Abstract
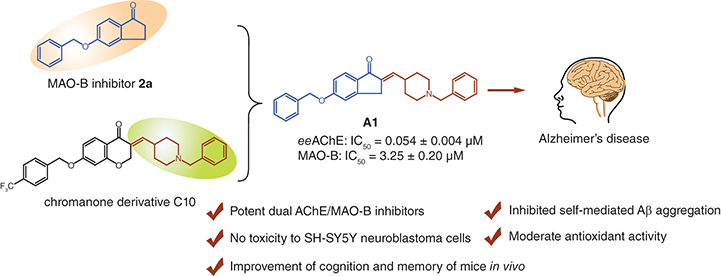
Aim: Based on a multitarget design strategy, a series of novel indanone-1-benzyl-1,2,3,6-tetrahydropyridin hybrids were identified for the potential treatment of Alzheimer's disease (AD). Results: These compounds exhibited significant inhibitory activities against acetylcholinesterase (AChE) and moderate inhibitory activities toward monoamine oxidase B (MAO-B). The optimal compound A1 possessed excellent dual AChE/MAO-B inhibition both in terms of potency (AChE: IC50 = 0.054 ± 0.004 μM; MAO-B: IC50 = 3.25 ± 0.20 μM), moderate inhibitory effects on self-mediated amyloid-β (Aβ) aggregation and antioxidant activity. In addition, compound A1 exhibited low neurotoxicity. More importantly, compound A1 showed significant cognitive and spatial memory improvements in the scopolamine-induced AD mouse model. Conclusion: All results suggest that compound A1 may become a promising lead of anti-AD drug for further development.
Graphical Abstract
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at:www.tandfonline.com/doi/full/10.2217/epi-2016-0184
Author contributions
J Xu and J Liu are responsible for the study's design, funding acquisition, progress monitoring, technical guidance provision, manuscript compilation and editing, as well as final revision. Z Zhu is responsible for providing technical guidance. Z Hu is responsible for design of new compounds, chemical synthesis and structural elucidation of most of the new compounds, as well as conducting molecular modeling studies, a part of the monoamine oxidase B and acetylcholinesterase assay and writing original draft. S Zhou conducted in vitro biological investigations and in vivo cognitive impairment experiments. J Li, X Li and Y Zhou conducted a portion of the synthesis as well as performing the monoamine oxidase B and acetylcholinesterase assay.
Financial disclosure
This study was supported by the National Natural Science Foundation of China (no. 81874289), ‘Double First-Class’ University Project (no. CPU2022PZQ12), China Pharmaceutical University. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosures
No writing assistance was utilized in the production of this manuscript.
