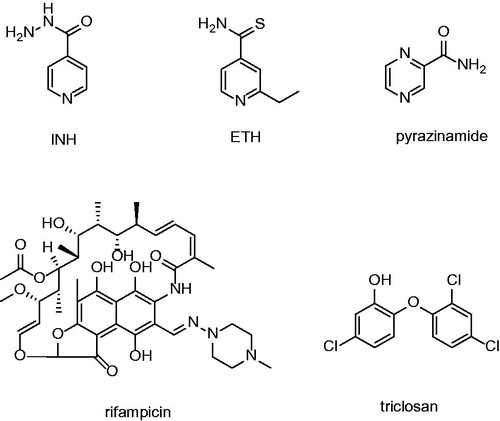
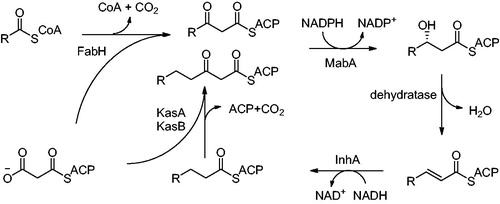
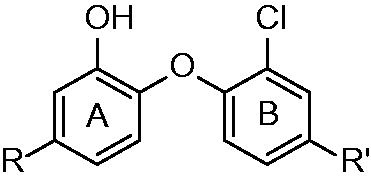
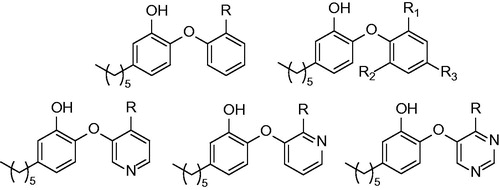
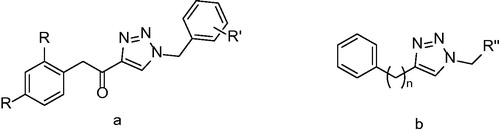
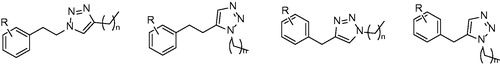
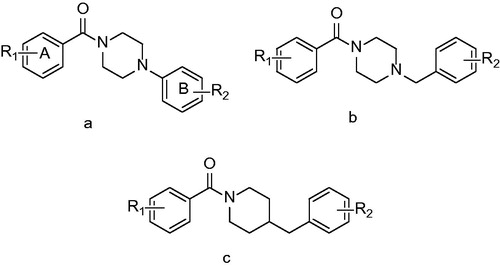
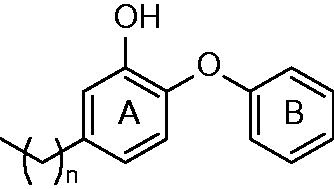
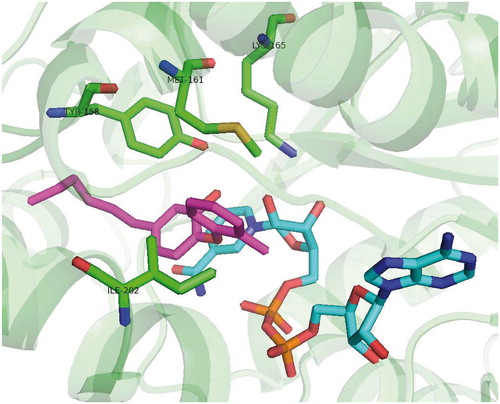
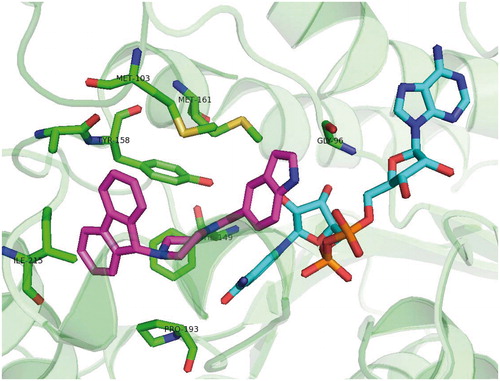
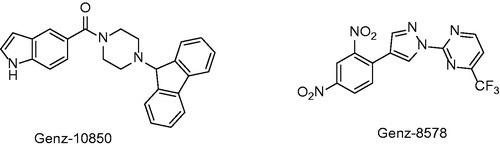
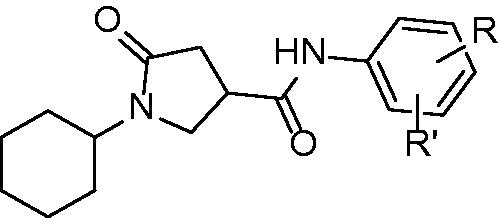
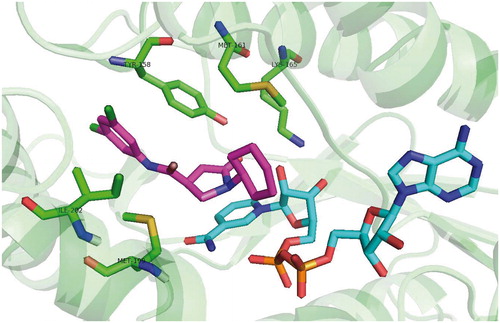
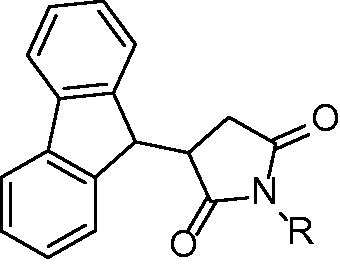
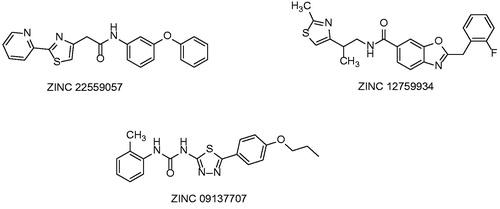
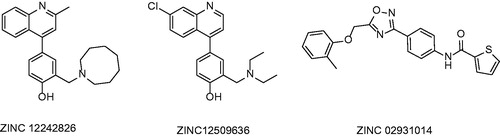
Figures & data
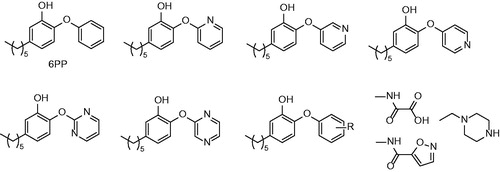
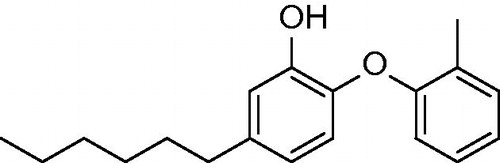
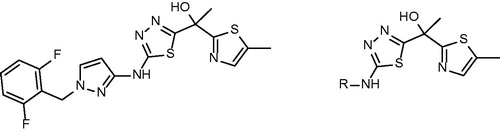
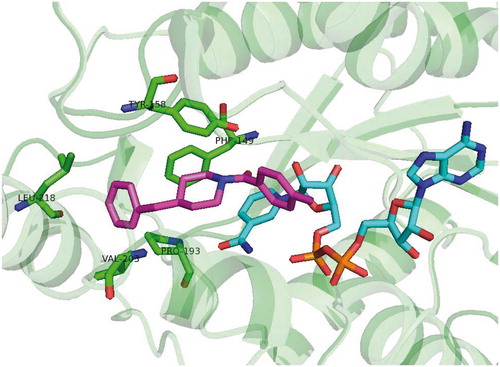
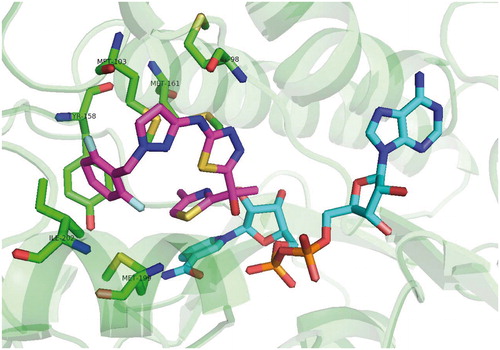
Table 1. Structures and in vitro results of notable InhA inhibitors.