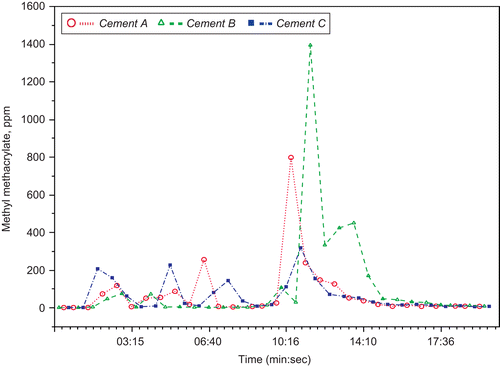
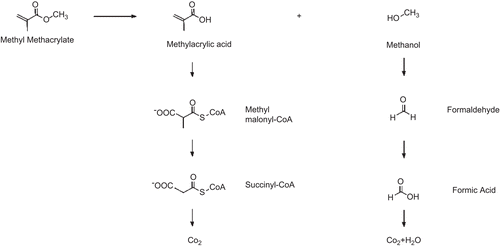
Figures & data
Table 1. Reactivity of MMA and other representative (meth)acrylates, respiratory sensitizers and irritants, dermal sensitizers, and non-sensitizers.
Table 2. Relative skin sensitization potency of contact allergens based on LLNA (ECETOC, 2008).
Table 3. Summary of skin sensitization studies of MMA.
Table 4. Summary of 19 case reports.