Abstract
Usually, syringomas display a wide spectrum of benign adnexal neoplasms derived from the intraepidermal part of eccrine sweat ducts. Women are more frequently affected than men. Syringomas mainly occur in the periorbital area, but also affect the scalp, forehead, neck, abdomen, and extremities. A variety of clinical presentations of syringomas have been published, as well as cases associated with Down`s syndrome and familial syringoma. The eruptive occurrence of multiple syringomas in healthy persons has been reported rarely. We report a small case series of patients with syringomas, highlighting the different clinicopathologic manifestations and variations, and review the recent literature.
Introduction
Usually, syringomas display a wide spectrum of benign adnexal neoplasms derived from the intraepidermal part of eccrine sweat ducts.Citation1 Women are more frequently affected than men. Syringomas mainly occur in the periorbital area, but also affect the scalp, forehead, neck, abdomen and extremities. A variety of clinical presentations of syringomas have been published, as well as cases associated with Down syndromeCitation1 and familial syringoma. The eruptive occurrence of multiple syringomas in healthy persons has been reported rarely. We report a small case series of patients with syringomas, highlighting the different clinicopathologic manifestations and variations, and review the recent literature.
Discussion
Histopathology findings and variants.
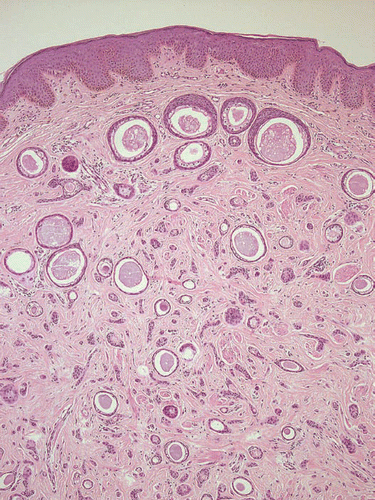
In general, syringomas present as symmetrical and well-delimited lesions located in the upper part of the dermis. Usually, there is no connection to the overlying epidermis. Syringomas are composed of single-to-double-layered epithelial cells with pale eosinophilic cytoplasm. Epithelial cells, forming nests, cords or tubules, with a typical comma-like tail characterize the neoplasm. Within the tubular lumen, PAS-positive eosinophilic material exists.Citation1 Four clinical variants have been distinguished, following the proposal of Friedman and Butler in 1987, as follows:Citation2 local, disseminated, syringoma associated with Down syndrome, and inherited forms. Thus, several additional entities of histomorphologic characteristic features have been described in the recent literature, as discussed in the following.
Eruptive syringomas are commonly very rare.Citation3 In a review of the literature by Soler-Carrillo et al.Citation3 Sixty-four cases (48 females and 16 males) were evaluated between 1872 and 1999; the ages ranged between 1 and 77 years. In general, women are more frequently affected with eruptive syringomas than men; in the patient cohort reviewed by Soler-Carrillo et al.,Citation3 90% were females. In most cases, syringomas are asymptomatic small papules, preferentially located on the neck, trunk, axillae, shoulders, abdomen and pubic area. The initial clinical diagnosis differs in most cases, in contrast to the classical finding of eye lid syringomas, where histopathologic confirmation is not required for diagnosis. Hence, definitive diagnosis of an eruptive syringomas can only be verified by histologic examination ( and ).
A unique syndrome that includes eruptive syringomas is the Nicolau-Balus syndrome, consisting of eruptive syringomas of the disseminated micropapular type, milium cysts and atrophoderma vermiculata.Citation4 Seven reports on eruptive syringomas in patients with Down syndrome have been published.Citation3,Citation5–Citation8 Further associations included urticaria pigmentosa, simulating milia and familial cases of eruptive syringomas.Citation3
Although the definition of syringomas as benign neoplasms of the eccrine duct is widely accepted, the pathomechanisms and the genuine neoplastic character of eruptive syringomas are controversial. As reported in two cases by Requena et al.Citation9 eruptive syringoma occur as a consequence of chronic inflammatory processes of the skin involving adnexal structures. The authors propose the term, “syringomatous dermatitis,” to outline the assumed underlying inflammatory reaction because the patients revealed eczematous dermatitis with residual lesions that could be histopathologically-diagnosed as an eccrine syringoma.Citation9
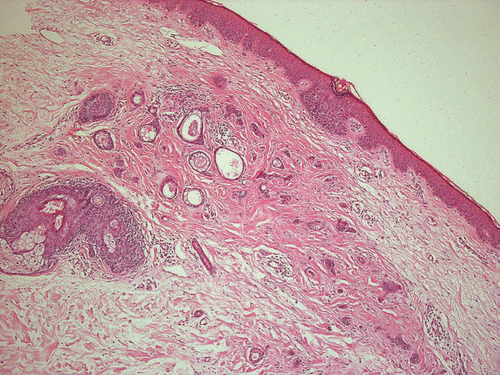
Supporting this theory of an inflammatory impulse leading to proliferation of parts of the eccrine duct epithelium, there are several reports of syringomas in scarring alopecia (),Citation10–Citation12 prurigo nodularis,Citation13 and following radiation therapy;Citation14 the inflammatory trigger has not been elucidated. Syringomatous changes have even been reported in melanocytic lesions, mostly as an incidental finding.Citation15–Citation17 As presented in our case series, syringomatous proliferations are found in scarring alopecia of the scalp due to lichen planopilaris and in association with a lentiginous melanocytic proliferation (patients #1 and #4).
In the recent literature, the influence of hormones on the development of syringomas is discussed. This has been confirmed by the observation that syringomas have a much higher incidence in females and occur frequently before and around puberty.Citation9 Further, pruritus is often associated with the menstrual cycle.Citation18 The influence of hormones on the syringomatous reaction process is further supported by observations of an aggravation or manifestation of vulvar syringomas during pregnancy. Citation19,Citation20 Vulvar syringomas most often present as multiple flesh-colored or brown papules on the labia majora of women in the third decade of life.Citation21 Vulvar syringomas are thought to be a common cause of a mild-to-severe pruritus of the vulva, and therefore should be included in the differential diagnosis of vulvar pruritus.Citation21 In case #2, syringomas were responsible for severe vulvar pruritus and also associated with chronic inflammatory bowel disease.
A very rare variant of syringomas is the so-called “milium-like” syringomas, first described in 1987 by Friedmann and Butler.Citation2 Because of the syringomas unique clinical and histologic pattern, they must be distinguished from simple miliae. Histopathologically, milium-like syringomas present as a keratinous cyst opening on the skin surface with numerous epithelial strands in the surrounding dermal tissue. Typically for syringomas, ductal structures with eosinophilic amorphous material can be found.Citation2 Only 31 cases of these syringoma variants have been reported in the literature. In one of these cases, an association between trisomy 21 and focal calcification was found.Citation22
Very rare cases of syringomas with an atypical appearance have been described as unilateral lesionsCitation23,Citation24 or simulating urticaria pigmentosaCitation25,Citation26 due to an increased content of mast cells. Furthermore, clear cell variants of syringomas have been described.Citation27 Clear cell syringomas represent rare histologic variants of syringomas with a bright and clear cytoplasm of the ductal epithelial cells due to increased content of intracellular glycogen. Clear-cell syringomas are clinically indistinguishable from ordinary syringomas and are thought to be a cutaneous marker for diabetes mellitus.Citation28–Citation30 None of the cases presented herein were milium-like or clear-cell-syringomas. In some cases, differential diagnosis between the so called plaque-type syringomas and microcystic adnexal carcinoma (MAC) of the skin can be very difficult, especially in superficial skin probes. MAC infiltrate much deeper into the dermis with regular involvement of subcutaneous structures and perineural and vascular-adventitial infiltration.Citation31 In order to avoid unnecessary and even disfiguring surgery, histopathologic diagnosis of these plaque-type syringomas is mandatory.Citation32
Most syringomas are harmless and benign, but of an aesthetically-disfiguring nature. Regression of lesions in adulthood has been observed, but is exceptional. Therapeutic efficacy is often frustrating, due to the slow growth and multicentric occurrence and the risk of scarring due to therapeutic procedures. Treatment modalities, such as topical or systemic retinoids, electrodissecation, laser ablation, cryosurgery, dermabrasion, electrocaustic therapy and chemical peelings have been attempted. However, all therapeutic options bear the risk of recurrence.Citation33–Citation35 Recently, there have been reports of successful treatments of syringoma with topical atropine.Citation36 Systematic approaches to all these therapeutic options are still lacking.
In summary, syringomas are benign neoplasms of the intraepidermal portion of the eccrine sweat duct and typically affect females in the facial regions, predominantly in the periorbital region. There have been a number of published clinical and histomorphologic variants (familial, milium-like, clear-cell, urticaria pigmentosa-like and vulvar), as well as many associated diseases and coincidental findings, such as Down syndrome or diabetes mellitus.
Materials and Methods
Nine patients with syringomas were referred to our department for evaluation between 2001 and 2008. All patients were females, with an age between 18 and 75 years (mean age, 50.5 years). The patient characteristics are summarized in .
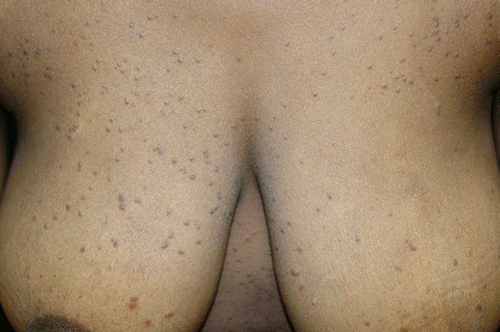
In the cases presented, syringomas appeared as single or disseminated papules, yellow to brown in color, on the face in six of nine cases. Five of these patients had slight-to-severe pruritus. An eruptive occurrence was diagnosed in only one case. Of note, this 18-year-old female patient (#3) presented with a 3-year history of multiple skin lesions (). The disease started with few papules on the anterior neck and subsequent spread to the entire chest, abdomen, back and upper extremities. The submammary region was also affected. Syringomas were not present on the face, the palmoplantar region, or the lower extremities. The lesions were nearly asymptomatic, except for mild and occasional pruritus. Previous treatments with topical corticosteroids and phototherapy yielded no persistent improvement.
No laboratory abnormalities were detected in any cases. No family history of skin diseases or syrinogomas was reported. The physical examinations revealed multiple, disseminated, brown, isomorphic lesions, 1–5 mm in diameter, without follicular distribution. The Darier sign was negative. The remainder of the physical examination was without pathologic findings.
In one case, a syringomatous proliferation was observed coincidentally in a skin biopsy from the capillitium, associated with severe lichen planopilaris (#1; ) in a further case (#4) as a dermal reaction pattern in an excision biopsy from a lentigo maligna. One patient had vulvar syringomas associated with severe vulvar pruritus (#2; ) and a coincidental occurrence of Crohn's disease. In two cases, an association with psoriasis vulgaris (#7) and polymorphic light eruption (#9) could be observed. None of the patients suffered from diabetes mellitus or trisomy 21.
Dermatopathologic findings.
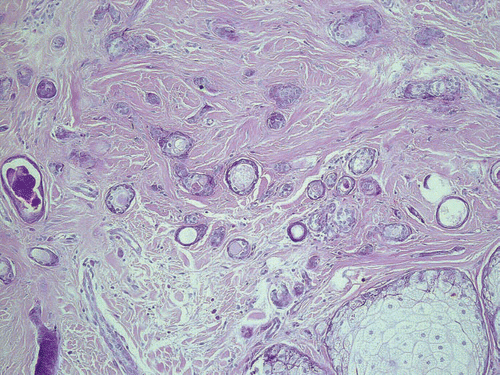
Skin biopsies of the patients presented were obtained either for diagnostic reasons or before laser ablation. All samples revealed a normal epidermis overlying a dermis filled with multiple cystic ducts and epithelial cords lined by one or two layers of polygonal epithelial cells with comma-like tails ( and ). Mild fibroplasia of the surrounding stroma was observed (). The ductal lumens were filled with amorphous, periodic acid-Schiff (PAS)-positive material (). Routinely, haematoxylin-eosin staining was performed; special stains included the PAS reaction and elastica-van-Giessonstain, as well as immunohistochemical stains with cytokeratin markers (MNF116 and AE1/AE3) epithelial cords lined by one or two layers of polygonal epithelial cells with comma-like tails ().
Conclusion
Syringomas occur frequently and predominantly affect women. These adnexal neoplasms are harmless and benign, but of an aesthetically-disfiguring nature since most often the face is affected. The histopathologic features are monomorphous with intriguing variations, e.g., clear-cell or milium-like variants. In the current literature, there is increasing evidence that the occurrence of eruptive syringomas or coincidental findings of these neoplasms in skin biopsies from scarring alopecia have to be discussed as an inflammatory dermal reaction pattern concerning the acrosyringium, the so-called syringomatous proliferation due to distinct inflammatory or hormonal stimuli.
As seen in the cases presented, most of the above-described variants of syringomas were found in the small patient cohort of our department. In agreement with recent reports, we also propose the term, reactive syringomatous proliferation, for most associations and coincidental findings of syringomas due to the absence of clinical features of a distinct clinicomorphologic entity.
Figures and Tables
Figure 1 Reactive syringomatous proliferation in conjunction with scarring alopecia due to lichen planopilaris (patient #1), H&E, ×200
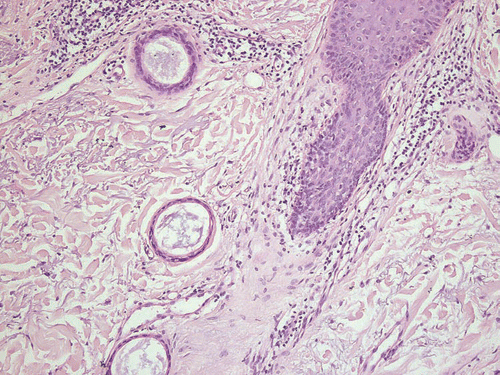
Figure 3 Representative lesion of an eruptively disseminating syringomatosis (patient #3), H&E, ×100.
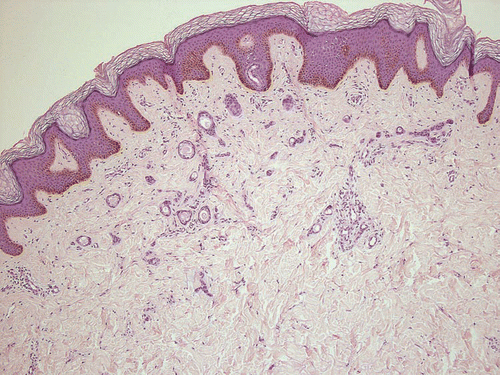
Figure 6 Epithelial cords lined by one or two layers of polygonal epithelial cells with comma-like tails, stained with cytokeratin marker AE1/AE3, ×100.
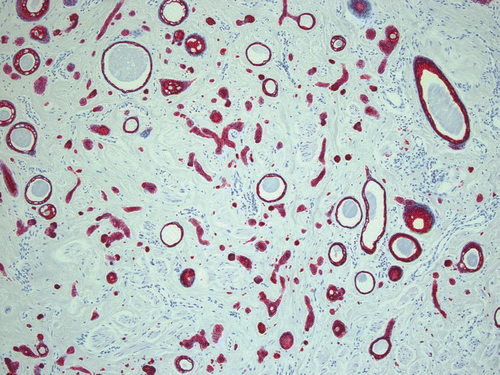
Table 1 Clinical data
Acknowledgements
We thank Anne Kerber for excellent histological and immunohistological stainings.
References
- Obaidat NA, Alsaad KO, Ghazarian D. Skin adnexal neoplasms—part 2: an approach to tumours of cutaneous sweat glands. J Clin Pathol 2007; 60:145 - 159
- Friedman SJ, Butler DF. Syringoma presenting as milia. J Am Acad Dermatol 1987; 16:310 - 314
- Soler-Carrillo J, Estrach T, Mascaro JM. Eruptive syringoma: 27 new cases and review of the literature. J Eur Acad Dermatol Venereol 2001; 15:242 - 246
- Dupre A, Bonafe JL, Christol B. Syringomas as a causative factor for cicatricial alopecia. Arch Dermatol 1981; 117:315
- Carey AB, Park HK, Burke WA. Multiple eruptive syringomas associated with Down's syndrome. J Am Acad Dermatol 1988; 19:759 - 760
- Draznin M. Hereditary syringomas: a case report. Dermatol Online J 2004; 10:19
- Jacobs S, Grussendorf-Conen EI. Disseminated eruptive syringomas in Down syndrome. Hautarzt 2004; 55:70 - 72
- Schepis C, et al. Eruptive syringomas with calcium deposits in a young woman with Down's syndrome. Dermatology 2001; 203:345 - 347
- Guitart J, Rosenbaum MM, Requena L. ‘Eruptive syringoma’: a misnomer for a reactive eccrine gland ductal proliferation?. J Cutan Pathol 2003; 30:202 - 205
- Barnhill RL, Goldberg B, Stenn KS. Proliferation of eccrine sweat ducts associated with alopecia areata. J Cutan Pathol 1988; 15:36 - 39
- Helm TN, et al. Occult syringoma associated with alopecia. Int J Dermatol 1992; 31:437 - 438
- Mehregan AH, Mehregan DA. Syringoma-like sweat duct proliferation in scalp alopecias. J Cutan Pathol 1990; 17:355 - 357
- Corredor F, Cohen PR, Tschen JA. Syringomatous changes of eccrine sweat ducts associated with prurigo nodularis. Am J Dermatopathol 1998; 20:296 - 301
- Yoshii N, et al. Syringoma-like eccrine sweat duct proliferation induced by radiation. J Dermatol 2006; 33:36 - 39
- Bhawan J, Malhotra R. Simultaneous occurrence of intradermal nevus and syringoma. Cutis 1983; 31:669 - 672
- Malhotra R, Bhawan J, Stadecker M. Association of syringoma and intradermal nevus. Int J Dermatol 1986; 25:397
- Nogita T, Kawashima M. Subclinical syringoma coexisting with nevocellular nevus on the vulva. J Dermatol 1993; 20:188 - 189
- Wallace ML, Smoller BR. Progesterone receptor positivity supports hormonal control of syringomas. J Cutan Pathol 1995; 22:442 - 445
- Turan C, et al. Vulvar syringoma exacerbated during pregnancy. Eur J Obstet Gynecol Reprod Biol 1996; 64:141 - 142
- Bal N, et al. Vulvar syringoma aggravated by pregnancy. Pathol Oncol Res 2003; 9:196 - 197
- Huang YH, et al. Vulvar syringoma: a clinicopathologic and immunohistologic study of 18 patients and results of treatment. J Am Acad Dermatol 2003; 48:735 - 739
- Seo SH, et al. A case of milium-like syringoma with focal calcification in Down syndrome. Br J Dermatol 2007; 157:612 - 614
- Wilms NA, Douglass MC. An unusual case of preponderantly right-sided syringomas. Arch Dermatol 1981; 117:308
- Yung CW, et al. Unilateral linear nevoidal syringoma. J Am Acad Dermatol 1981; 4:412 - 416
- Claudy AL. Adult-onset urticaria pigmentosa and eruptive syringomas. J Am Acad Dermatol 1988; 19:135
- Seifert HW. Association of disseminated syringoma and mast cells clinically resembling urticaria pigmentosa (author's transl). Z Hautkr 1981; 56:303 - 306
- Headington JT, Koski J, Murphy PJ. Clear cell glycogenosis in multiple syringomas. Description and enzyme histochemistry. Arch Dermatol 1972; 106:353 - 356
- Furue M, Hori Y, Nakabayashi Y. Clear-cell syringoma. Association with diabetes mellitus. Am J Dermatopathol 1984; 6:131 - 138
- Singh A, Mishra S. Clear cell syringoma—association with diabetes mellitus. Indian J Pathol Microbiol 2005; 48:356 - 357
- Krijnen RM, et al. Multiple clear-cell syringomas: a cutaneous marker for diabetes mellitus. Ned Tijdschr Geneeskd 1999; 143:1982 - 1983
- Henner MS, et al. Solitary syringoma. Report of five cases and clinicopathologic comparison with microcystic adnexal carcinoma of the skin. Am J Dermatopathol 1995; 17:465 - 470
- Suwattee P, et al. Plaque-type syringoma: two cases misdiagnosed as microcystic adnexal carcinoma. J Cutan Pathol 2007;
- Karam P, Benedetto AV. Intralesional electrodesiccation of syringomas. Dermatol Surg 1997; 23:921 - 924
- Mainitz M, Schmidt JB, Gebhart W. Response of multiple syringomas to isotretinoin. Acta Derm Venereol 1986; 66:51 - 55
- Metze D, Wigbels B, Hildebrand A. Familial syringoma: a rare clinical variant. Hautarzt 2001; 52:1045 - 1048
- Sanchez TS, et al. Eruptive pruritic syringomas: treatment with topical atropine. J Am Acad Dermatol 2001; 44:148 - 149