ABSTRACT
Prebiotics are good source of dietary fiber or a group of nutrients or non-digestible short chain carbohydrates which human body cannot digest and it stimulates the growth and activity of some friendly bacteria in your intestinal tract. Prebiotics fibers are linked to β-(2→1) fructosyl units and are universally agreed to be fructooligosaccharides (FOS) or oligofructose or oligosaccharide or inulin. FOS are oligosaccharides that occur naturally in a variety of plants such as garlic and onions, wheat, rye, chicory roots, jerusalem artichokes, nectarine, seaweed, sugar cane bagasse, cassava waste, rice bran, rice straw, apple pomace, papaya, beet root peels, flaxseed, cereals, and banana peels among many others. These are extracted from fruits, vegetables, seeds and cereals using the various novel techniques such as membrane ultra-filtration method, microwave extraction, Ultrasonic Microwave Assisted Extraction, and high-pressure solvent extraction method. FOS are non-digestible carbohydrates with low-calorie that improve the taste and texture of food products while immunity booster, nutraceutical properties, bone health, microbial properties and its prebiotic activity in digestive track are the limelight of current review article. In addition, the main beneficial physiological effects of FOS such as low carcinogenicity, improved mineral absorption in gut and a reduction in serum cholesterol levels, triacylglycerols and phospholipids are the major part of this review. Moreover, FOS is beneficial for long-term blood glucose and its sensitivity to insulin due to the higher contents of plasma-free fatty acids using low tissue glucose. Conclusively, FOS improves intestinal flora, with subsequent relief of constipation, reduce the risk of heart diseases and certain cancers, improved blood lipids in hyperlipidemia, suppressed the production of intestinal putrefactive-substances and make your digestive work better.
Overview
Fructo-oligosaccharides (FOS) are naturally occurring organic compounds found in fruits and vegetables. These contain 2–10 of monosaccharide units with glycosidic linkage and are widely used as a natural sugar. The sweetness level of FOS ranged from 30% to 50% also used to prepare natural sugar syrups. FOS naturally and commercially applied since the 1980s on the demand of low calorie and healthy food for consumer.[Citation1] FOS are indigestible compounds and natural prebiotics. These travels intact through the small intestine to large intestine, where these compounds stimulated the growth and activity of probiotics in the gastrointestinal tract and influence on bowel functioning through fermentation.[Citation2] Aspergillus niger produced FOS using transfructosylation of β-fructosidase or sucrose. The general formula for the resulting mixture is GFn, with a range from 1 to 5. In contrast, there are many other interactions in addition to inulin-derived from FOS with β (1→2) bound.[Citation3]
FOS endure the hydrolysis through saliva and intestinal digestive enzymes due to the formation of their glycosidic bonds. These are fermented in intestine by anaerobic bacteria, which contribute to enhance bioavailability of dietary fiber that used as low-caloric value. Furthermore, FOS is used as a functional ingredient in dairy and baking industries as it is more soluble than inulin. FOS is used in the manufacture of different food products as sweetener which enhances the taste and sweetness of the product.[Citation4]
Fructooligosaccharides are chain of plant sugars. Asparagus, soybeans and Jerusalem artichoke are the excellent source of the Fructooligosaccharides. FOS are used as natural compound in pharmaceutical and food industries. These compounds are extracted from fruits and vegetables such as onion, chicory roots, bananas, garlic, asparagus, leeks and jicama. These compounds are also present in cereal grains wheat and corn.[Citation5] Jerusalem artichoke and yacon are cultivated plants and are rich source of FOS.[Citation6] According to Juffrie [Citation7] dietary FOS improved digestive system and also reduced the frequency of diarrhea in infants.
Dietary FOS increase the number of fecal bifidobacteria in adults and elderly. Bifidobacteria is a probiotic that consumed to be antagonist of pathogenic bacteria and promote nonspecific stimulation of immune system. FOS showed beneficial perspectives on the host including immunomodulatory effects on the intestine by increasing the number of bifidobacteria. Dietary FOS are reported as functional compounds and have beneficial effects on the mucosal immune system. These are promoted the growth of lymphoid tissues that is associated with the intestine. [Citation8] Over time, people become more concerned about their diet and health. At the same time, many researches on food sector has led to increase food supplements and these increased the nutritional value of food. Prebiotics are nutrients that are not digested in the stomach.[Citation9] These are metabolized in the colon as well as stimulated the growth of beneficial bacteria and also reduced the growth of harmful bacteria. Oligosaccharides are known as prebiotic agents. These agents increase the growth of Lactobacillus and bifidobacteria in the large intestine and have beneficial biological and medicinal properties. These compounds are seeking commercial importance due to their functional and nutraceutical properties. [Citation10]
Prebiotics
Prebiotics have been described as a non-digestible food with beneficially effects on host by selectively stimulating the growth and activity of one or more bacteria in the large intestine. Many food oligosaccharides and polysaccharides as well as dietary fibers are claimed to have prebiotic activity, but not all dietary carbohydrates are prebiotic. There is a dire need to set clear criteria for classifying a food ingredient as prebiotic. Prebiotics are common and indigestible oligosaccharides. Prebiotics and probiotics are proposed to differentiate some colonial foods from the concept of attractive nutritional properties and symbiotic combination. [Citation11]
In another study, prebiotics are non-digestible food ingredients, which promote the growth of probiotics in the human gut. These are used as an alternative food ingredients to help in lowering bad cholesterol.[Citation12] Along with the food components, non-digestible foods such as oligosaccharides, polysaccharides as well as numerous proteins peptide and lipids are also runner prebiotics their compounds and chemical structure are not reacted with digestive enzymes called “colonial foods” (food ingredients which act as chemical reagent primarily in the colon and toward inside of large intestine). Therefore, host (gut) provides indirectly energy, metabolic substrates and essential micronutrients. In foods, prebiotics are allowed to accomplish the standards of non-digestible carbohydrates. Although some proteins peptides, plants and milk products are known to have partially indigestible and beneficial effects. Most of the catalytic minerals Fe and Ca are absorbed in the intestine, inspiring the host defense system. [Citation13]
Watson and Preedy [Citation14] reported the selective yeast of prebiotics and helped to maintain a balanced gut microflora, which was directly targeted at the intestine and used to promote the species. Other systemic effects are also found in the bloodstream after absorption of fermented products that are not established. Anaerobic proteolysis is likely to produce ammonia, amines which are potentially harmful compounds. Delzenne and Roberfroid [Citation15] studied that FOS are naturally present as nondigestible lipids in common food products that cannot be extensively investigated and colonial microbiota is mostly unknown due to lipid metabolism. Resistant starch is an indigestible carbohydrate including miscellaneous compounds, nondigestible oligosaccharides and non-starch polysaccharides such as cellulose and hemicellulose. However, it may be categorized as functional foods of gut.[Citation16] Most of the compounds in most colonial fermentation processes are nonspecific. Various bacterial species of metabolic activity are potentially beneficial and harmful.[Citation17]
The first introduced concept of non-digestible oligosaccharides have been classified as prebiotics.[Citation18] Similarly, the Dawood et al. [Citation19] classified prebiotics as “indigestible edible ingredients that are resistant to digestion and absorption, fermented by colonic microbiota, and contributing to colonic and host health.” These indigestible oligosaccharides are classified into prebiotics such as galacto-oligosaccharides, fructans, and lactulose.
Another study of Sanz et al. [Citation20] exposed the ability of bifidobacteria and lactobacillus to ferment prebiotic carbohydrates in these two strains. Furthermore, it is not clear which prebiotic carbohydrates is the most suitable substitute for selective development of specific strains. Recently, in the state of in vitro fermentation, several quantitative approaches have been formulated to eliminate the activity of prebiotics. These methods indicated that specific effects reproduce the comparability of a given prebiotic based on prebiotic growth rate dimension, short chain fatty acid production, substrate integration and microbial population. [Citation21]
There are prebiotic polysaccharides and oligosaccharides that can resist absorption and digestion in the small intestine but these are fermented by probiotic bacteria residing in the large intestine. In principle, oligosaccharides are short-chain carbohydrates such as galacto-oligosaccharides, FOS, and inulin. Food products with non-digestible ingredients have positive effects on animal gut by stimulating the activity and growth of beneficial native bacteria in the gastro intestinal tract and reduce the pathogenic one’s prebiotics. FOS are incorporated in many food products to enhance their functional and nutritional properties.[Citation22] Inulin, oligosaccharides, lactulose, maltodextrin, mannitol, raffinose, and sorbitol have been reported to possess these prebiotic characteristics. [Citation23]
Dietary modulation of the gut microflora through prebiotics can be designed to enhance the activity of the bifidobacteria and lactobacilli.[Citation24] Fermentable carbohydrates play an important role that are not stimulated and digested in the small intestine. Activity of some gram-positive bacteria and bifidobacteria is related to the human probiotic. When multifaceted carbohydrates enter the small intestine and go to the lower gut where these are present with colonic bacteria, except bacteria present in the large intestine, [Citation25] FOS, galacto-oligosaccharides, lactulose and inulin as well as its plant-derived protein, maltodextrin oligosaccharides. Moreover, non-digestible carbohydrates are broken down by gut bacteria and produces the energy in the form of monocarboxylic acid, butyric acid and propionic acid. [Citation26]
Food grades compounds contain saccharides with varying degrees of polymerization. The degree of polymerization (DP) in a large part of inulin is close to 14. These polysaccharides prepared in three different ways i.e., microbiological synthesis, enzymatic synthesis, extraction and enzymatic hydrolysis of polysaccharides. The majority of prebiotic oligosaccharides are widely available in the market and are manufactured on an industrial scale. Recently, most patents related to prebiotic oligosaccharides have been claimed and the field is growing steadily.[Citation27]
In scientific research, there is much interest in dietary modulation of the human gut. Therefore, prebiotic are added on the selection of processed foods to improve gut microbial activities and their composition. The stability of prebiotics during food processes, such as baking, pasteurization, extrusion, high temperature heating, low pH condition is very low. The presence of FOS around food may affect the stability of probiotics.[Citation28]
Sources and their applications
Fructo-oligosaccharides are compounds found naturally in many plants including tomatoes, garlic, onions, wheat, rye, chicory Root, Jerusalem artichokes, nectarine, seaweed, sugar cane bagasse, cassava waste, rice bran, maize bran, rice straw, apple pomace, papaya, beet root peels, and banana peels as shown in the (). [Citation45] In this study, the use of fructose oligosaccharides has been technically obtained through hydrolysis of sucrose, such as fructans, ketonic monosaccharide and 1,1-kestotetraose. FOS are same flavor of many low-energy ingredients (sucrose), its physical and chemical properties, and its ability to compete successfully in an inclusive selection of functional food, bread, cake, pastries and confectionary products. [Citation46]
Table 1. Different natural sources of FOS
FOS are good source of soluble dietary fiber. In this study, bacteria that ferment fibers and produce short chain fatty acids bacteria showed strong effects on the intestinal mucosa with specific emphasis on their relevance for inflammatory bowel diseases as compared to healthy individuals.[Citation47] FOS are good for long-term blood glucose and its sensitivity to insulin because of the high content of plasma – short chain fatty acids. However, enduring nutritional supplement with organic acid has been revealed in diet of mice and also in humans to lower blood glucose. [Citation48] FOS are used as a sweetener in comparison to microbial oligosaccharides.[Citation49] Fructo-oligosaccharides are a naturally occurring polyfructan which collaborate with microbial and chemical processes, are long-lived to prepare high-fructose syrups.[Citation50] Microbial inulinase converts inulin exchange to FOS and fructose, the oligomers of these oligosaccharides are therefore the safest alternative to sauerkraut desserts that have the potential to make such superior sweeteners as their beneficial individuality, calories that are mostly used in bakery products, safety for diabetics, lack of carcinogenicity and it stimulate.[Citation51] FOS helped to absorb minerals in the colon that increase bone mineral retention. Changes in fat formation have been linked to variables in the enterocytes brush boundary membranes. Current hypothesis is that dietary lipid compounds include the effects of Mg, Ca, Cu and Fe in the intestines, as well as some other bone properties.[Citation52]
Fructose is the major component of the structure FOS in β-(2-1) bonds. FOS is widely used as dietary fiber; nutritional sciences is more interest in dietary modulation of the gastrointestinal tract by FOS. In this research work, probiotic pediococcus and acidilactici have been isolated and identified by rDNA sequencing. The results revealed that the two clones possessed significantly enhanced growth in medium, containing FOS. [Citation53] Dincer and Kivanc [Citation54] studied the potential of three known probiotic strains such as two lactobacilli and one bifidobacterium. L refers to. B-4496, Lactobacillus acidophilus NRRL-1910, Bifidobacterium bifidum ATCC 15696 and Lactobacillus planarum NRRL B-4496 are able to confuse FOS in the roots of yacon.
Extraction methods
FOS was extracted from various food sources by using the different methods as present in the . In previous research work, the FOS is extracted from the Yacun root by membrane ultra-filtration process using the membranes of 10 to 30 kDa. In this process, the juice was extracted from the sample using the different various food process. Moreover, the remaining sugar was also extracted from the pomace of juice by addition of water at high temperature with different ratios. The filtered liquid solution and juice were passed through the polyethersulfone ultra-filtration membranes at optimum conditions for extract of FOS. The results showed that the infusion instability of sample was improved by increasing the pressure from 0.5 to 2 bars. Moreover, the water permeate flux before and after the ultrafiltration process was decreased. Therefore, membranes used in this study were able to retain the FOS according to their pore size. At the end, the purity of FOS in the sample was 24% and 18.4%.[Citation60]
Table 2. Different extraction of methods of FOS
In a similar research work, Siddeshwar et al. [Citation63] was used the membrane processing technology for the extraction of prebiotics from various vegetables and fruit sources. The HPLC was used to quantification of oligosaccharides components in various samples at 98°C for 2 h. The results indicated that raffinose content was observed higher in amount in onion 0.3015% as compared to other sources. Moreover, the garlic samples were also contained higher amount of maltotetraose content (0.3075%). Maltotriose content was observed in onion 0.1148%. The results concluded that the drying methods had no effect on the quantity of various oligosaccharides compared to fresh samples.
In another research work, burdock root was an excellent source of FOS as a bioactive compound. The FOS was extracted from the prepared sample using the microwave extraction method. In this extraction process, water and ethanol was used as a solvent. Moreover, for the detection of quantity of FOS in the samples were used the HPLC with high degrees of polymerization between 3 and 9, as well as other sugar components. The results indicated that the higher efficiency of FOS was estimated in the water extract solution as compared to the ethanol extract solution. [Citation44]
Lu et al. [Citation64] conducted a research work to extract the oligosaccharides from lotus seeds using the Ultrasonic Microwave Assisted Extraction (UMAE) method. In this study, the starch was separated from the prepared sample of lotus seeds using an innovative method to exclude the influence of ultrasonic degradation for the oligosaccharides yield. The results showed that the highest yield of samples were obtained at shortened extraction time.
Cruz-Cárdenas et al. [Citation65,Citation66] was conducted to identify the water soluble carbohydrates in the food context. For this purpose, high performance anion exchange chromatography with integrated pulsed amperometric detection (HPAEC-IPAD) was used to detect the various components of oligosaccharides in the pulp of mature fruits of different accessions. This fructan fingerprint points at the presence of one or more invertases that are able to use fructose and sucrose as alternative acceptor substrates.
Wichienchot et al. [Citation67] were conducted a study to determine the sugar composition such as, fructose, glucose, and a few more oligosaccharides in red meat and white patisserie dragon fruit using the dinitrosalicylic acid (DNS) assay. The results indicated that the sugar contents of white – and red-flesh dragon fruit analyzed by HPLC consisted mostly of glucose, fructose and some oligosaccharides. Oligosaccharides content was observed in red-flesh (89.6 ± 0.76 g/kg) which was slightly higher than white-flesh (86.2 ± 0.93 g/kg) but not significantly. Moreover, agro food wastes are most amenable to produce like that, in particular, functional oligosaccharides and other less abundant oligomers containing aldohexose series of carbohydrates, aldopentose, galactose and various sugar acids. [Citation58] Mass spectrometry method was used to measure the different oligosaccharides of molecular weight at 716, 700, 490 and 474 da. The oligosaccharides mixture showed that they were resistant to synthetic human gastric juice and hydrolysis with human amyloidosis, (4.04% to 34.88%) giving maximum hydrolysis. [Citation68]
Jovanovic-Malinovska et al. [Citation29] studied low-molecular-weight carbohydrates as well as sugar alcohols with mono, di and oligosaccharides, FOS and oligosaccharides, that are firmly present in many fruits and vegetables. In general, fruits contain more monosaccharides than vegetables. Xylitol and sorbitol were found in fruits, while mannitol was found only in watermelon. In addition, vegetables contain sorbitol, xylitol, and mannitol. Group analysis was useful to classify vegetables and fruits according to their satisfaction. Most fruits contain small amounts of FOS and most content was generated from FOS nectarines. The highest amounts of FOS are found in vegetables such as scallion and white onions in the high-friction of oligosaccharides and in the raffinose family of FOS. [Citation69]
Health perspectives
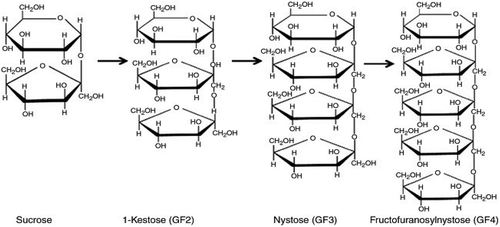
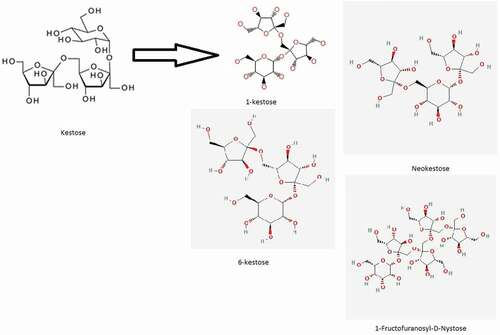
Fructans are important carbohydrates in which the glycosidic linkages are bound to fructosyl fructose links. These carbohydrates include inulin with β-(2,1)-D-fructofuranosyl units which is synthesized by fungi and present in plants. Furthermore, levan-type fructans is naturally occurring in the plants and it contain β-(6,2)-D-fructofuranosyl links which is synthesized by bacteria. Levan has a high molecular weight, which contains polymer up to 107 Da. In contradiction of levan, inulin is naturally found in the chicory root in the form of fructan polymer. The degree of polymerization (DP) varies with each prebiotic but between 2 and 60 in the fructan polymer. The native chicory contains about 10% of the fructan chain of inulin with DP of 2 to 5. FOS are formed by uniting two monosaccharides and can be joined together by a bond and merged into terminal glucosyl residue by an α-(1,2) linked maltose.[Citation70] The term FOS was primarily used for fructose oligomers that consist of one glucose unit and 2–4 fructose units bound together with by β-(2,1) glucosidic linkages[Citation71,Citation72] the FOS fructose oligomers are usually composed of 1-kestose (GF2), nystose (GF3) and 1 F-fructofuranosylnystose (GF4) as shown in the ().
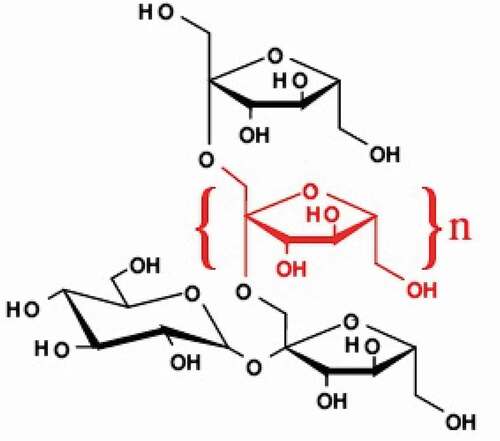
Inulin is naturally present in more than 3000 vegetables that widely distributed in many industrial plants. It is important part of the amount of food in our daily routine for many centuries, which contributed to the nutritional components and health benefits. Inulin is obtained from the natural sources including jerusalem artichoke, chicory roots, yacon, asparagus, dahlia tubers, onion, banana, leek, garlic and wheat.[Citation73] Naturally, inulin is made from sucrose. It is used in many savory snacks, cheese, breakfast cereals and bakery items as fat replacers to obtain the desired properties as it provides up to 25% to 35% more energy than digestible carbohydrates. Inulin is 10% sweeter than sucrose.[Citation74] Inulin is indigestible substance that provides nutritional benefits due to fermentation changes the bacteria and increases the absorption of calcium and magnesium. The structure of the chemical chair is inulin [GFn] which is fermentable Mono-, di-, oligo – saccharides and polyols, known as small carbohydrates as shown in , group of carbohydrates that are digested in the intestine where it draws water into large intestine to management of gastrointestinal diseases and disorders.[Citation75] Inulin stimulates the growth of microflora in the digestive system and is a suitable ingredient for the preparation of low-calorie foods to regulate blood sugar levels in human body.[Citation76]
Currently, dietary fiber is considered an important component to instruct health of humans and devoutness to foods rich in dietary fiber has been strengthened many times over because inulin has health-promoting properties. High intake of dietary fiber has been linked to a lower risk of heart disease. The initial properties of dietary fiber are impermeable for hydrolysis through secretions in digestive tract and its absorption in the small intestine while fermentation is preferable through intestinal microflora.[Citation77] inulin carbohydrate stored in plants which is composed of β-d-fructofuranosyl units linked to β-(2,1) the glycosidic linkages of inulin and is digested in the small intestine of humans due to the formation of anomeric C-2 but fermentation occurs in the colon. [Citation78] In practice, 90% of inulin is transferred to the large intestine and is digested by bacteria in the large intestine.[Citation79]
The low-calorie value is 1.50 to 6.30 kilocalorie per gram of inulin because indigestion of its components unlike monosaccharide moieties. Due to the activity of bacteria in the intestine, short chain fatty acid (SCFA) is produced by the colonic fermentation of inulin (which includes acetate, butyrate and propionate), bacterial gases and fuels.[Citation80] Simply, lactate and SCFAs enhance metabolism of energy in the host organism. According to their in-vivo and in-vitro studies, there results for inulin the increase the energy rate by 1.50 kilocalorie per gram and fructose used in the food packaging material.[Citation81] Furthermore, scientific researchers have proven that inulin energy is not more than 1 kilocalorie per gram for food labeling.[Citation82]
Crohn’s disease is a long-term condition that result causes inflammation in body along with lining in the digestive tract. Furthermore, ulcerative colitis causes inflammation or ulcer usually start in the small intestine and mainly causes inflammation in the large intestine. These diseases also known as the inflammatory bowel disease (IBD). In the Western world, 500 out of every 100,000 people are infected with IBD, according to a survey by the World Gastroenterology Organization. The inulin is used as a medicine to treat irritable bowel diseases (IBD) and colon cancer. Moreover, its ability to stimulate the growth of beneficial bacteria to reduce many factors of intestinal disorders.[Citation83] In the study of Mendis et al.[Citation84] indicate that numerous factors effect IBD but important factors such as, environmental, genetic and immunological. Probiotics and prebiotics are used for the treatment of chronic intestinal inflammation and improving the intestinal microflora.
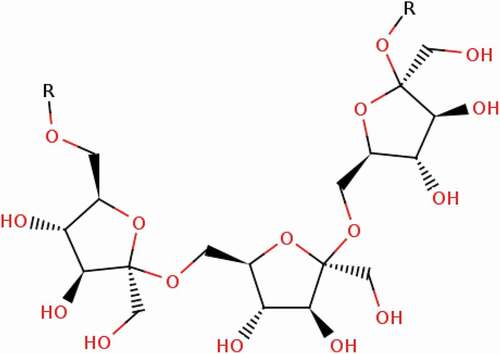
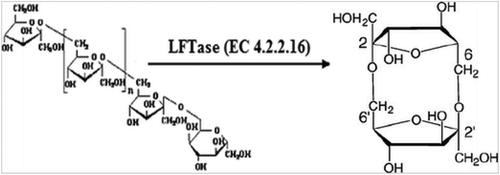
Levan is an essential component of FOS, it is naturally present in many plants in the form of polymers. It is isolated from different microbes. Its fructose is a homopolymer and its main carbon chain is connected by β-2,6 bonds as shown in the (). Its promising uses in the food and pharmaceutical industries, it acts as an anti-cancer & anti-inflammatory agent.[Citation85] Assimilation of certain oligosaccharides, polysaccharides and sugar alcohols is known to increase absorption of calcium. [Citation86] Di-fructose anhydrides (DFA) is a family of isomeric cyclic of fructose disaccharide derivatives of levan. Levan iss obtained from two fructose residues in the form of fructosyltransferase. Derivatives of DFA have been identified with levan such as β-D-fructofuranose-2′,6:2,6′-dianhydride and levan fructotransferase (LETase) as shown in the (). DFA is divided into four types, DFA I, DFA II, DFA III, and DFA IV. The chemical properties of DFA IV are different in each type due to its two fructose occupy a parallel position. Di-fructose anhydride IV (DFA IV) is obtained from the levan with LFTase. The Arthrobacter and pseudomonas were used in the production of DFA IV. DFA IV has a 45–55% sweetness of sucrose and a melting point of 177–178°C. DFA IV is not digested in intestinal enzymes so it is used in many food applications.[Citation87] However, according to Saito et al., [Citation88] rats were fed by different treatment of DFA IV with calcium to determine the mineral absorption. Mineral absorption was occurred in the gut microbes of rats. The results concluded that DFA IV can be used to prevent the osteoporosis in human.
These polysaccharides can lower LDL & HDL cholesterol levels is one of the most interesting health-related properties. Many drugs are presently advertised to lower cholesterol level in the blood and side effects occur because the body is very complex. Soluble fiber dissolves in water and works as anti-hyper lipidemic. This vegetable gum has high viscosity, which makes it tough to absorb water. Levan has both problems but, in many studies, works show that levan is a safe material for humans [Citation89] and the less viscosity streamlines preparation for making its consumption easy. It is also act as a hypo-cholesterolemic agent in the human body. In the research work of Yamamoto et al., [Citation90] high molecular weight levan was fed to rabbits. The consequences showed that it has an important effect on cholesterol level and aorta lipid deposits. Moreover, the results of the study of Zhang et al., [Citation91] show that the number of triglycerides in the adipose tissue of mice was reduced by supplementing of levan hydrolyzate. Levan also acts as anti-obesity agent and 100 mg of low viscosity levan solution is affect the one kilogram of bodyweight. Many efficacy studies indicated that the levan has no toxic effect and suggested that it is safe for human. Furthermore, similar study was conducted by Iizuka et al. [Citation92] which also indicates that levan directly effect on triglycerides level of mice.
A series of Kestoses oligosaccharides is prepared from the pyrolysis of sucrose. Kestoses are divided into three types according to their structures given below in . Neokestose, 1-kestose, and 6-kestose. Kestoses are oligomeric fructans consisting of one glucose and the main carbon chain is connected with fructose units β-(1→2) and β-(6→2) bonds of FOS. FOS linked with β-(2,6) as shown in the (), neokestose indicated that it has more bifidogenic effects than commercial FOS.[Citation93] When colorectal cancer patients ingested these compounds. They may come into contact cancer cells before the bifidobacteria can be fermented in the large intestine. According to the work of Lee et al., [Citation94] neokestos was used as a dietary anti-cancer agent (inhibiting the growth of cancer cells without affecting normal cell growth) to preventing colorectal cancer. Research work of Koga et al. [Citation95] indicated that the activity or growth of Faecalibacterium prausnitzii the kestose in the intestine professionally stimulates by kestose, thereby improving the gut modulation of gut bacteria and leading to an improvement in the symptoms of atopic dermatitis and beneficial bacteria in infants.
Restriction of an unusually low-carbohydrates, high-fat diet is called as ketogenic diet which helps to cure ketogenic diet and keep the body in normal condition called ketosis. In recent years, the intake of ketogenic-diet is a direct effect on sex drive hormones due to a decrease in carbs and an increase in ketosis. The keto diet causes a lack of magnesium in the body, which can cause flu-like symptoms in the initial stage. Carb is serving several key functions in body and brain development. Diminishing intake of carbohydrate cause the body uses ketones for nutrition instead of glucose. The ketogenic diet produces ketosis, which lowers glucose in the body. [Citation96]
In a study by Goldberg et al. [Citation97] indicated that ketogenic-diet is used as medicine in the diet of children to reduce the effect annexations. The results of neuro-protective properties showed that there was concern about the present prospective advantages of numerous brain disease such as, neurodegenerative diseases, encephalomyelitis disseminata, cancer, and insomnia. Nevertheless, ketosis is not used as a diet in medical trials to treat these diseases. Mostly, the ketogenic diets are used as weight loss medicine for obesity patients. Recent research by Bansal et al. [Citation98] show that the ketogenic diet has a strong effect on the obesity patients than conventional weight loss diets. The ketogenic diet has been shown to increase blood glucose levels in patients with diabetes in the early stages. Numerous tests show that some people have high cholesterol levels at the earlier stage of study, just to find out if cholesterol level drops after a couple weeks. Additionally, it does not take long to detect the effects of hypothyroidism in diabetes patients over a period.
The growing accuracy of facts underscores the importance of a ketogenic diet in hosting neurological diseases, diabetes, high cholesterol and obesity. Concerning neurological conditions, the ketogenic diet is recognized as an effective treatment for pharmaco-survival schizophrenia, but the new indication suggests that the ketogenic-diet may even lead to numerous scleroses, Alzheimer’s disease, Parkinson’s disease any other mitochondria-related diseases. These diseases have special molecular mechanism and properties. Numerous growth pathways can introduce the effect of the low-carb diet. Keto diet serves as an energy source, which can be lead Parkinson’s disorder due to increase in the metabolic alterations. [Citation99] Many research works have been reported the effect of keto diets. Free radicals from the keto diet reduce many physical disorders and improve the antioxidant balance, reducing your risk of ketoacidosis and endothelial dysfunction, as these diseases have specific molecular mechanisms and properties. [Citation100]
Many researchers suggested that ketogenic-diets are moderately effective in weight loss. This can make lose weight, conserve muscle mass and enhance the indication of various other diseases. [Citation101] The results also suggested that a ketogenic-diet is only improved by adjusting overall calorie intake. [Citation102] In the research by Bansal et al., [Citation98] people in keto lose 2.2 times more fat than low-calorie, low-fat diets. The amounts of triglyceride, there is also an increase in HDL lipids.
Conclusion
In current review, structure of fructo-oligosaccharides (FOS) has been comprehensively described in relation to its functional and nutraceutical properties. FOS has numerous derivatives such as Inulin, Levan, Kestoses or kesto-n-oses, and fructose. Additionally, health promoting potential of theses derivatives are also the major part of this review. FOS derivatives are beneficial for the colon health, as they stimulate the selective growth of bifidobacteria and lactobacilli and potentially inhibit microorganisms in intestinal cells, maintain the body balance of bacteria, a relief from constipation, and healthy intestinal microflora.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Prata, M. B.; Mussatto, S. I.; Rodrigues, L. R.; Teixeira, J. A. Fructooligosaccharide Production by Penicillium Expansum. Biotechnol Let. 2010, 32, 837–840.
- Cummings, J. H.; Macfarlane, G. T.; Englyst, H. N. Prebiotic Digestion and Fermentation. Ame. J Clin Nutr. 2001, 73(2), 415s–420s. DOI: https://doi.org/10.1093/ajcn/73.2.415s.
- Lorenzoni, A. S.; Aydos, L. F.; Klein, M. P.; Rodrigues, R. C.; Hertz, P. F. Fructooligosaccharides Synthesis by Highly Stable Immobilized β-fructofuranosidase from Aspergillus Aculeatus. Carb Polym. 2014, 103, 193–197. DOI: https://doi.org/10.1016/j.carbpol.2013.12.038.
- Kyu, M. T.; Dar, B.; San Aye, S.; matsuda, T. Prebiotic Oligosaccharides Prepared by Enzymatic Degradation of Dietary Fibers in Rice Grains. J Nut Sci Vitamin. 2019, 65(Supplement), S143–S147. DOI: https://doi.org/10.3177/jnsv.65.S143.
- Dumitriu, S.;, Ed. Polysaccharides: Structural Diversity and Functional Versatility. New York: CRC press: 2004, Pp. 855. ISBN. 978-0-8247-5480-8.
- Yıldız, S. The Metabolism of Fructooligosaccharides and Fructooligosaccharide-related Compounds in Plants. Food Rev Int. 2010, 27(1), 16–50. DOI: https://doi.org/10.1080/87559129.2010.518295.
- Juffrie, M. Fructooligosaccharide and Diarrhea. Biosci Microflora. 2002, 21(1), 31–34. DOI: https://doi.org/10.12938/bifidus1996.21.31.
- Pierre, F.; Perrin, P.; Champ, M.; Bornet, F.; Meflah, K.; Menanteau, J. Short-chain Fructo-oligosaccharides Reduce the Occurrence of Colon Tumors and Develop Gut-associated Lymphoid Tissue in Min Mice. Cancer Res. 1997, 57, 225–228.
- Bouhnik, Y.; Vahedi, K.; Achour, L.; Attar, A.; Salfati, J.; Pochart, P.; Rambaud, J. C. Short-chain Fructo-oligosaccharide Administration Dose-dependently Increases Fecal Bifidobacteria in Healthy Humans. J. Nutr. 1999, 129(1), 113–116. DOI: https://doi.org/10.1093/jn/129.1.113.
- Shukla, P. (Ed.). (2018). Applied Microbiology and Bioengineering: An Interdisciplinary Approach. USA: Academic Press.
- Delzenne, N. M.; Olivares, M.; Neyrinck, A. M.; Beaumont, M.; Kjølbæk, L.; Larsen, T. M.; Sanz, Y. Nutritional Interest of Dietary Fiber and Prebiotics in Obesity: Lessons from the MyNewGut Consortium. Clin. Nutr. 2020, 39(2), 414–424. DOI: https://doi.org/10.1016/j.clnu.2019.03.002.
- Anandharaj, M.; Sivasankari, B.; Rani, R. P. Corrigendum to “Effects of Probiotics, Prebiotics, and Synbiotics on Hypercholesterolemia: A Review.” Chin J Biol. 2020, 2020, 1–8. DOI: https://doi.org/10.1155/2020/8236703.
- Rayes, N.; Seehofer, D.; Neuhaus, P. Prebiotics, Probiotics, Synbiotics in Surgery—are They Only Trendy, Truly Effective or Even Dangerous? Langenbeck’s Arch. Surg. 2009, 394(3), 547–555. DOI: https://doi.org/10.1007/s00423-008-0445-9.
- Watson, R. R.; Preedy, V. R., Eds. Probiotics, Prebiotics, and Synbiotics: Bioactive Foods in Health Promotion. USA: Academic Press, 2015.
- Delzenne, N. M.; Roberfroid, M. R. Physiological Effects of Non-digestible Oligosaccharides. LWT Food Sci. Technol. 1994, 27(1), 1–6. DOI: https://doi.org/10.1006/fstl.1994.1001.
- Saeed, F.; Hussain, M.; Arshad, M. S.; Afzaal, M.; Munir, H.; Imran, M.; Tufail, T.; Anjum, F. M. Functional and Nutraceutical Properties of Maize Bran Cell Wall Non-starch Polysaccharides. Int. J. Food Prop. 2021, 24(1), 233–248. DOI: https://doi.org/10.1080/10942912.2020.1858864.
- Charalampopoulos, D.; Rastall, R. A. Eds. Prebiotics and Probiotics Science and Technology. Springer-Verlag New York: Springer Science & Business Media: Vol. 1, 2009.
- Roberfroid, M. B. Inulin-type Fructans: Functional Food Ingredients. J. Nutr. 2007, 137(11), 2493S–2502S. DOI: https://doi.org/10.1093/jn/137.11.2493S.
- Dawood, M. A.; Abo-Al-Ela, H. G.; Hasan, M. T. Modulation of Transcriptomic Profile in Aquatic Animals: Probiotics, Prebiotics and Synbiotics Scenarios. Fish Shellfish Immunol. 2020, 97, 268–282. DOI: https://doi.org/10.1016/j.fsi.2019.12.054.
- Sanz, M. L.; Gibson, G. R.; Rastall, R. A. Influence of Disaccharide Structure on Prebiotic Selectivity in Vitro. J. Agric. Food Chem. 2005, 53(13), 5192–5199. DOI: https://doi.org/10.1021/jf050276w.
- Al-Sheraji, S. H.; Ismail, A.; Manap, M. Y.; Mustafa, S.; Yusof, R. M.; Hassan, F. A. Prebiotics as Functional Foods: A Review. J. Funct. Foods. 2013, 5(4), 1542–1553. DOI: https://doi.org/10.1016/j.jff.2013.08.009.
- Vitali, B.; Ndagijimana, M.; Maccaferri, S.; Biagi, E.; Guerzoni, M. E.; Brigidi, P. An in Vitro Evaluation of the Effect of Probiotics and Prebiotics on the Metabolic Profile of Human Microbiota. Anaerobe. 2012, 18(4), 386–391. DOI: https://doi.org/10.1016/j.anaerobe.2012.04.014.
- Roberfroid, M.; Gibson, G. R.; Hoyles, L.; McCartney, A. L.; Rastall, R.; Rowland, I.; Guarner, F. Prebiotic Effects: Metabolic and Health Benefits. Br. J. Nutr. 2010, 104(S2), S1–S63. DOI: https://doi.org/10.1017/S0007114510003363.
- Manning, T. S.; Gibson, G. R. Prebiotics. Best Pract Res Clin Gastroenterol. 2004, 18(2), 287–298. DOI: https://doi.org/10.1016/j.bpg.2003.10.008.
- Crittenden, R. A.; Playne, M. Production, Properties and Applications of Food-grade Oligosaccharides. Trends Food Sci. Technol. 1996, 7(11), 353–361. DOI: https://doi.org/10.1016/S0924-2244(96)10038-8.
- Pandey, B. N.; Kulkarni, G. K. Biodiversity and Environment; SB Nangia. APH Publishing Corporation. 2006.
- BeMiller, J. N. Carbohydrate Chemistry for Food Scientists. Elsevier. 2018.
- Scott, K. P.; Grimaldi, R.; Cunningham, M.; Sarbini, S. R.; Wijeyesekera, A.; Tang, M. L.; Yang, K. Developments in Understanding and Applying Prebiotics in Research and Practice—an ISAPP Conference Paper. J. Appl. Microbiol. 2020, 128(4), 934–949. DOI: https://doi.org/10.1111/jam.14424.
- Jovanovic-Malinovska, R.; Kuzmanova, S.; Winkelhausen, E. Oligosaccharide Profile in Fruits and Vegetables as Sources of Prebiotics and Functional Foods. Int. J. Food Prop. 2014, 17(5), 949–965. DOI: https://doi.org/10.1080/10942912.2012.680221.
- Sridevi, V.; Sumathi, V.; Guru Prasad, M.; Kumar, S. M. Fructooligosaccharides-type Prebiotic: A Review. J. Pharm. Res. 2014, 8, 321–330.
- Mabrok, H.; Soliman, M.; Mohammad, M.; Husein, L. HPLC Profiles of Onion Fructooligosaccharides and Inulin and Their Prebiotic Effects on Modulating Key Markers of Colon Function, Calcium Metabolism and Bone Mass in Rat Model. J Biochem Physiol. 2018, 1(1), 10.
- Prayogi Sunu, D. S.; Mahfudz, L. D.; Yunianto, V. D. Prebiotic Activity of Garlic (Allium Sativum) Extract on Lactobacillus Acidophilus. Vet. World. 2019, 12(12), 2046. DOI: https://doi.org/10.14202/vetworld.2019.2046-2051.
- Pepe, O.; Ventorino, V.; Cavella, S.; Fagnano, M.; Brugno, R. Prebiotic Content of Bread Prepared with Flour from Immature Wheat Grain and Selected Dextran-producing Lactic Acid Bacteria. Appl. Environ. Microbiol. 2013, 79(12), 3779–3785. DOI: https://doi.org/10.1128/AEM.00502-13.
- Karppinen, S.; Myllymäki, O.; Forssell, P.; Poutanen, K. Fructan Content of Rye and Rye Products. Cereal Chem. 2003, 80(2), 168–171. DOI: https://doi.org/10.1094/CCHEM.2003.80.2.168.
- Koruri, S. S.; Banerjee, D.; Chowdhury, R.; Bhattacharya, P. Studies on Prebiotic Food Additive (Inulin) in Indian Dietary Fibre Sources-garlic (Allium Sativum), Wheat (Triticum Spp.), Oat (Avena Sativa) and Dalia (Bulgur). Int. J. Pharm. Pharm. Sci. 2014, 6, 278–282.
- Chikkerur, J.; Samanta, A. K.; Kolte, A. P.; Dhali, A.; Roy, S. Production of Short Chain Fructo-oligosaccharides from Inulin of Chicory Root Using Fungal Endoinulinase. Appl. Biochem. Biotechnol. 2020, 191(2), 695-715.
- Khuenpet, K.; Jittanit, W.; Sirisansaneeyakul, S.; Srichamnong, W. Inulin Powder Production from Jerusalem Artichoke (Helianthus Tuberosus L.) Tuber Powder and Its Application to Commercial Food Products. J. Food Process. Preserv. 2017, 41(4), e13097. DOI: https://doi.org/10.1111/jfpp.13097.
- Kim, S. W.; Hong, C. H.; Jeon, S. W.; Shin, H. J. High-yield Production of Biosugars from Gracilaria Verrucosa by Acid and Enzymatic Hydrolysis Processes. Bioresour. Technol. 2015, 196, 634–641. DOI: https://doi.org/10.1016/j.biortech.2015.08.016.
- Ganaie, M. A.; Soni, H.; Naikoo, G. A.; Oliveira, L. T. S.; Rawat, H. K.; Mehta, P. K.; Narain, N. Screening of Low-cost Agricultural Wastes to Maximize the Fructosyltransferase Production and Its Applicability in Generation of Fructooligosaccharides by Solid State Fermentation. Int. Biodeterior. Biodegrad. 2017, 118, 19–26. DOI: https://doi.org/10.1016/j.ibiod.2017.01.006.
- Lateef, A.; Gueguim-Kana, E. B. Utilization of Cassava Wastes in the Production of Fructosyltransferase by Rhizopus Stolonifer LAU 07. Romanian Biotechnol. Lett. 2012, 17, 7309–7316.
- Sangeetha, P. T.; Ramesh, M. N.; Prapulla, S. G. Production of Fructosyl Transferase by Aspergillus Oryzae CFR 202 in Solid-state Fermentation Using Agricultural By-products. Appl. Microbiol. Biotechnol. 2012, 65, 530–537.
- Sophonputtanaphoca, S.; Pridam, C.; Chinnak, J.; Nathong, M.; Juntipwong, P. Production of Non-digestible Oligosaccharides as Value-added By-products from Rice Straw. Agric. Nat. Resour. 2018, 52(2), 169–175. DOI: https://doi.org/10.1016/j.anres.2018.06.013.
- Rohin, M. A. K.; Abu Bakar, C. A.; Ali, A. M. Isolation and Characterization of Oligosaccharides Composition in Organically Grown Red Pitaya, White Pitaya and Papaya. Int. J. Pharm. Pharm. Sci. 2014, 6, 131–136.
- Li, J.; Liu, X.; Zhou, B.; Zhao, J.; Li, S. Determination of Fructooligosaccharides in Burdock Using HPLC and Microwave-assisted Extraction. J. Agric. Food Chem. 2013, 61(24), 5888–5892. DOI: https://doi.org/10.1021/jf400534n.
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S. J.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods. 2019, 8(3), 92. DOI: https://doi.org/10.3390/foods8030092.
- Chen, D.; Yang, X.; Yang, J.; Lai, G.; Yong, T.; Tang, X.; Wu, Q. Prebiotic Effect of Fructooligosaccharides from Morinda Officinalis on Alzheimer’s Disease in Rodent Models by Targeting the Microbiota-gut-brain Axis. Front Aging Neurosci. 2017, 9, 403. DOI: https://doi.org/10.3389/fnagi.2017.00403.
- Parada Venegas, D.; De la Fuente, M. K.; Landskron, G.; González, M. J.; Quera, R.; Dijkstra, G.; Hermoso, M. A. Short Chain Fatty Acids (Scfas)-mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. DOI: https://doi.org/10.3389/fimmu.2019.00277.
- Costa, G. T.; Guimarães, S. B.; Sampaio, H. A. D. C. Fructo-oligosaccharide Effects on Blood Glucose: An Overview. Acta Cirúrgica Brasileira. 2012, 27(3), 279–282. DOI: https://doi.org/10.1590/S0102-86502012000300013.
- Park, J. P.; Yun, J. W. Utilization of Chicory Roots for Microbial Endoinulinase Production. Lett Appl. Microbiol. 2001, 33(3), 183–187. DOI: https://doi.org/10.1046/j.1472-765x.2001.00977.x.
- Martins, G. N.; Ureta, M. M.; Tymczyszyn, E. E.; Castilho, P.; Gomez-Zavaglia, A. Technological Aspects of the Production of Fructo and Galacto-oligosaccharides. Enzymatic Synthesis and Hydrolysis. Front. Nutrit. 2019, 6, 78. DOI: https://doi.org/10.3389/fnut.2019.00078.
- Rastall, R.; Moreno, F. J.; Hernandez-Hernandez, O. Dietary Carbohydrate Digestibility and Metabolic Effects in Human Health. Front. Nutrit. 2019, 6, 164. DOI: https://doi.org/10.3389/fnut.2019.00164.
- Whisner, C. M.; Castillo, L. F. Prebiotics, Bone and Mineral Metabolism. Calcif. Tissue Int. 2018, 102(4), 443–479. DOI: https://doi.org/10.1007/s00223-017-0339-3.
- Petrov, K.; Petrova, P. Sugar Transport Systems Involved in Fructooligosaccharides Utilization by the Probiotic Bacterium Pediococcus Acidilactici. Comptes rendus de l’Académie bulgare des Sciences. 2017, 70(9), 1263-1270.
- Dincer, E.; Kivanc, M. Characterization of Lactobacillus Plantarum Strains Isolated from Turkish Pastırma and Possibility to Use of Food Industry. Food Sci. Technol. 2020, 40(2), 498–507. DOI: https://doi.org/10.1590/fst.05819.
- Manosroi, J.; Khositsuntiwong, N.; Manosroi, A. Biological Activities of Fructooligosaccharide (Fos)-containing Coix Lachryma-jobi Linn. Extract. J. Food Sci. Technol. 2014, 51(2), 341–346. DOI: https://doi.org/10.1007/s13197-011-0498-6.
- Wang, J.; Sporns, P.; Low, N. H. Analysis of Food Oligosaccharides Using MALDI-MS: Quantification of Fructooligosaccharides. J. Agric. Food Chem. 1999, 47(4), 1549–1557. DOI: https://doi.org/10.1021/jf9809380.
- Machado, M. T.; Eca, K. S.; Vieira, G. S.; Menegalli, F. C.; Martínez, J.; Hubinger, M. D. Prebiotic Oligosaccharides from Artichoke Industrial Waste: Evaluation of Different Extraction Methods. Ind. Crops Prod. 2015, 76, 141–148. DOI: https://doi.org/10.1016/j.indcrop.2015.06.047.
- Cano, M. E.; García-Martin, A.; Comendador Morales, P.; Wojtusik, M.; Santos, V. E.; Kovensky, J.; Ladero, M. Production of Oligosaccharides from Agrofood Wastes. Fermentation. 2020, 6(1), 31. DOI: https://doi.org/10.3390/fermentation6010031.
- Zeaiter, Z.; Regonesi, M. E.; Cavini, S.; Labra, M.; Sello, G.; Di Gennaro, P. Extraction and Characterization of Inulin-type Fructans from Artichoke Wastes and Their Effect on the Growth of Intestinal Bacteria Associated with Health. Biomed Res. Int. 2019, 2019, 1–8. DOI: https://doi.org/10.1155/2019/1083952.
- Brites, M. L.; Noreña, C. P. Z. Obtaining Fructooligosaccharides from Yacon (Smallanthus Sonchifolius) by an Ultrafiltration Process. Braz. J. Chem. Eng. 2016, 33(4), 1011–1020. DOI: https://doi.org/10.1590/0104-6632.20160334s20140010.
- Zhang, C.; Liu, Y. Sugaring-out-assisted Aqueous Two-phase Extraction of Fructooligosaccharides from Yacon (Smallanthus Sonchifolius). Sep. Sci. Technol. 2017, 52, 1531–1536.
- De Oliveira, A. J. B.; Gonçalves, R. A. C.; Chierrito, T. P. C.; dos Santos, M. M.; de Souza, L. M.; Gorin, P. A. J.; Iacomini, M. Structure and Degree of Polymerisation of Fructooligosaccharides Present in Roots and Leaves of Stevia Rebaudiana (Bert.) Bertoni. Food Chem. 2011, 129(2), 305–311. DOI: https://doi.org/10.1016/j.foodchem.2011.04.057.
- Siddeshwar, S.; Reddy, K. K.; Rao, T. M.; Arun, K.; Reddy, P. V. M. Screening and Estimation of Pre-biotic Oligosaccharides in Fruits and Vegetables. Curr. Trends Biotechnol. Pharm. 2008, 2, 183–191.
- Lu, X.; Zheng, Z.; Li, H.; Cao, R.; Zheng, Y.; Yu, H.; Xiao, J.; Miao, S.; Zheng, B. Optimization of Ultrasonic-microwave Assisted Extraction of Oligosaccharides from Lotus (Nelumbo Nucifera Gaertn.) Seeds. Ind. Crops Prod. 2017, 107, 546–557. DOI: https://doi.org/10.1016/j.indcrop.2017.05.060.
- Cruz-Cárdenas, C. I.; Miranda-Ham, M. L.; Castro-Concha, L. A.; Ku-Cauich, J. R.; Vergauwen, R.; Reijnders, T.; Escobedo-graciamedrano, R. M. Fructans and Other Water-soluble Carbohydrates in Vegetative Organs and Fruits of Different Musa Spp. Accessions. Front. Plant Sci. 2015, 6, 395. DOI: https://doi.org/10.3389/fpls.2015.00395.
- Rohrer, J. Analysis of carbohydrates by high-performance anion-exchange chromatography with pulsed amperometric detection (hpae-pad). Thermo Fisher Scientific, Technical Note, 2012. 20(1), 1–12.
- Wichienchot, S.; Jatupornpipat, M.; Rastall, R. A. Oligosaccharides of Pitaya (Dragon Fruit) Flesh and Their Prebiotic Properties. Food Chem. 2010, 120(3), 850–857. DOI: https://doi.org/10.1016/j.foodchem.2009.11.026.
- Kailemia, M. J.; Ruhaak, L. R.; Lebrilla, C. B.; Amster, I. J. Oligosaccharide Analysis by Mass Spectrometry: A Review of Recent Developments. Anal. Chem. 2014, 86(1), 196–212. DOI: https://doi.org/10.1021/ac403969n.
- Lenhart, A.; Chey, W. D. A Systematic Review of the Effects of Polyols on Gastrointestinal Health and Irritable Bowel Syndrome. Adv. Nutr. 2017, 8, 587–596.
- Barreteau, H.; Delattre, C.; Michaud, P. Production of Oligosaccharides as Promising New Food Additive Generation. Food Technol Biotechnol. 2006, 44(3), 323–333.
- Madlová, A.; Antošová, M.; Baráthová, M.; Polakovič, M.; Štefuca, V.; Báleš, V. Biotransformation of Sucrose to Fructooligosaccharides: The Choice of Microorganisms and Optimization of Process Conditions. In Progress in Biotechnology, Edited by Stanislaw Bielecki, Johannes Tramper, Jacek Polak. Poland: Elsevier: 2000; Vol. 17, pp 151–155.
- Silva, M. F.; Rigo, D.; Mossi, V.; Golunski, S.; de Oliveira Kuhn, G.; Di Luccio, M.; Treichel, H. Enzymatic Synthesis of Fructooligosaccharides by Inulinases from Aspergillus Niger and Kluyveromyces Marxianus NRRL Y-7571 in Aqueous–organic Medium. Food Chem. 2013, 138(1), 148–153. DOI: https://doi.org/10.1016/j.foodchem.2012.09.118.
- Awika, J. M.; Rose, D. J.; Simsek, S. Complementary Effects of Cereal and Pulse Polyphenols and Dietary Fiber on Chronic Inflammation and Gut Health. Food Funct. 2018, 9(3), 1389–1409. DOI: https://doi.org/10.1039/C7FO02011B.
- Cooper, P. D.; Rajapaksha, K. H.; Barclay, T. G.; Ginic-Markovic, M.; Gerson, A. R.; Petrovsky, N. Inulin Crystal Initiation via a Glucose-fructose Cross-link of Adjacent Polymer Chains: Atomic Force Microscopy and Static Molecular modelling. Carbohydr. Polym. 2015, 117, 964–972. DOI: https://doi.org/10.1016/j.carbpol.2014.10.022.
- Giarnetti, M.; Paradiso, V. M.; Caponio, F.; Summo, C.; Pasqualone, A. Fatreplacement in Shortbread Cookies Using an Emulsion Filled Gel Based on Inulinand Extra Virgin Olive Oil. LWT Food Sci. Technol. 2015, 63(1), 339–345. DOI: https://doi.org/10.1016/j.lwt.2015.03.063.
- Wichienchot, S.; Thammarutwasik, P.; Jongjareonrak, A.; Chansuwan, W.; Hmadhlu, P.; Hongpattarakere, T.; Ooraikul, B. Extraction and Analysis of Prebiotics from Selected Plants from Southern Thailand. Songklanakarin J. Sci. Technol. 2015, (2015, 33.
- Turner, N. D.; Lupton, J. R. Dietary Fiber. Advances in Nutrition. Int Rev J. 2011, 2, 151–152.
- Navarro, D. M.; Abelilla, J. J.; Stein, H. H. Structures and Characteristics of Carbohydrates in Diets Fed to Pigs: A Review. J Anim Sci Biotechnol. 2019, 10(1), 1–17. DOI: https://doi.org/10.1186/s40104-019-0345-6.
- Holscher, H. D. Dietary Fiber and Prebiotics and the Gastrointestinal Microbiota. Gut Microbes. 2017, 8(2), 172–184. DOI: https://doi.org/10.1080/19490976.2017.1290756.
- Baxter, N. T.; Schmidt, A. W.; Venkataraman, A.; Kim, K. S.; Waldron, C.; Schmidt, T. M. Dynamics of Human Gut Microbiota and Short-chain Fatty Acids in Response to Dietary Interventions with Three Fermentable Fibers. MBio. 2019, 10(1). DOI: https://doi.org/10.1128/mBio.02566-18.
- Roberfroid, M. B. Concepts in Functional Foods: The Case of Inulin And oligofructose. J. Nutr. 1999, 129(7), 1398S–1401S. DOI: https://doi.org/10.1093/jn/129.7.1398S.
- Flamm, G.; Glinsmann, W.; Kritchevsky, D.; Prosky, L.; Roberfroid, M. Inulin and Oligofructose as Dietary Fiber: A Review of the Evidence. Crit Rev Food Sci Nutr. 2001, 41, 353–362.
- Sartor, R. B. Therapeutic Manipulation of the Enteric Microflora in Inflammatory Bowel Diseases: Antibiotics, Probiotics, and Prebiotics. Gastroenterology. 2004, 126(6), 1620–1633. DOI: https://doi.org/10.1053/j.gastro.2004.03.024.
- Mendis, M.; Leclerc, E.; Simsek, S. Arabinoxylans, Gut Microbiota And immunity. Carbohydr. Polym. 2016, 139, 159–166. DOI: https://doi.org/10.1016/j.carbpol.2015.11.068.
- Phillips, G. O., & Williams, P. A. (Eds.). Handbook of hydrocolloids (pp. 53-64). 2000, Boca Raton, FL: CRC ress.
- Mineo, H.; Hara, H.; Kikuchi, H.; Sakurai, H.; Tomita, F. Various Indigestible Saccharides Enhance Net Calcium Transport from the Epithelium of the Small and Large Intestine of Rats in Vitro. J. Nutr. 2001, 131(12), 3243–3246. DOI: https://doi.org/10.1093/jn/131.12.3243.
- Ko, H.; Bae, J. H.; Sung, B. H.; Kim, M. J.; Park, S. H.; Sohn, J. H. Direct Production of Difructose Anhydride iV from Sucrose by Co-fermentation of Recombinant Yeasts. Sci. Rep. 2019, 9(1), 1–9. DOI: https://doi.org/10.1038/s41598-019-52373-5.
- Saito, K.; Hira, T.; Suzuki, T.; Hara, H.; Yokota, A.; Tomita, F. Effects of DFA IV in Rats: Calcium Absorption and Metabolism of DFA IV by Intestinal Microorganisms. Biosci., Biotechnol., Biochem. 1999, 63(4), 655–661. DOI: https://doi.org/10.1271/bbb.63.655.
- Calazans, G. M. T.; Lopes, C. E.; Lima, R. M. O. C.; De Franc, F. P. Antitumour Activities of Levans Produced by Zymomonas Mobilis Strains. Biotechnol. Lett. 1997, 19(1), 19–21. DOI: https://doi.org/10.1023/A:1018350617120.
- Yamamoto, K.; Isogai, Y.; Sato, H.; Taketomi, Y.; Murakami, M. Secreted Phospholipase A 2, Lipoprotein Hydrolysis, and Atherosclerosis: Integration with Lipidomics. Anal. Bioanal. Chem. 2011, 400(7), 1829. DOI: https://doi.org/10.1007/s00216-011-4864-z.
- Zhang, T.; Chen, J.; Tang, X.; Luo, Q.; Xu, D.; Yu, B. Interaction between Adipocytes and High-density Lipoprotein: New Insights into the Mechanism of Obesity-induced Dyslipidemia and Atherosclerosis. Lipids Health Dis. 2019, 18(1), 223. DOI: https://doi.org/10.1186/s12944-019-1170-9.
- Iizuka, M.; minamiura, N.; ogura, T. Utilization of Fructan. In Glycoenzymes; Ohnishi, M., Ed.; Japan Scientific Societies Press: Tokio, 2000; pp 241–258.
- Van den Ende, W. Novel Fructan Exohydrolase: Unique Properties and Applications for Human Health. J. Exp. Bot. 2018, (2018(69), 4227–4231. DOI: https://doi.org/10.1093/jxb/ery268.
- Lee, S. M.; Chang, J. Y.; Wu, J. S.; Sheu, D. C. Antineoplastic Effect of a Novel Chemopreventive Agent, Neokestose, on the Caco-2 Cell Line via Inhibition of Expression of Nuclear factor-κB and Cyclooxygenase-2. Mol. Med. Rep. 2015, 12(1), 1114–1118. DOI: https://doi.org/10.3892/mmr.2015.3507.
- Koga, Y.; Tokunaga, S.; Nagano, J.; Sato, F.; Konishi, K.; Tochio, T.; Kubo, C. Age-associated Effect of Kestose on Faecalibacterium Prausnitzii and Symptoms in the Atopic Dermatitis Infants. Pediatr. Res. 2016, 80(6), 844–851. DOI: https://doi.org/10.1038/pr.2016.167.
- Krishnasamy, S.; Abell, T. L. Diabetic Gastroparesis: Principles and Current Trends in Management. Diabetes Ther. 2018, 9(S1), 1–42. DOI: https://doi.org/10.1007/s13300-018-0454-9.
- Goldberg, E. L.; Shchukina, I.; Asher, J. L.; Sidorov, S.; Artyomov, M. N.; Dixit, V. D. Ketogenesis Activates Metabolically Protective γδ T Cells in Visceral Adipose Tissue. Nat Metab. 2020, 2018(2), 50–61. DOI: https://doi.org/10.1038/s42255-019-0160-6.
- Bansal, A.; Rashid, C.; Xin, F.; Li, C.; Polyak, E.; Duemler, A.; van der Meer, T.; Stefaniak, M.; Wajid, S.; Doliba, N.; Bartolomei, M. S. Sex- and Dose-Specific Effects of Maternal Bisphenol A Exposure on Pancreatic Islets of First- and Second-Generation Adult Mice Offspring. Environ. Health Perspect. 2017, 125(9), 097022. DOI:https://doi.org/10.1289/EHP1674.
- Muddapu, V. R.; Dharshini, S. A. P.; Chakravarthy, V. S.; Gromiha, M. M. Neurodegenerative Diseases–Is Metabolic Deficiency the Root Cause? Front Neurosci. 2020, 14. DOI: https://doi.org/10.3389/fnins.2020.00213.
- Jeszka-Skowron, M.; Oszust, K.; Zgoła-Grześkowiak, A.; Quality, F. M. Assessment of Goji Fruits, Cranberries, and Raisins Using Selected Markers. Eur. Food Res. Technol. 2018, 244(12), 2159–2168. DOI: https://doi.org/10.1007/s00217-018-3125-1.
- Innosa, D.; Ianni, A.; Palazzo, F.; Martino, F.; Bennato, F.; Grotta, L.; Martino, G. High Temperature and Heating Effect on the Oxidative Stability of Dietary Cholesterol in Different Real Food Systems Arising from Eggs. Eur. Food Res. Technol. 2019, 245(7), 1533–1538. DOI: https://doi.org/10.1007/s00217-019-03266-4.
- Harder, A. M.; Ardren, W. R.; Evans, A. N.; Futia, M. H.; Kraft, C. E.; Marsden, J. E.; Richter, C. A.; Rinchard, J.; Tillitt, D. E.; Christie, M. R. Thiamine Deficiency in Fishes: Causes, Consequences and Potential Solutions. Rev. Fish Biol. Fisheri. 2018, 28(4), 865–886. DOI:https://doi.org/10.1007/s11160-018-9538-x.