Abstract
The design criteria for matrices for encapsulation of cells for cell therapy include chemical, biological, engineering, marketing, regulatory, and financial constraints. What is required is a biocompatible material for culture of cells in three-dimensions (3-D) that offers ease of use, experimental flexibility to alter composition and compliance, and a composition that would permit a seamless transition from in vitro to in vivo use. The challenge is to replicate the complexity of the native extracellular matrix (ECM) environment with the minimum number of components necessary to allow cells to rebuild a given tissue. Our approach is to deconstruct the ECM to a few modular components that can be reassembled into biomimetic materials that meet these criteria. These semi-synthetic ECMs (sECMs) employ thiol-modified derivatives of hyaluronic acid (HA) that can form covalently crosslinked, biodegradable hydrogels. These sECMs are “living” biopolymers, meaning that they can be crosslinked in the presence of cells or tissues to enable cell therapy and tissue engineering. Moreover, the sECMs allow inclusion of the appropriate biological cues needed to simulate the complexity of the ECM of a given tissue. Taken together, the sECM technology offers a manufacturable, highly reproducible, flexible, FDA-approvable, and affordable vehicle for cell expansion and differentiation in 3-D.
Introduction
An ever-increasing number of hydrogels and fibrous scaffolds based on natural and synthetic polymers are being developed for the expansion of primary cells and progenitor cells in three dimensions (3-D).Citation1–Citation4 Among the naturally-derived biomaterials is an in situ-crosslinkable synthetic mimic of the extracellular matrix (sECM) that is based on chemically-modified hyaluronan (HA).Citation5 HA is a ubiquitous non-sulfated glycosaminoglycan with critical roles in morphogenesis,Citation6 and chemically-modified HA derivatives have generated considerable interest as biomaterials for tissue engineering.Citation7,Citation8 This versatile sECM offers user-controllable composition and compliance, to which a variety of growth factors or matricellular proteins may be added.Citation9,Citation10 The elastic moduli of hydrogels can be tailored to match the cell type to achieve optimal regeneration of a new tissue.Citation11,Citation12
Design criteria for ECM mimetics.
For a biomaterial to substitute for the ECM, it needs to recapitulate the principal functions of the natural ECM in orchestrating cell proliferation, migration, differentiation, angiogenesis and invasion.Citation13,Citation14 Recreating the interwoven set of biochemical and mechanical cues in the cellular microenvironmentCitation2,Citation15 and allowing full experimental versatility in fabrication methods for engineered microenvironmentsCitation1,Citation16,Citation17 requires consideration of a variety of design criteria. As summarized in , the performance criteria that should be met by a biomimetic ECM substitute should include: (1) experimentally variable composition, (2) experimentally variable compliance, (3) controllable biodegradability in vitro and in vivo, (4) multiple physical forms for in situ crosslinking during cell encapsulation and fabrication, (5) batch to batch consistency, (6) ease of use at physiological temperature and pH, (7) transparency for ease of visualization, (8) compatibility with high throughput screening (HTS) platforms, and (9) translational potential. It is important that the sECMs are modular in nature, allowing the user to add soluble factors, attachment peptides, matricellular proteins, crosslinkers, and macromomers in whatever combinationsCitation9 are dictated by the need to alter experimental parameters for testing hypotheses for a given cell type.
Most recently, we have underlined the importance of including the end-user needs as design criteria for materials to be used for cell therapyCitation5 and for development of drug evaluation tools.Citation18 These additional design criteria () are derived from the truism that “nothing from the lab ever reaches a patient unless it is commercialized.” Working backwards from this irrevocable reality, we recognized that commercialization—at least in the United States—requires significant financial commitment to developing the technology and to recruiting a team capable of reducing the technology to practice. Investor interest is highest for products for which a large market can be readily penetrated. Specifically, for biomaterials to be used in regenerative medicine, several key parameters must be been satisfied.
Deconstructing the ECM: A Chemist's Solution to a Bioengineering Problem
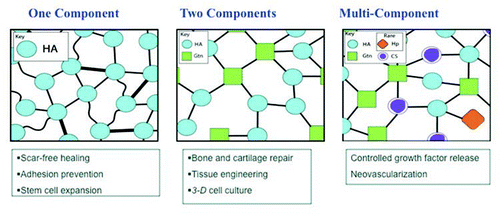
The native ECM is a heterogeneous network of soluble and fibrous proteins and glycosaminoglycans (GAGs), which is maintained by both covalent and non-covalent interactions (). In analogy to concrete, the ECM contains fibrillar collagen (rebar), proteoglycans (pebbles), hyaluronan (cement), and other minor components. To engineers and chemists, recapitulating the composition and structure of the ECM is a daunting yet irresistible challenge. To this end, we deconstructed the ECM into modular units containing one, two, or several chemically modified macromonomers that could be covalently crosslinked into a hydrogel network ().
The one-component solution.
In the last two decades, a host of new chemistries have been developed, resulting in a series of chemically-modified HA medical products that have addressed clinical needs with different levels of success. Citation8,Citation19 In addition, chemical modification techniques have allowed the modification of the surfaces of implantable devices and biomaterials with HA.Citation20
Chemically-modified HA derivatives were recently grouped into two categories.Citation21 When the modification resulted in a final form that could not form new chemical bonds in the presence of cells or tissues, and could only be manipulated by fabrication into different physical forms, these were classed as “monolithic” derivatives. In contrast, when new covalent bonds could be formed in the presence of cells or tissues, enabling a change in physical form during in vivo or in vitro biological use, these were classed as “living” derivatives. Thus, an in situ-crosslinkable material for adhesion prevention or cell encapsulation would be a “living” modification. This review focuses on living HA technologies and products and their uses for 3-D cell culture, wound repair, and tissue engineering.Citation22–Citation24
A synthetic extracellular matrix (sECM).
A novel approach to the preparation of a covalently-crosslinked sECM was recently described.Citation24,Citation25 The sECM material may be crosslinked in situ in the presence of cells in order to provide an injectable cell-seeded hydrogel for tissue repairCitation26 or without cells for the controlled, localized delivery of drugs or growth factors.Citation27–Citation29 Thiol-modified glycosaminoglycans and proteins are living macromonomers that can be crosslinked with biocompatible polyvalent electrophiles to form biocompatible, bioerodable hydrogels and porous sponges. This new technology offers a powerful set of new tools for culturing primary cells in three dimensions (3-D). Indeed, the applications of this technology include in vitro and in vivo growth of 3-D cellularized, vascularized tissues using films, sponges, and hydrogels.
The modular, clinically versatile and readily-manufactured sECM was made possible using hydrazide reagents that contain disulfide bonds,Citation30 affording thiol-modified proteins and GAGs can be prepared and crosslinked into hydrogels under cytocompatible physiological conditions.Citation31 The thiolated macromonomers may be subsequently crosslinked in air only to a hydrogel, which may be dried as a thin film or lyophilized to produce a porous sponge. Alternatively, crosslinking with difunctional electrophiles can be accomplished, in the presence or absence of cells, to give injectable and biocompatible hydrogels.Citation23,Citation26 Different physical properties and rates of biodegradation can be obtained by controlling several parameters for the biocompatible HA hydrogelsCitation32: (1) molecular weight of starting HA employed; (2) percentage DTPH modification; (3) concentration of HA-DTPH; (4) molecular weight of PEGDA; and (5) ratio of thiols to acrylates.
Photo-crosslinked HA: An alternative living polymer building block.
Methacrylate and methacryamide derivatized HA are living macromonomers that can be seeded with cells and photocrosslinked into hydrogels with either UV or visible light plus an initiator.Citation33–Citation36 The potential uses for these in situ photopolymerized hydrogels include prevention of adhesions and tissue regeneration; most recently, photopolymerized HA methacrylate was used for the expansion and differentiation of human embryonic stem cells.Citation37 For example, chondrocytes encapsulated in photocrosslinkable HA methacrylate retained the chondrocytic phenotype and synthesized cartilage matrix in vitro, and accelerated healing in an osteochondral defect model in vivo.Citation38 The poor cell attachment to these in situ photopolymerized HA hydrogels can be significantly enhanced by RGD modification.Citation33
Using the sECM platform in vivo.
The thiol modified HA component, first HA-DTPH but subsequently a carboxymethylated HA known as CMHA-S, was readily cross-linkable and yielded biocompatible hydrogels that did not support cell attachment and proliferation. Crosslinking was accomplished slowly via disulfide bond formation in air, or with better control of crosslinking time and hydrogel stiffness using cytocompatible bivalent cross-linkers such as polyethylene glycol diacrylate (PEGDA),Citation39 PEG divinyl sulfoneCitation40 or PEG bis(haloesters) and bis(haloamides).Citation41 This one-component gel has proven utility in scar-free wound healing, post-surgical adhesion prevention, and stem cell expansion. Specifically, the one-component gels were used in vivo for vocal fold repair,Citation42 to reduce post-operative intra-abdominal and pericardial adhesions,Citation43,Citation44 to eliminate postsurgical tendon adhesions,Citation45 to reduce airway stenosis,Citation46 to improve healing following endoscopic sinus surgery Citation47 and to promote healing following middle ear injury.Citation48
An example of the use of the PEGDA-crosslinked CMHA-S is illustrated in . Using a murine full-thickness wound repair model, we evaluate a one-component sECM composed of only the crosslinked carboxymethylated HA derivative. Bilateral injuries were formed on the flanks of each animal, allowing each animal to act as its own control. One site was left untreated, while a crosslinked hydrogel film of PEGDA-crosslinked CMHA-S was applied to the contralateral wound. A sheet of Tegaderm was laid across both wounds to prevent dessication and reduce microbial invasion, and a bandage was applied to retain the dressings in place. After six days, wound area was quantified, and then animals were sacrificed to perform histology. The treated wounds were over 45% re-epithelialized after six days, while the untreated wounds were only 12% healed. Moreover, wound filling, epidermal thickness, dermal thickness, and vascularization were also significantly enhanced in treated animals.
The two-component solution.
To obtain materials on which cells could readily attach and spread, we incorporated thiol-modified gelatin (Gtn-DTPH),Citation26,Citation49 RGD peptidesCitation50 and cysteine-containing domains of recombinant human fibronectinCitation51 into the hydrogel formulations. This produced the two-component gels shown in the middle panel of . Alternatively, simple addition of collagen provided a non-contracting 3-D hydrogel suitable for culture of human fibroblasts.Citation52 Such two-component hydrogels were tested with excellent results in three-dimensional (3-D) cell culture and tissue engineering applications.Citation23 Two quite different physical forms of the sECM were evaluated in vivo, as illustrated below.
First, we evaluated the use of an injectable, cell-free, two-component sECM hydrogel for repair of an osteochondral defect in a rabbit patellar groove defect ().Citation53 In untreated control defects, extensive scar tissue accumulated after twelve weeks. However, simply filling the defect with the sECM hydrogel resulted in regeneration of both new osteal and chondral tissues. Apparently, host-derived cells invaded the compliant hydrogel, remodeled it with matrix metalloproteinases, and secreted a new native ECM specific to either the bone or cartilage environment. Clearly, the simple HA-gelatin hydrogel is neither providing structurally or biochemically complete “instructions” for bone or for cartilage regeneration. Nonetheless, a biodegradable, osteo- and chondroconductive environment was provided by the hydrogel. Indeed, a more robust remodeling was accomplished by the addition of autologous MSCs to the hydrogel, suggesting that localized cell delivery and retention can add an important dimension to the regenerative process.Citation45 Still, the fundamental role of the hydrogel is to enhance the natural biological repair processes mediated by endogenous cells.
Second, we tested the hypothesis that the stiffness of the ECM substitute was important in the reparative context. For this study, we employed a rat femoral defect model, using either a hydrogel form of the cell-free two-component sECM Extracel™, or a lyophilized sponge of Extracel™.Citation54 We also tested whether the gels and sponges containing human demineralized bone matrix (DBM), a xenogeneic bone-derived growth factor mixture, would further enhance bone repair. As shown in , the untreated control injury showed little new bone formation after eight weeks. In contrast, we found that the stiffer sECM sponge was superior at all time points to the softer sECM hydrogel in generating new bone. In addition, the addition of DBM to either material gave a statistically significant improvement in healing.Citation54 Perhaps unexpectedly, stiffness was more important than growth factors; a sponge without DBM was more effective than a gel with DBM. In the end, the optimal treatment consisted of the Extracel” sponge plus DBM, which showed >95% closure of the defect and filling of the defect with healthy bone tissue after eight weeks.
Adding complexity one step at a time-sulfated GAG-containing sECMs.
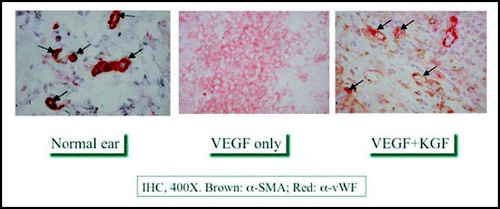
Engineered tissues ultimately require a blood supply, so that thiolated heparin (HP-DTPH) and chondroitin sulfate (CS-DTPH) macromonomers were developed to provide the biomimetic equivalent of heparan sulfate proteoglycans. We demonstrated that these hydrogels provided spatiotemporal control of growth factor release over a time course of weeks to months. Basic fibroblast growth factor (bFGF) was released from an HA-based sECM in vitro with a half-life of one month;Citation27 when released from a CS-based sECM in vivo, bFGF showed dose-dependent acceleration of re-epithelialization and improved formation of vascularized dermal tissue in the repair of large acute wounds in diabetic mice.Citation55 Dual delivery of vascular endothelial growth factor (VEGF) with bFGF,Citation56 with angiopoetin-1 (Ang-1), and with keratinocyte growth factor (KGF)Citation57 led to enhanced neovascularization in vivo. For example, shows mature vasculogenesis in a mouse ear pinnae model when both KGF and VEGF from a heparan proteoglycan-mimetic sECM hydrogel, similar to what is seen in native vasculature. In contrast, VEGF-only hydrogels generated only immature leaky angiogenic sprouts.
The Translational Challenge
The translational challenge in replicating the native ECM environment requires co-optimization of biological, structural, and practical design parameters. Too often, biologists and engineers have minimized the importance of the financial, production, and regulatory constraints when developing a biomaterial for reparative or regenerative use in the clinic. This has resulted in many false starts. In contrast, a marketing approach driven by the needs of the end-users places these crucial and limiting pragmatic factors as part of the initial design criteria. In this way, we have advocated using the minimum number of components necessary to allow cells to rebuild a given tissue.
The message of this rearrangement of the discovery process for new biomaterials is summarized in . The physiological and structural aspects of rebuilding tissues from cells—from whatever source—are immensely complicated. We must embrace this complexity, but it cannot dominate the design of a clinical material. Moreover, one formulation cannot fulfill every need, from stem cell expansion and delivery for neural tissues to creating a structurally robust material for large-scale osteochondral defect repair. We should engineer flexibility, so that a single suite of approvable materials can be exploited for multiple physician uses in a variety of clinical niches. Finally, for regenerative medicine to succeed as a reality, we must deliver simplicity. The penetration of new technology into any market is not determined only by improvement in patient outcome —the true limiting factors are cost, familiarity, and ease of use. We believe that the living sECM technology addresses a balance between the physiological and the practical requirement, and that its uses for clinical, veterinary, and laboratory research needs will continue to expand in the decades to come.
Abbreviations
| abbreviations | = |
Figures and Tables
Figure 2 Additional design criteria for a synthetic extracellular matrix optimized for reparative medicine.

Figure 3 The native extracellular matrix is a heterogeneous networked complex of proteins and glycosaminoglycans that is maintained by multiple covalent and non-covalent interactions.
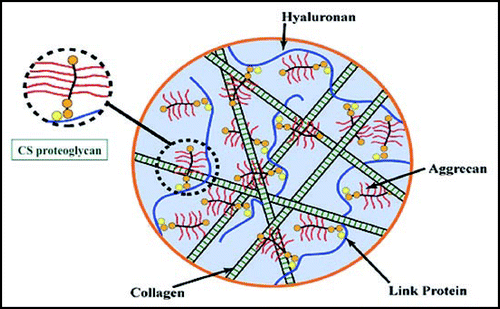
Figure 5 (A) Wound repair model and evaluation of a one-component sECM composed of only a crosslinked HA derivative. (B) Improved histological outcomes include wound filling, production of a vascularized dermis, and regeneration of a new epithelium complete with stratum corneum.
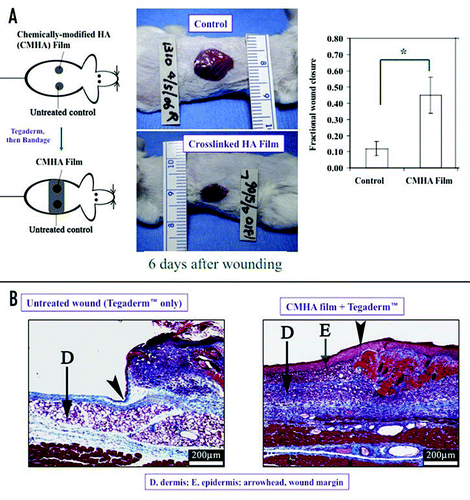
Figure 6 Cartilage repair in an osteochondral defect with a cell-free two-component sECM hydrogel. Left, rabbit patellar groove defect with no treatment results in extensive scar tissue after three months. Right, filling the defect with Extracel™ hydrogel results in regeneration of both new osteal and chondral tissues by invasion of the sECM by host-derived cells.
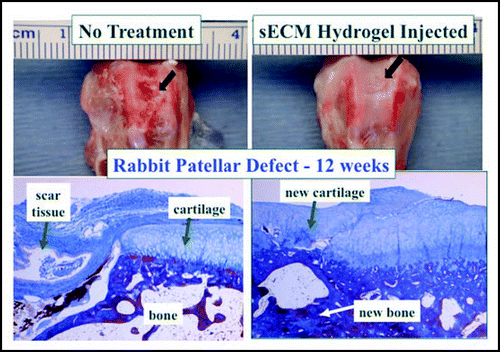
Figure 7 Bone repair in a femoral defect model with a cell-free two-component Extracel™ lyophilized sponge containing human demineralized bone matrix (DBM). Above, untreated control injury results in little defect closure (autoradiogram) and extensive scar tissue (histology). Below, optimal treatment with Extracel™ sponge plus DBM shows complete closure of the defect and filling of the defect with healthy bone tissue.

Acknowledgements
We thank the Utah Centers of Excellence Program for the primary financial support for this work. Additional resources from NIH Grant R01 DC4336 from the National Institute of Deafness and other Communicative Disorders, and from the NSF Frontiers in Innovative Biological Research Grant EF0526854 contributed to the conceptualization and implementation of the sECMs described herein.
References
- Cushing MC, Anseth KS. Materials science. Hydrogel cell cultures. Science 2007; 316:1133 - 1134
- Drury JL, Mooney DJ. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 2003; 24:4337 - 4351
- Dawson E, Mapili G, Erickson K, Taqvi S, Roy K. Biomaterials for stem cell differentiation. Adv Drug Deliv Rev 2008; 60:215 - 228
- Hoffman AS. Hydrogels for biomedical application. Adv Drug Del Rev 2002; 43:3 - 12
- Prestwich GD. Simplifying the extracellular matrix for 3-D cell culture and tissue engineering: A pragmatic approach. J Cell Biochem 2007; 101:1370 - 1383
- Toole BP. Hyaluronan in morphogenesis. Semin Cell Dev Biol 2001; 12:79 - 87
- Allison D, Grande-Allen K. Hyaluronan: a powerful tissue engineering tool. Biomaterials 2006; 12:2131 - 2140
- Shu XZ, Prestwich GD. Therapeutic biomaterials from chemically modified hyaluronan. Chemistry and Biology of Hyaluronan 2004; Amsterdam Elsevier Press 475 - 504
- Serban MA. Making modular extracellular matrices: solutions for the puzzle. Methods 2008; In press
- Serban MA, Liu Y, Prestwich GD. Effects of synthetic extracellular matrices on primary human fibroblast behavior. Acta Biomaterialia 2008; 4:67 - 75
- Discher D, Janmey P, Wang Y-l. Tissue cells feel and respond to the stiffness of their substrate. Science 2005; 310:1139 - 1143
- Ghosh K, Pan Z, Guan E, Ge S, Liu Y, Nakamura T, Ren X-D, Rafailovich MH, Clark RAF. Cell adaptation to a physiologically relevant ECM mimic with different viscoelastic properties. Biomaterials 2007; 28:671 - 679
- Geiger B, Bershadsky A, Pankov R, Yamada KM. Transmembrane crosstalk between the extracellular matrix-cytoskeleton crosstalk. Nat Rev Mol Cell Biol 2001; 2:793 - 805
- Lutolf MP, Hubbell JA. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat Biotechnol 2005; 23:47 - 55
- Griffith L, Swartz M. Capturing complex 3D tissue physiology in vitro. Nat Rev Molec Cell Biol 2006; 7:211 - 224
- Yeh J, Ling Y, Karp JM, Gantz J, Chandawarkar A, Eng G, Blumling J III, Langer R, Khademhosseini A. Micromolding of shape-controlled, harvestable cell-laden hydrogels. Biomaterials 2006; 27:5391 - 5398
- Bhadriraju K, Chen CS. Engineering cellular microenvironments to improve cell-based drug testing. Drug Discov Today 2002; 7:612 - 620
- Prestwich GD. Evaluating drug toxicity and efficacy in three dimensions: using synthetic extracellular matrices in drug discovery. Acc Chem Res 2008; 41:139 - 148
- Kuo JW. Practical Aspects of Hyaluronan-Based Medical Products 2006; CRC Taylor & Francis
- Morra M. Engineering of biomaterials surfaces by hyaluronan. Biomacromolecules 2005; 6:1205 - 1223
- Prestwich GD, Kuo J-w. Chemically-modified HA for therapy and regenerative medicine. Curr Pharm Biotech 2008; In press
- Prestwich GD. Biomaterials from chemically-modified hyaluronan. Glycoforum 2001; http://glycoforum.gr.jp/science/hyaluronan/HA18/HA18E.html.
- Shu XZ, Liu Y, Palumbo F, Luo Y, Prestwich GD. In situ crosslinkable glycosaminoglycan hydrogels for tissue engineering. Biomaterials 2004; 25:1139 - 1148
- Prestwich GD, Shu XZ, Liu Y, Cai S, Walsh JF, Hughes CW, Ahmad S, Kirker KR, Yu B, Orlandi RR, Park AH, Thibeault SL, Duflo S, Smith ME. Injectable synthetic extracellular matrices for tissue engineering and repair. Adv Exp Med Biol 2006; 585:125 - 133
- Shu XZ, Liu Y, Prestwich GD. Balazs E A, Hascall V C. Injectable, in situ-crosslinkable biomimetic hydrogels for tissue engineering. Hyaluronan: Structure, Metabolism, Biological Activities, Therapeutic Applications 2005; Edgewater, New Jersey Matrix Biology Institute 415 - 419
- Shu XZ, Ahmad S, Liu Y, Prestwich GD. Synthesis and evaluation of injectable, in situ crosslinkable synthetic extracellular matrices sECMs. for tissue engineering. J Biomed Mater Res 2006; 79A:902 - 912
- Cai S, Liu Y, Shu XZ, Prestwich GD. Injectable glycosaminoglycan hydrogels for controlled release of basic fibroblast growth factor. Biomaterials 2005; 26:6054 - 6067
- Peattie RA, Rieke ER, Hewett EM, Fisher RJ, Shu XZ, Prestwich GD. Dual growth factor-induced angiogenesis in vivo using hyaluronan hydrogel implants. Biomaterials 2006; 27:1868 - 1875
- Liu Y, Li H, Shu XZ, Gray SD, Prestwich GD. Crosslinked hyaluronan hydrogels containing mitomycin C reduce post-operative abdominal adhesions. Fertil & Steril 2005; 83:1275 - 1283
- Pouyani T, Prestwich GD. US Patent 5,652,347 1997; pp 5,616,568
- Shu XZ, Liu Y, Luo Y, Robert MC, Prestwich GD. Disulfide crosslinked hyaluronan hydrogel. Biomacromolecules 2002; 3:1304 - 1311
- Liu Y, Shu XZ, Prestwich GD. Biocompatibility and stability of disulfide-crosslinked hyaluronan films. Biomaterials 2005; 26:4737 - 4746
- Park YD, Tirelli N, Hubbell JA. Photopolymerized hyaluronic acid-based hydrogels and interpenetrating networks. Biomaterials 2003; 24:893 - 900
- Inukai M, Jin Y, Yomota C, Yonese M. Preparation and characterization of hyaluronate-hydroxyethyl acrylate blend hydrogel for controlled release device. Chem Pharm Bull 2000; 48:850 - 854
- Leach JB, Bivens KA, Patrick CW Jr. Photocrosslinked hyaluronic acid hydrogels: natural, biodegradable tissue engineering scaffolds. Biotechnol Bioeng 2003; 82:578 - 589
- Smeds KA, Pfister-Serres A, Hatchell DL, Grinstaff MW. Synthesis of novel polysaccharide hydrogel. J M S - Pure Appl Chem A 1999; 36:981 - 989
- Gerecht S, Burdick J, Ferreira L, Townsend S, Langer R, Vunjak-Novakovic G. Hyaluronic acid hydrogel for controlled self-renewal and differentiation of human embryonic stem cells. Proc Natl Acad Sci USA 2007; 104:11298 - 11303
- Nettles DL, Vail TP, Morgan MT, Grinstaff MW, Setton LA. Photocrosslinkable hyaluronan as a scaffold for articular cartilage repair. Ann Biomed Eng 2004; 32:391 - 397
- Shu XZ, Liu Y, Palumbo FS, Luo Y, Prestwich GD. In situ crosslinkable hyaluronan hydrogels for tissue engineering. Biomaterials 2004; 25:1339 - 1348
- Ghosh K, Shu XZ, Mou R, Lombardi J, Prestwich GD, Rafailovich MH, Clark RAF. Rheological characterization of in situ cross-linkable hyaluronan hydrogels. Biomacromolecules 2005; 6:2857 - 2865
- Vanderhooft JL, Mann BK, Prestwich GD. Synthesis and characterization of novel thiolreactive polyethylene glycol. crosslinkers for biomaterials. Biomacromolecules 2007; 8:2883 - 2889
- Hansen JK, Thibeault SL, Walsh JF, Shu XZ, Prestwich GD. In vivo engineering of the vocal fold ECM with injectable HA hydrogels: Early effects on tissue repair and biomechanics in a rabbit model. Ann Otol Rhinol Laryngol 2005; 114:662 - 670
- Liu Y, Shu XZ, Prestwich GD. Reduced postoperative intra-abdominal adhesions using Carbylan-SX, a semisynthetic glycosaminoglycan hydrogel. Fertil Steril 2007; 87:940 - 948
- Connors RC, Muir JJ, Liu Y, Reiss GR, Kouretas PC, Whitten MG, Sorenson TK, Prestwich GD, Bull DA. Postoperative pericardial adhesion prevention using Carbylan-SX in a rabbit model. J Surg Res 2007; 140:237 - 242
- Liu Y, Skardal A, Shu XZ, Prestwich GD. Prevention of peritendinous adhesions following flexor tendon injury with Carbylan™-SX, a semisynthetic glycosaminoglycan hydrogel. J Orthopedic Res 2008; 562 - 569
- Sondrup C, Liu Y, Shu XZ, Prestwich GD, Smith ME. Bioactive stents in the prevention of airway stenosis. Otolaryngol Head & Neck Surg 2006; 135:28 - 35
- Orlandi RR, Shu XZ, McGill L, Petersen E, Prestwich GD. Structural variations in a single hyaluronan derivative significantly alter wound-healing effects in the rabbit maxillary sinus. Laryngoscope 2007; 117:1288 - 1295
- Park AH, Jackson A, Hunter L, McGill L, Simonsen SE, Alder SC, Shu XZ, Prestwich GD. Crosslinked hydrogels for middle ear packing. Otol Neurotol 2006;
- Shu XZ, Liu Y, Palumbo F, Prestwich GD. Disulfide-crosslinked hyaluronan-gelatin hydrogel films: a covalent mimic of the extracellular matrix for in vitro cell growth. Biomaterials 2003; 24:3825 - 3834
- Shu XZ, Ghosh K, Liu Y, Palumbo FS, Luo Y, Clark RA, Prestwich GD. Attachment and spreading of fibroblasts on an RGD peptide-modified injectable hyaluronan hydrogel. J Biomed Mat Res 2004; 68A:365 - 375
- Ghosh K, Ren X-D, Shu XZ, Prestwich GD, Clark RAF. Fibronectin functional domains coupled to hyaluronan stimulate adult human dermal fibroblast responses critical for wound healing. Tissue Eng 2006; 12:601 - 613
- Mehra T, Ghosh K, Shu XZ, Prestwich GD, Clark RAF. Molecular stenting with a cross-linked hyaluronan inhibits collagen gel contraction. J Invest Dermatol 2006; 126:2202 - 2209
- Liu Y, Shu XZ, Prestwich GD. Osteochondral defect repair with autologous bone marrow-derived mesenchymal stem cells in an injectable, in situ, cross-linked synthetic extracellular matrix. Tissue Eng 2006; 12:3405 - 3416
- Liu Y, Ahmad S, Shu XZ, Sanders RK, Kopesec SA, Prestwich GD. Accelerated repair of cortical bone defects using a synthetic extracellular matrix to deliver human demineralized bone matrix. J Orthoped Res 2006; 24:1454 - 1462
- Liu Y, Cai S, Shu XZ, Shelby J, Prestwich GD. Release of basic fibroblast growth factor from a crosslinked glycosaminoglycan hydrogel promotes wound healing. Wound Rep Reg 2007; 15:245 - 251
- Pike DB, Cai S, Pomraning KR, Firpo MA, Fisher RJ, Shu XZ, Prestwich GD, Peattie RA. Heparin-regulated release of growth factors in vitro and angiogenic response in vivo to implanted hyaluronan hydrogels containing VEGF and bFGF. Biomaterials 2006; 27:5242 - 5251
- Hosack L, Firpo MA, Scott JA, Prestwich GD, Peattie RA. Microvascular maturity elicited in tissue treated with cytokine-loaded hyaluronan-based hydrogels. Biomaterials 2008; 2336 - 2347