Figures & data
Table 1. The main advantages and disadvantages of various preparation methods of CQDs.
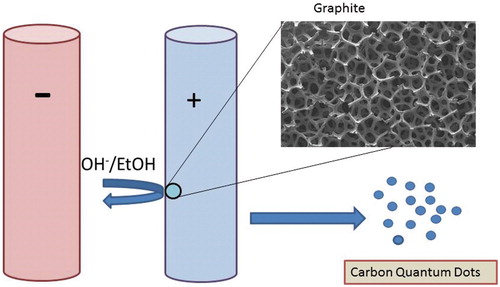
Figure 3. HR-TEM images of CQDs obtained from electrochemical methods. (Reprinted from Ref. [Citation31] Copyright 2016, with permission from Elsevier).
![Figure 3. HR-TEM images of CQDs obtained from electrochemical methods. (Reprinted from Ref. [Citation31] Copyright 2016, with permission from Elsevier).](/cms/asset/15345890-0a17-4495-a630-ed60f296b20f/ianb_a_1377725_f0003_b.jpg)
Figure 4. Processing diagram for the synthesis of multicolour photoluminescent carbon dots. (Reprinted from Ref. [Citation22] with permission from Copyright 2009 John Wiley and Sons).
![Figure 4. Processing diagram for the synthesis of multicolour photoluminescent carbon dots. (Reprinted from Ref. [Citation22] with permission from Copyright 2009 John Wiley and Sons).](/cms/asset/c07d5991-058c-4662-b24c-102aa9b9d869/ianb_a_1377725_f0004_c.jpg)
Figure 5. (a) TEM image of 5 nm-CQDS obtained from glucose; (b, c) photographs of CQDs dispersals in water with visible light and UV (365 nm, centre) illumination, respectively; (d–g) fluorescent microscope images of CQDs under diverse excitation: d, e, f and g for 360, 390, 470 and 540 nm, respectively. (Reprinted from Ref. [Citation11] Copyright 2011, with permission from Elsevier).
![Figure 5. (a) TEM image of 5 nm-CQDS obtained from glucose; (b, c) photographs of CQDs dispersals in water with visible light and UV (365 nm, centre) illumination, respectively; (d–g) fluorescent microscope images of CQDs under diverse excitation: d, e, f and g for 360, 390, 470 and 540 nm, respectively. (Reprinted from Ref. [Citation11] Copyright 2011, with permission from Elsevier).](/cms/asset/375bbd0d-ff2d-4251-9526-be01f3efbdbc/ianb_a_1377725_f0005_c.jpg)
Figure 6. (a) Schematic depiction of producing of CQDs by MAH technique. (Reprinted with permission from Ref. [Citation41] Copyright (2012) American Chemical Society); (b) processing diagram of the one-step preparation of luminescent CQDs in PEG200N solvent (Reprinted from Ref. [Citation46] with permission from The Royal Society of Chemistry); (c) schematic representation of laser ablation experimental setup. (Reprinted from Ref. [Citation45] with permission from The Royal Society of Chemistry).
![Figure 6. (a) Schematic depiction of producing of CQDs by MAH technique. (Reprinted with permission from Ref. [Citation41] Copyright (2012) American Chemical Society); (b) processing diagram of the one-step preparation of luminescent CQDs in PEG200N solvent (Reprinted from Ref. [Citation46] with permission from The Royal Society of Chemistry); (c) schematic representation of laser ablation experimental setup. (Reprinted from Ref. [Citation45] with permission from The Royal Society of Chemistry).](/cms/asset/1c8530f5-2eb4-4b85-9ad2-dbf7cfbac403/ianb_a_1377725_f0006_c.jpg)
Figure 7. Schematic illustration of limited reaction in nanoreactors for synthesis of CQDs. (Reprinted from Ref. [Citation37] with permission from The Royal Society of Chemistry).
![Figure 7. Schematic illustration of limited reaction in nanoreactors for synthesis of CQDs. (Reprinted from Ref. [Citation37] with permission from The Royal Society of Chemistry).](/cms/asset/422efdfc-e8d2-496b-a8eb-9030cf24e0bc/ianb_a_1377725_f0007_c.jpg)
Figure 8. (a) Diagram of the synthesis of CQDs unzipped from PAN@PMMA core–shell nanoparticles. Numerous n-CQDs were unzipped from one polymeric nanoparticle and exhibited diverse PL behaviours at different pyrolysis temperatures. (b, c) TEM images of the PAN@PMMA core–shell nanoparticles. Scale bars are 200 nm (b) and 50 nm (c), respectively. (d) DLS curve of the PAN@PMMA core–shell nanoparticles; the inset exhibits a photograph of a PAN@PMMA microemulsion. (Reprinted from Ref. [Citation49] with permission from The Royal Society of Chemistry).
![Figure 8. (a) Diagram of the synthesis of CQDs unzipped from PAN@PMMA core–shell nanoparticles. Numerous n-CQDs were unzipped from one polymeric nanoparticle and exhibited diverse PL behaviours at different pyrolysis temperatures. (b, c) TEM images of the PAN@PMMA core–shell nanoparticles. Scale bars are 200 nm (b) and 50 nm (c), respectively. (d) DLS curve of the PAN@PMMA core–shell nanoparticles; the inset exhibits a photograph of a PAN@PMMA microemulsion. (Reprinted from Ref. [Citation49] with permission from The Royal Society of Chemistry).](/cms/asset/5347682a-0e1f-48ad-b50a-dce4b50702ca/ianb_a_1377725_f0008_c.jpg)
Figure 9. Schematic illustration of producing soluble CQDs with adjustable sizes from single-chain polymeric nanoparticles. (Reprinted from Ref. [Citation51] with permission from The Royal Society of Chemistry).
![Figure 9. Schematic illustration of producing soluble CQDs with adjustable sizes from single-chain polymeric nanoparticles. (Reprinted from Ref. [Citation51] with permission from The Royal Society of Chemistry).](/cms/asset/60831eae-ad43-4837-922e-a7d7cfc9a2fd/ianb_a_1377725_f0009_c.jpg)
Figure 10. (a) Four typical sizes of CQDs illuminated by white (left; usual lamp; from left to right the colors are pale green, pale yellow, yellow and red, respectively) and UV light (right; 365 nm; from left to right the colors are blue, green, yellow and red, respectively); (b) four typical sizes of CQDs: from left to right the colors are red, black, green and blue lines are related to the PL spectra for blue-, green-, yellow- and red-emission CQDs, respectively; (c) correlation between PL properties and CQDs size; (d) the dependence of HOMO–LUMO gap on the size of the CQDs. (Reprinted from Ref. [Citation30] with permission from Copyright 2010 John Wiley and Sons).
![Figure 10. (a) Four typical sizes of CQDs illuminated by white (left; usual lamp; from left to right the colors are pale green, pale yellow, yellow and red, respectively) and UV light (right; 365 nm; from left to right the colors are blue, green, yellow and red, respectively); (b) four typical sizes of CQDs: from left to right the colors are red, black, green and blue lines are related to the PL spectra for blue-, green-, yellow- and red-emission CQDs, respectively; (c) correlation between PL properties and CQDs size; (d) the dependence of HOMO–LUMO gap on the size of the CQDs. (Reprinted from Ref. [Citation30] with permission from Copyright 2010 John Wiley and Sons).](/cms/asset/0c9d7111-8e8f-4acc-82a8-1486abba23fe/ianb_a_1377725_f0010_c.jpg)
Figure 11. (a) Schematic representation of the light-excited fluorescence modulation of spiropyran-modified CQDs. (Reprinted from Ref. [Citation60] with permission from The Royal Society of Chemistry.); (b) schematic diagram for the preparation of photoluminescent CDs, flexible CD film and CDs/silica particles. (Reprinted from Ref. [Citation52] with permission from Copyright 2011 John Wiley and Sons).
![Figure 11. (a) Schematic representation of the light-excited fluorescence modulation of spiropyran-modified CQDs. (Reprinted from Ref. [Citation60] with permission from The Royal Society of Chemistry.); (b) schematic diagram for the preparation of photoluminescent CDs, flexible CD film and CDs/silica particles. (Reprinted from Ref. [Citation52] with permission from Copyright 2011 John Wiley and Sons).](/cms/asset/ed5d3ce9-9884-4eb2-8a6e-45bca6cf309c/ianb_a_1377725_f0011_c.jpg)
Figure 12. (a) Diagram illustration of UV/Vis absorption and PL emission spectra (recorded for increasingly longer excitation wavelengths from 320 to 520 nm in 20 nm increments) of CQDs surface-passivated with PEG1500N in water. In the inset, the emission spectral intensities are normalized. (b) Optical photograph attained under excitation at 365 nm. (Reprinted with permission from Ref.[Citation22] Copyright 2009 John Wiley and Sons).
![Figure 12. (a) Diagram illustration of UV/Vis absorption and PL emission spectra (recorded for increasingly longer excitation wavelengths from 320 to 520 nm in 20 nm increments) of CQDs surface-passivated with PEG1500N in water. In the inset, the emission spectral intensities are normalized. (b) Optical photograph attained under excitation at 365 nm. (Reprinted with permission from Ref.[Citation22] Copyright 2009 John Wiley and Sons).](/cms/asset/c1102c40-5fcd-4726-9272-5a51bafd56b7/ianb_a_1377725_f0012_c.jpg)
Figure 13. PL spectra of CQDs exited by N-IR (a) CQDs obtained from glucose/NaOH; (b) CQDs obtained from glucose/HCl. (Reprinted from Ref. [Citation11] Copyright 2011, with permission from Elsevier).
![Figure 13. PL spectra of CQDs exited by N-IR (a) CQDs obtained from glucose/NaOH; (b) CQDs obtained from glucose/HCl. (Reprinted from Ref. [Citation11] Copyright 2011, with permission from Elsevier).](/cms/asset/b86e102f-6447-4415-b060-0bfd85926947/ianb_a_1377725_f0013_c.jpg)
Figure 14. Luminescence images (all scale bars 20 nm) of the CQDs with (a) argon ion laser excitation at 458 nm and (b) femtosecond pulsed laser excitation at 800 nm; (c) is an overlap of (a) and (b). (Reprinted with permission from Ref. [Citation14] Copyright (2007) American Chemical Society).
![Figure 14. Luminescence images (all scale bars 20 nm) of the CQDs with (a) argon ion laser excitation at 458 nm and (b) femtosecond pulsed laser excitation at 800 nm; (c) is an overlap of (a) and (b). (Reprinted with permission from Ref. [Citation14] Copyright (2007) American Chemical Society).](/cms/asset/109b22fc-6f4e-4adb-8dbd-de84d0f8751d/ianb_a_1377725_f0014_c.jpg)
Figure 15. Intravenous injection of C-Dots: (a) bright field, (b) as-detected fluorescence (Bl, bladder; Ur, urine) and (c) colour-coded images. The same order is used for the images of the dissected kidneys (a′–c′) and liver (a″–c″). (Reprinted with permission from Ref. [Citation42] Copyright (2009) American Chemical Society).
![Figure 15. Intravenous injection of C-Dots: (a) bright field, (b) as-detected fluorescence (Bl, bladder; Ur, urine) and (c) colour-coded images. The same order is used for the images of the dissected kidneys (a′–c′) and liver (a″–c″). (Reprinted with permission from Ref. [Citation42] Copyright (2009) American Chemical Society).](/cms/asset/5cc7bf26-c47e-48ce-8dab-2d8a1e843e85/ianb_a_1377725_f0015_c.jpg)
Figure 16. (a, b) SEM image of photocatalysts for SiO2/CQDs and TiO2 CQDs; insets exhibit the corresponding HRTEM images; (c) correlation between MB concentration and reaction time for diverse catalysts: SiO2/CQDs, TiO2/CQDs, SiO2 NPs, TiO2 NPs and CQDs. (Reprinted with permission from Ref. [Citation30] Copyright 2010 John Wiley and Sons).
![Figure 16. (a, b) SEM image of photocatalysts for SiO2/CQDs and TiO2 CQDs; insets exhibit the corresponding HRTEM images; (c) correlation between MB concentration and reaction time for diverse catalysts: SiO2/CQDs, TiO2/CQDs, SiO2 NPs, TiO2 NPs and CQDs. (Reprinted with permission from Ref. [Citation30] Copyright 2010 John Wiley and Sons).](/cms/asset/1ba6d6de-34bd-4d99-be5d-cfd709814635/ianb_a_1377725_f0016_b.jpg)
Figure 17. Illustration of catalytic mechanism for TiO2/CQDs under visible daylight lamp. (Reprinted with permission from Ref. [Citation30] Copyright 2010 John Wiley and Sons).
![Figure 17. Illustration of catalytic mechanism for TiO2/CQDs under visible daylight lamp. (Reprinted with permission from Ref. [Citation30] Copyright 2010 John Wiley and Sons).](/cms/asset/15856214-bd21-4a3c-8be4-056822100036/ianb_a_1377725_f0017_b.jpg)
Figure 18. Graphical depiction of FRET-based ratiometric sensing of mitochondrial H2O2 in living cell by the nano-probe. (Reprinted with permission from Ref. [Citation83] Copyright 2013 John Wiley and Sons).
![Figure 18. Graphical depiction of FRET-based ratiometric sensing of mitochondrial H2O2 in living cell by the nano-probe. (Reprinted with permission from Ref. [Citation83] Copyright 2013 John Wiley and Sons).](/cms/asset/92cb578d-c3b3-4d6c-9f91-4bad0b1477c9/ianb_a_1377725_f0018_c.jpg)
Figure 19. (a) Preparation procedure for CQDs-Oxa and its applications in bioimaging and theranostics. (Reprinted with permission from Ref. [Citation92] Copyright 2014 John Wiley and Sons.); (b) graphical depiction of the preparation of luminescent HCQDs and the loading of DOX. (Reprinted from Ref. [Citation16] Copyright 2013, with permission from Elsevier).
![Figure 19. (a) Preparation procedure for CQDs-Oxa and its applications in bioimaging and theranostics. (Reprinted with permission from Ref. [Citation92] Copyright 2014 John Wiley and Sons.); (b) graphical depiction of the preparation of luminescent HCQDs and the loading of DOX. (Reprinted from Ref. [Citation16] Copyright 2013, with permission from Elsevier).](/cms/asset/66d69d86-d8a6-48e7-aacf-a68e0b21e98d/ianb_a_1377725_f0019_c.jpg)