Abstract
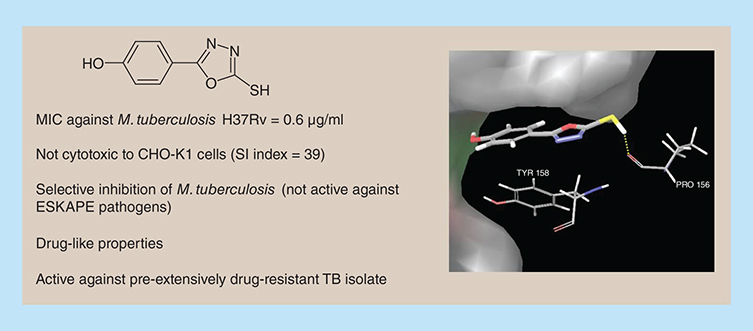
Aim: In recent times, heterocyclic chemotypes are being explored for the development of new antimycobacterials that target the drug-resistant tuberculosis. Here, we are disclosing the 5-substitued 2-mercapto-1,3,4-oxadiazoles as potent antitubercular agents. Methodology: A small library of 2-mercapto-1,3,4-oxadiazoles was synthesized using various acids. The compounds were evaluated for antituberculosis activity against M. tuberculosis H37Rv. Results: Compound 8j was identified as antitubercular lead with MIC of 0.6 μg/ml against M. tuberculosis H37Rv. This compound was nontoxic to CHO-K1 cells and showed selectivity index of 39. Of note, 8j showed antitubercular activity against pre-extensively drug-resistant clinical isolate of Mycobacterium with MIC of 2 μg/ml. Conclusion: This study provides potent antitubercular agent which can be further optimized to discover novel antibiotics.
Graphical abstract
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at:www.tandfonline.com/doi/full/10.2217/epi-2016-0184
Financial & competing interests disclosure
The financial support through a research grant (GAP0584) from Department of Science and Technology, Government of India is acknowledged. Vitthal B Makane and Manjulika Shukla thanks the Council of Scientific and Industrial Research (CSIR), and UGC, Government of India, for research fellowship. This research work has CSIR-IICT communication number IICT/pubs./2018/307. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations.
