Abstract
Psoriasis is a chronic inflammatory disease affecting up to 3% of the general population. The prevalence of nail involvement in psoriasis patients varies between 15% and 79%. While the nails represent a small portion of the body surface area, psoriasis in these areas can have a disproportionate influence on a patient’s physical and psychosocial activities. Differential diagnosis between an onychomycosis and a psoriatic nail could be challenging; nevertheless, coexistence of onychomycosis and nail psoriasis also occurs and both are common disorders in the general population. Nail psoriasis can be difficult to treat. Treatment of nail psoriasis should consider the body surface area of skin disease, psoriatic arthritis, severity of nail disease, and the impairment in the quality of life. All patients should be tested for onychomycosis before starting a therapy. This recommendation is underlined by the fact that nail psoriasis is mostly treated by immunosuppressive drugs, like steroids, methotrexate, or biologics, which may aggravate mycotic nail infections. Conventional systemic therapy, such as use of steroids, cyclosporine, methotrexate, and retinoid in the long term, can cause organ toxicities. Currently, use of apremilast and tofacitinib favors an early healing of nail psoriasis because they act directly on the pathogenic targets, distressing the inflammatory signals associated with the initiation and maintenance of the disease activity, and as with several conventional synthetic disease modifying antirheumatic drugs, they are characterized by the convenience of oral administration. The number of treatment options has increased considerably in recent years; however, given the heterogeneity of the disease, the therapy should be personalized to individual cases.
Keywords:
Introduction
Psoriasis is a chronic inflammatory disease affecting up to 3% of the general population. The disease is characterized by epidermal hyper-proliferation resulting in erythematous-squamous skin plaques that may cover large body areas; in ~30% of patients it is characterized by a seronegative spondyloarthritis.Citation1
Psoriasis is considered a multiorgan disorder that requires a multidisciplinary approach and an appropriate management that takes into consideration a number of comorbidities. In fact, several studies revealed the association between psoriasis and a number of disease-related comorbidities including blood hypertension and cardiovascular diseases, obesity, type II diabetes, nonalcoholic fatty liver disease, anxiety, depression, and inflammatory bowel disease.Citation2–Citation6
The disorder, in its entirety, is associated with a high degree of morbidity including a notable impact on social relationships, mental health, and work-related activities.Citation6,Citation7 The disease can have a substantial negative influence on a patient’s quality of life (QOL), especially psoriasis of vastly visible areas of the body including face, hands, scalp, and nails, and it is associated with physical impairment and pain. Psoriasis affects a patient’s QOL sometimes more than other chronic diseases such as diabetes, cardiovascular disease, and rheumatoid arthritis.Citation5,Citation8–Citation10
While the most specific features of psoriasis are skin manifestations, nails are commonly involved.Citation11 The prevalence of nail involvement in psoriasis patients varies between 15% and 79%.Citation11–Citation13 It is uncommon in children, and the prevalence ranges from 7% to 13%, whereas in adult patients, nail psoriasis is common – in the absence of skin and joint diseases – and 5%–10% of adult patients are reported to be affected.Citation13
Approximately 90% of psoriatic patients develop nail psoriasis during their lifetimes, and it is not related to gender or age.Citation14,Citation15 McGonagle et al described that nail, scalp, and intergluteal skin involvement are unfavorable signs and are often predictors of Psoriatic Arthritis (PsA) evolution.Citation16 Nail involvement is frequently observed in association with psoriatic arthritis, and several studies have described an incidence of 80%.Citation14–Citation18 Nail psoriasis is often related to a protracted duration of psoriasis and severity of skin and joint involvement.Citation19 Furthermore, psoriatic nail disease may be considered as a risk factor for the development of psoriatic arthritis.Citation20
Clinical features of nail psoriasis
Nardo Zaias, in 1969, firstly described the pathophysiology of nail psoriasis.Citation21 The nail bed, nail matrix, hyponychium, and nail folds can be affected by nail psoriasis. The most observed forms are psoriasis of the nail matrix, nail bed, and nail fold.Citation12 Pitting, leukonychia, red spots of the lunula, transverse grooves (Beau’s lines), and crumbling of the nail plates are the typical signs of psoriasis of the nail matrix.Citation21 Oil-drop discoloration, splinter hemorrhages involving the distal third of the nail plate, subungual hyperkeratosis, and/or detachment of the nail plate from the nail bed (onycholysis) are the characteristic marks of the nail bed involvement.Citation12 Psoriasis of the periungual region is characterized by paronychia.Citation12
The severity of nail psoriasis is evaluated by Nail Psoriasis Severity Index (NAPSI) which is a numeric, reproducible, objective, and simple tool. According to this index, each nail is divided into four quadrants, each of which is assessed for the presence of any signs of psoriasis in the nail matrix such as pitting, leukonychia, red spots in the lunula, nail plate crumbling and nail bed as oil-drop discoloration, onycholysis, hyperkeratosis, and splinter hemorrhages.Citation22 This scale is used to evaluate the severity of nail bed psoriasis and nail matrix psoriasis based on the area of involvement in the nail unit. NAPSI is useful during clinical trials for assessing response to treatment of patients with psoriatic nails.Citation22
Different clinical presentations are associated with nail psoriasis according to the nail structure apparatus. All the pathognomonic signs of nail psoriasis are not exclusive and may be found in several other nail disorders. Differential diagnosis between an onychomycosis and a psoriatic nail could be challenging; nevertheless, coexistence of onychomycosis and nail psoriasis also occurs and both are common disorders in the general population.Citation23
Klaassen et al reported in their study a higher prevalence of onychomycosis in patients affected by psoriasis of the nail compared to the non-affected population.Citation18 Several studies hypothesized that morphological defects in psoriatic nails are predisposing factors for onychomycosis and that onychomycosis could act as a Koebner phenomenon for the development of the psoriatic nails.Citation24 In healthy nails, the nail plate acts as a natural barrier counteracting the development of fungal infections, whereas in psoriatic patients, the defective nail plate may be predisposed to fungal infection.Citation25 Several studies have shown that patients with the highest NAPSI scores were most likely to test positive for fungal colonization.Citation26,Citation27
Pathogenesis
Psoriasis seems to be a multifactorial disorder whose rigorous underlying mechanism is still uncertain, and environmental factors, genetic susceptibility, abnormal function of keratinocytes, and dysregulation of innate and acquired immune response are all assumed.Citation28,Citation29
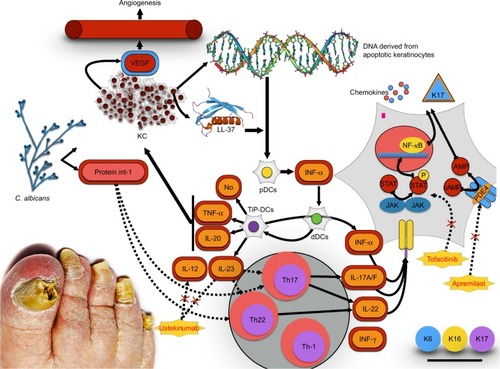
Certain infections, such as bacterial and fungal infections, especially Candida albicans have been shown to be involved and to play a role in the exacerbation and maintenance of the disease. in fact, psoriasis is a systemic inflammatory disease in which dysregulation of the immune system results in overexpression of inflammatory cytokines. Some of these cytokines are involved in host defense against common infections, including Candida.Citation30–Citation33 Candida can stimulate the production of superantigens, determining non-specific T-cell activation and secretion of cytokines that can initiate the psoriatic process.Citation34,Citation35 Consequently, candidiasis is a documented trigger for psoriasis exacerbations and persistence. Candida could act as a trigger for the exacerbation of skin and nail psoriasis through the same LL-37 (cathelicidin) pathway; in particular, in the psoriatic nail, Candida could activate the antimicrobial peptide, LL-37, produced by epithelial nail bed cells that induce interleukin (IL)-23 production by dendritic cells and macrophages, which consequentially activate Th17, determining the cytokine overflow theory and acting as a trigger for the exacerbation of nail psoriasis.Citation30–Citation36
Previous findings demonstrated an increased expression of tumor necrosis factor (TNF)-α, nuclear factor-kappa B, IL-6, and IL-8 in psoriasis-affected nails, which is consistent with the findings of a study on lesional psoriatic skin.Citation37
Rashmi et al described an imbalanced cytokine milieu in psoriatic lesions, with the presence of increased levels of TNF-α, interferon-α, IL-2, IL-6, IL-8, IL-12, and leukemic inhibitory factor-1 and reduced levels of IL-1, IL-4, IL-5, and IL-10.Citation38 The critical role of IL-23/Th17 axis at tissue level is indicated by the increased levels of IL-23, IL-23R and Th17 cytokines revealed in psoriatic skin, especially in lesional versus non-lesional skin.Citation39
IL-10 is an anti-inflammatory and immunosuppressive cytokine that plays an important role in the regulation of the immune response. Several studies demonstrated a down-regulation of IL-10 in psoriatic skin lesions.Citation40 In contrast, Saulite et al found an increased expression of IL-10 in the affected nail bed suggesting unique pathways of psoriatic nail disease and the nail as an immune-privileged site.Citation41–Citation43 However, those results suggest that although the milieu of some of the distinctive inflammatory cytokines and chemokines appears to be consistent with that described in psoriatic skin lesions, there seems to be some distinctive peculiarities for nail psoriasis, thereby confirming the hypothesis that nails act as immune-privileged sites. Different therapeutic responses to monotherapy are due to the distinctive anatomic characteristics of the nail ().Citation41
Figure 1 The cytokines axis in psoriasis. IL-23-, IL-17-axis-related mediators are overexpressed in lesional psoriatic skin and nails. TNFα-/iNOS-producing dendritic cells (TiP-DCs) are activated by various cells and stimuli including Candida albicans. Candida activates Th17 and Th22 to produce IL-22 and IL-17A/F. Keratinocytes are the key-responding cells to this pathway.
Therapeutic strategies
There are limited therapeutic strategies for the management of nail psoriasis. Most conventional treatments have been used for nail psoriasis, but their efficacies are limited and can cause side effects; relapses are common and any noticeable nail improvement will take a long time.Citation37,Citation44–Citation46
Therapeutic management is based on clinical presentation, as well as patient-related factors. Most patients have mild nail psoriasis without arthropathic disease or severe skin psoriasis.Citation28 These patients may be suggested topical therapy, while systemic therapy is suggested in patients affected by severe nail psoriasis and in those with major impact on QOL or with moderate-to-severe psoriatic skin lesions or PsA. Age, burden of disease experienced, comorbidities, individual patient medication preferences, and risks of treatment should be considered for the treatment strategy.
Topical treatments are steroids, vitamin D3 analog calcipotriol or tacalcitol or calcitriol used in monotherapy or in combination with corticosteroids, tazarotene, topical calcineurin inhibitors, 5-fluorouracil. Vitamin D analogs normalize epidermal cell proliferation and differentiation, as well as production and release of proinflammatory cytokines. 1,25(OH)D has been shown to have an antiproliferative effect on keratinocytes.Citation47 In particular, low concentration of vitamin D stimulates keratinocyte proliferation in vitro, while at higher pharmacological doses, a clear inhibitory influence has become evident.Citation48
Furthermore, vitamin D acts as an immunomodulator inhibiting the proliferation of T lymphocytes and stimulates the generation of CD25+/CD4+ Tregs, a phenotype of T cells endorsing tolerance and inhibiting immunity after stimulation with antigen.Citation48 Additionally, vitamin D stimulates the expression of C-C chemokine receptor type 10 on the surface of T lymphocytes, which is involved in T-cell mediated skin inflammation, determining T lymphocytes migration from dermal blood vessels to epidermal keratinocytes.Citation48 In conclusion, vitamin D aids to defend from opportunistic infections, inducing autophagy and supporting the innate skin barrier, thereby stimulating endogenous antimicrobial peptides expression.Citation48
Recent studies have shown that 1,25(OH)D values are significantly lower in psoriatic patients than in control healthy subjects.Citation47 Vitamin D3 derivatives appear to be more successful in treating nail bed psoriasis than nail matrix.Citation47 Tazarotene is a retinoid that has been shown to have antiproliferative, differentiation normalizing, and anti-inflammatory effects, and it is also approved for the treatment of psoriasis.Citation48–Citation53 Topical calcineurin inhibitors such as cyclosporine and tacrolimus are effective in treating both nail matrix signs of nail psoriasis (pitting) and nail bed signs (hyperkeratosis, onycholysis, crumbling, and oil-drop discoloration). No local or systemic side effects were reported during the clinical trial except for a yellowish discoloration after long-term application of cyclosporine.
Topical therapy for nail psoriasis has the evident advantage of treating the nail apparatus without exposing the rest of the organs to the risk of adverse events; however, in patients affected by severe skin psoriasis or PsA, systemic treatments offer a valuable alternative. Conventional systemic therapy, such as use of cyclosporine, methotrexate, and retinoid in the long term, can cause organ toxicities.Citation54
Methotrexate has been shown to improve NAPSI score in several studies; however, its use is limited by a wide range of potential side effects, including hepatotoxicity, ulcerative stomatitis, lymphopenia, nausea, and low white blood cell count.Citation55–Citation58 The calcineurin inhibitor cyclosporine is an immunosuppressive drug characterized by reasonable efficacy in the treatment of nail bed and nail matrix signs of psoriasis. Unfortunately, serious adverse reactions such as renal dysfunction, hypertension, fatigue, headache, paresthesia, hypertrichosis, gingival hyperplasia, and gastrointestinal disorders have been reported. Furthermore, the prolonged use of cyclosporine may play a role in the development of renal failure and several malignancies.Citation59,Citation60
Acitretin, which belongs to the retinoid family, regulates cellular differentiation and also possesses anti-inflammatory properties. Acitretin has been shown to have a moderate efficacy particularly on nail signs of psoriasis, although its efficacy is limited by common side effects including cheilitis, dry mouth, and skin exfoliation.Citation61,Citation62 New molecules have been studied to improve the QOL of psoriasis patients focusing on the new discoveries. Psoriasis is characterized by anomalous immune response and determined by self-cytokine networks.Citation63 The importance of these adverse effects has stimulated the development of new therapies characterized by high affinity and a safety profile.
Historically, anti-TNF-α biological agents such as infliximab, adalimumab, golimumab, certolizumab as well as etanercept, a recombinant TNF-α decoy receptor, have been used to treat psoriasis. TNF-α is a proinflammatory cytokine that plays a major role in psoriasis stimulating keratinocyte proliferation, inflammatory infiltrate in the epidermis and preventing keratinocyte apoptosis. The Anti-TNF-α family has been demonstrated to be a reasonably safe and effective treatment for plaque-type psoriasis, PsA, and nail psoriasis. Infections, such as mycobacterium tuberculosis, demyelinating diseases, congestive heart failure, induction of the formation of autologous antibodies, and production of antibodies neutralizing anti-TNF-α drugs were described as adverse reactions related to the anti-TNF-α agent.Citation64
Infliximab, a chimeric anti-TNF-α immunoglobulin (Ig) G1 monoclonal antibody that consists of human antibody constant regions and murine variable regions, was approved for PsA and plaque-type psoriasis in 2005/2006. Several studies have shown the valuable effect of infliximab on nail psoriasis – both nail bed and nail matrix psoriasis. Also in patients with severe nail psoriasis at baseline, an excellent degree of response can be achieved after 22 weeks of therapy.Citation65 The data indicate that infliximab is effective for psoriatic nail disease in the context of severe skin and/or joint involvement.
Adalimumab is a fully human anti-TNF-α IgG1 monoclonal antibody. Several studies have emphasized adalimumab to be an effective choice of treatment for psoriasis of the nail bed and nail plate. A prospective study reported a decreased incidence of onychomycosis in patients treated with adalimumab for 24 weeks. Significant improvement was noted as early as week 12 with regard to both fingernails and toenails in patients treated with adalimumab.Citation66
Etanercept is a fusion protein of the TNF-α receptor and Fc end of the IgG1 antibody. Barrera et al reported an average reduction of 51% in the NAPSI score in 562 patients enrolled in this study after 54 weeks of therapy.Citation67 The efficacy of etanercept on nail psoriasis was evident after 24 weeks of treatment, with greater improvements observed after 48 weeks.Citation68,Citation69 In conclusion, treatment of nail psoriasis with etanercept should be considered in the context of treating moderate-to-severe psoriasis with the aim of achieving improvement of both skin and psoriasis nail lesions.
Golimumab is a human monoclonal anti-TNF-α antibody. The randomized controlled trial, GO-REVEAL study, showed a mean NAPSI improvement of 52% at 52 weeks.Citation70 Substantial improvements in nail symptoms, evaluated by the NAPSI and physician’s global assessment of psoriatic nail disease, were observed in golimumab-treated patients as early as 24 weeks and were preserved or improved through 52 weeks.Citation70 Golimumab should be considered in patients with nail and joint involvement as an advantageous treatment strategy.
Certolizumab pegol is a humanized mouse monoclonal antibody to TNF-α that is chemically modified by PEGylation, and consequentially it has a prolonged half-life in patients. Certolizumab confirmed the NAPSI improvement of the other anti-TNF drugs, the clinical trial showed that the NAPSI change from baseline at week 24 was −52% with certolizumab pegol 200 mg every 2 weeks and −59% with certolizumab pegol 400 mg every 4 weeks versus −32% with placebo.Citation71 Certolizumab pegol should be considered in patients with nail and joint involvement. Advances in understanding the pathogenesis of the disease have led to the development of new treatment strategies.Citation72,Citation73 Immunological research has recently pointed out the central role of IL-17 and IL-23 in the pathogenic pathway of psoriasis.
Ustekinumab is a human anti-IL-12/23 IgG1 monoclonal antibody. Rich et al described a mean NAPSI improvement of 46.5% within 24 weeks both in nail bed and nail matrix psoriasis.Citation74 PHOENIX-1 study was a placebo-controlled study carried out in 766 psoriasis patients treated with ustekinumab 45 mg, of whom 70% were affected by nail psoriasis. Nail improvement was higher in patients with a good Psoriasis Area Severity Index (PASI) response and the improvement in the NAPSI scale ranged from 30% to 57% at 24 weeks.Citation74
Several other studies have been performed that have confirmed these results.Citation75,Citation76 Studies on ustekinumab demonstrated nail responses similar to those obtained with other biologics (about 24 weeks of treatments). Contraindications and adverse events of ustekinumab are similar to anti-TNFα treatments.Citation77
IL-17, firstly described by Yao et al in 1995,Citation78 is a family of proinflammatory cytokines that consists of IL-17A, IL-17B, IL-17C, IL-17D, IL-17E, and IL-17F, secreted by T cells, natural killer cells, mast cells, and neutrophils.Citation78 IL-17A is produced predominantly by Th17, a subset of CD4+ T cells. Anomalous production of IL-17A is strongly implicated in the pathogenesis of psoriasis and other autoimmune diseases such as rheumatoid arthritis, chronic noninfectious uveitis, and Crohn’s disease. Its neutralization has potential utility in other autoimmune disorders.Citation79,Citation80
Based on the evidence available, regarding the role of Th17 cells in psoriasis and psoriatic arthritis, three therapeutic agents against IL-17A have been developed: secukinumab, ixekizumab, and brodalumab. Significant reductions in NAPSI scores were observed using ixekizumab as early as 2 weeks, through 20 weeks, and in an open-label extension.Citation81,Citation82 Complete remission of nail disease was achieved in a high proportion of patients: 43% at week 44 and 51% at week 68.Citation82 Although few data are available, due to the recent approval of the drug, ixekizumab should be considered in patients affected by severe skin psoriasis.
Apremilast is a selective inhibitor of phosphodiesterase 4 that breaks down cyclic adenosine monophosphate (cAMP). An increase in cAMP levels downregulates the expressions of TNFα, IL-17, IL-23, and many others, and upregulates the anti-inflammatory IL-10.Citation83
A 50% reduction in NAPSI was achieved for 33.3% and 14.9% of patients given apremilast (30 mg) twice a day and placebo, respectively, at 16 weeks. At week 32 (an open-label study), a 50% reduction in NAPSI score response was observed in 45.2% of patients treated with apremilast by Papp et al in a Phase III, multicenter, double-blind, placebo-controlled study.Citation83
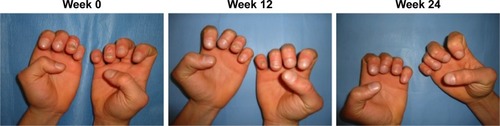
ESTEEM 1 and ESTEEM 2 evidenced a target NAPSI improvement of 43% and 60%, respectively, after 32 weeks of therapy, which was maintained through week 52.Citation84,Citation85 Apremilast has been shown to be efficient in the treatment of both nail matrix and nail bed psoriasis (). Furthermore, apremilast was generally well tolerated during Phase III clinical trials, and in particular, most adverse events were mild or moderate in intensity and the incidence rate did not appear to increase over time.Citation44 The convenience of oral administration of apremilast is associated with its safety profile, and the tolerability of the side effect profile makes this medication an attractive choice for clinicians.Citation83,Citation84 Apremilast should be considered in patients with joint and skin involvement. Apremilast is not only effective for moderate-to-severe psoriasis or PsA but it is also characterized by a good safety profile and does not require laboratory monitoring as with traditional biological agents.
Figure 2 Patient affected by nail psoriasis at baseline (week 0). The same patient after 12 weeks and 24 weeks of treatment with apremilast.
Tofacitinib is an oral Janus kinase inhibitor that recently has been investigated for psoriasis. Papp et al during Phase III studies described significant dose-dependent improvements in the NAPSI score with tofacitinib treatment in patients affected by nail psoriasis, suggesting that the JAK pathway is a critical therapeutic target of the disease.Citation86 It also offers the convenience of oral administration. In conclusion, oral tofacitinib is an effective, well-tolerated treatment option for moderate-to-severe chronic plaque psoriasis.
Discussion
While the nails represent a small portion of the body surface area, psoriasis in this area can have a disproportionate influence on a patient’s physical and psychosocial activities.Citation6 This review highlighted the primary importance of the nail involvement in the psoriatic disease. Furthermore, we emphasize the role of Candida in inducing and maintaining inflammation through IL-17; moreover, keratinocytes express IL-17 receptor and are involved in the production of aberrant K17.Citation35,Citation41
Therefore, we suggest ruling out onychomycosis in patients with nail abnormalities before concluding that the patients are affected by nail psoriasis and, in psoriatic patients, excluding the comorbidities with a fungal infection before starting treatment. This recommendation is underlined by the fact that nail psoriasis is mostly treated with immunosuppressive drugs, like steroids, methotrexate, or biologics, which may aggravate mycotic nail infections.Citation44 To manage a patient co-affected by onychomycosis and psoriasis, we suggest to initially treat the fungal infection for at least 3 months using a systemic antifungal therapy and to avoid local topical steroids and subsequently treat the psoriatic disease.Citation24,Citation62
The commonly used disease modifying antirheumatic drugs (DMARDs) can cause adverse effects that require monitoring,Citation54 while anti-TNF-α (eg, adalimumab, etanercept, infliximab, certolizumab pegol, and golimumab) can cause infections, malignancies, and production of antidrug antibodies that can impact effectiveness; administration of TNF-α is through injection or infusion.Citation64,Citation66,Citation70,Citation71
Currently, two recent meta-analyses – regarding adalimumab, etanercept, golimumab, and infliximab – showed that differences in terms of efficacy of these drugs cannot be demonstrated.Citation87,Citation88 Additionally, in one of these studies, no significant differences in terms of safety of these drugs were established.Citation87 Though the mechanism of action of the anti-TNF biologic drugs is similar, some molecular differentiations might justify the successful switch from one to other.
Currently, use of apremilast and tofacitinib favors an early healing of nail psoriasis because they act directly on the pathogenic targets, distressing the inflammatory signals associated with the initiation and maintenance of the disease activity without altering the microbial flora and, as with several conventional synthetic DMARDs, they are characterized by the convenience of oral administration.Citation44,Citation84 The Medical Board of the National Psoriasis Foundation recommends systemic treatments, including apremilast, for patients with major skin and nail disease or skin, nail, and joint disease.Citation44
Conclusion
This review underlines that the number of treatments for PsA and psoriasis has increased considerably in recent years and will probably lead to the authorization of new therapies in the future. Given the heterogeneity of the disease, the therapy should be personalized to individual cases. Principal features of these strategies should consider optimizing QOL, eliciting inclinations and goals, balancing risks and benefits, setting priorities, discontinuing needless medications, and starting beneficial medications, however, prioritizing simple regimens and integrating care. Models of care should be custom-made on patient necessity. Achieving this transition will require a change from disease-specific to patient-specific models of care delivery.Citation88
Further investigations including prognostic biomarkers and patient stratification are necessary to allow clinicians the possibility of making better use of various biologic and non-biologic treatment options available.
Disclosure
The authors report no conflicts of interest in this work.
References
- ParisiRSymmonsDPGriffithsCEAshcroftDMIdentification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project teamGlobal epidemiology of psoriasis: a systematic review of incidence and prevalenceJ Invest Dermatol20131332 377 38523014338
- GordonKBStroberBEUnderstanding therapeutic pathways and comorbidities in psoriasisSemin Cutan Med Surg2014332 Suppl 2 S20 S2324979540
- DavidoviciBBSattarNPrinzJPsoriasis and systemic inflammatory diseases: potential mechanistic links between skin disease and co-morbid conditionsJ Invest Dermatol20101307 1785 179620445552
- GottliebABDannFComorbidities in patients with psoriasisAm J Med200912212 1150.e1 e9
- GisondiPFerrazziAGirolomoniGMetabolic comorbidities and psoriasisActa Dermatovenerol Croat2010184 297 30421251450
- EspositoMSaracenoRGiuntaAMaccaroneMChimentiSAn Italian study on psoriasis and depressionDermatology20062122 123 12716484818
- EspositoMFaleriSBabinoGFrom patients’ needs to treatment outcomes in psoriasis: results from the ‘pSORRIDI’ experienceJ Int Med Res2016441 Suppl 1 95 9927683149
- ThamSNLimJJTayShChiewYFChuaTNTanETanTClinical observations on nail changes in psoriasisAnn Acad Med Singapore1988174 482 4853265604
- BaranRde BerkerDARHolzbergMThomasLBaran and Dawber’s Diseases of the Nail and Their Management4th edWiley-Blackwell2012
- HeydendaelVMDe BorgieCASpulsPIBossuytPMBosJDDe RieMAThe burden of psoriasis is not determined by disease severity onlyJ Investig Dermatol Symp Proc20049 131 135
- ReichKApproach to managing patients with nail psoriasisJ Eur Acad Dermatol Venereol200923Suppl 1 15 2119686381
- PaschMCNail psoriasis: a review of treatment optionsDrugs201676 675 70527041288
- de JongEMSeegersBAGulinckMKBoezemanJBvan de KerkhofPCPsoriasis of the nails associated with disability in a large number of patients: results of a recent interview with 1,728 patientsDermatology19961934 300 3038993953
- LawryMBiological therapy and nail psoriasisDermatol Ther2007201 60 6717403261
- ArmestoSEsteveACoto-SeguraPNail psoriasis in individuals with psoriasis vulgaris: a study of 661 patientsActas Dermosifiliogr20111025 365 372 Spanish21514549
- McGonagleDAshZDickieLMcDermottMAydinSZThe early phase of psoriatic arthritisAnn Rheum Dis201170Suppl 1 i71 i7621339224
- LangleyRGDaudenETreatment and management of psoriasis with nail involvement: a focus on biologic therapyDermatology2010221Suppl 1 29 4220733313
- KlaassenKMvan de KerkhofPCPaschMCNail psoriasis: a questionnaire-based surveyBr J Dermatol20131692 314 31923550612
- JiaravuthisanMMSassevilleDVenderRBMurphyFMuhnCYPsoriasis of the nail: anatomy, pathology, clinical presentation, and a review of the literature on therapyJ Am Acad Dermatol2007571 1 2717572277
- LangenbruchARadtkeMAKrenselMJacobiAReichKAugustinMNail involvement as a predictor of concomitant psoriatic arthritis in patients with psoriasisBr J Dermatol20141715 1123 112825040629
- ZaiasNPsoriasis of the nail. A clinical-pathologic studyArch Dermatol1969995 567 5795780963
- RichPScherRKNail psoriasis severity index: a useful tool for evaluation of nail psoriasisJ Am Acad Dermatol2003492 206 21212894066
- SzepietowskiJCSalomonJDo fungi play a role in psoriatic nails?Mycoses2007506 437 44217944702
- RigopoulosDPapanagiotouVDanielR3rdPiracciniBMOnychomycosis in patients with nail psoriasis: a point to point discussionMycoses2017601 6 1027523738
- ElewskiBEOnychomycosis: pathogenesis, diagnosis, and managementClin Microbiol Rev1998113 415 4299665975
- ZaiasNEscovarSXZaiacMNFinger and toenail onycholysisJ Eur Acad Dermatol Venereol2015295 848 85325512134
- ZisovaLValtchevVSotiriouEGospodinovDMateevGOnychomycosis in patients with psoriasis: a multicentre studyMycoses2012552 143 14721771106
- NestleFOKaplanDHBarkerJPsoriasisN Engl J Med20093615 496 50919641206
- MakRKHundhausenCNestleFOProgress in understanding the immunopathogenesis of psoriasisActas Dermosifiliogr2009100Suppl 2 2 1320096156
- De LucaAZelanteTD’AngeloCIL-22 defines a novel immune pathway of antifungal resistanceMucosal Immunol201034 361 37320445503
- KisandKBøe WolffASPodkrajsekKTChronic mucocutaneous candidiasis in APECED or thymoma patients correlates with autoimmunity to Th17-associated cytokinesJ Exp Med20102072 299 30820123959
- HupplerARContiHRHernández-SantosNDarvilleTBiswasPSGaffenSLRole of neutrophils in IL-17-dependent immunity to mucosal candidiasisJ Immunol20141924 1745 175224442441
- KagamiSRizzoHLKurtzSEMillerLSBlauveltAIL-23 and IL-17A, but not IL-12 and IL-22, are required for optimal skin host defense against Candida albicansJ Immunol20101859 5453 546220921529
- WaldmanAGilharADuekLBerdicevskyIIncidence of Candida in psoriasis – a study on the fungal flora of psoriatic patientsMycoses2001443–4 77 8111413927
- Taheri SarvtinMShokohiTHajheydariZYazdaniJHedayatiMTEvaluation of candidal colonization and specific humoral responses against Candida albicans in patients with psoriasisInt J Dermatol20145312 e555 e56025427068
- FryLBakerBSTriggering psoriasis: the role of infections and medicationsClin Dermatol2007256 606 61518021899
- GoldminzAMAuSCKimNGottliebABLizzulPFNF-κB: an essential transcription factor in psoriasisJ Dermatol Sci2013692 89 9423219896
- RashmiRRaoKSBasavarajKHA comprehensive review of biomarkers in psoriasisClin Exp Dermatol2009346 658 66319558584
- Di MeglioPPereraGKNestleFOThe multitasking organ: recent insights into skin immune functionImmunity2011356 857 86922195743
- IyerSSChengGRole of interleukin 10 transcriptional regulation in inflammation and autoimmune diseaseCrit Rev Immunol2012321 23 6322428854
- SauliteIPilmaneMKisisJExpression of antimicrobial peptides in nail psoriasis and normal nailsActa Derm Venereol2017975 644 64527983743
- McGonagleDBenjaminMTanALThe pathogenesis of psoriatic arthritis and associated nail disease: not autoimmune after all?Curr Opin Rheumatol2009214 340 34719424069
- GniadeckiRNext-generation antipsoriatic drugs: small molecules joinBr J Dermatol20151736 1355 1356
- CrowleyJJWeinbergJMWuJJRobertsonADVan VoorheesASNational Psoriasis FoundationTreatment of nail psoriasis: best practice recommendations from the Medical Board of the National Psoriasis FoundationJAMA Dermatol20151511 87 9425471223
- SagginiAChimentiSChiricozziAIL-6 as a druggable target in psoriasis: focus on pustular variantsJ Immunol Res20142014 96406925126586
- ManhartRRichPNail PsoriasisClin Exp Rheumatol201533 S7 S13
- Marquez BalbasGSanchez ReganaMUmbert MilletPTacalcitol ointment for the treatment of nail psoriasisJ Dermatolog Treat2009205 308 31019367480
- ReichrathJVitamin D and the skin: an ancient friend, revisitedExp Dermatol2007167 618 62517576242
- ScherRKStillerMZhuYITazarotene 0.1% gel in the treatment of fingernail psoriasis: a double-blind, randomized, vehicle-controlled studyCutis2001685 355 35811766122
- BianchiLSodaRDiluvioLChimentiSTazarotene 0.1% gel for psoriasis of the fingernails and toenails: an open, prospective studyBr J Dermatol20031491 207 20912890227
- CampioneEPaternòEJCostanzaGTazarotene as alternative topical treatment for onychomycosisDrug Des Devel Ther20159 879 886
- SodaRDiluvioLBianchiLChimentiSTreatment of trachyonychia with tazaroteneClin Exp Dermatol2005303 301 30215807700
- Fischer-LevanciniCSanchez-ReganaMLlambiFCollgrosHExposito-SerranoVUmbert-MilletPNail psoriasis: treatment with tazarotene 0.1% hydrophilic ointmentActas Dermosifiliogr20121038 725 728 Spanish22818395
- BlauveltAArmstrongAWKruegerGGEssential truths for the care and management of moderate-to-severe psoriasisJ Drugs Dermatol2015148 805 81226267724
- GumuselMOzdemirMMevlitogluIBodurSEvaluation of the efficacy of methotrexate and cyclosporine therapies on psoriatic nails: a one-blind, randomized studyJ Eur Acad Dermatol Venereol2011259 1080 108421118309
- ReichKLangleyRGPappKAA 52-week trial comparing briakinumab with methotrexate in patients with psoriasisN Engl J Med201136517 1586 159622029980
- Sanchez-ReganaMSola-OrtigosaJAlsina-GibertMVidal-FernandezMUmbert-MilletPNail psoriasis: a retrospective study on the effectiveness of systemic treatments (classical and biological therapy)J Eur Acad Dermatol Venereol2011255 579 58621198950
- DemirsoyEOKıranRSalmanSEffectiveness of systemic treatment agents on psoriatic nails: a comparative studyJ Drugs Dermatol2013129 1039 104324002153
- SyutoTAbeMIshibuchiHIshikawaOSuccessful treatment of psoriatic nails with low-dose cyclosporine administrationEur J Dermatol2007173 248 249
- KaranikolasGNKoukliEMKatsaliraAAdalimumab or cyclosporine as monotherapy and in combination in severe psoriatic arthritis: results from a prospective 12-month nonrandomized unblinded clinical trialJ Rheumatol20113811 2466 247421885499
- RicceriFPescitelliLTripoLBassiAPrignanoFTreatment of severe nail psoriasis with acitretin: an impressive therapeutic resultDermatol Ther2013261 77 7823384024
- TostiARicottiCRomanelliPCameliNPiracciniBMEvaluation of the efficacy of acitretin therapy for nail psoriasisArch Dermatol20091453 269 27119289755
- LowesMASuarez-FarinasMKruegerJGImmunology of psoriasisAnnu Rev Immunol201432 227 25524655295
- PirowskaMMGoździalskaALipko-GodlewskaSAuto-immunogenicity during anti-TNF therapy in patients with psoriasis and psoriatic arthritisPostepy Dermatol Alergol2015324 250 25426366147
- BianchiLBergaminAde FeliceCCapriottiEChimentiSRemission and time of resolution of nail psoriasis during infliximab therapyJ Am Acad Dermatol2005524 736 73715793554
- RigopoulosDGregoriouSLazaridouETreatment of nail psoriasis with adalimumab: an open label unblinded studyJ Eur Acad Dermatol Venereol2010245 530 53419807825
- BarreraMVHabicheynSMendiolaMVHerrera CeballosEEtanercept in the treatment and retreatment of psoriasis in daily clinical practiceEur J Dermatol2008186 683 68719049936
- RallisEStavropoulouERigopoulosDVerrosCRapid response of nail psoriasis to etanerceptJ Rheumatol2008353 544 54518322982
- CoelhoJDDiamantinoFLestreSFerreiraAMTreatment of severe nail psoriasis with etanerceptIndian J Dermatol Venereol Leprol2011771 72 7421220889
- KavanaughAvan der HeijdeDMcInnesIBGolimumab in psoriatic arthritis: one-year clinical efficacy, radiographic, and safety results from a phase III, randomized, placebo-controlled trialArthritis Rheum2012648 2504 251722378566
- MeasePJFleischmannRDeodharAAEffect of certolizumab pegol on signs and symptoms in patients with psoriatic arthritis: 24-week results of a Phase 3 double-blind randomised placebo-controlled study (RAPID-PsA)Ann Rheum Dis2014731 48 5523942868
- ParkHLiZYangXOA distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17Nat Immunol2005611 1133 114116200068
- CaiYFlemingCYanJDermal γδ T cells – a new player in the pathogenesis of psoriasisInt Immunopharmacol2013163 388 39123499509
- RichPBourcierMSofenHPHOENIX 1 investigatorsUstekinumab improves nail disease in patients with moderate-to-severe psoriasis: results from PHOENIX 1Br J Dermatol20141702 398 40724117389
- RigopoulosDGregoriouSMakrisMIoannidesDEfficacy of ustekinumab in nail psoriasis and improvement in nail-associated quality of life in a population treated with ustekinumab for cutaneous psoriasis: an open prospective unblinded studyDermatology20112234 325 32922134046
- PatsatsiAKyriakouASotiriadisDUstekinumab in nail psoriasis: an open-label, uncontrolled, nonrandomized studyJ Dermatol Treat2013242 96 100
- IgarashiAKatoTKatoMSongMNakagawaHJapanese Ustekinumab Study GroupEfficacy and safety of ustekinumab in Japanese patients with moderate-to severe plaque-type psoriasis: long-term results from a phase 2/3 clinical trialJ Dermatol2012393 242 25221955098
- YaoZPainterSLFanslowWCHuman IL-17: a novel cytokine derived from T cellsJ Immunol199515512 5483 54867499828
- GaffenSLJainRGargAVCuaDJThe IL-23-IL-17 immune axis: from mechanisms to therapeutic testingNat Rev Immunol2014149 585 60025145755
- GaffenSLThe role of interleukin-17 in the pathogenesis of rheumatoid arthritisCurr Rheumatol Rep2009115 365 37019772832
- LeonardiCMathesonRZachariaeCAnti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasisN Engl J Med201236613 1190 119922455413
- LangleyRGRichPMenterAImprovement of scalp and nail lesions with ixekizumab in a phase 2 trial in patients with chronic plaque psoriasisJ Eur Acad Dermatol Venereol2015299 1763 177025693783
- ThaçiDKimballAFoleyPApremilast, an oral phosphodiesterase 4 inhibitor, improves patient-reported outcomes in the treatment of moderate to severe psoriasis: results of two phase III randomized, controlled trialsJ Eur Acad Dermatol Venereol2016313 498 50627538241
- PappKReichKLeonardiCLApremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1)J Am Acad Dermatol2015731 37 4926089047
- RichPGooderhamMBachelezHApremilast, an oral phosphodiesterase 4 inhibitor, in patients with difficult-to-treat nail and scalp psoriasis: results of 2 phase III randomized, controlled trials (ESTEEM 1 and ESTEEM 2)J Am Acad Dermatol2016741 134 14226549249
- PappKAMenterMAAbeMOPT Pivotal 1 and OPT Pivotal 2 investigators. Tofacitinib, an oral Janus kinase inhibitor, for the treatment of chronic plaque psoriasis: results from two randomized, placebo-controlled, phase III trialsBr J Dermatol20151734 949 96126149717
- Fénix-CaballeroSAlegre-del ReyEJCastaño-LaraRPuigventós-LatorreFBorrero-RubioJMLópez-VallejoJFDirect and indirect comparison of the efficacy and safety of adalimumab, etanercept, infliximab and golimumab in psoriatic arthritisJ Clin Pharm Ther2013384 286 29323590560
- ThorlundKDruytsEAviña-ZubietaJAMillsEJAnti-tumor necrosis factor (TNF) drugs for the treatment of psoriatic arthritis: an indirect comparison meta-analysisBiologics20126 417 42723271892
- BiermanASTinettiMEPrecision medicine to precision care: managing multimorbidityLancet201638810061 2721 272327924764
