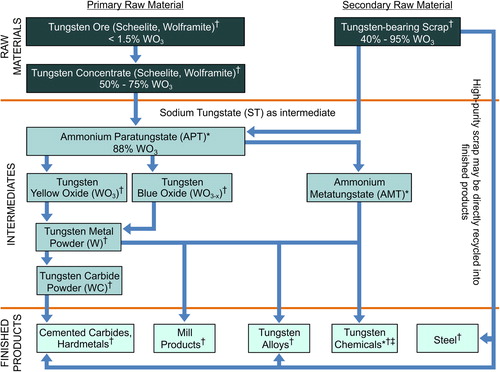
Figures & data
Table 1. Categories for ranking the reliability of studies or data from the literature or reports, based on the approach described by CitationKlimisch et al. (1997).
Table 2. Search strings, PubMed® MeSH query translations, and returned results for tungsten and tungstate publications from January 1, 2004 to May 19, 2014.
Table 3. Tungsten substance nomenclature and solubility in water.
Table 4. Standard toxicity testing summary for water-soluble and sparingly water-soluble tungsten substances.
Table 5. Acute oral, dermal, and inhalation studies on tungsten substances.
Table 6. Genotoxicity studies on tungsten substances.
Table 7. Studies on nano-tungsten substances.