Abstract
Background
There is an ongoing debate about the appropriate spirometric criterion for airway obstruction to detect COPD. Furthermore, the association of different criteria with comorbidity prevalence and inflammatory biomarkers in advanced age is unclear.
Materials and methods
Spirometry was performed in a population-based study (n=2,256) covering an age range of 41–90 years. COPD was spirometrically determined either by a fixed ratio (FR) of <0.7 for forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) or by FEV1/FVC below the lower limit of normal (LLN). Comorbidity prevalences and circulating biomarker levels (C-reactive protein [CRP], interleukin [IL]-6) were compared between subjects with or without COPD by the two criteria using logistic and multiple regression models, adjusting for sex and age.
Results
The prevalence of spirometrically defined COPD by FR increased with age from 10% in subjects aged <65 years to 26% in subjects aged ≥75 years. For LLN-defined COPD, it remained below 10% for all age groups. Overall, COPD diagnosis was not associated with specific comorbidities, except for a lower prevalence of obesity in both FR- and LLN-defined cases. Both CRP and IL-6 tended to be higher in cases by both criteria.
Conclusion
In a population-based cohort of adults up to the age of 90 years, the prevalence of spirometrically defined COPD was higher for the FR criterion than for the LLN criterion. This difference increased with age. Neither prevalences of common comorbidities nor levels of the biomarkers, CRP or IL-6, were conclusively associated with the selection of the COPD criterion. Results have to be considered in light of the predominantly mild cases of airway obstruction in the examined study population.
Background
According to current recommendations, a clinical diagnosis of COPD is primarily based on the detection of airflow obstruction in spirometric measures.Citation1,Citation2 However, the spirometric criteria defining airflow obstruction in COPD are still subject to lively debate.Citation3,Citation4 The Global Initiative for Chronic Obstructive Lung Disease recommends the use of a fixed ratio (FR) of 0.7 for postbronchodilator forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) to confirm the diagnosis.Citation1 This approach has been criticized as arbitrary and argued to particularly overestimate prevalences in advanced age.Citation5 Alternatively, the use of the fifth percentile of the predicted value (lower limit of normal, LLN) for FEV1/FVC has been proposed by respiratory societies, eg, the American Thoracic Society and European Respiratory Society.Citation6,Citation7 It has been shown that the two COPD definitions lead to deviating prevalences.Citation8 In addition, data and subjects in advanced age beyond 75 years are often underrepresented in the studied population-based samples.
Besides airflow obstruction, COPD is accompanied by a variety of systemic impairments.Citation9 Comorbidities of COPD include cardiovascular disease, diabetes, depression, and cancer,Citation10 and it has been reported that COPD may accelerate the progression of some of these comorbidities.Citation9 Moreover, COPD is characterized by low-grade systemic inflammation.Citation11 Different biomarkers such as C-reactive protein (CRP)Citation12 and interleukin (IL)-6Citation13 have been associated with a poor prognosis in COPD participants. Celli et alCitation14 showed that using a whole panel of inflammatory biomarkers (eg, CRP, IL-6, IL-8, and fibrinogen) improved the clinical prediction of mortality in COPD participants. Still, in how far circulating biomarker levels are affected by spirometric criteria is less investigated.
The aim of this investigation was 1) to quantify the impact of disease definition by different spirometric criteria (FR vs LLN) on COPD prevalence in a population-based sample and 2) to evaluate the association of FR vs LLN criteria with comorbidity prevalences and selected inflammatory biomarkers.
Materials and methods
Study participants
Within a population-based cohort from the Augsburg region (KORA, Cooperative Health Research in the Augsburg Region, Germany),Citation15 two follow-up studies, KORA-F4Citation16 and KORA-Age,Citation17 were launched from the MONICA/KORA surveys S1–S4. The KORA-F4 and KORA-Age studies were approved by the ethics committee of the Bavarian Medical Association, and written informed consent was obtained from all participants. Details of the KORA platform and surveys have been described earlier.Citation15,Citation18 Briefly, spirometry was performed in 1,321 subjects aged 41–63 years in KORA-F4 (2006–2008) and in 935 subjects aged 65–90 years in KORA-Age (2009). Measurement conditions for spirometry and the examiners were the same in both studies. Standing height and weight were measured with subjects wearing only light clothes and no shoes.
Spirometry
Details on spirometric measurements have been reported earlier.Citation16 In short, standard spirometry without bronchodilation was performed in line with the American Thoracic Society/European Respiratory Society recommendations in an upright sitting position, while subjects were wearing nose clips using a pneumotachograph-type spirometer (MasterScope PC; CareFusion, Höchberg, Germany). The participants performed at least three spirometric maneuvers in order to obtain a minimum of two acceptable and reproducible values. Subjects were classified as having COPD if FEV1/FVC was <0.7 for the FR criterion and if FEV1/FVC was < LLN for the LLN criterion. Reference values of the Global Lungs Initiative for FVC, FEV1, and FEV1/FVC were used.Citation19 The severity of COPD was graded according to the stages of disease based on FEV1 results into categories as defined by the Global Initiative for Chronic Obstructive Lung Disease.Citation1
Comorbidities and biomarkers
Self-reported information on myocardial infarction (MI), stroke, cancer, diabetes, inflammatory joint disease/rheumatic disease, gastrointestinal disease (GID), and COPD/chronic bronchitis diagnosis was collected in standardized interviews, which were partly performed via telephone and self-administered questionnaires within the comprehensive KORA assessments.Citation20 Information on current medication was assessed by asking all study participants to bring original packaging of their medications used during the last 7 days before the study examination.
Hypertension was defined as blood pressure values ≥140/90 mmHg measured during the visit and/or intake of antihypertensive medication, given that the participant was aware of having hypertension as assessed in the standardized interview or questionnaire. Diabetes mellitus was defined as having a self-reported physician diagnosis of diabetes and/or intake of antidiabetic medication. The presence of an anxiety disorder was defined using the Generalized Anxiety Disorder Scale-7Citation21 in the standardized interview or questionnaire with a score of ≥10 indicating the presence of an anxiety disorder. Depression was assessed using the depression module of the Brief Patient Health Questionnaire-D (PHQ-D) in KORA-F4 (age 41–63 years), whereas in KORA-Age (age 65–90 years) it was assessed using the Geriatric Depression Scale-15 (GDS-15) during the standardized interview, where participants with a score of ≥10 were classified as being depressive.Citation22 Smoking habits and symptoms of chronic bronchitis, defined as cough and sputum on most days during 3 or more months per year, were assessed within the standardized interview or questionnaire. Furthermore, information on the presence of a physician diagnosis of COPD or chronic bronchitis was assessed during the standardized interview. Information on medication against obstructive respiratory disease comprised sympathomimetics, anticholinergics, xanthines, leukotriene receptor antagonists, and corticosteroids.
Serum and ethylenediaminetetraacetic acid (EDTA) plasma samples were obtained from all study participants and were stored at −80°C until analysis. Levels of high-sensitivity CRP were measured in serum (KORA-Age) or EDTA plasma samples (KORA-F4) using Cardio Phase® hsCRP (Siemens, Eschborn, Germany). Serum IL-6 was measured using Quantikine HS Elisa (R&D Systems, Inc., Minneapolis, MN, USA). IL-6 measurements were available only for the KORA-Age cohort.
Statistical analysis
Prevalences are expressed as numbers and percentages of subjects with the trait. Subject characteristics and spirometric indices are expressed as mean (standard deviation). Basic study results are presented according to three age groups (<65 years/≥65 years and <75 years/≥75 years) and spirometric results on the basis of FEV1/FVC categories. As the distributions of the biomarker levels were skewed, results were log10 transformed before further analysis. For the outcome variables of comorbidities, self-reported physician diagnosis of COPD/chronic bronchitis, use of lung medication, chronic bronchitis symptoms, and ever-smoking logistic regression models were generated including sex and age as factors. The influence of the following dichotomous variables was then tested by separately including them into these basic models: 1) cases vs healthy subjects by the FR criterion, 2) cases vs healthy subjects by the LLN criterion, and 3) cases by the FR criterion only vs cases by the LLN criterion. All LLN-defined COPD cases were included in the respective FR-defined group except for one subject in the age group of 40–44 years who was excluded from these analyses. For the outcome variables, log10(CRP) and log10(IL-6) general linear models were generated including sex and age as predictors. The influence of the dichotomous variables for COPD definition was then tested analogously. Analyses were performed using the commercially available software SAS 9.3 (SAS Institute Inc., Cary, NC, USA) and Statgraphics Centurion XVII (Statpoint Technologies Inc., Warrenton, VA, USA); additional plots were done using SigmaPlot 12.0 (Systat Software Inc., San Jose, CA, USA).
Results
In total, 2,256 individuals were included: 1,321 from KORA-F4 (618 men and 703 women) and 935 from KORA-Age (474 men and 461 women). summarizes descriptive data of our study participants from both cohorts separately and combined. All study participants from KORA-F4 were younger than 65 years (41–63 years), and all participants from KORA-Age were 65 years and older (65–90 years).
Table 1 General characteristics of the KORA-F4 and KORA-Age study participants with spdata
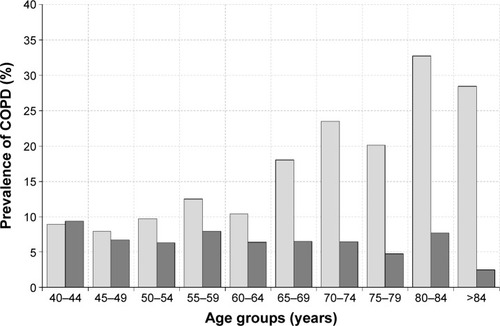
The prevalence of COPD defined by spirometry without bronchodilation according to the FR criterion increased with age from ~10% for the age group below 65 years to 21% for the age group between 65 years and 75 years, and finally 26% for individuals in the age group of 75 years and older (), but was predominantly confined to mild and moderate stages of COPD. Severe and very severe COPD grades were rare in our population-based study (nine cases in total) and did not differ between FR and LLN criteria for all age groups. The prevalence of COPD on the basis of the LLN criterion remained stable below 10% up to an age of 90 years. The drifting apart with increasing age between both COPD definitions, depending on the diagnostic criteria used (FR vs LLN), became most prominent at age beyond 65 years ().
Table 2 COPD based on FR (FEV1/FVC <0.7) and LLN (FEV1/FVC < LLN) criteria by age and sex (N, % of group)
Figure 1 Prevalence of spirometrically defined COPD based on FR (light gray) or LLN (dark gray) criterion.
Abbreviations: FR, fixed ratio; LLN, lower limit of normal.
As shown in , the presence of a self-reported physician diagnosis of COPD/chronic bronchitis in cases of spirometrically defined COPD was generally rather low ranging between 14% and 38%. However, it was predominantly higher for LLN-defined COPD than for the FR criterion as was true for the current use of lung medication and the presence of chronic bronchitis symptoms. In the logistic regression model, this difference between the spirometric criteria was observed for all three aspects (). Regarding ever smoking, differences of cases vs no COPD by both criteria were most prominent in the youngest age group (), while no differences between the two COPD criteria were observed.
Table 3 Frequency (%) of self-reported physician diagnosis of COPD or chronic bronchitis, current use of lung medication, symptoms of chronic bronchitis, and ever smoking in subjects classified by FR (FEV1/FVC <0.7) and LLN (FEV1/FVC < LLN) criteria
Table 4 Odds ratio estimates (95% CI) for lung-associated parameters by spirometric criterion (FR: FEV1/FVC <0.7; LLN: FEV1/FVC <lower limit of normal) from adjusted logistic regression models
shows the odds ratio estimates for the influence of COPD according to FR and LLN criteria on disease prevalence from the logistic regression models; primary prevalences of comorbidities are given in Figure S1A–C. By trend, differences between subjects with and without COPD were most prominent in males for the LLN criterion for MI, cancer, stroke, and anxiety in the middle and oldest age group and for GID in the youngest age group. Although a similar trend was observed for GID in females in the youngest age group, other comorbidities did not show consistent tendencies in women. On the other hand, as shown in , the prevalence of obesity was consistently lower in COPD cases, which was independent of the criterion. Beyond this observation, no firm pattern was observed regarding the prevalence of the examined comorbidities. Still, the occurrence of two or more comorbidities was lower in subjects with vs without COPD defined by the LLN criterion ().
Table 5 Odds ratio estimates (95% CI) for comorbidities by spirometric criterion (FR: FEV1/FVC <0.7; LLN: FEV1/FVC < LLN) from adjusted logistic regression models
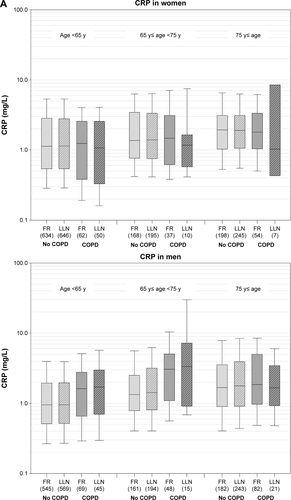
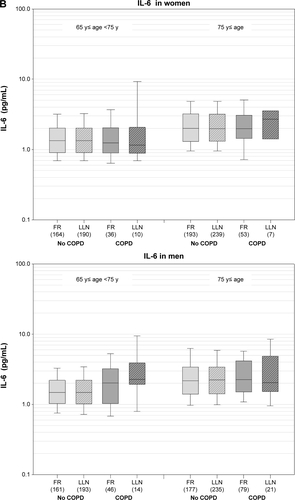
The estimates for the effect of COPD on levels of CRP and IL-6 are presented in ; median CRP and IL-6 levels are shown in Figure S2A and B. Levels of both bio-markers slightly increased with age in all participants without COPD, but consistent differences between FR- and LLN-defined COPD were not detectable. Still, both CRP and IL-6 tended to be higher in cases by both criteria ().
Table 6 Parameter estimates (95% CI) for CRP and IL-6 levels from general linear models by FR and LLN criterion
Discussion
In epidemiology, the definition of COPD disease status is frequently based on either spirometric results, often without bronchodilation, or questionnaire data of physician diagnosis. In this population-based observational study, the impact of disease definition by different spirometric criteria (FR vs LLN) on COPD prevalence was evident and increased with age (). The tendency of COPD being possibly over-diagnosed by the FR criterion becomes more pronounced in advanced age, most prominently beyond 65 years in our sample. However, neither the prevalence of common comorbidities nor levels of the inflammatory biomarkers CRP and IL-6 were affected by the selection of the COPD criterion. Our results have to be considered in light of the fact that in our epidemiological cohort we predominantly observed participants with mild-to-moderate airflow obstruction based on spirometry without bronchodilation.
The prevalence of respiratory obstruction we observed for both spirometric criteria compared well with recent population-based analyses that were performed in a similar fashion. For example, in a large Canadian sample, an overall obstruction prevalence of 17% by the FR criterion has been reported for the age range of 40–93 years,Citation23 Turkeshi et alCitation24 found an FR-based prevalence of 27% in a Belgian sample of very old adults aged 80 years or older. However, the prevalence of ~7% we found for LLN was slightly lower than the range of 9%–11% reported in these studies.
Unexpectedly, study participants with spirometrically defined COPD generally did not suffer from more comorbidities than participants of the same age without COPD irrespective of the applied spirometric criterion. Although a tendency toward higher prevalences of MI, cancer, stroke, and anxiety was observed in participants with LLN-defined COPD compared with participants without LLN-defined COPD, there was no distinct difference after taking account of the sex and age in logistic regression models. On the contrary, we found a lower prevalence of obesity in COPD subjects for both criteria, which might be explained by a higher fraction of smokers among these subjects.Citation25 None of the other comorbidities evaluated were more prevalent among COPD participants, which is in contrast to findings from other studies.Citation26–Citation28 The lack of a difference may partly be explained by the fact that our study was conducted in the general population with predominantly mild cases as opposed to a clinical setting with more severe stages of COPD. The prevalences we found in spirometrically defined COPD for MI, stroke, anxiety, and diabetes compared well with findings described by Divo et alCitation29 in a recent analysis of COPD patients based on the BODE registry, when considering comparable age groups. However, our prevalences for hypertension were markedly higher, while those for depression were lower, which might be attributed to differences in the assessment of those comorbidities. Furthermore, the COPD cases as defined in our study were predominantly mild and mostly not yet confirmed by a physician, as indicated by the rather poor overlap with physician diagnoses, () suggesting early undiagnosed cases to a large degree. Moreover, the increased fraction of ever smokers especially in the youngest age group points to the fact that this factor is of importance predominantly in COPD at comparably young age. It might be speculated that possible concomitant systemic impairments in these cases are in a comparably early stage and thus are not or not yet diagnosed by a physician. Alternatively, a limitation in airway function may precede the affections of other organs that are later described as comorbidities in COPD. The exploration of a possible sequence in this regard, however, would require a longitudinal evaluation, which was beyond the scope of the current cross-sectional study.
In recent years, systemic inflammation has come into the focus of COPD research, and increased serum levels of IL-6 and CRP in COPD participants have been reported.Citation30 Although we found a tendency toward elevated values for both markers in COPD participants, the overall effect was comparatively small. The lack of findings, particularly in the aged, may, in our case, be partly explained by harvesting, where subjects in poor health did not survive or did not participate in the study as they were unable to get into the study center. A recent study by Silva et alCitation31 did not find markedly elevated CRP in nonsmoking COPD patients compared to a control group indicating the possible impact of active smoking on this parameter. However, the tendency toward higher IL-6 levels in LLN-defined COPD cases in older age for males suggests that this criterion may be diagnostically more specific in identifying participants with (early stage) systemic inflammation than the FR and needs confirmation in longitudinal settings.
Consequently, these findings indicate that the application of the LLN criterion in epidemiological or clinical settings will result in an identification of subjects with more concomitant typical characteristics of COPD, eg, chronic bronchitis symptoms, and a higher probability of a physician diagnosis of COPD compared to FR (). However, since no consistent differences in comorbidity prevalences or biomarker levels were observed, the preference of one criterion over the other in this regard could not be derived from our study. Beyond the FEV1/FVC ratio, the use of more advanced lung function measurements such as body plethysmographyCitation32 could provide a refined characterization of patient subgroups with a higher number or specific patterns of comorbidities.
The limitation of the current study might be the fact that the definition of COPD cases here, as a rather common epidemiological approach, was solely based on spirometric results as opposed to a usually more comprehensive approach in clinical practice including anamnesis, symptoms, risk factors, and individual clinical history. However, the restriction to different interpretations of a spirometric measure allowed us to specifically focus on the impact of this particular component of the diagnostic sequence in predominantly early, often undiagnosed cases of airway obstruction according to our research question. A clear strength of our study is the inclusion of participants from the general population up to an advanced age of 90 years, an age group that is often underrepresented in COPD studies.
Conclusion
In a population-based sample of adults in middle and advanced age, we found that the prevalence of spirometrically defined COPD was higher when using the FR criterion as opposed to LLN and this difference increased with increasing age. However, for the predominantly mild cases observed in our cohort, there was no conclusive impact of the different spirometric criteria on the prevalence of comorbidities and on common inflammatory biomarker levels.
Acknowledgments
This work was supported by the Competence Network Asthma and COPD (ASCONET), network COSYCONET (subproject 2, BMBF FKZ 01GI0882), and the KORA-Age project (BMBF FKZ 01ET0713 and 01ET1003A) funded by the German Federal Ministry of Education and Research (BMBF). The KORA study was initiated and financed by the Helmholtz Zentrum München – German Research Center for Environmental Health, which was funded by the German Federal Ministry of Education and Research (BMBF) and by the State of Bavaria. Furthermore, KORA research was supported by the Munich Center of Health Sciences (MC-Health), Ludwig-Maximilians-Universität, as part of LMUinnovativ.
Supplementary materials
Figure S1 (A–C) Prevalence of comorbidities in subjects without COPD (light grey) or with COPD (dark grey) by FR (FEV1/FVC <0.7; solid color) and LLN (FEV1/FVC <lower limit of normal; hatched) criterion (percentages).
Abbreviations: FR, fixed ratio; LLN, lower limit of normal; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; y, years.
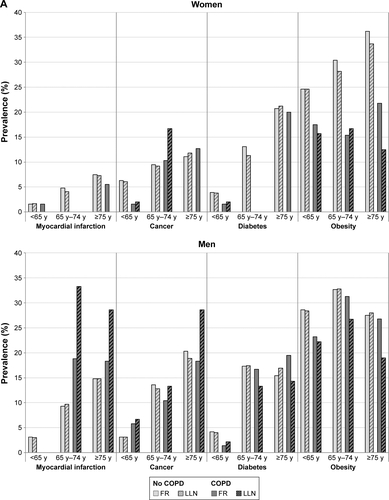
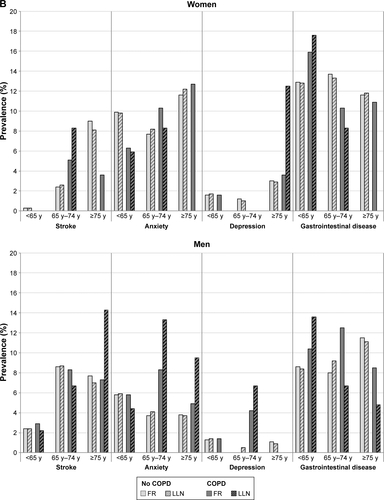
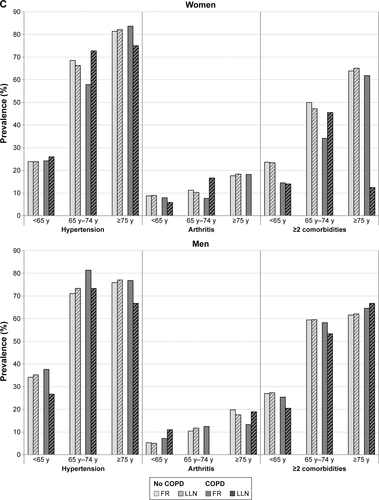
Figure S2 (A–B) Levels of CRP and IL-6 in subjects without COPD (light grey) and COPD cases (dark grey) based on FR (FEV1/FVC <0.7; solid color) or LLN (FEV1/FVC <lower limit of normal; hatched) criterion (number of subjects per group are given in brackets).
Abbreviations: FR, fixed ratio; LLN, lower limit of normal; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; y, years.
Disclosure
The authors report no conflicts of interest in this work.
References
- Global Initiative for Chronic Obstructive Lung Disease (GOLD) [homepage on the Internet]Global Strategy for the Diagnosis, Management and Prevention of COPD2015 Available from: http://www.goldcopd.org/Accessed October 14, 2015
- NardiniSAnnesi-MaesanoIDel DonnoMThe AIMAR recommendations for early diagnosis of chronic obstructive respiratory disease based on the WHO/GARD modelMultidiscip Respir Med2014914625473523
- QuanjerPHCorrectly defining criteria for diagnosing chronic obstructive pulmonary disease mattersAm J Respir Crit Care Med2014189223024428654
- RennardSVestboJAgustiAReply: correctly defining criteria for diagnosing chronic obstructive pulmonary disease mattersAm J Respir Crit Care Med2014189223023124428653
- QuanjerPHEnrightPLMillerMRThe need to change the method for defining mild airway obstructionEur Respir J201137372072221357929
- Lung function testing: selection of reference values and interpretative strategies. American Thoracic SocietyAm Rev Respir Dis19911445120212181952453
- PellegrinoRViegiGBrusascoVInterpretative strategies for lung function testsEur Respir J200526594896816264058
- Mohamed HoeseinFAZanenPLammersJWLower limit of normal or FEV1/FVC <0.70 in diagnosing COPD: an evidence-based reviewRespir Med2011105690791521295958
- DivoMCoteCde TorresJPBODE Collaborative GroupComorbidities and risk of mortality in patients with chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2012186215516122561964
- RabeKFWedzichaJAWoutersEFEuropean Respiratory Monograph 59: COPD and Comorbidity59LausanneEuropean Respiratory Society2013
- FabbriLMRabeKFFrom COPD to chronic systemic inflammatory syndrome?Lancet2007370958979779917765529
- DahlMVestboJLangePBojesenSETybjaerg-HansenANordestgaardBGC-reactive protein as a predictor of prognosis in chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2007175325025517053205
- MehrotraNFreireAXBauerDCHealth ABC StudyPredictors of mortality in elderly subjects with obstructive airway disease: the PILE scoreAnn Epidemiol201020322323220159492
- CelliBRLocantoreNYatesJECLIPSE InvestigatorsInflammatory biomarkers improve clinical prediction of mortality in chronic obstructive pulmonary diseaseAm J Respir Crit Care Med2012185101065107222427534
- HolleRHappichMLöwelHWichmannHEMONICA/KORA Study GroupKORA – a research platform for population based health researchGesundheitswesen200567suppl 1S19S2516032513
- KarraschSFlexederCBehrJKORA Study GroupSpirometric reference values for advanced age from a south German populationRespiration201385321021922797415
- PetersADöringALadwigKHMultimorbidity and successful aging: the population-based KORA-Age studyZ Gerontol Geriatr20114424154 German22270973
- LöwelHDöringASchneiderAMONICA/KORA Study GroupThe MONICA Augsburg surveys – basis for prospective cohort studiesGesundheitswesen200567suppl 1S13S1816032512
- QuanjerPHHallGLStanojevicSColeTJStocksJGlobal LungsIAge- and height-based prediction bias in spirometry reference equationsEur Respir J201240119019722183491
- KirchbergerIMeisingerCHeierMPatterns of multimorbidity in the aged population. Results from the KORA-age studyPLoS One201271e3055622291986
- SpitzerRJKroenkeKWilliamsJBWLöweBA brief measure for assessing generalized anxiety disorderArch Intern Med2006166101092109716717171
- YesavageJASheikhJIGeriatric depression scale (GDS). Recent evidence and development of a shorter versionClin Gerontol198651–2165173
- van DijkWTanWLiPCanCOLD Study GroupClinical relevance of fixed ratio vs lower limit of normal of FEV1/FVC in COPD: patient-reported outcomes from the CanCOLD cohortAnn Fam Med2015131414825583891
- TurkeshiEVaesBAndreevaEAirflow limitation by the Global lungs initiative equations in a cohort of very old adultsEur Respir J201546112313225882799
- MolariusASeidellJCKuulasmaaKDobsonAJSansSSmoking and relative body weight: an international perspective from the WHO MONICA ProjectJ Epidemiol Community Health19975132522609229053
- IslamMMValderasJMYenLDawdaPJowseyTMcRaeISMultimorbidity and comorbidity of chronic diseases among the senior Australians: prevalence and patternsPLoS One201491e8378324421905
- SmithMWrobelJEpidemiology and clinical impact of major comorbidities in patients with COPDInt J Chron Obstruct Pulmon Dis2014987188825210449
- TzanakisNHillasGPerlikosFTsiligianniIManaging comorbidities in COPDInt J Chron Obstruct Pulmon Dis2015109510925609943
- DivoMJCasanovaCMarinJMBODE Collaborative GroupCOPD comorbidities networkEur Respir J201546364065026160874
- BaldiSBruschiCMaestriRThe mediating role of cytokine IL-6 on the relationship of FEV1 upon 6-minute walk distance in chronic obstructive pulmonary diseaseInt J Chron Obstruct Pulmon Dis201491091109925336940
- SilvaDRGazzanaMBKnorstMMC-reactive protein levels in stable COPD patients: a case-control studyInt J Chron Obstruct Pulmon Dis2015101719172526357470
- CrieeCPSorichterSSmithHJWorking Group for Body Plethysmography of the German Society for Pneumology and Respiratory CareBody plethysmography – its principles and clinical useRespir Med2011105795997121356587
