ABSTRACT
Mumps outbreaks among previously vaccinated young adults raise concerns regarding waning vaccine immunity. This study identified, described and assessed the changing incidence of mumps cases following mumps-containing vaccination (MMR/MMRV) in a non-mumps outbreak setting. Potential cases between 1996 and 2018 were identified by the international classification of disease codes or by mumps laboratory test orders among Kaiser Permanente Northern California members. Medical charts were reviewed to confirm diagnoses, timing relative to vaccination and clinical characteristics. Among 474 potential cases, 257 (54.2%) were confirmed after chart review. A third of the cases were <10 years old at diagnosis and 48% were over 25 years. Most cases (92.2%) had parotitis and 5% of males had orchitis. Mumps rates decreased from 8.5 to 1.8/1,000,000 person-years as time since the second MMR/MMRV dose increased from <2 years to ≥10 years. Similarly, rates decreased from 16.3 to 3/1,000,000 person-years after at least 1 dose of MMR/MMRV. Mumps rates were higher among children aged ≤10 years compared with older age groups. In conclusion, in the context of a non-outbreak setting, this study suggests that waning of vaccine immunity to mumps appeared to have minimal clinical impact.
Introduction
Mumps is a contagious vaccine-preventable viral disease, usually spread through saliva or mucus from the mouth, nose, or throat. The most common clinical presentation in symptomatic cases is parotitis. Other nonspecific symptoms include fever, headache, myalgia, and malaise. Prior to the widespread use of mumps-containing vaccines, the infection can be asymptomatic in about 30% of those who become infectedCitation1. Generally, the disease is mild, but serious complications such as orchitis, encephalitis, and deafness can occur.
In the pre-vaccine era, there were approximately 186,000 cases reported each year in the US.Citation2,Citation3 Following the institution of routine mumps vaccination in 1967, mumps cases decreased by 99% in the US, and annual mumps cases continued to decline following the expansion of the measles mumps and rubella (MMR) vaccine program in 1989.Citation4 As part of the childhood immunization schedule, currently, the Advisory Committee on Immunization Practice (ACIP) in the US. recommends one dose of MMR or measles mumps, rubella and varicella vaccine (MMRV) at ages 12–15 months and a second dose at ages 4–6 years.
Despite high coverage of the recommended two doses of MMR/MMRV vaccine (~94% among US. children entering kindergarten),Citation5 the US, and other developed countries have seen mumps outbreaks in recent years.Citation2,Citation6-14 Recent mumps outbreaks have seen a shift in the burden of disease away from traditional unvaccinated young children toward vaccinated adolescents and young adults aged 18–24 years on college campuses or in settings where people reside in close contact.Citation15-17 As a result, the ACIP, during its October 2017 meeting, recommended a third dose of a mumps virus-containing vaccine for persons previously vaccinated with 2 doses who are identified by public health authorities as being part of a group or population at increased risk for acquiring mumps because of an outbreak.Citation18
The shift in the epidemiology of mumps during recent outbreaks has raised concerns regarding the long-term effectiveness of the MMR/MMRV vaccine to protect against mumps. Studies conducted during outbreaks among college students or people living in close contact suggest that waning of the mumps component of the MMR/MMRV vaccine may have contributed to the outbreaks because most cases were fully vaccinated several years prior.Citation7,Citation8,Citation10,Citation15,Citation17,Citation19-22 Some previous studies estimated that the odds of mumps increased by 10%36% for every year that had passed since the second dose of MMR.Citation20,Citation22 Other studies suggested a significant risk increase 10 years after receiving a measles-containing vaccine.Citation8,Citation21
There are limited data on the waning of mumps vaccine immunity in a non-outbreak setting.Citation23,Citation24 It is not clear whether rates of mumps increase in a non-outbreak setting as the time since vaccination increases. The objective of this study was to identify and describe mumps cases in a non-outbreak setting, and to assess the changing incidence of mumps over 20 years following MMR/MMRV vaccination.
Materials and methods
Study setting, population, and design
The study setting was Kaiser Permanente Northern California (KPNC), an integrated healthcare delivery organization that provides comprehensive care to approximately 4 million members. Members receive almost all medical care at KPNC-owned facilities, including clinics, hospitals, pharmacies, and laboratories. Members receive all their routine vaccinations free of charge. KPNC members are similar to the broad catchment population in Northern California in terms of sociodemographic characteristics, except the extremes of income distribution are underrepresented.Citation25
We conducted a retrospective cohort study among KPNC members to identify and describe mumps cases diagnosed between January 1, 1996 and December 31, 2018. Potential mumps cases were identified by International Classification of Disease (ICD) 9th edition code 072, ICD 10th edition code B26 or by laboratory test orders for mumps (polymerase chain reaction [PCR] or serology). We reviewed the electronic and paper medical charts of all potential cases to confirm diagnoses, minimize or avoid false-positive diagnoses and to determine the onset date of mumps and vaccination date. Chart reviews were conducted by trained medical chart abstractors using a standard form, followed by reviews from three members of the research team, including a physician researcher, to determine final case status. Final determination was made regardless of whether the patient had a laboratory confirmation. We also collected clinical characteristics on all confirmed cases. We required that cases be KPNC members in the year the diagnosis was made.
Statistical analysis
In the context of a non-outbreak, the number of mumps cases was low and did not allow for direct estimation of waning vaccine effectiveness. Instead, we calculated mumps incidence rates in relation to time since the last MMR/MMRV vaccination for at least one and for two doses as an indication of whether mumps vaccine immunity wanes over time. Time since last MMR/MMRV vaccine dose was categorized as <2 years, 2 to <5 years, 5 to <10 years, and ≥10 years. The numerator of the rate for each time category was the number of mumps cases and the denominator (person-years) was the sum of all KPNC members as of mid-June of each year across that specific time period. An increase in rates as the time from vaccination increased would suggest waning of vaccine immunity. We also described the sociodemographic and clinical characteristics of confirmed cases.
The study was approved by the KPNC Institutional Review Board with a waiver of written informed consent because the study had no direct contact with study participants.
Results
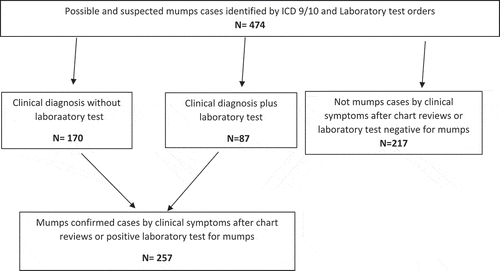
We identified 474 potential mumps cases between 1996 and 2018. After the medical chart review, 257 (54.2%) cases were confirmed (). Of these, 170 (66%) were confirmed clinically without a laboratory test and 87 (34%) were confirmed by clinical symptoms and a positive laboratory test. Among the 87 cases with laboratory results, 66 (75.9%) were identified after a serology test and 21 (24.1%) after a PCR test. Mumps cases were 42.8% female and mean age was 26.5 years. About a third of the cases were <10 years old, 48% were over 25 years old and few were between the ages of 14–24 years (). Clinically, most cases (92.2%) had parotitis and 5.4% of males had orchitis (). There were no cases of mumps encephalitis or meningitis.
Table 1. Socio-demographic characteristics of mumps cases. Kaiser Permanente Northern California, 1996–2018
Table 2. Clinical characteristics of mumps cases. Kaiser Permanente Northern California, 1996–2018
MMR/MMRV vaccination status was not available for half the cases. Of those with vaccination information, 28 cases (10.9%) received only 1 dose, 99 (38.5) received 2 doses and 3 (1.2%) received 3 doses. Vaccination dates were available for 115 (44.7%) mumps cases with at least 1 dose (). Most mumps cases were diagnosed within 5 years of receiving either one or two doses of MMR/MMRV (). Among people with vaccine information, regardless of whether one or two of doses of MMR/MMRV were administered, mumps rates decreased from 16.3 to 3/1,000,000 person-years as time since vaccination increased from <2 years to ≥10 years (). Similarly, after the 2nd dose, mumps rates declined from 8.5 to 1.8/1,000,000 person-years as time since vaccination increased from <2 years to ≥10 years. Mumps rates varied by age and by time since vaccination. Those aged <10 years and 15–19 years had higher rates of mumps shortly after vaccination (<2 years) than they did following longer times since vaccination (2- <5 years or 5 – <10 years) (Supplemental Table).
Table 3. Mumps rates by time since last measles, mumps and rubella/measle, mumps rubella, and varicella vaccine. Kaiser Permanente Northern California, 1996–2018
Discussion
In the context of non-outbreak settings, within a large geographically diverse population of all ages and living situations receiving routine health care mumps was rarely diagnosed. Despite KPNC’s current annual membership of >4 million people, our study identified only 474 potential cases over a 22-year study period. Further, mumps rates were high within 2 years or less after a dose of MMR/MMRV vaccine and decreased as the time from vaccine increased. This finding is in contrast to previous studies conducted in the context of mumps outbreaksCitation8,Citation20,Citation22 showing an increased risk of mumps as the time from last vaccine dose increased. If vaccine immunity waned substantially over time, we would expect to see an increase in rates of mumps as the time from vaccination increased rather than seeing a decrease in rates.
Our results are similar to those of some recent studies that did not find strong evidence of waning in non-outbreak settings. A prospective study that analyzed blood samples from 98 subjects 7 years and 17 years after the second dose of mumps-containing vaccine found no statistical difference in total mumps virus-specific IgG levels between the two time points.Citation26 Similarly, Date et al. also reported a non-significant lower geometric mean of mumps virus-neutralizing antibodies >16 years after vaccination compared to 1–5 years after vaccination.Citation27 Although immune correlates of protection against mumps have not been defined, virus-neutralizing antibody may be a plausible marker. Non-significant differences in antibodies levels as time since vaccine increases may suggest that antibody titers do not significantly wane.
Studies have reported that time since vaccination is a risk factor for mumps acquisition,Citation15,Citation19-22,Citation28 however this may not be the only factor. Previous mumps outbreaks occurred in closed-contact or crowded settings without the substantial transmission in the greater community. Thus, prolonged direct contact with the mumps virus in a vaccinated population with a vaccine that has less than 100% efficacy might overwhelm the protection provided by the vaccine, leading to a possible outbreak.Citation29 While some studiesCitation30,Citation31 have proposed immune escape by circulating mumps viruses as a contributing factor for current outbreaks, other studies did not find evidence that this plays a strong role in the occurrence of mumps outbreaks.Citation32-34 More studies are needed to determine the root cause of mumps outbreaks.Citation35
Consistent with previous epidemiological studies that have reported a high burden of disease among young adults,Citation4,Citation36 our study similarly found that a high proportion of the cases were <10 years of age or ≥25 years of age. Our observation that there was a high rate of mumps among the younger age group within 2 years following vaccination may be related to vaccine failure rather than waning. However, follow-up studies with immunological data would be necessary to confirm.
To the best of our knowledge, this is the first study to identify and describe mumps cases and determine rates in a non-outbreak setting. The study was strengthened by the long follow-up time and the availability of medical histories which allowed for a thorough review to confirm diagnosis and timing of diagnosis in relation to vaccination. Furthermore, the study included people from all ages and race/ethnicity living in different settings across northern California. Finally, we chart reviewed every potential case, which was a necessity since chart review confirmed only 54% of the potential cases. This finding highlights that epidemiological studies of mumps cannot rely solely on ICD codes to accurately identify and confirm mumps cases. Future studies should include a chart review of all potential cases identified through ICD codes to minimize misclassification of cases.
Our study was limited by missing vaccination dates for many of the mumps cases, which led to their total exclusion from our rate calculations. Since nearly half our cases were >25 years of age and MMR is recommended for ages 1 and 4–6 years, this was not unexpected. It is unlikely that these exclusions significantly impacted mumps rates because we only included people for whom we had vaccine information. The proportion of cases between 18 and 24 years of age was small in our study possibly because this age group might receive their care at college health-care centers and may not appear in our system if they are diagnosed with mumps at school. The small number of cases with complete vaccine data did not allow us to directly assess for waning using regression analyses. Our findings that rates of mumps did not increase with time since vaccination assumed that exposure to mumps viruses did not vary significantly by age group. We were not able to verify this assumption in the present study. Finally, we may have missed some cases because we only included medically attended cases. Subclinical and mild cases that did not seek care were misclassified as non-cases, which may have attenuated our rate estimates.
Conclusion
This study found that within a health-care setting providing routine clinical care over a 22-year time period, mumps was rarely diagnosed. In this study, we did not see evidence of increased rates of mumps as the time from vaccination increased. In the context of a non-outbreak setting, this study suggests that waning of vaccine immunity to mumps appeared to have minimal clinical impact.
Disclosure of potential conflicts of interest
Dr Klein has received research grant support from Merck, GlaxoSmithKline, Sanofi Pasteur, MedImmune, Pfizer, Protein Sciences (now Sanofi Pasteur), and Dynavax for unrelated studies. All other co-authors do not have a commercial or other association that might pose a conflict of interest (e.g., pharmaceutical stock ownership, consultancy, advisory board membership, relevant patents, or research funding).
Supplemental Material
Download MS Word (26 KB)Supplementary material
Supplemental data for this article can be accessed online at https://doi.org/10.1080/21645515.2020.1756153.
Additional information
Funding
References
- Rota JS, Rosen JB, Doll MK, McNall RJ, McGrew M, Williams N, Lopareva EN, Barskey AE, Punsalang A, Rota PA, et al. Comparison of the sensitivity of laboratory diagnostic methods from a well-characterized outbreak of mumps in New York city in 2009. Clin Vaccine Immunol. 2013;20(3):391–96. doi:https://doi.org/10.1128/CVI.00660-12.
- CDC. Mumps cases and outbreaks. 2019 [ Updated 2017 Sep 17, accessed 2019 Sep 23. https://www.cdc.gov/mumps/outbreaks.html
- McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS; Centers for Disease C, Prevention. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: summary recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2013;62(RR–04):1–34.
- Dayan GH, Quinlisk MP, Parker AA, Barskey AE, Harris ML, Schwartz JMH, Hunt K, Finley CG, Leschinsky DP, O’Keefe AL. Recent resurgence of mumps in the United States. N Engl J Med. 2008;358(15):1580–89. doi:https://doi.org/10.1056/NEJMoa0706589.
- Seither R, Calhoun K, Street EJ, Mellerson J, Knighton CL, Tippins A, Underwood JM. Vaccination coverage for selected vaccines, exemption rates, and provisional enrollment among children in Kindergarten - United States, 201617 school year. MMWR Morb Mortal Wkly Rep. 2017;66(40):1073–80. doi:https://doi.org/10.15585/mmwr.mm6640a3.
- Barskey AE, Glasser JW, LeBaron CW. Mumps resurgences in the United States: A historical perspective on unexpected elements. Vaccine. 2009;27(44):6186–95. doi:https://doi.org/10.1016/j.vaccine.2009.06.109.
- Barskey AE, Schulte C, Rosen JB, Handschur EF, Rausch-Phung E, Doll MK, Cummings KP, Alleyne EO, High P, Lawler J, et al. Mumps outbreak in Orthodox Jewish communities in the United States. N Engl J Med. 2012;367(18):1704–13. doi:https://doi.org/10.1056/NEJMoa1202865.
- Cardemil CV, Dahl RM, James L, Wannemuehler K, Gary HE, Shah M, Marin M, Riley J, Feikin DR, Patel M, et al. Effectiveness of a third dose of MMR vaccine for mumps outbreak control. N Engl J Med. 2017;377(10):947–56. doi:https://doi.org/10.1056/NEJMoa1703309.
- Yung CF, Andrews N, Bukasa A, Brown KE, Ramsay M. Mumps complications and effects of mumps vaccination, England and Wales, 2002-2006. Emerg Infect Dis. 2011;17(4):661–67. quiz 766. doi:https://doi.org/10.3201/eid1704.101461.
- Cohen C, White JM, Savage EJ, Glynn JR, Choi Y, Andrews N, Brown D, Ramsay ME. Vaccine effectiveness estimates, 2004-2005 mumps outbreak, England. Emerg Infect Dis. 2007;13(1):12–17. doi:https://doi.org/10.3201/eid1301.060649.
- Sane J, Gouma S, Koopmans M, de Melker H, Swaan C, van Binnendijk R, Hahné S. Epidemic of mumps among vaccinated persons, The Netherlands, 2009-2012. Emerg Infect Dis. 2014;20(4):643–48. doi:https://doi.org/10.3201/eid2004.131681.
- Eriksen J, Davidkin I, Kafatos G, ANDREWS N, BARBARA C, COHEN D, DUKS A, GRISKEVICIUS A, JOHANSEN K, BARTHA K, et al. Seroepidemiology of mumps in Europe (1996-2008): why do outbreaks occur in highly vaccinated populations? Epidemiol Infect. 2013;141(3):651–66. doi:https://doi.org/10.1017/S0950268812001136.
- Anis E, Grotto I, Moerman L, Warshavsky B, Slater PE, Lev B. Mumps outbreak in Israel’s highly vaccinated society: are two doses enough? Epidemiol Infect. 2012;140(3):439–46. doi:https://doi.org/10.1017/S095026881100063X.
- Kuehn B. Mumps in migrant detention centers. JAMA. 2019;322:1344.
- Centers for Disease C, Prevention. Mumps outbreak on a university campus–California, 2011. MMWR Morb Mortal Wkly Rep. 2012;61(48):986–89.
- Albertson JP, Clegg WJ, Reid HD, Arbise BS, Pryde J, Vaid A, Thompson-Brown R, Echols F. Mumps Outbreak at a University and Recommendation for a Third Dose of Measles-Mumps-Rubella Vaccine - Illinois, 2015-2016. MMWR Morb Mortal Wkly Rep. 2016;65(29):731–34. doi:https://doi.org/10.15585/mmwr.mm6529a2.
- Centers for Disease C, Prevention. Update: mumps outbreak - New York and New Jersey, June 2009-January 2010. MMWR Morb Mortal Wkly Rep. 2010;59(5):125–29.
- Marin M, Marlow M, Moore KL, Patel M. Recommendation of the advisory committee on immunization practices for use of a third dose of mumps virus-containing vaccine in persons at increased risk for mumps during an outbreak. MMWR Morb Mortal Wkly Rep. 2018;67(1):33–38. doi:https://doi.org/10.15585/mmwr.mm6701a7.
- Braeye T, Linina I, De Roy R, Hutse V, Wauters M, Cox P, Mak R. Mumps increase in Flanders, Belgium, 2012-2013: results from temporary mandatory notification and a cohort study among university students. Vaccine. 2014;32(35):4393–98. doi:https://doi.org/10.1016/j.vaccine.2014.06.069.
- Cortese MM, Jordan HT, Curns AT, Quinlan P, Ens K, Denning P, Dayan G. Mumps vaccine performance among university students during a mumps outbreak. Clin Infect Dis. 2008;46(8):1172–80. doi:https://doi.org/10.1086/529141.
- Marin M, Quinlisk P, Shimabukuro T, Sawhney C, Brown C, Lebaron CW. Mumps vaccination coverage and vaccine effectiveness in a large outbreak among college students–Iowa, 2006. Vaccine. 2008;26(29–30):3601–07. doi:https://doi.org/10.1016/j.vaccine.2008.04.075.
- Vygen S, Fischer A, Meurice L, Mounchetrou Njoya I, Gregoris M, Ndiaye B, Ghenassia A, Poujol I, Stahl JP, Antona D, et al. Waning immunity against mumps in vaccinated young adults, France 2013. Euro Surveill. 2016;21(10):30156. doi:https://doi.org/10.2807/1560-7917.ES.2016.21.10.30156.
- Seagle EE, Bednarczyk RA, Hill T, Fiebelkorn AP, Hickman CJ, Icenogle JP, Belongia EA, McLean HQ. Measles, mumps, and rubella antibody patterns of persistence and rate of decline following the second dose of the MMR vaccine. Vaccine. 2018;36(6):818–26. doi:https://doi.org/10.1016/j.vaccine.2017.12.075.
- LeBaron CW, Forghani B, Beck C, Brown C, Bi D, Cossen C, Sullivan B. Persistence of mumps antibodies after 2 doses of measles-mumps-rubella vaccine. J Infect Dis. 2009;199(4):552–60. doi:https://doi.org/10.1086/596207.
- Gordon N How does the adult kaiser permanente membership in northern california compare with the larger community? 2006.
- Kennedy RB, Ovsyannikova IG, Thomas A, Larrabee BR, Rubin S, Poland GA. Differential durability of immune responses to measles and mumps following MMR vaccination. Vaccine. 2019;37(13):1775–84. doi:https://doi.org/10.1016/j.vaccine.2019.02.030.
- Date AA, Kyaw MH, Rue AM, Julie K, Obrecht L, Krohn T, Rowland J, Rubin S, Safranek TJ, Bellini WJ, et al. Long-term persistence of mumps antibody after receipt of 2 measles-mumps-rubella (MMR) vaccinations and antibody response after a third MMR vaccination among a university population. J Infect Dis. 2008;197(12):1662–68. doi:https://doi.org/10.1086/588197.
- Lewnard JA, Grad YH. Vaccine waning and mumps re-emergence in the United States. Sci Transl Med. 2018;10:433. doi:https://doi.org/10.1126/scitranslmed.aao5945.
- Fiebelkorn AP, Rosen JB, Brown C, Zimmerman CM, Renshowitz H, D'Andrea C, Gallagher KM, Harpaz R, Zucker JR. Environmental factors potentially associated with mumps transmission in yeshivas during a mumps outbreak among highly vaccinated students: Brooklyn, New York, 2009-2010. Hum Vaccin Immunother. 2013;9(1):189–94. doi:https://doi.org/10.4161/hv.22415.
- Nojd J, Tecle T, Samuelsson A, Orvell C. Mumps virus neutralizing antibodies do not protect against reinfection with a heterologous mumps virus genotype. Vaccine. 2001;19(13–14):1727–31. doi:https://doi.org/10.1016/S0264-410X(00)00392-3.
- Crowley B, Afzal MA. Mumps virus reinfection–clinical findings and serological vagaries. Commun Dis Public Health. 2002;5:311–13.
- Rubin SA, Qi L, Audet SA, Sullivan B, Carbone KM, Bellini WJ, Rota PA, Sirota L, Beeler J. Antibody induced by immunization with the Jeryl Lynn mumps vaccine strain effectively neutralizes a heterologous wild-type mumps virus associated with a large outbreak. J Infect Dis. 2008;198(4):508–15.
- Rubin SA, Link MA, Sauder CJ, Zhang C, Ngo L, Rima BK, Duprex WP. Recent mumps outbreaks in vaccinated populations: no evidence of immune escape. J Virol. 2012;86(1):615–20. doi:https://doi.org/10.1128/JVI.06125-11.
- Wohl S, Metsky HC, Schaffner SF, Piantadosi A, BurnsID M, Lewnard JA, Chak B, Krasilnikova LA, Siddle KJ, Matranga CB, et al. Combining genomics and epidemiology to track mumps virus transmission in the United States. PLoS Biol. 2020;18:2. doi:https://doi.org/10.1371/journal.pbio.3000611.
- Principi N, Esposito S. Mumps outbreaks: A problem in need of solutions. J Infect. 2018;76(6):503–06. doi:https://doi.org/10.1016/j.jinf.2018.03.002.
- Greenland K, Whelan J, Fanoy E, Borgert M, Hulshof K, Yap K-B, Swaan C, Donker T, van Binnendijk R, de Melker H, et al. Mumps outbreak among vaccinated university students associated with a large party, the Netherlands, 2010. Vaccine. 2012;30(31):4676–80. doi:https://doi.org/10.1016/j.vaccine.2012.04.083.