Abstract
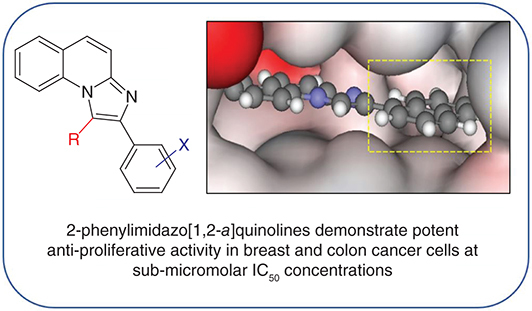
Background: It has been demonstrated that the lead compound 2-phenylimidazo[1,2-a]quinoline 1a selectively inhibits CYP1 enzymes. Additionally, CYP1 inhibition has been linked to inducing antiproliferative effects in various breast cancer cell lines as well as relieving drug resistance caused by CYP1 upregulation. Materials & methods: Herein, 54 novel analogs of 2-phenylimidazo[1,2-a]quinoline 1a have been synthesized with varied substitution on the phenyl and imidazole rings. Antiproliferative testing was conducted using 3H thymidine uptake assays. Results: 2-Phenylimidazo[1,2-a]quinoline 1a and phenyl-substituted analogs 1c (3-OMe), 1n (2,3-napthalene) displayed excellent anti-proliferative activities, demonstrating their potency against cancer cell lines for the first time. Molecular modeling suggested that 1c and 1n bind similarly to 1a in the CYP1 binding site.
Graphical abstract
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at:www.tandfonline.com/doi/full/10.2217/epi-2016-0184
Author contributions
LI Pilkington: conceptualization, development of methodology, data acquisition and analysis, project supervision and administration, funding acquisition, critical review and revision of this article. D Barker: development of methodology and analysis, critical review and revision of this article E Leung: data acquisition and analysis, critical review and revision of this article. H Jones-Moore: data acquisition, critical review and revision of this article. SWP Rees: data acquisition and analysis, drafting of this article. All authors have read and agreed to the published version of this article.
Financial & competing Interests disclosure
The authors would like to thank and acknowledge those that funded this research; Lottery Health Research (New Zealand, Post-Doctoral Fellowship, LI Pilkington), RSNZ (The Royal Society of New Zealand) Rutherford Foundation (Post-Doctoral Fellowship, LI Pilkington) and the University of Auckland (New Zealand, Doctoral Scholarship SWP Rees). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in this article apart from those disclosed.
No writing assistance was utilized in the production of this article.
