Figures & data
Scheme 1 Synthesis of imidazolidin-2-ones 10-17 and 33-37 and tetrahydropyrimidin-2(1H)-ones 42-45. Reaction reagents and conditions: (i) AcOH, reflux; (ii) Pd/C 5%, H2, THF, 50°C; (iii) 2-chloroethyl isocyanate, CHCl3, reflux; (iv) 2-chloroethyl isocyanate (8 éq), microwaves, 82°C, 20 W; (v) 3-chloropropyl isocyanate, CHCl3, reflux; (vi) Cs2CO3, CH3CN, reflux; (vii) Na2CO3, CH3CN, reflux.
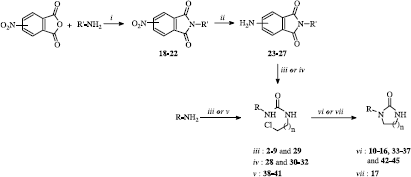
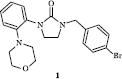
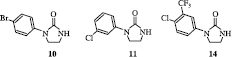
Table I. Inhibition of the mouse splenocyte ConA-induced proliferation by N-substituted imidazolidin-2-ones 10-17 and 33-37 and tetrahydropyrimidin-2(1H)-ones 42-45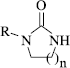 .
.