ABSTRACT
The need for alternative treatments for multiple sclerosis (MS) has triggered copious amounts of research into microbial therapies focused on manipulating the microbiota–gut–brain axis. This comprehensive review was intended to present and systematically evaluate the current clinical and preclinical evidence for various probiotic and commensal gut microbial therapies as treatments for MS, using the Bradford Hill criteria (BHC) as a multi-parameter assessment rubric. Literature searches were performed to identify a total of 37 relevant studies (6 human, 31 animal), including 28 probiotic therapy and 9 commensal therapy studies. In addition to presenting qualitative summaries of these findings, therapeutic evidence for each bacterial formulation was assessed using the BHC to generate summative scores. These scores, which encompassed study quality, replication, and other considerations, were used to rank the most promising therapies and highlight deficiencies. Several therapeutic formulations, including VSL#3, Lactobacillus paracasei, Bifidobacterium animalis, E. coli Nissle 1917, and Prevotella histicola, emerged as the most promising. In contrast, a number of other therapies were hindered by limited evidence of replicable findings and other criteria, which need to be addressed by future studies in order to harness gut microbial therapies to ultimately provide cheaper, safer, and more durable treatments for MS.
Introduction
Multiple sclerosis
Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system (CNS) characterized by neuroinflammation, myelin sheath degeneration, axonal loss, and blood–brain barrier (BBB) deterioration.Citation1,Citation2 Globally, about 2.8 million people are estimated to live with MS.Citation3 The disease typically follows four different clinical courses: clinically isolated syndrome (CIS), relapsing-remitting MS (RRMS), primary progressive MS (PPMS), and secondary progressive MS (SPMS), with progressive forms being most severe and refractory to treatment.Citation1 As MS progresses, most patients are challenged with chronic pain and fatigue, gradual sensorimotor impairments, bowel and bladder dysfunction, cognitive changes, and overall diminished quality of life.Citation1,Citation2
There are currently 15 FDA-approved disease-modifying therapies (DMTs) used to decrease the severity and frequency of MS relapses.Citation4–6 These DMTs are generally effective at mitigating MS pathology by suppressing various aspects of the immune system, but they are also expensive,Citation2 often accompanied by an array of side effects,Citation5 and demonstrate decreased efficacy over time.Citation6–9 As such, improved, alternative MS treatments are warranted.
Like most chronic diseases, susceptibility to MS is driven by both genetic and environmental components, with the latter including well-documented risk factors like Epstein–Barr virus infection, vitamin D-insufficiency, and smoking.Citation10,Citation11 An emerging putative pseudo-environmental risk factor for MS and other chronic diseases is an imbalance (dysbiosis) in the gut microbiome, a complex ecosystem of trillions of microorganisms inhabiting our intestinal tracts. A generalized mechanism proposed is the bidirectional communication between the CNS and gut by way of the so-called microbiota-gut-brain axis (MGBA).Citation1,Citation12–14 Dysbiosis within the gut can promote effector T cell phenotypes toward proinflammatory pathways that subsequently increase intestinal barrier permeability.Citation15–20 This enables the release of microbial antigens and intestinal immune cells into circulation, further promoting systemic, low-level inflammation which may contribute to the weakened BBB tight junctions and enhanced T cell autoreactivity observed in MS.Citation15,Citation16,Citation18–20 The directionality of whether MS contributes to, as opposed to results from, this dysbiosis, however, is still unclear. Nevertheless, multiple studiesCitation16,Citation18–20 have characterized MS gut microbiomes as distinct from their healthy control-counterparts, generally possessing an elevated relative abundance of microrganisms associated with inflammation that when transplanted into mice have been shown to exacerbate an experimental animal model of MS, experimental autoimmune encephalomyelitis (EAE).Citation19
Commensal and probiotic gut bacterial therapies
Given the putative role of gut dysbiosis in promoting MS susceptibility, an attractive therapeutic approach would be to restore balance of the microbiome, and/or to take advantage of the intimate cross-talk between the immune system and gut microorganisms to inhibit or skew autoimmune responses.Citation21–24 Hence, gut targeted microbial therapies have been gaining traction as alternative or supplemental treatment options for a variety of conditions, including MS.
Gut bacterial commensals are generally beneficial organisms naturally comprising the gut microbiome to maintain a healthy host environment.Citation22 Whereas probiotics are defined by the International Scientific Association for Probiotics and Prebiotics (ISAPP) as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.”Citation25 For the purposes of this review, these are defined as live bacteria that can be supplemented into the microbiome to elicit beneficial changes in the commensal microbial community structure, and/or exert direct beneficial effects on the host.Citation22,Citation26,Citation27 It should be noted, however, that the exact distinction between gut bacterial commensals and bacterial probiotics remains arbitrary and far from uniform across studies, organizations, and regulatory guidelines. Nevertheless, both commensals and probiotics have important roles in digestive and immune health, including nutrient and vitamin synthesis, metabolism of host dietary products, intestinal barrier reinforcement, prevention of pathogenic microbe colonization, and anti-inflammatory immunoregulation.Citation21,Citation22,Citation26,Citation27Consequently, both have the therapeutic potential to mitigate MS pathology through modulation of the MGBA. This, in combination with accessibility and relatively low costs, makes probiotic and commensal therapies attractive alternative MS treatment candidates.
Alternative MS therapies: where do we stand?
A number of recent reviews have attempted to compile the growing body of evidence for gut microbiome-targeted therapies, though most of these are far from comprehensive. Citation12,Citation26–34 To date, there have been two published reviews that evaluate the clinical utility of probiotics and explore possible underlying mechanisms, including one systematic review of two clinical and five preclinical studies,Citation31 and one recent study with a meta-analysis of three clinical studies and systematic review of 22 preclinical studies.Citation33 Though valuable for highlighting probiotic therapeutic efficacy, both of these reviews are exclusively focused on probiotics without consideration of commensal therapy, and neither ranked the current evidence of each specific gut bacterial formulations using a quantitative objective rubric. Addressing the latter is particularly important, given that it helps to identify stronger and weaker areas within the field, highlight discrepancies, and ultimately provide direction for future research.
Accordingly, in this comprehensive review, we attempt to (1) compile the current clinical and preclinical evidence of MS mitigation by probiotic and commensal therapies; and (2) systematically rank the evidence of each gut-bacterial formulation using the Bradford Hill criteria (see below). In doing so, we aim to identify the most promising emerging therapies, as well as to highlight existing shortcomings in the field and emphasize specific foci for future studies.
Methods
This comprehensive review was originally intended as a systematic review, and therefore registered in the International Prospective Register of Systematic Reviews (PROSPERO; ID# CRD42020206819) following the initial search, but prior to screening articles.
Search strategies
Searches were conducted by two authors (LB & TM) on August 27, 2020 and January 4, 2021 using four databases: OvidMEDLINE, CINAHL, PubMed, and Web of Science. One paper was identified separately by one author (DK) outside of the search strategy.Citation35 Search strategies were tailored to each database using keywords, MeSH and MH headings, truncation, and an English Language filter (Supp. File 1A).
Selection criteria
Studies from all years were included in this review if they (1) were written or available in English, (2) investigated the effects of probiotic and/or commensal therapy on MS or an MS animal model severity and progression, and (3) utilized an experimental/intervention-based study design. Studies were excluded if they did not meet the inclusion criteria and/or used a non-intervention/experimental study design, including cohorts, cross-sectional studies, case–control studies, case series, and case reports.
Operational definitions
As mentioned above, the distinction between bacterial probiotics and commensals is not well defined, particularly as it applies to MS. For the purposes of this review, a bacterial therapeutic was considered “probiotic” when meeting evidence level 1–2 based on World Gastroenterology Organization guidelines from the Oxford Center for Evidence-Based Medicine, and “commensal” if falling at evidence level 3 and below where RCTs are lacking.Citation36 Study interventions were therefore classified as “probiotic therapy” if researchers supplemented with the following putative probiotics: Lactobacillus spp., Bifidobacterium spp., Escherichia coli Nissle 1917 (E. coli Nissle 1917), Enterococcus faecium (E. faecium), or Streptococcus thermophilus (S. thermopohilis); or “commensal therapy” if researchers supplemented with any other species of commensal bacteria, including Prevotella spp., Akkermansia spp., Pediococcus acidilactici (P. acidilactici), Clostridium butyricum (C. butyricum), and Bacteroides fragilis (B. fragilis).
There were two animal studies that were exceptions to these classifications, for the following reasons. Both studies introduced putative probiotic Lactobacillus spp. via stable colonization by a single inoculation rather than continuous treatment, which is more representative of commensal therapy than a probiotic therapy.Citation37,Citation38Additionally, the bacterial strains used in these two studies are not strains recognized as probiotics, but are instead isolates from commensal murine gut microbiota. These two studies were hence classified as commensal therapy.
Data extraction
Following screening studies for relevance against the selection criteria (LB, TM, & DK), data were extracted from the included studies by two authors (LB & TM) (Supp. File 1). The study metrics extracted included first author, year of publication, DOI, location of study, study design, sample, intervention, duration of study, MS model (for animal studies), measurements/outcomes, statistical methods, and power (for human studies). The study measurement/outcomes extracted included clinical parameters of MS/EAE severity and progression, immune and metabolic indices, microbiome and metabolome parameters, and mechanistic or correlative findings.
Evaluating quality and evidence of included studies
Included studies were subject to quality and risk of bias (ROB) assessments using the Cochrane ROB toolCitation39 for human studies and SYRCLE toolCitation40 for animal studies. High quality was assigned to studies with a low ROB, including randomized controlled trials (RCTs) and animal studies that explicitly stated using randomization and blinding measures. Medium quality was assigned to studies with an uncertain ROB, including non-RCT human studies and animal studies that did not explicitly state using randomization and/or blinding measures. Low quality was assigned to studies with a high ROB, including studies with considerable confounding, in addition to not explicitly stating the use of randomization or blinding. These quality assessments were factored into the summative evaluation of each bacterial therapy; therefore, no studies were excluded from analysis on the basis of ROB.
The overall quality and strength of therapeutic evidence provided by each bacterial formulation was assessed using the Bradford Hill criteria (BHC), which includes the following: temporal relationship, strength of relationship, dose–response relationship, replication of findings, biological plausibility, cessation of exposure, specificity of association, and coherence between multiple approaches.Citation41,Citation42 The descriptions and numerical designations of each BHC can be found in . Sufficient evidence (Yes or No) was determined for each criterion (except for replication, see below) and assigned a score of 1 for yes, followed by summation across all criteria to yield a final “BH score” for each therapy. Replication of findings was the most heavily weighted criterion, and was scored as follows: 3 = replicated in human and animal studies, 2 = replicated by different groups, 1 = replicated by the same group, 0 = not replicated, −2 = conflicting findings (not considering lack of effect as conflicting with positive). The calculations are detailed in Supplemental File 2 and summarized in .
Table 1. Bradford Hill criteria numerical designations and descriptions
Table 2. Human studies of probiotic therapies: summary of study characteristics and major findings
Table 3. Animal studies of probiotic therapies: summary of study characteristics and major findings
Table 4. Animal studies of commensal therapies: summary of study characteristics and major findings
Table 5. Bradford Hill criteria evaluation of probiotic and commensal therapies
Table 6. Bradford Hill criteria (BHC) warranting most attention for future probiotic and commensal therapy studies
Results
A. Study characteristics
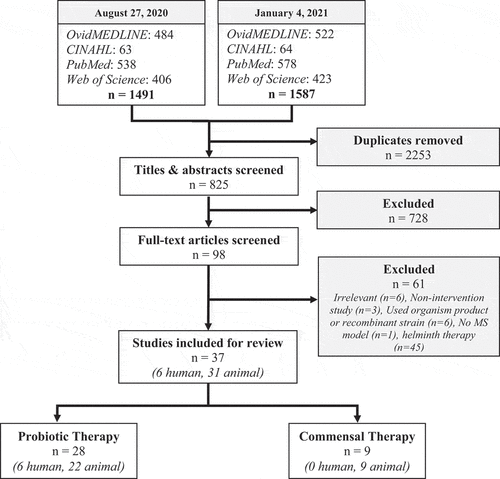
The study characteristics and major findings of the included studies are summarized in . A total of 770 de-duplicated articles were found by the initial search, 55 additional articles were found by the second search, and one articleCitation76 was found by an author (DK) outside of the search strategy. A total of 37 studiesCitation35,Citation37,Citation38Citation43–76 (6 human, 31 animal) were included for analysis in this review based on the stated selection criteria (see Methods) (; Supp. File 1B). Of these 37 studies, 28 (6 human, 22 animal) investigated the effects of probiotic therapy and 9 (0 human, 9 animal) utilized commensal therapy. Studies were conducted between 1998 and 2020 in the following countries: USA, Iran, Japan, England, Netherlands, Spain, Russia, Italy, Sweden, France, China, and Republic of Korea.
Figure 1. Search strategies, selection criteria, and inclusion of articles. Articles from four separate databases were identified and screened for inclusion. Note that helminth therapy studies were included in the search strategy but manually removed during the eligibility stage of the second search
Risk of bias
Overall, 10 studies (4 human, 6 animal) were deemed “high quality” based on the low risk of bias determined using the Cochrane ROB and SYRCLE tools (Supp. File 1D and 1 F). Most (n = 24) of the studies (0 human, 24 animal) were classified as “medium quality” due to study design limitations or failure to disclose randomization and/or blinding efforts. The remaining studies (n = 3) were classified as “low quality,” including two human studies that had important baseline characteristic differences between groups, substantial risk of confounding due to concurrent use of a DMT (glatiramer acetate), and compared the results to healthy controls rather than untreated MS patient controlsCitation46,Citation47,; and one animal study that was not powered to perform statistical analysis lacked clarity regarding the control groups and did not explicitly state the use of randomization or blinding measures.Citation72
Study design
Four human studies were structured as double-blind, placebo-controlled RCTs,Citation60,Citation61,Citation63,Citation64 and the remaining two were prospective cohort studies.Citation46,Citation47, The animal models of MS included EAE (n = 27), cuprizone-Citation45,Citation71 or gliotoxin-induced demyelination,Citation52 and Theiler’s murine encephalomyelitis virus-induced demyelinating disease (TMEV-IDD).Citation43
Subjects
Human subjects were studied exclusively in the RRMS stage. Expanded disability status scores (EDSS) were all ≤4.5 as a maximum for inclusion. Male and female participants ranged from 18 to 60 y old. Animal subjects included both male and female rats (Lewis and Wistar) and mice (C57BL/6, SJL, and PWD/PhJ) ranging in age from 2 weeks to 13 months, with a mean of 7–8 weeks old. Various genetically modified C57BL/6 mice were used, including HLA-DR3.DQ8 transgenicCitation56,Citation57,Citation58 and IL-10-,Citation48 TLR-2-,Citation49 and TLR-9-Citation49 deficient strains.
Interventions
Probiotic therapy involved individual strains and various combinations (including Lacto-mix, Lactibiane iki, IRT5, Protexin, VSL3/VSL#3/Vivomixx/LBS) of Lactobacillus spp. including L. casei, L. paracasei, L. plantarum, L. reuteri, L. delbrueckii subsp. bulgaricus, L. crispatus, L. rhamnosus, L. murinus, L. brevis, L. helveticus, L. lactis subsp. lactis, L. fermentum, L. acidophilus, and L. salivarius, Bifidobactererium ssp. including B. animalis, B. B94, B. breve, B. longum, B. bifidum, B. infantis, B. lactis, B. subtilis as well as E. coli Nissle 1917, E. faecium, and S. thermophilus. All four human RCT studies utilized probiotic combinations at 2 × 109 CFU/mL ranging from 12 weeks to 6 months duration, while the prospective cohorts used 3.6 × 1012 CFU/d for 2 months. The animal studies administered a mean of 109 CFU/mL (range of 108–1010 CFU/mL) for 2–7 weeks duration. Probiotics were administered prophylactically,Citation54,Citation59, therapeutically,Citation43,Citation44,Citation46,Citation47,Citation50,Citation52,Citation60,Citation61,Citation63,Citation64,Citation66,Citation75,Citation76 or both.Citation45,Citation51,Citation55,Citation69,Citation72,
There were no human studies using commensal therapy identified. Animal studies used Prevotella histicola (P. histicola),Citation56,Citation57,Citation58 P. acidilactici,Citation70C. butyricum,Citation65Akkermansia muciniphila (A. muciniphila),Citation35 L. reuteriCitation37,Citation38, (by stable colonization), Allobaculum,Citation38 and B. fragilisCitation62 at a dosage ranging 5 × 106 to 1 × 1010 CFU/mL for one administration or 2–5 weeks. Commensals were administered prophylactically,Citation37,Citation38,Citation62,Citation65 therapeutically,Citation35 or both.Citation56,Citation57,Citation58,Citation70
Measurements and outcomes
The most common measurements for clinical parameters for human studies included EDSS, mental health and quality of life assessments (Beck Depression Inventory (BDI), General Health Questionnaire-28 (GHQ-28), Depression Anxiety Stress Scale (DASS), Fatigue Severity Scale (FSS), McGill Pain Questionnaire (MPQ)). Notably, none of the human studies assessed MRI lesions. For animal studies, clinical signs of motor disability and associated quantitative variables (EAE incidence, onset, duration, and clinical scores; motor function, coordination, and activity for other MS models), histopathology (demyelination, CNS infiltration), BBB and intestinal permeability, and weight loss. Cytokine analysis, oxidative stress/antioxidant markers, and immunophenotyping were the most commonly measured immune/metabolic indices. Microbiome and metabolome assessments were primarily measured using fecal microbiome analysis, and fecal/serum short-chain fatty acid (SCFA) production. Gene expression and adoptive transfer experiments comprised additional mechanistic and correlative findings.
B. Probiotic therapy
Major trends
The major findings of each probiotic therapy study included can be found in for human and animal studies, respectively. A qualitative summary of these studies is provided below, followed by a semi-quantiative ranked evaluation using BH criteria.
Clinical studies
Four of the human probiotic therapy studies included were double-blind, placebo-controlled RCTs, and thus all were classified as “high quality” studies.Citation60,Citation61,Citation63,Citation64 Probiotic therapy produced modest decreases in EDSS that, while sometimes statistically significant, were not found to be clinically significant based on the authors’ designation of an EDSS change of ≥1.0 point for levels less than 5.5 or ≥0.5 point for levels greater than 5.5.Citation60,Citation64 The impact on EDSS seemed more pronounced in the shorter, 12 week study, suggesting that the observed benefits may only be transient.Citation60 Probiotic therapy did, however, lead to marked improvements in quality of life as measured through the BDI, GHQ-28, DASS, FSS, and MPQ assessments.Citation60,Citation63,Citation64 The proinflammatory cytokines that were measured (IL-6, IL-8, and TNFα) were consistently reduced in the probiotic treatment groups, as were several oxidative stress markers (hs-CRP, MDA).Citation60,Citation61,Citation63,Citation64 Anti-inflammatory cytokines and antioxidants were measured, showing elevated IL-10 and plasma nitric oxide.Citation60,Citation64
Two additional prospective cohort studies used therapy with the probiotic mixture VSL#3.Citation46,Citation47 While neither study focused on clinical outcome, both found that VSL#3 elicited changes in the peripheral immune response consistent with an immune regulatory state, including phenotypic changes in monocytes and dendritic cells and decreased expression of the MS risk allele HLA-DQA1. Additionally, these studies found an increased relative abundance of Lactobacillus, Bifidobacterium, Streptococcus spp. in stool, which is consistent with the species that were administered in probiotic form in the VSL#3 formulation. Further, one study found an increased relative abundance of Collinsela and Veillonellaceae family members that are typically depleted in MS gut microbiomes, as well as a decreased relative abundance of Akkermansia, Blautia, and Dorea genera, which are typically enriched.Citation46 Notably, these differences display an inverse relationship in MS patient cohort gut microbiomes, suggesting that VSL#3 may restore the MS-dysbiotic state.
Preclinical studies
The majority (26 out of 30) of animal studies used the EAE model. This is an important consideration, since it is a model driven by an autoimmune response, thus immunomodulation is the most likely mode of action for any effects on clinical disease. More studies investigated the effects of Lactobacillus spp.Citation48–50,Citation52,Citation53,Citation59,Citation67,Citation68,Citation71–76 and probiotic combinationsCitation43–45,Citation48,Citation50,Citation53,Citation69 rather than Bifidobacterium spp.Citation51–53,Citation68 and E. coli Nissle 1917,Citation54,Citation55 and just a single study utilized E. faecium.Citation66
Proportionally, Lactobacillus spp. tended to outperform Bifidobacterium spp. in reducing EAE incidence, onset, clinical score, duration, demyelination, immune cell infiltration, and motor activity. Positive clinical outcomes were observed in about 64% of Lactobacillus spp. studies (n = 14), 25% of Bifidobacterium spp. studies (n = 4), 100% of E. coli Nissle 1917 studies (n = 2), and about 81% of probiotic combination studies (n= 11), while E. faecium was shown to be at least as effective as the standard MS DMT glatiramer acetate.Citation66 L. casei Shirota,Citation68,Citation72,Citation73 and B. animalisCitation50–53 were the least clinically successful therapies among the studies investigated, while L. paracasei,Citation48,Citation49 L. plantarum,Citation48,Citation50 and E. coli Nissle 1917Citation54,Citation55 appeared the most successful. Notably, despite disparate outcomes, VSL#3Citation45 and VivomixxCitation43,Citation44 contain the same probiotic formulation, although importantly each study used a different model: cuprizone-induced demyelination/remyelination vs. TMEV-IDD vs. EAE, respectively, with the former (cuprizone) lacking a strong immune-mediated component. Furthermore, L. paracaseiCitation49 and the combinations of L. crispatus and L. rhamnosusCitation53 and B. animalis subsp. lactis strainsCitation53 were able to elicit clinical benefits whether administered live or heat-killed, while Lacto-mixCitation48 was only effective when live probiotic organisms were used. Five studies demonstrated a dose–response relationship,Citation44,Citation48,Citation50,Citation53,Citation54 and three studies provided evidence for the combinatorial effects of probiotics.Citation43,Citation45,Citation69 Only ~32% of the animal studies (n = 22) reported primarily no effect or exacerbation of clinical disease.Citation51,Citation59,Citation68,Citation72–75 It should also be noted that only four of the studies were classified as high quality, all of which reported positive results with probiotics.Citation44,Citation45,Citation50,Citation53
The majority of studies reported favorable secondary immunological findings, with elevated levels of anti-inflammatory cytokines (IL-10, IL-4, TGF-β)Citation43,Citation48,Citation50,Citation53,Citation55,Citation59,Citation69,Citation71,Citation74 and CD4+ CD25+ FoxP3+ TregsCitation44,Citation50,Citation54,Citation55,Citation69,Citation74 and reduced levels of proinflammatory cytokines (IL-17, IL-1, IL-6, TNF-α, IFN-γ),Citation43,Citation48,Citation50,Citation53,Citation55,Citation67,Citation69,Citation71,Citation75 chemokines (CCL3, CCL4, CXCL5, CXCL13),Citation49 and TH1 and TH17 cells.Citation43,Citation54,Citation55,Citation67,Citation69,Citation74,Citation75 Multiple Lactobacillus spp. and probiotic combinations also demonstrated decreased antigen-specific T cell proliferation.Citation44,Citation48,Citation50,Citation53,Citation69 Putatively beneficial microbiome changes included an increased relative abundance of Firmicutes, Bacteriodetes, Proteobacteria phyla and Sutterella, Bifidobacterium, Streptococcus, Lactobacillus, and Prevotella spp.Citation43,Citation44,Citation75 Two studies measured SCFA production and found increased levels in both serum and feces.Citation43,Citation45 Four studies reported increased expression of TH2 and Treg regulators (GATA3, Foxp3),Citation50 miR-25,Citation71 antimicrobial peptides (Reg3g, Reg3b),Citation55 and tight junction proteins (Claudin-8, ZO-I);Citation55 and decreased expression of TH1 and TH17 regulators (Tbet, RORγt),Citation44,Citation50 miR-155,Citation71 and the IDO gene,Citation71 a potential marker of MS/EAE relapses. Furthermore, Lavasani et al. performed an adoptive transfer experiment of CD4+ CD25 + T cells from mesenteric lymph nodes of the probiotic Lacto-mix group and found that the recipient mice had suppressed EAE symptoms and elevated IL-10 levels.Citation48 These effects, however, were eliminated when tested in IL-10-deficient mice.Citation48 Notably, Sanchez et al. also included an adoptive transfer experiment of splenic and mesenteric lymph node leukocytes of heat-killed L. paracasei–treated donor mice into recipient mice and found no such effects.Citation49 Additional mechanistic findings included decreased intestinal barrier permeabilityCitation55 and oligodendrocyte differentiation enhancement.Citation45
BHC scores and rankings
The BH score calculations and findings for probiotic therapy are detailed in Supplemental File 2, and summarized in . Given the large number of studies and treatments, we do not discuss each individually, but instead highlight and contrast some of the key findings below.
One probiotic treatment approach emerged as the most strongly supported (BH score = 9), namely the VSL#3 multi-species formulation, which was assessed in two human and three animal studies. Two out of three animal studies reported significant clinical improvement in the EAE model. The third study used the cuprizone demyelination model and reported a lack of clinical improvement, but some favorable histologic changes. The human studies did not measure clinical parameters, but reported immunological and microbiological changes that would be consistent with favorable immune modulation. Hence, this particular approach satisfied seven out of eight BH criteria (BHC #1, 3–8), with high evidence for replication (). In contrast, another combination treatment (L. acidophilus, L. casei, L. fermentum, and B. bifidum) was used in two high-quality human RCTs, but lacked supporting mechanistic and/or animal model studies (BH score = 5; ). Additional promising probiotic treatments with high BH scores included B. animalis, L. paracasei, and E. coli Nissle 1917, each receiving a BH score of 7; and L. plantarum, Lacto-mix, L. crispatus & L. rhamnosus, and the B. animalis combination therapy, all of which received a BH score of 6.
The majority of the remaining microbial treatments were characterized by low BH scores, resulting from a paucity of studies, lack of mechanistic evidence, and/or presence of conflicting evidence. The latter is exemplified by L. casei (BH score = 1), which was examined in six animal studies, but showed evidence of disease exacerbation or lack of effect in four of those studies, resulting in a deduction of 2 points for BHC #4. We note that this interpretation is confounded by the fact that different strains/isolates were used across these different studies, highlighting the need for careful standardization and interpretation.
BHC deficiencies
Using the Bradford Hill criteria, several therapies had considerable evidence for strength of relationship, dose–response relationship, biological plausibility, and coherence. Future studies should focus on strengthening these areas further by investigating dosing effects, establishing more direct evidence of MGBA involvement with more probiotic organisms and combinations, addressing alternative explanations, and repeating interventions in various contexts. Additionally, more evidence is needed to fulfill the remaining Bradford Hill criteria categories (BHC #s 1, 4, 6, and 7), including more before-and-after analyses, using a standardized protocol to facilitate comparisons across studies and research groups, and investigating cessation effects (). Strengthening the specificity of association by comparing the effects of live versus heat-killed organisms and their soluble products, and the inclusion of more mechanistic experiments is also recommended. Future studies and reviews should also consider the taxonomic reclassification of Lactobacillus spp. when referring to those probiotic organisms.Citation77
Lastly, to move toward translational application, wherein probiotics are stringently defined as conferring a known benefit to human health, disease-specific usage should be assessed in well-powered RCTs to provide clinically relevant guidance. Notably, a defined benefit to MS patient health should not be limited to clinical outcome, but also include secondary parameters such as quality of life, since it is plausible that probiotic therapy may improve the well-known GI-associated MS symptomatology (e.g. constipation) rather than affecting overall disease progression directly; and mental health, since depression has been identified as a risk factor for RRMS disability and relapses.Citation78,Citation79,Citation80
C. Commensal therapy
Major trends
The major findings for each of the commensal therapy animal studies included in this review can be found in . A qualitative summary of these studies is provided below, followed by a semi-quantiative ranked evaluation using BH criteria.
No human studies were identified for commensal therapy in this review, so the below trends are limited to preclinical findings. All but two (L. reuteriCitation37,Citation38 and AllobaculumCitation38) of the commensal organisms studied among the nine studies were shown to delay EAE onset and decrease clinical scores, incidence, inflammatory CNS infiltration, and demyelination. P. histicola was one of two commensals represented in more than one study and exhibited positive outcomes in each.Citation56–58 One study also reported reduced astrocytosis and microglial activation in the brain and spinal cord of P. histicola-treated mice,Citation58 while a sister study found that P. histicola helped to strengthen the MGBA by decreasing BBB permeability and restoring gut permeability.Citation56 Similar to probiotic therapy, reduced proinflammatory and increased anti-inflammatory immune responses were observed in each of the commensal studies. Specifically, studies found decreased IL-17- and IFN-γ-producing CD4 + T cells,Citation56,Citation58,Citation65 TH17 cells,Citation65 and IL-17,Citation56,Citation62,Citation70 IFN-γ,Citation56,Citation70 IL-23,Citation56 and IL-1270 cytokines; and increased IL-10,Citation56,Citation62,Citation70 TGF-β,Citation56 and Tregs.Citation56–58,Citation65 These results were consistent across all three P. histicola treatments.Citation56,Citation57,Citation58 L. reuteri was also represented in multiple studies and was shown to exacerbate EAE in both, either when administered alone (in the context of a normal microbiome)Citation37 or in combination with Allobaculum (in a dual-colonization gnotobiotic model).Citation38
For studies that analyzed the microbiome, commensal therapy groups had a general microbiome shift toward pre-EAE states following treatment, including an increased relative abundance of Bacteriodetes, Firmicutes, Prevotella spp., and Lactobacillus spp.Citation56,Citation57,Citation65 Mechanistically, the adoptive transfer of splenocytes from P. histicola-treated mice led to decreased EAE incidence in recipient mice.Citation56 Similarly, the adoptive transfer of FoxP3+ cells from wild-type B. fragilis-treated mice, resulted in decreased EAE clinical scores and increased levels of IL-10 in recipient mice.Citation62 These findings were not observed in the recipient mice receiving cells from polysaccharide A (PSA)-deficient B. fragilis-treated mice, suggesting that PSA is requisite for EAE protection. Separately, treatment with C. butyricum was reported to suppress phosphorylation of p38 MAPK and JNK signaling pathways – which are typically elevated in EAE – in the spinal cords of mice.Citation65 One commensal was also found to be at least as effective as two different DMTs (glatiramer acetateCitation57 and IFNβCitation58).
BHC scores and rankings
The BH score calculations and findings for commensal therapy are detailed in Supplemental File 2, and summarized in .
Treatment with P. histicola had fairly strong evidence (BH score = 7), but fell short across several BH categories (BHC #s 4 and 6), as it lacked human studies and replication by independent groups (). Another promising treatment with a high BH score was B. fragilis (BH score = 5), which scored points in BHC # 1, 2, 5, 7, and 8 owing to an adoptive transfer experiment, but lacked replication and evidence of dose-response and cessation effects. The remaining treatments were characterized by low BH scores (ranging 1–4) comprised of points in BHC # 1, 2, 3, and/or 5, once again resulting from a paucity of studies, lack of mechanistic evidence, and/or presence of conflicting evidence, as observed with L. reuteri (BH score = 1), which was found to exacerbate the disease in three of the five studies, leading to a 2-point deduction for BHC #4. As noted with L. casei for probiotic therapy, this interpretation is confounded by the use of different strains/isolates and modes of treatment (stable commensal colonization vs. daily gavage) across studies and would benefit from careful standardization ().
BHC deficiencies
Using the Bradford Hill criteria, commensal therapy had strong evidence for temporal relationship, specificity of association, and biological plausibility, but was lacking in the remaining categories (BHC #s 2–4, 6, and 8; ). Additional studies replicating current findings and testing more commensal organisms and combinations should be the main focus of future studies, since there were few commensal therapy studies overall and only P. histicola and L. reuteri were used in more than one study. Future studies should also focus on confirming colonization of the commensal organisms in the gut to reduce confounding and strengthen the specificity of association (BHC #7). Testing the effects of live versus heat-killed organisms and their products should also be prioritized, as these findings may contribute to the elucidating the underlying therapeutic mechanisms. For instance, subsequent studies of B. fragilis by the same research group utilized only the B. fragilis PSA symbiosis factor rather than administering live, wild-type B. fragilis and found similar reductions in EAE severity, as well as protection against EAE demyelination and inflammatory responses, providing a key molecular mechanism in support of the action of the live bacterium.Citation81–84
Other recommendations reflect those of probiotic therapy, namely controlling for alternative explanations, supporting immunological and microbiological findings with mechanistic experiments, and adding standardization to promote study design consistency and ease of comparison across studies.
Discussion
The purpose of this comprehensive review was to compile, summarize, and systematically rank the current evidence for probiotic and commensal therapeutic efficacy in MS and its preclinical models in an effort to identify weaker areas that should be addressed in future studies. A total of 37 studies were evaluated, including 28 for probiotic therapy and 9 for commensal therapy. The probiotic formulations VSL#3 (BH score = 9), B. animalis, L. paracasei, and E. coli Nissle 1917 (BH scores = 7) ranked highest due to their fulfillment of at least six of the eight Bradford Hill criteria. For commensal therapy – which suffered from a complete absence of clinical studies – the highest rankings went to P. histicola (BH score = 7) and B. fragilis (BH score = 5).
Animal studies demonstrated generally higher efficacy for reducing disease severity and progression with probiotic therapy than did the human studies, which is not unexpected, given the known shortcomings of the animal models, and the expected difficulties in translating basic science findings into therapy. The disconnect between human and animal studies could also be due to the difference and extent of the clinical markers measured, as clinical studies only measured MS severity through EDSS and questionnaires, while the pre-clinical studies were able to investigate EAE and the other MS models more comprehensively. MRI evaluation is a powerful, unbiased, and quantitative surrogate for MS severity and progression, and this was conspicuously lacking in the human studies. Additionally, human studies were in all likelihood underpowered to detect potentially subtle effects of probiotic treatments, and confounded by multiple environmental variables (e.g. diet, host baseline differences) that are impossible to control in this setting.
Replication of findings (BHC #4) was one of the most deficient Bradford Hill criteria across studies, with only 25% (n = 28) of the formulations receiving points and two of them (L. reuteri and L. casei) losing points. Another almost uniformly unfulfilled criterion was cessation of exposure, only addressed by six formulations. Both probiotic and commensal therapies would benefit from additional replication, testing more organism combinations, improved mechanistic evidence and comparison of live versus heat-killed organisms and their soluble products, and protocol standardization to enable improved contextual comparison across studies.
Limitations
Study limitations
There were several limitations to accurately assessing the efficacies of both therapies. First, there was widespread study design variability in the species and/or strain, dosage, duration of intervention, timeline, and sample characteristics. These variations would have been beneficial to external validation if the same strains were used across studies, but instead posed a challenge for assessing therapeutic utility. Standardized protocols outlining the optimal dosage, timeline, and duration for different organisms would be helpful for mitigating this issue, as was the focus of a review on probiotic therapy that concluded 109 CFU for 8–12 weeks duration produced the most favorable results.Citation31
Another study design issue was exclusion criteria and control of confounding variables, since some of the human studies did not account for diet or stress, which can alter gut microbial composition and subsequently influence MGBA interactions and concurrent DMT use, which could overshadow the true therapeutic efficacy if synergism exists between the two.Citation34 Additionally, genetic variability was also mostly unaccounted for in both human and animal studies (since the latter for the most part used a single strain of mouse). Furthermore, none of the human studies were conducted long enough to span the average remission period of 12–18 months, so the true impact of each therapy on reducing the severity of MS cannot be revealed with certainty.Citation1,Citation85 As for animal studies, none of these can accurately capture the complexity of spontaneous MS and its various forms in humans.Citation86–88
Only five studies tested the effects of live versus heat-killed organisms or their products.Citation48,Citation49,Citation53,Citation56,Citation62 This distinction is important, since equivalent efficacy with heat-killed organisms would help to reduce any associated risks of therapy posed by live microbiota and likely improve treatment uptake and adherence in patients. Furthermore, this effect likely differs across organisms. For instance, L. paracasei,Citation49 a combination of L. crispatus and L. rhamnosus,Citation53 and a combination of two B. animalis subsp. lactis strainsCitation53 did not need to be viable for EAE suppression, while P. histicolaCitation56 and Lacto-mixCitation48 did. Additionally, protection from EAE elicited by B. fragilis required the expression PSA by this bacterium, indicating that this bacterial product alone can play an important role in EAE protection.Citation62 Indeed, follow-up studies confirmed that live B. fragilis is not required, while PSA is sufficient to elicit a therapeutic effects.Citation81–84 Future studies should prioritize these distinctions to help optimize efficacy and therapeutic success.
Another limitation of this review was the quality and risk of bias for the studies included. Most of the animal studies included were classified as “medium quality” with “uncertain” bias due to the lack of explicitly stated randomization and blinding measures used. A lack of randomization can subject the results to inadvertent confounding and chance findings.Citation39,Citation40 Animals that live together in the same cage or area of a room may have more similar characteristics to each other than compared to a different cage or area importantly including the basal composition of their gut microbiomes. Additionally, a lack of blinding can introduce both performance and detection bias, as a researcher or caretaker’s knowledge of the treatment group can cause them to subconsciously act differently toward one group, such as providing extra care to sicker animals.Citation39,Citation40 It is entirely possible that these studies did in fact incorporate these measures into their protocol, but since it was not stated it was considered to be absent for this review. Other issues regarding quality include the premise that the positive findings observed across studies may simply reflect consistency of confounding variables and/or publication bias, rather than the therapy itself.
Review limitations
As for the risk of bias for this review, there were several methodological flaws in the assessment of therapeutic efficacy. Personal judgment was required for assessing each therapy’s fulfillment of the Bradford Hill criteria and there were no pre-defined guidelines as to what should be considered “sufficient evidence.” The scoring system was implemented as an arbitrary method to facilitate comparison of the efficacies in the context of the recommendation criterion and not intended to be a comprehensive assessment of the therapies. Regardless, the aim of this review was to highlight areas within each therapy that should be strengthened in future studies, so the risk of bias in this sense seems low. Separately, a few of the Bradford Hill criteria may be less important for establishing efficacy, causing the therapeutic to be penalized for lacking evidence in a non-applicable category. For example, a threshold effect rather than a dose–response relationship might be necessary for observing beneficial effects. Cessation of exposure may also not be necessary to demonstrate, since these therapies would theoretically be lifelong as is the case for DMTs. Accordingly, another avenue for future research could be establishing a minimum set of probiotic specific criteria for comprehensively evaluating therapeutic strategies in both animal and human studies.
Other limitations of this review were related to the search and screening process for identifying relevant studies. First, the use of an English language filter in our search strategy imposed obvious restrictions on the number of studies included and extent of available evidence. Second, the operational definitions we established limited the scope of probiotic and commensal therapies to only live or heat-killed bacteria, excluding any mechanistic evidence that may have been generated in studies that used only probiotic/commensal strain-soluble products. The BH score for B fragilis, for example, could have been improved had the follow-up studies that focused on the B. fragilis PSA symbiosis factor been eligible for inclusion.Citation81–84 Lastly, we did not contact the authors of studies classified as medium or low quality for clarification of missing methodological data (i.e. randomization, blinding). Attaining such information could have altered our quality assessments and evaluations for BHC #2. Regardless of these limitations, this review was intended as a resource to guide and optimize future probiotic and commensal therapy studies by highlighting both emerging therapies and study shortcomings, rather than to firmly conclude the therapeutic utility of specific formulations.
Conclusion
In this comprehensive review, we used a Bradford Hill criteria scoring approach to provide a multi-parameter assessment and ranking of evidence for specific gut microbial therapies, with the overall goal of identifying and highlighting areas of need for future research (see ). Several formulations emerged as having the most promise, including VSL#3, B. animalis, L. paracasei, and E. coli Nissle 1917 for probiotics; and P. histicola and B. fragilis for commensals. However, many other therapies fell short across a number of criteria, notably replication of findings. Other Bradford Hill criteria lacking evidence were temporal relationship and specificity of association for probiotic therapy, and strength of relationship, dose–response relationship, and coherence for commensal therapy. Future studies should prioritize addressing these shortcomings through better control of confounding, supporting immunological and microbiological findings with mechanistic experiments, improved standardization of protocols and therapeutic formutions, and the other suggestions discussed in this review. Focusing on these areas is necessary to make progress toward clinical implementation, since cheaper, safer, and more durable treatments for MS are in demand.
Supplemental Material
Download Zip (158.1 KB)Disclosure Statement
No potential conflict of interest was reported by the author(s).
Supplementary material
Supplemental data for this article can be accessed on the publisher’s website.
Additional information
Funding
References
- Baecher-Allan C, Kaskow BJ, Weiner HL. Multiple sclerosis: mechanisms and immunotherapy. Neuron. 2018;97(4):742–27. doi:10.1016/j.neuron.2018.01.021.
- Robertson D, Moreo N. Disease-modifying therapies in multiple sclerosis: overview and treatment considerations. Fed Pract. 2016;33:28–34.
- Walton C, King R, Rechtman L, Kaye W, Leray E, Marrie RA, Robertson N, La Rocca N, Uitdehaag B, van der Mei I, et al. Rising prevalence of multiple sclerosis worldwide: insights from the Atlas of MS, third edition. Mult Scler. 2020;26(14):1816–1821. doi:10.1177/1352458520970841
- Förster M, Küry P, Aktas O, Warnke C, Havla J, Hohlfeld R, Mares J, Hartung H-P, Kremer D. Managing risks with immune therapies in multiple sclerosis. Drug Saf. 2019;42(5):633–647. doi:10.1007/s40264-018-0782-8
- Harding K, Williams O, Willis M, Hrastelj J, Rimmer A, Joseph F, Tomassini V, Wardle M, Pickersgill T, Robertson N, Tallantyre E. Clinical outcomes of escalation vs early intensive disease-modifying therapy in patients with multiple sclerosis. JAMA Neurol. 2019;76(5):536. doi:10.1001/jamaneurol.2018.4905
- Yano H, Gonzalez C, Healy BC, Glanz BI, Weiner HL, Chitnis T. Discontinuation of disease-modifying therapy for patients with relapsing-remitting multiple sclerosis: effect on clinical and MRI outcomes. Mult Scler Relat Disord. 2019;35:119–127. doi:10.1016/j.msard.2019.07.021.
- Baroncini D, Zaffaroni M, Moiola L, Lorefice L, Fenu G, Iaffaldano P, Simone M, Fanelli F, Patti F, D’Amico E, et al. Long-term follow-up of pediatric MS patients starting treatment with injectable first-line agents: a multicentre, Italian, retrospective, observational study. Mult Scler. 2019;25(3):399–407. doi:10.1177/1352458518754364
- Gajofatto A, Bacchetti P, Grimes B, High A, Waubant E. Switching first-line disease-modifying therapy after failure: impact on the course of relapsing–remitting multiple sclerosis. Mult Scler. 2009;15(1):50–58. doi:10.1177/1352458508096687.
- Hua LH, Harris H, Conway D, Thompson NR. Changes in patient-reported outcomes between continuers and discontinuers of disease modifying therapy in patients with multiple sclerosis over age 60. Mult Scler Relat Disord. 2019;30:252–256. doi:10.1016/j.msard.2019.02.028.
- Marrie RA. Environmental risk factors in multiple sclerosis aetiology. Lancet Neurol. 2004;3(12):709–718. doi:10.1016/S1474-4422(04)00933-0.
- Ramagopalan SV, Dobson R, Meier UC, Giovannoni G. Multiple sclerosis: risk factors, prodromes, and potential causal pathways. Lancet Neurol. 2010;9(7):727–739. doi:10.1016/S1474-4422(10)70094-6.
- Boziki MK, Kesidou E, Theotokis P, Mentis A-FA, Karafoulidou E, Melnikov M, Sviridova A, Rogovski V, Boyko A, Grigoriadis N. Microbiome in multiple sclerosis: where are we, what we know and do not know. Brain Sci. 2020;10(4):234. doi:10.3390/brainsci10040234
- Galland L. The gut microbiome and the brain. J Med Food. 2014;17(12):1261–1272. doi:10.1089/jmf.2014.7000.
- Camara-Lemarroy CR, Metz LM, Yong VW. Focus on the gut-brain axis: multiple sclerosis, the intestinal barrier and the microbiome. WJG. 2018;24(37):4217–4223. doi:10.3748/wjg.v24.i37.4217.
- Ochoa-Repáraz J, Mielcarz DW, Ditrio LE, Burroughs AR, Foureau DM, Haque-Begum S, Kasper LH. Role of gut commensal microflora in the development of experimental autoimmune encephalomyelitis. J Immunol. 2009;183(10):6041–6050. doi:10.4049/jimmunol.0900747
- Jangi S, Gandhi R, Cox LM, Li N, von Glehn F, Yan R, Patel B, Mazzola MA, Liu S, Glanz BL, et al. Alterations of the human gut microbiome in multiple sclerosis. Nat Commun. 2016;7(1):12015. doi:10.1038/ncomms12015
- Camara-Lemarroy CR, Metz L, Meddings JB, Sharkey KA, Wee Yong V. The intestinal barrier in multiple sclerosis: implications for pathophysiology and therapeutics. Brain. 2018;141(7):1900–1916. doi:10.1093/brain/awy131.
- Miyake S, Kim S, Suda W, Oshima K, Nakamura M, Matsuoka T, Chihara N, Tomita A, Sato W, Kim S-W, et al. Dysbiosis in the gut microbiota of patients with multiple sclerosis, with a striking depletion of species belonging to Clostridia XIVa and IV clusters. Wilson BA, ed. PLoS ONE. 2015;10(9):e0137429. doi:10.1371/journal.pone.0137429
- Berer K, Gerdes LA, Cekanaviciute E, Jia X, Xiao L, Xia Z, Liu C, Klotz L, Stauffer U, Baranzini SE, et al. Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice. Proc Natl Acad Sci USA. 2017;114(40):10719–10724. doi:10.1073/pnas.1711233114
- Chen J, Chia N, Kalari KR, Yao JZ, Novotna M, Paz Soldan MM, Luckey DH, Marietta EV, Jeraldo PR, Chen X, et al. Multiple sclerosis patients have a distinct gut microbiota compared to healthy controls. Sci Rep. 2016;6(1):28484. doi:10.1038/srep28484
- Chang C-J, Lin T-L, Tsai Y-L, Wu T-R, Lai W-F, Lu C-C, Lai H-C. Next generation probiotics in disease amelioration. J Food Drug Anal. 2019;27(3):615–622. doi:10.1016/j.jfda.2018.12.011
- Xu H, Liu M, Cao J, Li X, Fan D, Xia Y, Lu X, Li J, Ju D, Zhao H. The dynamic interplay between the gut microbiota and autoimmune diseases. J Immunol Res. 2019;2019:1–14. doi:10.1155/2019/7546047
- Smallwood TB, Giacomin PR, Loukas A, Mulvenna JP, Clark RJ, Miles JJ. Helminth immunomodulation in autoimmune disease. Front Immunol. 2017;8:453. doi:10.3389/fimmu.2017.00453.
- Correale J. Helminth/parasite treatment of multiple sclerosis. Curr Treat Options Neurol. 2014;16(6):296. doi:10.1007/s11940-014-0296-3.
- Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506–514. doi:10.1038/nrgastro.2014.66
- Dargahi N, Johnson J, Donkor O, Vasiljevic T, Apostolopoulos V. Immunomodulatory effects of probiotics: can they be used to treat allergies and autoimmune diseases? Maturitas. 2019;119:25–38. doi:10.1016/j.maturitas.2018.11.002.
- Bach J-F. The hygiene hypothesis in autoimmunity: the role of pathogens and commensals. Nat Rev Immunol. 2018;18(2):105–120. doi:10.1038/nri.2017.111.
- Calvo-Barreiro L, Eixarch H, Montalban X, Espejo C. Combined therapies to treat complex diseases: the role of the gut microbiota in multiple sclerosis. Autoimmun Rev. 2018;17(2):165–174. doi:10.1016/j.autrev.2017.11.019.
- Guarner F, Bourdet-Sicard R, Brandtzaeg P, Gill HS, McGuirk P, van Eden W, Versalovic J, Weinstock JV, Rook GA. Mechanisms of Disease: the hygiene hypothesis revisited. Nat Rev Gastroenterol Hepatol. 2006;3(5):275–284. doi:10.1038/ncpgasthep0471
- Liu Y, Alookaran J, Rhoads J. Probiotics in autoimmune and inflammatory disorders. Nutrients. 2018;10(10):1537. doi:10.3390/nu10101537.
- Morshedi M, Hashemi R, Moazzen S, Sahebkar A, Hosseinifard E-S. Immunomodulatory and anti-inflammatory effects of probiotics in multiple sclerosis: a systematic review. J Neuroinflammation. 2019;16(1):231. doi:10.1186/s12974-019-1611-4.
- Zoledziewska M. The gut microbiota perspective for interventions in MS. Autoimmun Rev. 2019;18(8):814–824. doi:10.1016/j.autrev.2019.03.016.
- Jiang J, Chu C, Wu C, Wang C, Zhang C, Li T, Zhai Q, Yu L, Tian F, Chen W. Efficacy of probiotics in multiple sclerosis: a systematic review of preclinical trials and meta-analysis of randomized controlled trials. Food Funct. 2021;12(6):2354–2377. doi:10.1039/D0FO03203D
- Kohl HM, Castillo AR, Ochoa-Repáraz J. The microbiome as a therapeutic target for multiple sclerosis: can genetically engineered probiotics treat the disease? Diseases. 2020;8(3):33. doi:10.3390/diseases8030033.
- Liu S, Rezende RM, Moreira TG, Tankou SK, Cox LM, Wu M, Song A, Dhang FH, Wei Z, Costamagna G, Weiner HL. Oral administration of mir-30d from feces of MS patients suppresses MS-like symptoms in mice by expanding Akkermansia muciniphila. Cell Host Microbe. 2019;26(6):779–794.e8. doi:10.1016/j.chom.2019.10.008
- Guarner F, Sanders ME, Eliakim R, Fedorak R, Gangl A, Garisch J, Kaufmann P, Karakan T, Khan AG, Kim N, et al. World Gastroenterology Organisation Global Guidelines: probiotics and prebiotics. World Gastroenterology Organisation. 2017. [accessed 2021 Feb 1] https://www.worldgastroenterology.org/UserFiles/file/guidelines/probiotics-and-prebiotics-english-2017.pdf
- Montgomery TL, Künstner A, Kennedy JJ, Fang Q, Asarian L, Culp-Hill R, D’Alessandro A, Teuscher C, Busch H, Krementsov DN. Interactions between host genetics and gut microbiota determine susceptibility to CNS autoimmunity. Proc Natl Acad Sci USA. 2020;117(44):27516–27527. doi:10.1073/pnas.2002817117
- Miyauchi E, Kim S-W, Suda W, Kawasumi M, Onawa S, Taguchi-Atarashi N, Morita H, Taylor TD, Hattori M, Ohno H. Gut microorganisms act together to exacerbate inflammation in spinal cords. Nature. 2020;585(7823):102–106. doi:10.1038/s41586-020-2634-9
- Higgins JPT, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JAC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343(oct182):d5928–d5928. doi:10.1136/bmj.d5928
- Hooijmans CR, Rovers MM, De Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14(1):43. doi:10.1186/1471-2288-14-43.
- Hill AB. The environment and disease: association or causation? Proc R Soc Med. 1965;58(5):295–300. doi:10.1177/003591576505800503.
- Gordis L. Epidemiology.5th ed.Philadelphia (PA):Elsevier/Saunders;2014
- Mestre L, Carrillo-Salinas FJ, Feliú A, Mecha M, Alonso G, Espejo C, Calvo-Barreiro L, Luque-García JL, Estevez H, Villar LM, Guaza C. How oral probiotics affect the severity of an experimental model of progressive multiple sclerosis? Bringing commensal bacteria into the neurodegenerative process. Gut Microbes. 2020;12(1):1813532. doi:10.1080/19490976.2020.1813532
- Calvo-Barreiro L, Eixarch H, Ponce-Alonso M, Castillo M, Lebrón-Galán R, Mestre L, Guaza C, Clemente D, del Campo R, Montalban X, Espejo C. A commercial probiotic induces tolerogenic and reduces pathogenic responses in experimental autoimmune encephalomyelitis. Cells. 2020;9(4):906. doi:10.3390/cells9040906
- McMurran CE, Guzman de la Fuente A, Penalva R, Ben Menachem-Zidon O, Dombrowski Y, Falconer J, Gonzalez GA, Zhao C, Krause FN, Young AMH, et al. The microbiota regulates murine inflammatory responses to toxin-induced CNS demyelination but has minimal impact on remyelination. Proc Natl Acad Sci USA. 2019;116(50):25311–25321. doi:10.1073/pnas.1905787116
- Tankou SK, Regev K, Healy BC, Tjon E, Laghi L, Cox LM, Kivisäkk P, Pierre IV, Hrishikesh L, Gandhi R, et al. A probiotic modulates the microbiome and immunity in multiple sclerosis. Ann Neurol. 2018;83(6):1147–1161. doi:10.1002/ana.25244
- Tankou SK, Regev K, Healy BC, Cox LM, Tjon E, Kivisakk P, Vanande IP, Cook S, Gandhi R, Glanz B, et al. Investigation of probiotics in multiple sclerosis. Mult Scler. 2018;24(1):58–63. doi:10.1177/1352458517737390
- Lavasani S, Dzhambazov B, Nouri M, Fåk F, Buske S, Molin G, Thorlacius H, Alenfall J, Jeppsson B, Weström B. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. Unutmaz D, ed. PLoS ONE. 2010;5(2):e9009. doi:10.1371/journal.pone.0009009
- Sanchez JMS, Doty DJ, DePaula-Silva AB, Brown DG, Bell R, Klag KA, Truong A, Libbey JE, Round JL, Fujinami RS. Molecular patterns from a human gut-derived Lactobacillus strain suppress pathogenic infiltration of leukocytes into the central nervous system. J Neuroinflammation. 2020;17(1):291. doi:10.1186/s12974-020-01959-2
- Salehipour Z, Haghmorad D, Sankian M, Rastin M, Nosratabadi R, Soltan Dallal MM, Tabasi N, Khazaee M, Nasiraii LR, Mahmoudi M. Bifidobacterium animalis in combination with human origin of Lactobacillus plantarum ameliorate neuroinflammation in experimental model of multiple sclerosis by altering CD4+ T cell subset balance. Biomedicine Pharmacother. 2017;95:1535–1548. doi:10.1016/j.biopha.2017.08.117
- Ezendam J, De Klerk A, Gremmer ER, Van Loveren H. Effects of Bifidobacterium animalis administered during lactation on allergic and autoimmune responses in rodents: immune effects of probiotics. Clin Exp Immunol. 2008;154(3):424–431. doi:10.1111/j.1365-2249.2008.03788.x.
- Goudarzvand M, Rasouli Koohi S, Khodaii Z, Soleymanzadeh Moghadam S. Probiotics Lactobacillus plantarum and Bifidobacterium B94: cognitive function in demyelinated model. Med J Islam Repub Iran. 2016;30:391.
- Consonni A, Cordiglieri C, Rinaldi E, Marolda R, Ravanelli I, Guidesi E, Elli M, Mantegazza R, Baggi F. Administration of Bifidobacterium and Lactobacillus strains modulates experimental myasthenia gravis and experimental encephalomyelitis in Lewis rats. Oncotarget. 2018;9(32):22269–22287. doi:10.18632/oncotarget.25170
- Libbey JE, Sanchez JMS, Fleming BA, Doty DJ, DePaula-Silva AB, Mulvey MA, Fujinami RS. Modulation of experimental autoimmune encephalomyelitis through colonisation of the gut with Escherichia coli. Benef Microbes. 2020;11(7):669–684. doi:10.3920/BM2020.0012
- Secher T, Kassem S, Benamar M, Bernard I, Boury M, Barreau F, Oswald E, Saoudi A. Oral administration of the probiotic strain Escherichia coli Nissle 1917 reduces susceptibility to neuroinflammation and repairs experimental autoimmune encephalomyelitis-induced intestinal barrier dysfunction. Front Immunol. 2017;8:1096. doi:10.3389/fimmu.2017.01096
- Mangalam A, Shahi SK, Luckey D, Karau M, Marietta E, Luo N, Choung RS, Ju J, Sompallae R, Gibson-Corley K, et al. Human gut-derived commensal bacteria suppress CNS inflammatory and demyelinating disease. Cell Rep. 2017;20(6):1269–1277. doi:10.1016/j.celrep.2017.07.031
- Shahi SK, Freedman SN, Murra AC, Zarei K, Sompallae R, Gibson-Corley KN, Karandikar NJ, Murray JA, Mangalam AK. Prevotella histicola, a human gut commensal, is as potent as Copaxone® in an animal model of multiple sclerosis. Front Immunol. 2019;10:462. doi:10.3389/fimmu.2019.00462
- Shahi SK, Jensen SN, Murra AC, Tang N, Guo H, Gibson-Corley KN, Zhang J, Karandikar NJ, Murray JA, Mangalam AK. Human commensal Prevotella histicola ameliorates disease as effectively as interferon-beta in the experimental autoimmune encephalomyelitis. Front Immunol. 2020;11:578648. doi:10.3389/fimmu.2020.578648
- Maassen CBM, van Holten JCAM, Balk F, den Bak‐Glashouwer MJH, Leer R, Laman JD, Boersma WJA, Claassen E. Orally administered Lactobacillus strains differentially affect the direction and efficacy of the immune response. Veterinary Quarterly. 1998;20(sup3):81–83. doi:10.1080/01652176.1998.9694976
- Kouchaki E, Tamtaji OR, Salami M, Bahmani F, Daneshvar Kakhaki R, Akbari E, Tajabadi-Ebrahimi M, Jafari P, Asemi Z. Clinical and metabolic response to probiotic supplementation in patients with multiple sclerosis: a randomized, double-blind, placebo-controlled trial. Clin Nutrition. 2017;36(5):1245–1249. doi:10.1016/j.clnu.2016.08.015
- Tamtaji OR, Kouchaki E, Salami M, Aghadavod E, Akbari E, Tajabadi-Ebrahimi M, Asemi Z. The effects of probiotic supplementation on gene expression related to inflammation, insulin, and lipids in patients with multiple sclerosis: a randomized, double-blind, placebo-controlled trial. J Am Coll Nutr. 2017;36(8):660–665. doi:10.1080/07315724.2017.1347074
- Ochoa-Repáraz J, Mielcarz DW, Ditrio LE, Burroughs AR, Begum-Haque S, Dasgupta S, Kasper DL, Kasper LH. Central nervous system demyelinating disease protection by the human commensal Bacteroides fragilis depends on polysaccharide a expression. JI. 2010;185(7):4101–4108. doi:10.4049/jimmunol.1001443
- Rahimlou M, Hosseini SA, Majdinasab N, Haghighizadeh MH, Husain D. Effects of long-term administration of Multi-Strain Probiotic on circulating levels of BDNF, NGF, IL-6 and mental health in patients with multiple sclerosis: a randomized, double-blind, placebo-controlled trial. Nutr Neurosci. 2020;1–12. Published online June 5. doi:10.1080/1028415X.2020.1758887.
- Salami M, Kouchaki E, Asemi Z, Tamtaji OR. How probiotic bacteria influence the motor and mental behaviors as well as immunological and oxidative biomarkers in multiple sclerosis? A double blind clinical trial. J Funct Foods. 2019;52:8–13. doi:10.1016/j.jff.2018.10.023.
- Chen H, Ma X, Liu Y, Ma L, Chen Z, Lin X, Si L, Ma Xueying, Chen X. Gut microbiota interventions with Clostridium butyricum and norfloxacin modulate immune response in experimental autoimmune encephalomyelitis mice. Front Immunol. 2019;10:1662. doi:10.3389/fimmu.2019.01662
- Abdurasulova IN, Matsulevich AV, Tarasova EA, Kudryavtsev IV, Serebrjakova MK, Ermolenko EI, Bisaga GN, Klimenko VM, Suvorov AN. Enterococcus faecium strain L-3 and glatiramer acetate ameliorate experimental allergic encephalomyelitis in rats by affecting different populations of immune cells. Benef Microbes. 2016;7(5):719–729. doi:10.3920/BM2016.0018
- Yamashita M, Ukibe K, Matsubara Y, Hosoya T, Sakai F, Kon S, Arima Y, Murakami M, Nakagawa H, Miyazaki T. Lactobacillus helveticus SBT2171 attenuates experimental autoimmune encephalomyelitis in mice. Front Microbiol. 2018;8:2596. doi:10.3389/fmicb.2017.02596
- Kobayashi T, Kato I, Nanno M, Shida K, Shibuya K, Matsuoka Y, Onoue M. Oral administration of probiotic bacteria, Lactobacillus casei and Bifidobacterium breve, does not exacerbate neurological symptoms in experimental autoimmune encephalomyelitis. Immunopharmacol Immunotoxicol. 2010;32(1):116–124. doi:10.3109/08923970903200716
- Kwon H-K, Kim G-C, Kim Y, Hwang W, Jash A, Sahoo A, Kim J-E, Nam JH, Im S-H. Amelioration of experimental autoimmune encephalomyelitis by probiotic mixture is mediated by a shift in T helper cell immune response. Clinical Immunol. 2013;146(3):217–227. doi:10.1016/j.clim.2013.01.001
- Takata K, Kinoshita M, Okuno T, Moriya M, Kohda T, Honorat JA, Sugimoto T, Kumanogoh A, Kayama H, Takeda K, et al. The lactic acid bacterium Pediococcus acidilactici suppresses autoimmune encephalomyelitis by inducing IL-10-producing regulatory T cells. Ashour HM, ed. PLoS ONE. 2011;6(11):e27644. doi:10.1371/journal.pone.0027644
- Gharehkhani Digehsara S, Name N, Esfandiari B, Karim E, Taheri S, Tajabadi-Ebrahimi M, Arasteh J. Effects of Lactobacillus casei strain T2 (LBRC-M10783) on the modulation of Th17/Treg and evaluation of miR-155, miR-25, and IDO-1 expression in a cuprizone-induced C57BL/6 mouse model of demyelination. Inflammation. 2021;44(1):334–343. doi:10.1007/s10753-020-01339-1
- Ezendam J, Van Loveren H. Lactobacillus casei Shirota administered during lactation increases the duration of autoimmunity in rats and enhances lung inflammation in mice. Br J Nutr. 2008;99(1):83–90. doi:10.1017/S0007114507803412.
- Baken KA, Ezendam J, Gremmer ER, de Klerk A, Pennings JLA, Matthee B, Peijnenburg AACM, van Loveren H. Evaluation of immunomodulation by Lactobacillus casei Shirota: immune function, autoimmunity and gene expression. Int J Food Microbiol. 2006;112(1):8–18. doi:10.1016/j.ijfoodmicro.2006.06.009
- Kobayashi T, Suzuki T, Kaji R, Serata M, Nagata T, Ando M, Iizuka R, Tsujibe S, Murakami J, Kiyoshima-Shibata J, et al. Probiotic upregulation of peripheral IL-17 responses does not exacerbate neurological symptoms in experimental autoimmune encephalomyelitis mouse models. Immunopharmacol Immunotoxicol. 2012;34(3):423–433. doi:10.3109/08923973.2010.617755
- He B, Hoang TK, Tian X, Taylor CM, Blanchard E, Luo M, Bhattacharjee MB, Freeborn J, Park S, Couturier J, et al. Lactobacillus reuteri reduces the severity of experimental autoimmune encephalomyelitis in mice by modulating gut microbiota. Front Immunol. 2019;10:385. doi:10.3389/fimmu.2019.00385
- Johanson DM, Goertz JE, Marin IA, Costello J, Overall CC, Gaultier A. Experimental autoimmune encephalomyelitis is associated with changes of the microbiota composition in the gastrointestinal tract. Sci Rep. 2020;10(1):15183. doi:10.1038/s41598-020-72197-y.
- Zheng J, Wittouck S, Salvetti E, Franz CMAP, Harris HMB, Mattarelli P, O’Toole PW, Pot B, Vandamme P, Walter J, et al. A taxonomic note on the genus Lactobacillus: description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int J Syst Evol Microbiol. 2020;70(4):2782–2858. doi:10.1099/ijsem.0.004107
- Bsteh G, Ehling R, Lutterotti A, Hegen H, Di Pauli F, Auer M, Deisenhammer F, Reindl M, Berger T. Long term clinical prognostic factors in relapsing-remitting multiple sclerosis: insights from a 10-year observational study. Meuth SG, ed. PLoS ONE. 2016;11(7):e0158978. doi:10.1371/journal.pone.0158978
- McIvor GP, Riklan M, Reznikoff M. Depression in multiple sclerosis as a function of length and severity of illness, age, remissions, and perceived social support. J Clin Psychol. 1984;40(4):1028–1033. doi:10.1002/1097-4679(198407)40:4<1028::AID-JCLP2270400427>3.0.CO;2-1.
- Moore P, Hirsta C, Harding KE, Clarkson H, Pickersgilla TP, Robertson NP. Multiple sclerosis relapses and depression. J Psychosom Res. 2012;73(4):272–276. doi:10.1016/j.jpsychores.2012.08.004.
- Ochoa-Repáraz J, Mielcarz DW, Wang Y, Begum-Haque S, Dasgupta S, Kasper DL, Kasper LH. A polysaccharide from the human commensal Bacteroides fragilis protects against CNS demyelinating disease. Mucosal Immunol. 2010;3(5):487–495. doi:10.1038/mi.2010.29
- Telesford KM, Yan W, Ochoa-Reparaz J, Pant A, Kircher C, Christy MA, Begum-Haque S, Kasper DL, Kasper LH. A commensal symbiotic factor derived from Bacteroides fragilis promotes human CD39 + Foxp3 + T cells and T reg function. Gut Microbes. 2015;6(4):234–242. doi:10.1080/19490976.2015.1056973
- Wang Y, Begum-Haque S, Telesford KM, Ochoa-Repáraz J, Christy M, Kasper EJ, Kasper DL, Robson SC, Kasper LH. A commensal bacterial product elicits and modulates migratory capacity of CD39 + CD4 T regulatory subsets in the suppression of neuroinflammation. Gut Microbes. 2014;5(4):552–561. doi:10.4161/gmic.29797
- Wang Y, Telesford KM, Ochoa-Repáraz J, Haque-Begum S, Christy M, Kasper EJ, Wang L, Wu Y, Robson SC, Kasper DL, Kasper LH. An intestinal commensal symbiosis factor controls neuroinflammation via TLR2-mediated CD39 signalling. Nat Commun. 2014;5(1):4432. doi:10.1038/ncomms5432
- Rolak LA. Multiple sclerosis: it’s not the disease you thought it was. Clin Med Res. 2003;1(1):57–60. doi:10.3121/cmr.1.1.57.
- Palumbo S, Pellegrini S. Experimental in vivo models of multiple sclerosis: state of the art. In: Zagon IS, McLaughlin PJ, editors. Multiple Sclerosis: perspectives in Treatment and Pathogenesis. Brisbane (AU): Codon Publications; 2017. p. 173–183. http://www.ncbi.nlm.nih.gov/books/NBK470145
- Torkildsen Ø, Brunborg LA, Myhr K-M, Bø L. The cuprizone model for demyelination. Acta Neurol Scand. 2008;117(s188):72–76. doi:10.1111/j.1600-0404.2008.01036.x.
- Dal Canto MC, Kim BS, Miller SD, Melvold RW. Theiler’s murine encephalomyelitis virus (TMEV)-induced demyelination: a model for human multiple sclerosis. Methods. 1996;10(3):453–461. doi:10.1006/meth.1996.0123.

