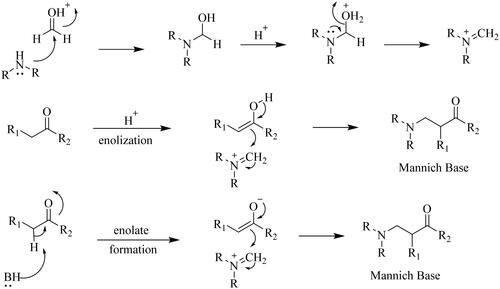
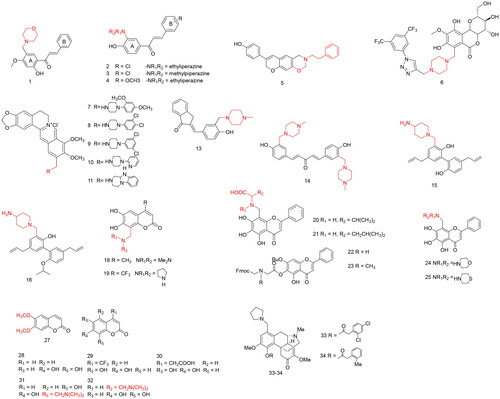
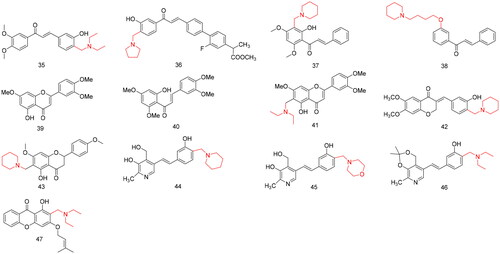
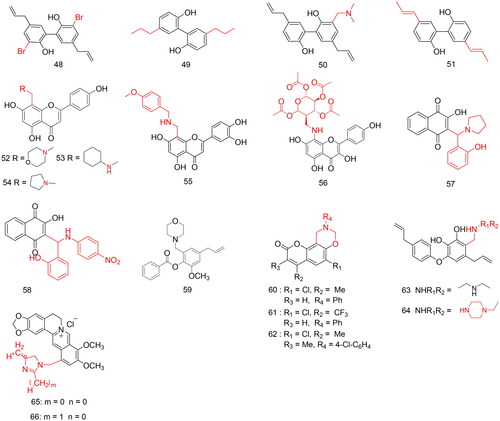
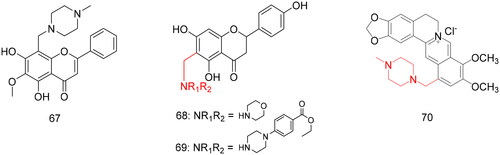
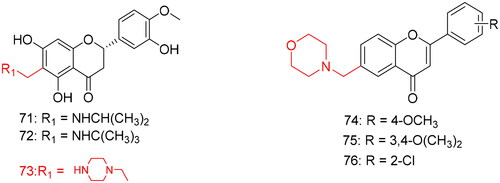
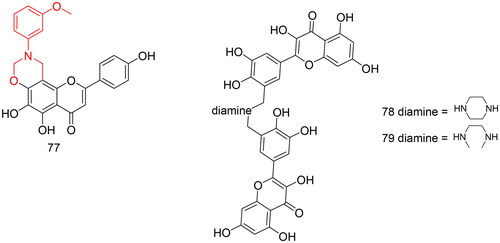
Figures & data
Table 1. Applications in modification of natural products for antitumor activity.
Table 2. Application in modification of natural products for anti-Alzheimer’s disease.
Table 3. Application in modification of natural products for antimicrobial activity.
Table 4. Application in modification of natural products for antidiabetic activity.