Correction to: Dose-finding study of luseogliflozin in Japanese patients with type 2 diabetes mellitus: a 12-week, randomized, double-blind, placebo-controlled, phase II study
Seino Y, Sasaki T, Fukatsu A, Ubukata M, Sakai S, Samukawa Y. Dose-finding study of luseogliflozin in Japanese patients with type 2 diabetes mellitus: a 12-week, randomized, double-blind, placebo-controlled, phase II study. Current Medical Research and Opinion 2014;30(07):1231-44.
Following publication of this article, an error has been identified in .
The corrected is shown below.
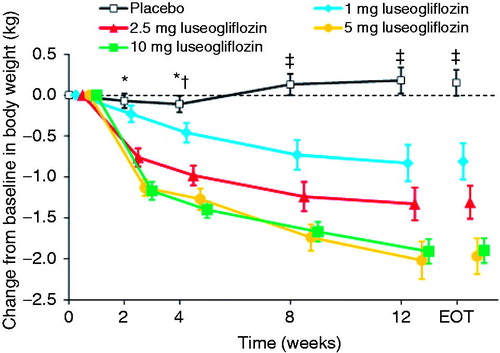
Figure 5. Changes in body weight from baseline to each visit or end of treatment. Values are means with standard error. The last observation carried forward method was applied to data at EOT. Differences between each luseogliflozin group and placebo were analyzed by the unrestricted least significant difference method. *P < 0.001 for 2.5, 5, and 10 mg luseogliflozin vs. placebo. †P < 0.05 for 1 mg luseogliflozin vs. placebo. ‡P < 0.001 for all luseogliflozin groups vs. placebo. All data are shown for the full analysis set. EOT, end of treatment.