta Anaesthesiol Scand 2007;51:347-58
- Kastrup M, Braun J, Kaffarnik M, et al. Catecholamine dosing and survival in adult intensive care unit patients. World J Surg 2013;37:766-73